Structural Elucidation of Three Novel Kaempferol O-tri-Glycosides that Are Involved in the Defense Response of Hybrid Ornithogalum to Pectobacterium carotovorum
Abstract
:1. Introduction
2. Results and Discussion
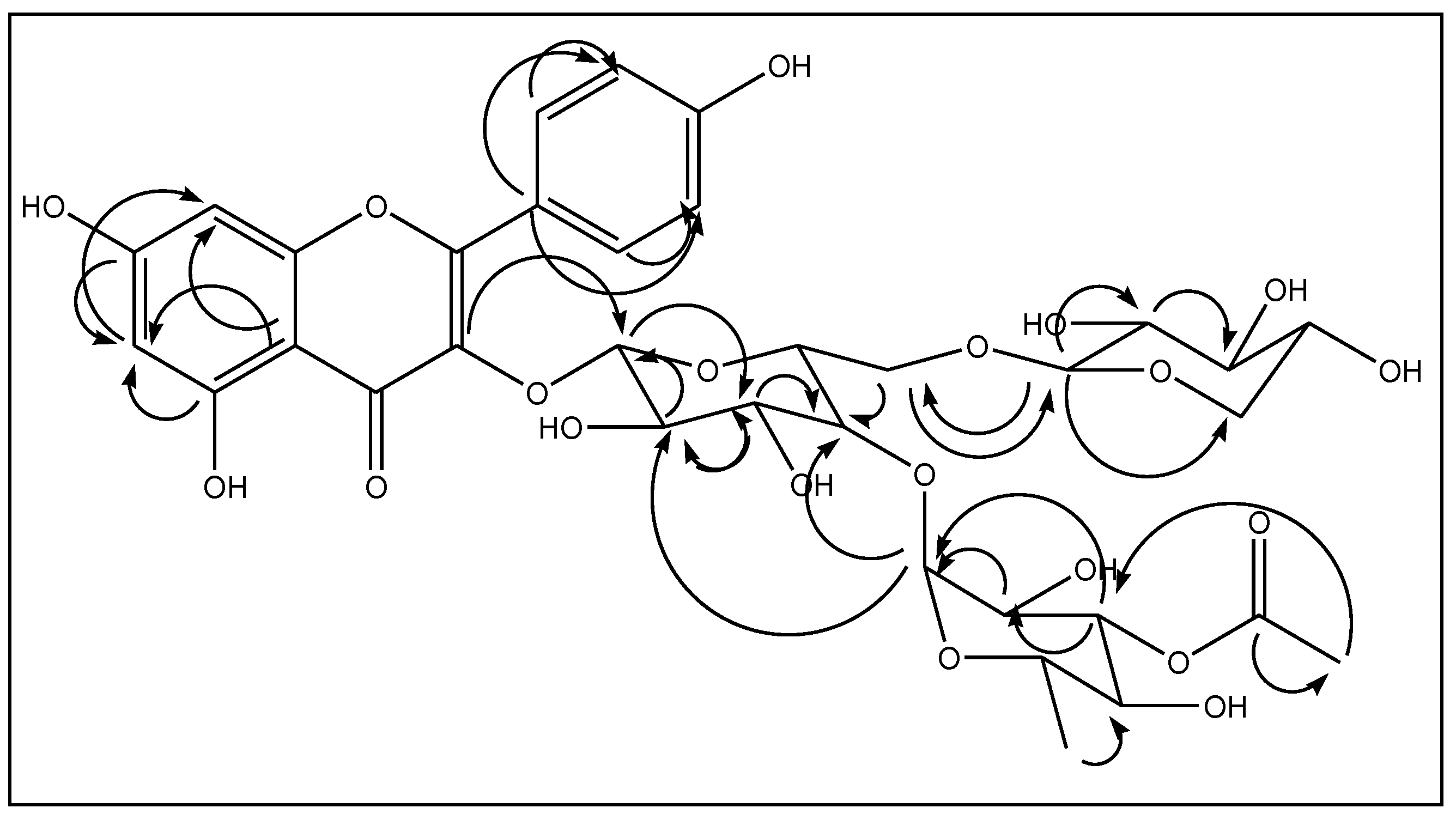
2.1. Mass Spectrometry of Compound 1
2.2. NMR Analyses of Compound 1
2.3. Spectral Analysis of Compound 2
2.4. Spectral Analysis of Compound 3
3. Experimental
3.1. General
3.2. Plant Material, Establishment of Cell Cultures, Plants and Bacterial Infection
3.3. Extraction and Separation
3.4. Reversed Phase High-Performance Liquid Chromatography (RP-LC) Assay of Kaempferol
3.5. Solid Phase Extraction (SPE)
3.6. Kaempferol O-tri-glycoside (1)
3.7. Kaempferol O-tri-glycoside (2)
3.8. Kaempferol O-tri-glycoside (3)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Manning, J.C.; Forest, F.; Devey, D.S.; Fay, M.F.; Goldblatt, P. A molecular phylogeny and a revised classification of Ornithogaloideae (Hyacinthaceae) based on an analysis of four plastid DNA regions. Taxon 2009, 58, 77–107. [Google Scholar] [CrossRef]
- De Villiers, S.M.; Kamo, K.; Thomson, J.A.; Bornman, C.H.; Berger, D.K. Biolistic transformation of chincherinchee (Ornithogalum) and regeneration of transgenic plants. Physiol. Plant. 2000, 109, 450–455. [Google Scholar] [CrossRef]
- Littlejohn, G.M. Star of bethlehem Ornithogalum. In Flower Breeding and Genetics; Anderson, N.O., Ed.; Springer: Amsterdam, The Netherlands, 2006; pp. 739–752. [Google Scholar]
- Martinez-Azori n, M.; Crespo, M.B.; Juan, A. Taxonomic revision of Ornithogalum subg. Ornithogalum (Hyacinthaceae) in the Iberian Peninsula and the Balearic Islands. Plant Syst. Evol. 2010, 289, 181–211. [Google Scholar] [CrossRef]
- Cohen, A.; Lipsky, A.N.G.; Manulis, S.; Arazi, T.; Ion, A.; Stav, R.; Sandler-Ziv, D.; Fintea, C.; Yedidia, I. Molecular Breeding of Ornithogalum for Erwinia Resistance. In Proceedings of the Xth IS on Flower Bulbs and Herbaceous Perennials, Acta Hort, The Netherlands, 2011; pp. 49–58. [Google Scholar]
- Golan, A.; Kerem, Z.; Tun, O.M.; Luzzatto, T.; Lipsky, A.; Yedidia, I. Combining flow cytometry and gfp reporter gene for quantitative evaluation of Pectpbacterium carotovorum ssp carotovorum in Ornithogalum dubium plantlets. J. Appl. Microbiol. 2010, 108, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, A.; Joshi, J.R.; Carmi, N.; Yedidia, I. Expression levels of antimicrobial peptide tachyplesin I in transgenic Ornithogalum lines affect the resistance to Pectobacterium infection. J. Biotechnol. 2016, 238, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Joshi, J.R.; Burdman, S.; Yedidia, I. Elucidating the mechanism by which plant derived small molecules affect virulence determinants of the genus Pectobacterium. FEBS J. 2016, 283, 145–146. [Google Scholar]
- Chen, H.; Jones, A.D.; Howe, G.A. Constitutive activation of the jasmonate signaling pathway enhances the production of secondary metabolites in tomato. FEBS Lett. 2006, 580, 2540–2546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rusterucci, C.; Zhao, Z.; Haines, K.; Mellersh, D.; Neumann, A.; Cameron, R. Age-related resistance to Pseudomonas syringae pv. tomato is associated with the transition to flowering in Arabidopsis and is effective against Peronospora parasitica. Physiol. Mol. Plant Pathol. 2005, 66, 222–231. [Google Scholar] [CrossRef]
- Wasternack, C.; Strnad, M. Jasmonates are signals in the biosynthesis of secondary metabolites—Pathways, transcription factors and applied aspects—A brief review. New Biotechnol. 2019, 48, 1–11. [Google Scholar] [CrossRef]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef]
- Luzzatto, T.; Golan, A.; Yishay, M.; Bilkis, I.; Ben-Ari, J.; Yedidia, I. Priming of antimicrobial phenolics during induced resistance response towards Pectobacterium carotovorum in the ornamental monocot calla lily. J. Agric. Food Chem. 2007, 55, 10315–10322. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M. Polyphenols as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouysegu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar] [CrossRef] [PubMed]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammerschmidt, R. Phytoalexins: What Have We Learned After 60 Years? Ann. Rev. Phytopathol. 1999, 37, 285–306. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Takahashi, S.; Waki, T. Formation of flavonoid metabolons: Functional significance of protein-protein interactions and impact on flavonoid chemodiversity. Front. Plant Sci. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B. Arsenal for survival: Secondary plant products. Taxon 2000, 49, 435–449. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Kerem, Z.; Chetrit, D.; Shoseyov, O.; Regev-Shoshani, G. Protection of lipids from oxidation by epicatechin, trans-resveratrol, and gallic and caffeic acids in intestinal model systems. J. Agric. Food Chem. 2006, 54, 10288–10293. [Google Scholar] [CrossRef]
- Treutter, D. Significance of flavonoids in plant resistance: A review. Environ. Chem. Lett. 2006, 4, 147–157. [Google Scholar] [CrossRef]
- Joshi, J.R.; Yedidia, I. Breeding for resistance to soft rot disease in Ornithogalum. Acta Hortic. 2017, 1171, 279–284. [Google Scholar] [CrossRef]
- Lipsky, A.; Cohen, A.; Ion, A.; Yedidia, I. Genetic transformation of Ornithogalum via particle bombardment and generation of Pectobacterium carotovorum-resistant plants. Plant Sci. 2014, 228, 150–158. [Google Scholar] [CrossRef]
- Daayf, F.; BelRhlid, R.; Belanger, R.R. Methyl ester of p-coumaric acid: A phytoalexin-like compound from long English cucumber leaves. J. Chem. Ecol. 1997, 23, 1517–1526. [Google Scholar] [CrossRef]
- Yedidia, I.; Shoresh, M.; Kerem, Z.; Benhamou, N.; Kapulnik, Y.; Chet, I. Concomitant induction of systemic resistance to Pseudomonas spingae pv. lachrymans in cucumber by Trichoderma asperellum (T-203) and accumulation of phytoalexins. Appl. Environ. Microbiol. 2003, 69, 7343–7353. [Google Scholar] [CrossRef]
Sample Availability: Not available. |



| 1 | 2 | 3 | ||||
|---|---|---|---|---|---|---|
| 13C | 1H | 13C | 1H | 13C | 1H | |
| Kaempferol | ||||||
| 2 | 158.54 | — | 158.57 | — | 158.61 | — |
| 3 | 134.53 | — | 134.51 | — | 134.56 | — |
| 4 | 179.38 | — | 179.32 | — | 179.37 | — |
| 4a | 106.04 | — | 106.00 | — | 106.13 | — |
| 5 | 163.23 | — | 163.22 | — | 163.25 | — |
| 6 | 99.84 | 6.18 (d, 2) | 99.89 | 6.17 (d, 2) | 99.80 | 6.19 (d, 2) |
| 7 | 165.90 | — | 165.83 | — | 165.69 | — |
| 8 | 94.80 | 6.39 (d, 2) | 94.87 | 6.39 (d, 2) | 94.79 | 6.40 (d, 2) |
| 8a | 158.49 | — | 158.47 | — | 158.54 | — |
| 1′ | 123.10 | — | 123.08 | — | 123.09 | — |
| 2′, 6′ | 132.24 | 8.08 (AA’XX’) | 132.24 | 8.07 (AA’XX’) | 132.23 | 8.07 (AA’XX’) |
| 3′, 5′ | 116.20 | 6.90 (AA’XX’) | 116.16 | 6.89 (AA’XX’) | 116.22 | 6.91 (AA’XX’) |
| 4′ | 161.41 | — | 161.42 | — | 161.43 | — |
| 1 | 2 | 3 | ||||
|---|---|---|---|---|---|---|
| 13C | 1H | 13C | 1H | 13C | 1H | |
| Glucose | ||||||
| 1 | 100.26 | 5.65 (d, 7.5) | 100.34 | 5.64 (d, 7.5) | 100.25 | 5.64 (d, 7.5) |
| 2 | 80.51 | 3.61 (dd, 9.5,7.5) | 79.72 | 3.60 (dd, 9.5,7.5) | 80.41 | 3.62 (dd, 9.5,7.5) |
| 3 | 78.60 | 3.57 (dd, 9.5, 9) | 78.57 | 3.57 (dd, 9.5, 9) | 78.44 | 3.57 (dd, 9.5, 9) |
| 4 | 71.83 | 3.31 a | 71.84 | 3.29 a | 71.85 | 3.30 a |
| 5 | 77.58 | 3.41 (ddd, 9.5, 6, 2) | 77.58 | 3.40 (ddd, 9.5, 6, 2) | 77.58 | 3.41 (ddd, 9.5, 6, 2) |
| 6 | 69.46 | 3.57 (dd, 12, 6) | 69.47 | 3.56 (dd, 12, 6) | 69.47 | 3.58 (dd, 12, 6) |
| 3.90 (dd, 12, 2) | 3.90 (dd, 12, 2) | 3.90 (dd, 12, 2) | ||||
| Xylose | ||||||
| 1 | 105.14 | 4.07 (d, 7.5) | 105.14 | 4.06 (d, 7.5) | 105.15 | 4.07 (d, 7.5) |
| 2 | 74.76 | 2.99 (dd, 9, 7.5) | 74.75 | 2.99 (dd, 9, 7.5) | 74.75 | 3.00 (dd, 9, 7.5) |
| 3 | 77.42 | 3.08 (t, 9) | 77.42 | 3.09 (t, 9) | 77.41 | 3.09 (t, 9) |
| 4 | 71.02 | 3.34 a | 71.02 | 3.33 a | 71.04 | 3.34 a |
| 5 | 66.54 | 2.88 (dd, 11.5, 10) | 66.54 | 2.87 (dd, 11.5, 10) | 66.53 | 2.88 (dd, 11.5, 10) |
| 3.64 (dd, 11.5, 5) | 3.64 (dd, 11.5, 5) | 3.65 (dd, 11.5, 5) | ||||
| Rhamnose | ||||||
| 1 | 102.70 | 5.20 (d, 2) | 100.25 | 5.21 (d, 2) | 100.07 | 5.20 (d, 2) |
| 2 | 69.99 | 4.18 (dd, 3.5, 2) | 74.22 | 5.23 (dd, 3.5, 2) | 71.44 b | 5.42 (dd, 3.5, 2) |
| 3 | 75.98 | 5.05 (dd, 10, 3.5) | 70.52 | 3.97 (dd, 10, 3.5) | 73.50 | 5.18 (dd, 10, 3.5) |
| 4 | 71.30 | 3.55 (t, 10) | 74.27 | 3.30 (t, 10) | 71.51 b | 3.46 (t, 10) |
| 5 | 70.10 | 4.20 (dd, 10, 6) | 70.10 | 4.10 (dd, 10, 6) | 70.02 | 4.24 (dd, 10, 6) |
| 6 | 17.58 | 1.00 (d, 6, 3H) | 17.61 | 1.00 (d, 6, 3H) | 17.55 | 1.02 (d, 6, 3H) |
| 2-Ac | — | — | 20.98 | 2.09 (s, 3H) | 20.94 | 2.01 (s, 3H) |
| — | — | 172.31 | — | 171.79 | — | |
| 3-Ac | 21.16 | 2.11 (s, 3H) | — | — | 20.75 | 2.09 (s, 3H) |
| 172.82 | — | — | — | 172.41 | — | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yedidia, I.; Schultz, K.; Golan, A.; Gottlieb, H.E.; Kerem, Z. Structural Elucidation of Three Novel Kaempferol O-tri-Glycosides that Are Involved in the Defense Response of Hybrid Ornithogalum to Pectobacterium carotovorum. Molecules 2019, 24, 2910. https://doi.org/10.3390/molecules24162910
Yedidia I, Schultz K, Golan A, Gottlieb HE, Kerem Z. Structural Elucidation of Three Novel Kaempferol O-tri-Glycosides that Are Involved in the Defense Response of Hybrid Ornithogalum to Pectobacterium carotovorum. Molecules. 2019; 24(16):2910. https://doi.org/10.3390/molecules24162910
Chicago/Turabian StyleYedidia, Iris, Keren Schultz, Avner Golan, Hugo E. Gottlieb, and Zohar Kerem. 2019. "Structural Elucidation of Three Novel Kaempferol O-tri-Glycosides that Are Involved in the Defense Response of Hybrid Ornithogalum to Pectobacterium carotovorum" Molecules 24, no. 16: 2910. https://doi.org/10.3390/molecules24162910





