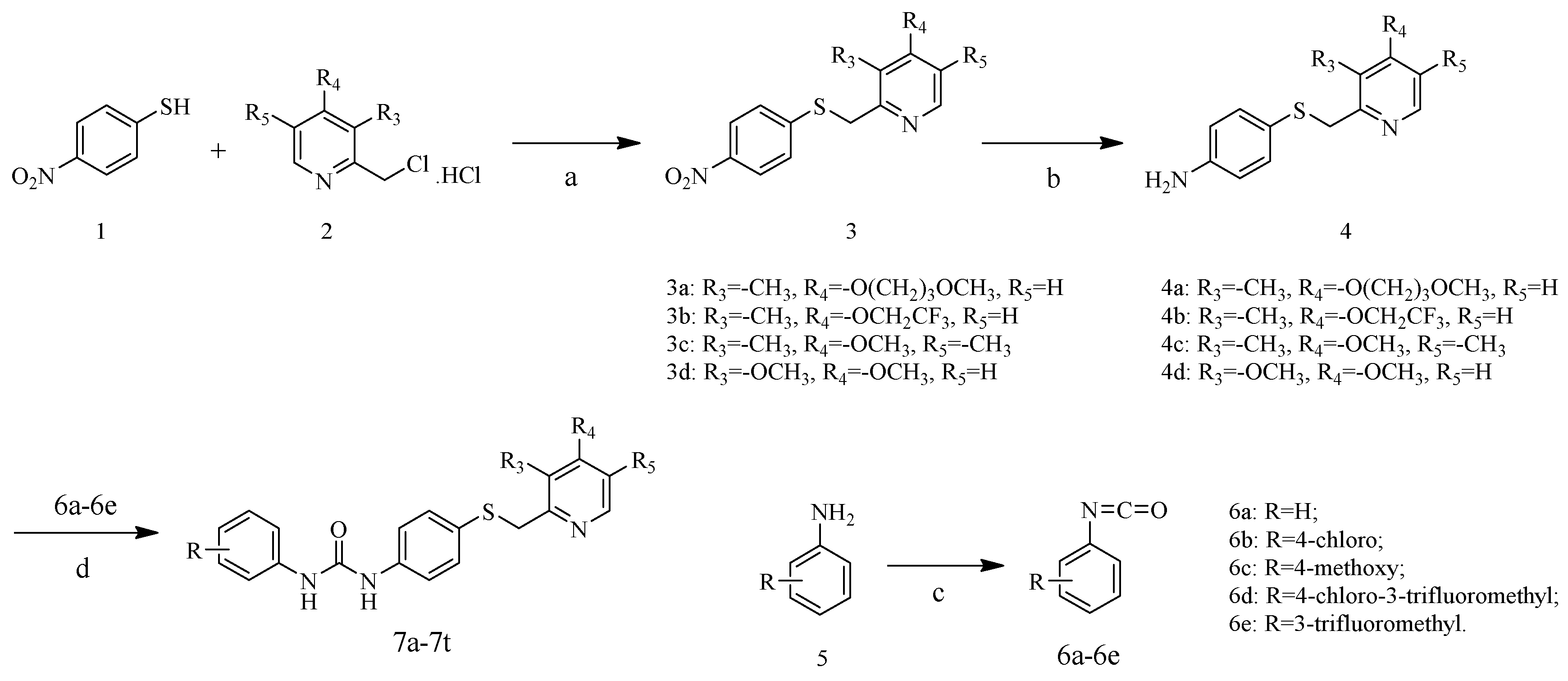
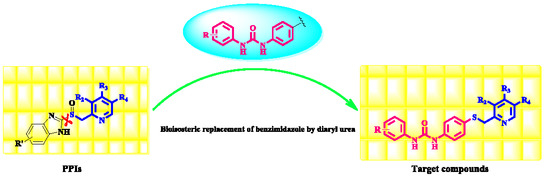
All reagents were obtained from commercial suppliers and used without further purification. Reaction progress was monitored by thin layer chromatography (TLC) on silica gel plates. The spots were visualized by ultraviolet (UV) light (254 nm). The column chromatography was performed using 200−300 mesh silica gel (Qingdao PUKE, Qingdao, China). Melting points were obtained by X-5 micro-melting point apparatus (Beijing Zhongyi Boteng Technology Co., Ltd., Beijing, China) and were uncorrected. 1H-NMR and 13C-NMR spectra were recorded on Bruker NMR spectrophotometers (Karlsruhe, Germany) using DMSO-d6 as the solvent and TMS as the internal standard. Mass spectra were measured with an electrospray (ESI-MS) on a Waters spectrometer (Waters Corporation, Milford, MA, USA). High resolution mass spectrometry (HRMS) analyses were performed on an Agilent Technologies 6530 Accurate-Mass Q-TOF Mass Spectrometer (Santa Clara, CA, USA). The purities were determined by high-performance liquid chromatography (HPLC) using an Agilent 1100 series HPLC (Santa Clara, CA, USA).
3.1.3. General Procedure for the Preparation of the Target Compounds (7a–7t)
To a solution of BTC (1 mmol) in CH2Cl2 (20 mL) was added dropwise to primary aromatic amine 5 (1 mmol) in CH2Cl2 (20 mL) followed by the dropwise addition of triethylamine (1 mL) in CH2Cl2 (10 mL). The solvent was evaporated. The resulting residue was dissolved in CH2Cl2 (20 mL), and intermediates (4a–4d) (1 mmol) in CH2Cl2 (10 mL) was added dropwise. The mixture was stirred for about 3 h, monitored by TLC. After the reaction completed, the solvent was washed with water and brine, then dried over anhydrous magnesium sulfate. The mixture was filtered, the filtrate was evaporated and purified by silica gel chromatography (CH2Cl2/MeOH = 60/1, v/v) to obtain target compounds.
1-{4-{{[4-(3-Methoxypropoxy)-3-methylpyridin-2-yl]methyl}thio}phenyl}-3-phenylurea (7a)
![Molecules 24 02108 i001]()
Compound 7a was prepared according to the general procedure by using compound 4a (0.32 g, 1 mmol) and aniline (0.10 g, 1 mmol), obtained a white solid (0.20 g) in 45.1% yield. m.p. 105.2–106.8 °C. IR (KBr, cm−1): υ 3421.1, 2922.7, 2852.4, 1596.1, 1545.1, 1492.4, 1460.8, 1440.3, 1398.2, 1385.2, 1309.3, 1231.3, 1174.2, 1092.1, 1006.7, 894.6, 832.0, 799.2, 751.9, 693.4, 617.3, 507.5. 1H-NMR (400 MHz, DMSO-d6) δ 8.74 (s, 1H), 8.69 (s, 1H), 8.17 (d, J = 5.6 Hz, 1H), 7.46 (s, 1H), 7.44 (s, 1H), 7.41 (s, 1H), 7.39 (s, 1H), 7.32 (s, 1H), 7.31–7.29 (m, 1H), 7.28 (s, 1H), 7.26 (s, 1H), 6.97 (t, J = 7.3 Hz, 1H), 6.90 (d, J = 5.7 Hz, 1H), 4.22 (s, 2H), 4.09 (t, J = 6.2 Hz, 2H), 3.48 (t, J = 6.2 Hz, 2H), 3.25 (s, 3H), 2.12 (s, 3H), 2.02–1.94 (m, 2H). 13C-NMR (101 MHz, DMSO-d6) δ 163.17, 156.31, 152.87, 147.91, 140.07, 131.77, 129.23, 128.07, 122.34, 120.30, 119.09, 118.69, 106.49, 68.79, 65.48, 58.44, 31.14, 29.16, 10.88. ESI-MS (m/z): 438.4 ([M + H]+), 460.2 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C24H27N3O3S, 438.1846 ([M + H]+), found: 438.1856 ([M + H]+). Purity (HPLC): 99.27%.
1-(4-Chlorophenyl)-3-{4-{{[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methyl}thio}phenyl}urea (7b)
![Molecules 24 02108 i002]()
Compound 7b was prepared according to the general procedure by using compound 4a (0.32 g, 1 mmol) and 4-chloroaniline (0.13 g, 1 mmol), obtained a white solid (0.36 g) in 75.3% yield. m.p. 191.7–192.5 °C. IR (KBr, cm−1): υ 3428.1, 2923.1, 2852.9, 1631.8, 1490.8, 1398.9, 1384.8, 1298.9, 1273.5, 1237.0, 1174.2, 1121.4, 1086.2, 1008.0, 881.2, 832.1, 702.8, 619.7, 506.0. 1H-NMR (400 MHz, DMSO-d6) δ 8.83 (s, 1H), 8.77 (s, 1H), 8.17 (d, J = 5.6 Hz, 1H), 7.49 (d, J = 2.2 Hz, 1H), 7.47 (d, J = 2.1 Hz, 1H), 7.40 (d, J = 2.0 Hz, 1H), 7.39 (d, J = 2.1 Hz, 1H), 7.34 (s, 1H), 7.33 (s, 1H), 7.32 (d, J = 2.1 Hz, 1H), 7.31 (s, 1H), 6.90 (d, J = 5.7 Hz, 1H), 4.22 (s, 2H), 4.08 (t, J = 6.2 Hz, 2H), 3.48 (t, J = 6.2 Hz, 2H), 3.25 (s, 3H), 2.12 (s, 3H), 2.00–1.94 (m, 2H). 13C-NMR (101 MHz, DMSO-d6) δ 163.09, 156.35, 152.77, 147.99, 139.09, 138.88, 131.62, 129.07, 128.41, 125.86, 120.22, 119.24, 106.47, 68.80, 65.45, 58.43, 31.14, 29.16, 10.89. ESI-MS (m/z): 473.3 ([M + H]+), 495.2 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C24H26ClN3O3S, 472.1456 ([M + H]+), found: 472.1467 ([M + H]+). Purity (HPLC): 98.66%.
1-(4-Methoxyphenyl)-3-{4-{{[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methyl}thio}phenyl}urea (7c)
![Molecules 24 02108 i003]()
Compound 7c was prepared according to the general procedure by using compound 4a (0.32 g, 1 mmol) and 4-methoxyaniline (0.12 g, 1 mmol), obtained a white solid (0.22 g) in 46.3% yield. m.p. 144.8–146.6 °C. IR (KBr, cm−1): υ 3428.6, 2984.7, 2923.0, 2852.7, 1635.5, 1599.5, 1562.6, 1510.7, 1492.7, 1461.8, 1441.3, 1398.1, 1289.8, 1245.2, 1173.5, 1120.3, 1093.5, 1035.2, 1005.6, 800.0, 617.1, 548.4, 522.7. 1H-NMR (400 MHz, DMSO-d6) δ 8.64 (s, 1H), 8.47 (s, 1H), 8.16 (d, J = 5.7 Hz, 1H), 7.39 (d, J = 1.9 Hz, 1H), 7.37 (d, J = 2.2 Hz, 1H), 7.35 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.3 Hz, 1H), 7.30 (d, J = 2.1 Hz, 1H), 7.29 (d, J = 2.0 Hz, 1H), 6.89 (d, J = 5.6 Hz, 1H), 6.87 (d, J = 2.2 Hz, 1H), 6.86 (d, J = 2.2 Hz, 1H), 4.21 (s, 2H), 4.08 (t, J = 6.2 Hz, 2H), 3.71 (s, 3H), 3.48 (t, J = 6.2 Hz, 2H), 3.25 (s, 3H), 2.12 (s, 3H), 2.00–1.94 (m, 2H). 13C-NMR (101 MHz, DMSO-d6) δ 163.06, 156.41, 154.97, 153.07, 148.01, 139.37, 133.09, 131.78, 127.82, 120.52, 120.22, 118.98, 114.44, 106.44, 68.79, 65.42, 58.43, 55.63, 31.14, 29.16, 10.88. ESI-MS (m/z): 468.4 ([M + H]+), 490.2 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C25H29N3O4S, 468.1952 ([M + H]+), found: 468.1959 ([M + H]+). Purity (HPLC): 98.91%.
1-[4-Chloro-3-(trifluoromethyl)phenyl]-3-{4-{{[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methyl}thio}phenyl}urea (7d)
![Molecules 24 02108 i004]()
Compound 7d was prepared according to the general procedure by using compound 4a (0.32 g, 1 mmol) and 3-chloro-4-(trifluoromethyl)aniline (0.20 g, 1 mmol), obtained a white solid (0.24 g) in 43.8% yield. m.p. 136.0–137.2 °C. IR (KBr, cm−1): υ 3425.2, 2922.0, 2852.7, 1590.1, 1546.1, 1482.2, 1463.1, 1384.5, 1306.6, 1175.4, 1117.7, 1034.4, 820.6. 1H-NMR (400 MHz, DMSO-d6) δ 9.16 (s, 1H), 8.89 (s, 1H), 8.16 (d, J = 5.6 Hz, 1H), 8.10 (d, J = 2.2 Hz, 1H), 7.63 (d, J = 2.2 Hz, 1H), 7.62 (s, 1H), 7.42 (d, J = 2.0 Hz, 1H), 7.40 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.1 Hz, 1H), 7.32 (d, J = 2.0 Hz, 1H), 6.89 (d, J = 5.7 Hz, 1H), 4.23 (s, 2H), 4.09 (t, J = 6.2 Hz, 2H), 3.48 (t, J = 6.2 Hz, 2H), 3.25 (s, 3H), 2.13 (s, 3H), 2.00–1.94 (m, 2H). 13C-NMR (101 MHz, DMSO-d6) δ 163.08, 156.34, 152.76, 148.01, 139.76, 138.48, 132.45, 131.46, 128.91, 123.55, 122.80, 120.24, 119.57, 117.23, 106.48, 68.80, 65.44, 58.43, 31.14, 29.16, 10.89. ESI-MS (m/z): 540.2 ([M + H]+), 562.0 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C25H25ClF3N3O3S, 540.1330 ([M + H]+), found: 540.1320 ([M + H]+). Purity (HPLC): 97.33%.
1-{4-{{[4-(3-Methoxypropoxy)-3-methylpyridin-2-yl]methyl}thio}phenyl}-3-[3-(trifluoromethyl)phenyl]urea (7e)
![Molecules 24 02108 i005]()
Compound 7e was prepared according to the general procedure by using compound 4a (0.32 g, 1 mmol) and 3-(trifluoromethyl)aniline (0.16 g, 1 mmol), obtained a white solid (0.27 g) in 53.1% yield. m.p. 136.1–137.9 °C. IR (KBr, cm−1): υ 3327.8, 2958.7, 2927.8, 2859.0, 2377.4, 2350.2, 2311.0, 1724.0, 1648.5, 1585.5, 1552.2, 1492.5, 1465.2, 1397.5, 1338.6, 1295.8, 1230.8, 1166.2, 1116.1, 1092.4, 1068.9, 1005.4, 890.0, 804.1, 732.9, 699.2, 602.3, 505.8. 1H-NMR (400 MHz, DMSO-d6) δ 9.05 (s, 1H), 8.84 (s, 1H), 8.17 (d, J = 5.6 Hz, 1H), 8.09 (s, 0H), 8.00 (d, J = 2.3 Hz, 1H), 7.57 (d, J = 8.4 Hz, 1H), 7.51 (t, J = 7.9 Hz, 1H), 7.42 (d, J = 2.0 Hz, 1H), 7.40 (s, 1H), 7.33 (d, J = 2.1 Hz, 1H), 7.32 (d, J = 2.1 Hz, 1H), 7.30 (s, 1H), 6.90 (d, J = 5.7 Hz, 1H), 4.23 (s, 2H), 4.09 (t, J = 6.2 Hz, 2H), 3.48 (t, J = 6.2 Hz, 2H), 3.25 (s, 3H), 2.13 (s, 3H), 2.01–1.94 (m, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 163.11, 156.32, 152.85, 147.97, 140.98, 138.68, 131.55, 130.34, 129.10, 128.64, 122.30, 120.25, 119.40, 118.53, 106.46, 68.79, 65.44, 58.42, 31.13, 29.16, 10.88. ESI-MS (m/z): 506.3 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C25H26F3N3O3S, 506.1720 ([M + H]+), found: 506.1728 ([M + H]+). Purity (HPLC): 97.09%.
1-{4-{{[3-Methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl]methyl}thio}phenyl}-3-phenylurea (7f)
![Molecules 24 02108 i006]()
Compound 7f was prepared according to the general procedure by using compound 4b (0.33 g, 1 mmol) and aniline (0.10 g, 1 mmol), obtained a white solid (0.18 g) in 39.9% yield. m.p. 143.3–145.1 °C. IR (KBr, cm−1): υ 3424.1, 2923.9, 2852.6, 1687.8, 1639.9, 1600.0, 1548.5, 1495.9, 1441.2, 1399.4, 1384.7, 1307.8, 1284.4, 1266.4, 1232.1, 1176.6, 1112.2, 970.6, 915.9, 854.7, 836.1, 801.5, 783.1, 751.0, 696.2, 657.1, 618.8, 574.4. 1H-NMR (400 MHz, DMSO-d6) δ 8.73 (s, 1H), 8.67 (s, 1H), 8.24 (d, J = 5.7 Hz, 1H), 7.46 (s, 1H), 7.44 (s, 1H), 7.41 (s, 1H), 7.39 (s, 1H), 7.32 (s, 1H), 7.30 (s, 1H), 7.28 (s, 1H), 7.26 (s, 1H), 7.03 (d, J = 5.7 Hz, 1H), 6.97 (t, J = 7.3 Hz, 1H), 4.89 (q, J = 8.7 Hz, 2H), 4.25 (s, 2H), 2.16 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 161.62, 157.24, 152.87, 148.00, 140.05, 139.22, 131.90, 129.23, 127.85, 125.66, 122.90, 122.35, 120.44, 119.10, 118.71, 107.07, 64.92, 31.13, 10.74. ESI-MS (m/z): 448.4 ([M + H]+), 470.2 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C22H20F3N3O2S, 448.1301 ([M + H]+), found: 448.1295 ([M + H]+). Purity (HPLC): 97.04%.
1-(4-Chlorophenyl)-3-{4-{{[3-methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl]methyl}thio}phenyl}urea (7g)
![Molecules 24 02108 i007]()
Compound 7g was prepared according to the general procedure by using compound 4b (0.33 g, 1 mmol) and 4-chloroaniline (0.13 g, 1 mmol), obtained a white solid (0.29 g) in 61.0% yield. m.p. 203.6–205.2 °C. IR (KBr, cm−1): υ 3424.1, 2984.9, 2923.1, 2851.9, 2350.0, 2311.0, 1611.4, 1548.6, 1491.9, 1440.3, 1399.6, 1384.9, 1370.1, 1311.1, 1268.3, 1172.6, 1111.7, 1051.4, 1004.4, 897.2, 798.3, 668.5, 615.4. 1H-NMR (400 MHz, DMSO-d6) δ 8.81 (s, 1H), 8.75 (s, 1H), 8.23 (d, J = 5.7 Hz, 1H), 7.48 (d, J = 2.1 Hz, 1H), 7.47 (d, J = 2.2 Hz, 1H), 7.40 (d, J = 2.0 Hz, 1H), 7.38 (s, 1H), 7.33 (d, J = 2.1 Hz, 1H), 7.32 (d, J = 2.2 Hz, 1H), 7.31 (s, 1H), 7.30 (d, J = 1.9 Hz, 1H), 7.03 (d, J = 5.7 Hz, 1H), 4.88 (q, J = 8.7 Hz, 2H), 4.25 (s, 2H), 2.16 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 161.62, 157.22, 152.77, 148.00, 139.08, 138.98, 131.80, 129.07, 128.11, 125.87, 122.90, 120.43, 120.23, 119.23, 107.08, 65.09, 31.14, 10.74. ESI-MS (m/z): 482.6 ([M + H]+), 504.3 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C22H19ClF3N3O2S, 482.0911 ([M + H]+), found: 482.0916 ([M + H]+). Purity (HPLC): 99.19%.
1-(4-Methoxyphenyl)-3-{4-{{[3-methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl]methyl}thio}phenyl}urea (7h)
![Molecules 24 02108 i008]()
Compound 7h was prepared according to the general procedure by using compound 4b (0.33 g, 1 mmol) and 4-methoxyaniline (0.12 g, 1 mmol), obtained a white solid (0.22 g) in 45.9% yield. m.p. 171.3–172.1 °C. IR (KBr, cm−1): υ 3383.8, 2922.1, 2851.4, 2377.6, 2349.6, 1703.0, 1656.7, 1619.2, 1591.3, 1546.4, 1511.1, 1492.7, 1465.5, 1399.3, 1312.1, 1264.5, 1231.2, 1175.1, 1112.8, 1040.3, 1006.7, 970.2, 918.1, 897.2, 831.5, 799.7, 658.3. 1H-NMR (400 MHz, ) δ 8.64 (s, 1H), 8.47 (s, 1H), 8.16 (d, J = 5.7 Hz, 1H), 7.39 (d, J = 1.9 Hz, 1H), 7.37 (d, J = 2.2 Hz, 1H), 7.35 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.3 Hz, 1H), 7.30 (d, J = 2.1 Hz, 1H), 7.29 (d, J = 2.0 Hz, 1H), 6.89 (d, J = 5.6 Hz, 1H), 6.87 (d, J = 2.2 Hz, 1H), 6.86 (d, J = 2.2 Hz, 1H), 4.91-4.85 (m, 2H), 4.21 (s, 2H), 3.71 (s, 3H), 2.12 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 161.84, 156.03, 154.81, 153.88, 153.39, 148.09, 133.72, 133.38, 125.65, 121.84, 120.37, 120.33, 115.40, 114.43, 108.04, 65.10, 55.63, 31.13, 10.44. ESI-MS (m/z): 478.3 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C23H22F3N3O3S, 478.1407 ([M + H]+), found: 478.1408 ([M + H]+). Purity (HPLC): 99.66%.
1-[4-Chloro-3-(trifluoromethyl)phenyl]-3-{4-{{[3-methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl]methyl}thio}phenyl}urea (7i)
![Molecules 24 02108 i009]()
Compound 7i was prepared according to the general procedure by using compound 4b (0.33 g, 1 mmol) and 3-chloro-4-(trifluoromethyl)aniline (0.20 g, 1 mmol), obtained a white solid (0.27 g) in 49.5% yield. m.p. 142.0–143.0 °C. IR (KBr, cm−1): υ 3422.3, 2922.1, 2852.6, 1587.4, 1547.6, 1480.8, 1419.0, 1309.8, 1263.9, 1177.2, 1111.0, 1035.5, 974.7, 819.3, 618.3. 1H-NMR (400 MHz, DMSO-d6) δ 9.17 (s, 1H), 8.90 (s, 1H), 8.24 (d, J = 5.7 Hz, 1H), 8.10 (d, J = 2.2 Hz, 1H), 7.64 (dd, J = 8.9, 2.2 Hz, 1H), 7.61 (d, J = 8.7 Hz, 1H), 7.42 (d, J = 2.0 Hz, 1H), 7.40 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.2 Hz, 1H), 7.32 (d, J = 2.1 Hz, 1H), 7.04 (d, J = 5.7 Hz, 1H), 4.89 (q, J = 8.7 Hz, 2H), 2.16 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 161.61, 157.18, 152.75, 148.00, 139.74, 138.60, 132.42, 131.64, 128.58, 127.33, 127.02, 125.65, 123.53, 122.80, 120.44, 119.55, 117.28, 117.22, 107.07, 64.79, 39.69, 10.72. ESI-MS (m/z): 550.1, 552.1, 553.1 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C23H18ClF6N3O2S, 550.0785 ([M + H]+), found: 550.0769 ([M + H]+). Purity (HPLC): 99.97%.
1-{4-{{[3-Methyl-4-(2,2,2-trifluoroethoxy)pyridin-2-yl]methyl}thio}phenyl}-3-[3-(trifluoromethyl)phenyl]urea (7j)
![Molecules 24 02108 i010]()
Compound 7j was prepared according to the general procedure C by using compound 4b (0.33 g, 1 mmol) and 3-(trifluoromethyl)aniline (0.16 g, 1 mmol), obtained a white solid (0.34 g) in 66.1% yield. m.p. 174.7–176.4 °C. IR (KBr, cm−1): υ 3421.4, 2985.6, 2924.1, 2852.9, 2349.2, 2311.0, 1614.9, 1491.8, 1445.1, 1399.1, 1339.8, 1313.1, 1288.0, 1264.6, 1231.2, 1173.2, 1114.6, 1071.2, 1006.3, 976.0, 832.4, 798.2, 700.6, 616.4. 1H-NMR (400 MHz, DMSO-d6) δ 9.07 (s, 1H), 8.87 (s, 1H), 8.24 (d, J = 5.7 Hz, 1H), 8.01 (d, J = 2.0 Hz, 1H), 7.60–7.54 (m, 1H), 7.51 (t, J = 7.9 Hz, 1H), 7.43 (d, J = 2.0 Hz, 1H), 7.41 (d, J = 2.1 Hz, 1H), 7.33 (d, J = 2.1 Hz, 1H), 7.33–7.31 (m, 1H), 7.30 (d, J = 1.7 Hz, 1H), 7.04 (d, J = 5.7 Hz, 1H), 4.89 (q, J = 8.7 Hz, 2H), 4.26 (s, 2H), 2.16 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 161.66, 157.16, 152.86, 147.96, 140.97, 138.80, 131.76, 130.35, 130.15, 129.84, 128.31, 122.30, 120.47, 119.40, 118.54, 114.59, 107.09, 65.09, 49.05, 31.13, 10.73. ESI-MS (m/z): 516.2 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C23H19F6N3O2S, 516.1175 ([M + H]+), found: 516.1174 ([M + H]+). Purity (HPLC): 97.29%.
1-{4-{[(4-Methoxy-3,5-dimethylpyridin-2-yl)methyl]thio}phenyl}-3-phenylurea (7k)
![Molecules 24 02108 i011]()
Compound 7k was prepared according to the general procedure by using compound 4c (0.27 g, 1 mmol) and aniline (0.10 g, 1 mmol), obtained a white solid (0.23 g) in 57.3% yield. m.p. 188.1–188.9 °C. IR (KBr, cm−1): υ 3422.4, 2923.8, 2852.4, 2351.0, 2321.9, 1644.0, 1597.4, 1553.7, 1494.2, 1441.5, 1398.0, 1312.9, 1270.7, 1237.5, 1173.3, 1127.7, 1073.5, 1002.5, 798.1, 755.7, 738.0, 694.0, 616.4. 1H-NMR (400 MHz, DMSO-d6) δ 8.72 (s, 1H), 8.67 (s, 1H), 8.12 (s, 1H), 7.45 (d, J = 1.3 Hz, 1H), 7.44-7.42 (m, 1H), 7.40 (d, J = 2.1 Hz, 1H), 7.39 (d, J = 2.1 Hz, 1H), 7.31 (d, J = 2.2 Hz, 1H), 7.29 (d, J = 2.1 Hz, 1H), 7.28 (s, 1H), 7.26 (d, J = 1.6 Hz, 1H), 6.97 (t, J = 7.4 Hz, 1H), 4.21 (s, 2H), 3.70 (s, 3H), 2.18 (s, 6H). 13C-NMR (101 MHz, DMSO-d6) δ 163.86, 155.80, 152.86, 148.97, 140.05, 139.16, 131.86, 129.24, 127.98, 125.19, 125.03, 122.35, 119.11, 118.70, 60.17, 31.15, 13.38, 11.34. ESI-MS (m/z): 394.6 ([M + H]+), 416.3 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C22H23N3O2S, 394.1584 ([M + H]+), found: 394.1586 ([M + H]+). Purity (HPLC): 99.89%.
1-(4-Chlorophenyl)-3-{4-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]thio}phenyl}urea (7l)
![Molecules 24 02108 i012]()
Compound 7l was prepared according to the general procedure by using compound 4c (0.27 g, 1 mmol) and 4-chloroaniline (0.13 g, 1 mmol), obtained a white solid (0.28 g) in 66.2% yield. m.p. 206.8–208.2 °C. IR (KBr, cm−1): υ 3422.5, 2923.0, 2852.0, 2377.1, 2349.6, 2310.8, 1630.4, 1547.7, 1491.7, 1439.7, 1399.2, 1385.1, 1309.9, 1270.9, 1235.5, 1173.0, 1124.1, 1051.3, 1004.6, 832.1, 798.2, 702.1, 668.3, 617.0. 1H-NMR (400 MHz, DMSO-d6) δ 8.81 (s, 1H), 8.75 (s, 1H), 8.11 (s, 1H), 7.48 (d, J = 2.1 Hz, 1H), 7.46 (d, J = 2.2 Hz, 1H), 7.40 (d, J = 2.0 Hz, 1H), 7.38 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.1 Hz, 1H), 7.32 (s, 1H), 7.31 (s, 1H), 7.30 (d, J = 2.1 Hz, 1H), 4.21 (s, 2H), 3.70 (s, 3H), 2.18 (s, 3H), 2.18 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 163.86, 155.79, 152.77, 148.97, 139.08, 138.94, 131.76, 129.07, 127.98, 128.25, 125.87, 125.19, 125.03, 120.23, 119.25, 60.17, 31.14, 13.37, 11.33. ESI-MS (m/z): 428.7 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C22H22ClN3O2S, 428.1194 ([M + H]+), found: 428.1199 ([M + H]+). Purity (HPLC): 99.53%.
1-{4-{[(4-Methoxy-3,5-dimethylpyridin-2-yl)methyl)]thio}phenyl}-3-(4-methoxyphenyl)urea (7m)
![Molecules 24 02108 i013]()
Compound 7m was prepared according to the general procedure by using compound 4c (0.27 g, 1 mmol) and 4-methoxyaniline (0.12 g, 1 mmol), obtained a white solid (0.21 g) in 49.2% yield. m.p. 171.4–172.6 °C. IR (KBr, cm−1): υ 3422.5, 2921.1, 2850.5, 1642.5, 1593.2, 1547.5, 1493.7, 1468.2, 1439.0, 1397.7, 1311.5, 1292.4, 1270.1, 1240.4, 1173.4, 1073.2, 1053.2, 1031.3, 1003.9, 828.2, 797.9, 616.3. 1H-NMR (400 MHz, DMSO-d6) δ 8.68 (s, 1H), 8.50 (s, 1H), 8.15 (s, 1H), 7.39 (d, J = 1.9 Hz, 1H), 7.37 (d, J = 2.1 Hz, 1H), 7.35 (d, J = 2.0 Hz, 1H), 7.33 (d, J = 2.2 Hz, 1H), 7.29 (d, J = 2.1 Hz, 1H), 7.27 (d, J = 1.9 Hz, 1H), 6.87 (d, J = 2.3 Hz, 1H), 6.86 (d, J = 2.2 Hz, 1H), 4.21 (s, 2H), 3.73 (s, 3H), 3.71 (s, 3H), 2.20 (s, 3H), 2.17 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 164.47, 155.40, 154.98, 153.08, 148.27, 139.64, 133.09, 132.28, 127.19, 125.70, 125.46, 125.41, 120.49, 118.95, 118.40, 114.46, 60.30, 55.65, 31.14, 13.45, 11.35. ESI-MS (m/z): 424.3 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C23H25N3O3S, 424.1689 ([M + H]+), found: 424.1698 ([M + H]+). Purity (HPLC): 96.88%.
1-[4-Chloro-3-(trifluoromethyl)phenyl]-3-{4-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]thio}phenyl}urea (7n)
![Molecules 24 02108 i014]()
Compound 7n was prepared according to the general procedure by using compound 4c (0.27 g, 1 mmol) and 3-chloro-4-(trifluoromethyl)aniline (0.20 g, 1 mmol), obtained a white solid (0.35 g) in 70.1% yield. m.p. 152.3–153.1 °C. IR (KBr, cm−1): υ 3422.2, 2921.6, 2852.2, 1719.2, 1593.8, 1546.4, 1480.4, 1419.8, 1384.6, 1311.1, 1265.2, 1229.0, 1174.6, 1130.8, 1073.5, 1031.7, 823.3, 619.9. 1H-NMR (400 MHz, DMSO-d6) δ 9.18 (s, 1H), 8.91 (s, 1H), 8.12 (s, 1H), 8.10 (d, J = 2.0 Hz, 1H), 7.64 (d, J = 8.9 Hz, 1H), 7.62 (s, 1H), 7.42 (d, J = 2.1 Hz, 1H), 7.40 (d, J = 2.1 Hz, 1H), 7.33 (s, 1H), 7.31 (d, J = 1.9 Hz, 1H), 4.22 (s, 2H), 3.70 (s, 3H), 2.19 (s, 3H), 2.18 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 164.07, 154.69, 154.49, 153.03, 149.00, 140.01, 132.84, 132.38, 126.45, 126.36, 124.20, 123.34, 122.49, 121.03, 115.40, 60.18, 31.13, 13.43, 11.00. ESI-MS (m/z): 496.1; 497.1; 498.1; 499.1 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C23H21ClF3N3O2S, 496.1068 ([M + H]+), found: 496.1066 ([M + H]+). Purity (HPLC): 98.56%.
1-{4-{[(4-Methoxy-3,5-dimethylpyridin-2-yl)methyl]thio}phenyl}-3-[3-(trifluoromethyl)pHenyl]urea (7o)
![Molecules 24 02108 i015]()
Compound 7o was prepared according to the general procedure by using compound 4c (0.27 g, 1 mmol) and 3-(trifluoromethyl)aniline (0.16 g, 1 mmol), obtained a white solid (0.30 g) in 64.7% yield. m.p. 156.9–158.1 °C. IR (KBr, cm−1): υ 3420.6, 2984.8, 2922.8, 2851.8, 2350.3, 2321.1, 1609.8, 1491.8, 1443.2, 1398.4, 1369.9, 1338.2, 1311.7, 1271.2, 1229.2, 1172.1, 1124.2, 1072.1, 1002.9, 797.9, 698.2, 616.0. 1H-NMR (400 MHz, DMSO-d6) δ 9.04 (s, 1H), 8.84 (s, 1H), 8.12 (s, 1H), 8.00 (d, J = 2.0 Hz, 1H), 7.57 (d, J = 8.8 Hz, 1H), 7.51 (t, J = 7.8 Hz, 1H), 7.42 (d, J = 1.9 Hz, 1H), 7.40 (d, J = 2.2 Hz, 1H), 7.33 (s, 1H), 7.32 (s, 1H), 7.30 (d, J = 2.6 Hz, 1H), 4.22 (s, 2H), 3.70 (s, 3H), 2.19 (s, 3H), 2.18 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 163.88, 155.76, 152.85, 148.95, 140.97, 138.73, 131.69, 130.35, 128.51, 125.21, 125.05, 123.31, 122.32, 119.43, 118.58, 114.64, 60.16, 31.13, 13.36, 11.33. ESI-MS (m/z): 462.3 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C23H22F3N3O2S, 462.1458 ([M + H]+), found: 462.1469 ([M + H]+). Purity (HPLC): 99.79%.
1-{4-{[(3,4-Dimethoxypyridin-2-yl)methyl]thio}phenyl}-3-phenylurea (7p)
![Molecules 24 02108 i016]()
Compound 7p was prepared according to the general procedure by using compound 4d (0.28 g, 1 mmol) and aniline (0.10 g, 1 mmol), obtained a white solid (0.18 g) in 45.8% yield. m.p. 127.7–128.5 °C. IR (KBr, cm−1): υ 3287.2, 2937.3, 1654.0, 1593.7, 1548.0, 1487.0, 1442.7, 1421.2, 1379.4, 1297.9, 1270.4, 1231.6, 1175.0, 1071.7, 997.3, 932.8, 829.0, 782.9, 742.9, 692.4, 618.5, 516.5. 1H-NMR (400 MHz, DMSO-d6) δ 8.72 (s, 1H), 8.67 (s, 1H), 8.12 (d, J = 5.5 Hz, 1H), 7.49–7.45 (m, 1H), 7.44 (s, 1H), 7.41 (d, J = 2.0 Hz, 1H), 7.40 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.0 Hz, 1H), 7.31 (d, J = 1.9 Hz, 1H), 7.29 (d, J = 7.7 Hz, 1H), 7.28–7.25 (m, 1H), 7.03 (d, J = 5.5 Hz, 1H), 6.97 (t, J = 7.3 Hz, 1H), 4.17 (s, 2H), 3.87 (s, 3H), 3.74 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 158.52, 152.88, 151.53, 145.72, 143.40, 138.98, 131.39, 129.24, 128.54, 122.33, 119.17, 118.69, 108.29, 60.99, 56.34, 36.37, 31.14. ESI-MS (m/z): 396.3 ([M + H]+), 418.2 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C21H21N3O3S, 396.1376 ([M + H]+), found: 396.1380 ([M + H]+). Purity (HPLC): 99.90%.
1-(4-Chlorophenyl)-3-{4-{[(3,4-dimethoxypyridin-2-yl)methyl]thio}phenyl}urea (7q)
![Molecules 24 02108 i017]()
Compound 7q was prepared according to the general procedure by using compound 4d (0.28 g, 1 mmol) and 4-chloroaniline (0.13 g, 1 mmol), obtained a white solid (0.21 g) in 49.1% yield. m.p. 141.7–142.9 °C. IR (KBr, cm−1): υ 3345.3, 3096.8, 2924.2, 2852.2, 1711.6, 1631.2, 1590.8, 1535.1, 1490.0, 1449.2, 1427.9, 1399.4, 1300.6, 1284.5, 1237.1, 1195.2, 1174.0, 1087.1, 1067.2, 996.9, 828.3, 703.0, 509.1. 1H-NMR (400 MHz, DMSO-d6) δ 8.83 (s, 1H), 8.76 (s, 1H), 8.11 (d, J = 5.5 Hz, 1H), 7.49 (s, 1H), 7.47 (s, 1H), 7.41 (s, 1H), 7.39 (s, 1H), 7.34-7.31 (m, 2H), 7.03 (d, J = 5.5 Hz, 1H), 4.17 (s, 2H), 3.87 (s, 3H), 3.74 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 158.52, 152.78, 151.50, 145.71, 143.40, 139.10, 138.75, 131.29, 129.07, 128.79, 125.85, 120.21, 119.30, 108.30, 60.99, 56.34, 36.29, 31.14. ESI-MS (m/z): 430.6 ([M + H]+), 452.1 ([M + Na]+). HRMS (ESI) (m/z): Calcd. for C21H20ClN3O3S, 430.0987 ([M + H]+), found: 430.0993 ([M + H]+). Purity (HPLC): 99.33%.
1-{4-{[(3,4-Dimethoxypyridin-2-yl)methyl]thio}phenyl}-3-(4-methoxyphenyl)urea (7r)
![Molecules 24 02108 i018]()
Compound 7r was prepared according to the general procedure by using compound 4d (0.28 g, 1 mmol) and 4-methoxyaniline (0.12 g, 1 mmol), obtained a white solid (0.20 g) in 47.9% yield. m.p. 179.0–180.6 °C. IR (KBr, cm−1): υ 3428.5, 2985.2, 2923.2, 2851.3, 1630.7, 1587.2, 1557.0, 1510.3, 1490.9, 1442.6, 1398.6, 1299.7, 1270.7, 1232.4, 1173.6, 1072.2, 1033.0, 1000.9, 934.0, 829.1, 799.5, 617.4, 549.0, 523.3. 1H-NMR (400 MHz, DMSO-d6) δ 8.62 (s, 1H), 8.46 (s, 1H), 8.11 (d, J = 5.5 Hz, 1H), 7.39 (d, J = 2.0 Hz, 1H), 7.38 (d, J = 2.2 Hz, 1H), 7.35 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.2 Hz, 1H), 7.31 (d, J = 2.2 Hz, 1H), 7.30 (d, J = 2.0 Hz, 1H), 7.03 (d, J = 5.5 Hz, 1H), 6.87 (d, J = 2.2 Hz, 1H), 6.85 (d, J = 2.2 Hz, 1H), 4.15 (s, 2H), 3.87 (s, 3H), 3.74 (s, 3H), 3.71 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 158.51, 154.98, 153.07, 151.55, 145.73, 143.40, 139.22, 133.08, 131.45, 128.24, 120.54, 119.06, 114.46, 108.30, 60.99, 56.35, 55.65, 36.43, 31.15. ESI-MS (m/z): 426.3 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C22H23N3O4S, 426.1482 ([M + H]+), found: 426.1489 ([M + H]+). Purity (HPLC): 98.84%.
1-[4-Chloro-3-(trifluoromethyl)phenyl]-3-{4-{[(3,4-dimethoxypyridin-2-yl)methyl]thio}pHenyl}urea (7s)
![Molecules 24 02108 i019]()
Compound 7s was prepared according to the general procedure by using compound 4d (0.28 g, 1 mmol) and 3-chloro-4-(trifluoromethyl)aniline (0.20 g, 1 mmol), obtained a white solid (0.32 g) in 64.5% yield. m.p. 188.1–189.2 °C. IR (KBr, cm−1): υ 3425.5, 2921.9, 2852.4, 1589.9, 1545.2, 1485.2, 1419.2, 1384.4, 1306.1, 1229.2, 1175.6, 1132.0, 1068.8, 1033.0, 824.9. 1H-NMR (400 MHz, DMSO-d6) δ 9.15 (s, 1H), 8.88 (s, 1H), 8.11 (d, J = 5.5 Hz, 1H), 8.10 (d, J = 2.2 Hz, 1H), 7.64 (d, J = 8.8 Hz, 1H), 7.62–7.58 (m, 1H), 7.42 (d, J = 2.0 Hz, 1H), 7.41 (d, J = 2.2 Hz, 1H), 7.34 (d, J = 2.2 Hz, 1H), 7.33 (d, J = 2.0 Hz, 1H), 7.03 (d, J = 5.5 Hz, 1H), 4.17 (s, 2H), 3.88 (s, 3H), 3.74 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 158.52, 152.76, 151.47, 145.73, 143.40, 139.76, 138.36, 132.44, 131.12, 129.28, 123.53, 122.78, 119.62, 117.24, 108.32, 61.00, 56.35, 36.17, 31.14. ESI-MS (m/z): 498.2 ([M + H]+). HRMS (ESI) (m/z): Calcd. for C22H19ClF3N3O3S, 498.0861 ([M + H]+), found: 498.0844 ([M + H]+). Purity (HPLC): 98.10%.
1-{4-{[(3,4-Dimethoxypyridin-2-yl)methyl]thio}phenyl}-3-[3-(trifluoromethyl)phenyl]urea (7t)
![Molecules 24 02108 i020]()
Compound 7t was prepared according to the general procedure by using compound 4d (0.28 g, 1 mmol) and 3-(trifluoromethyl)aniline (0.16 g, 1 mmol), obtained a white solid (0.21 g) in 44.3% yield. m.p. 198.4–199.8 °C. IR (KBr, cm−1): υ 3422.2, 2985.4, 2377.5, 2349.8, 2320.7, 2024.8, 1712.9, 1594.1, 1564.4, 1537.2, 1491.3, 1445.8, 1399.4, 1370.2, 1316.3, 1273.6, 1230.1, 1173.8, 1124.9, 1068.3, 1002.1, 932.4, 892.8, 828.2, 798.3, 743.3, 697.8, 615.6. 1H-NMR (400 MHz, DMSO-d6) δ 9.04 (s, 1H), 8.83 (s, 1H), 8.11 (d, J = 5.5 Hz, 1H), 8.01 (d, J = 2.0 Hz, 1H), 7.60–7.54 (m, 1H), 7.51 (t, J = 7.9 Hz, 1H), 7.43 (d, J = 2.0 Hz, 1H), 7.41 (d, J = 2.2 Hz, 1H), 7.34 (d, J = 2.2 Hz, 1H), 7.32 (d, J = 2.5 Hz, 1H), 7.30 (s, 1H), 7.03 (d, J = 5.6 Hz, 1H), 4.17 (s, 2H), 3.88 (s, 3H), 3.75 (s, 3H). 13C-NMR (101 MHz, DMSO-d6) δ 158.52, 152.87, 151.50, 145.72, 143.41, 140.98, 138.55, 131.22, 130.34, 129.08, 122.31, 119.49, 118.56, 114.65, 108.31, 60.99, 56.34, 36.24, 31.12. ESI-MS (m/z): 464.2 ([M + H]+). Purity (HPLC): 98.85%.