Review of Ethnomedicinal, Phytochemical and Pharmacological Properties of Lannea schweinfurthii (Engl.) Engl.
Abstract
:1. Introduction
2. Research Methodology
3. Taxonomy, Distribution and Description of L. schweinfurthii
4. Medicinal Uses of Lannea schweinfurthii
5. Phytochemistry of Lannea schweinfurthii
6. Pharmacological Properties and Safety Evaluation of L. schweinfurthii
6.1. Acetylcholinesterase Inhibitory Activities
6.2. Anti-Apoptotic Activities
6.3. Antibacterial Activities
6.4. Antiviral Activities
6.5. Anti-giardial Activities
6.6. Anti-inflammatory Activities
6.7. Antioxidant Activities
6.8. Antiplasmodial Activities
6.9. Antitrypanosomal Activities
6.10. Hepatoprotective Activities
6.11. Larvicidal Activities
6.12. Cytotoxicity and Toxicity Activities
7. Conclusions
Funding
Conflicts of Interest
References
- Koekemoer, M.; Steyn, H.M.; Bester, S.P. Guide to Plant Families of Southern Africa; South African National Biodiversity Institute: Pretoria, South Africa, 2014. [Google Scholar]
- Okoth, D.A. Phytochemistry and Bioactive Natural Products from Lannea alata, Lannea rivae, Lannea schimperi and Lannea schweinfurthii (Anacardiaceae). Ph.D. Thesis, University of Kwazulu-Natal, Pietermaritsburg, South Africa, 201. [Google Scholar]
- Palmer, E.; Pitman, P. Trees of Southern Africa Covering all Known Indigenous Species in the Republic of South Africa, South West Africa, Botswana, Lesotho and Swaziland; A.A. Balkema: Cape Town, South Africa, 1972. [Google Scholar]
- Coates Palgrave, M. Keith Coates Palgrave Trees of Southern Africa; Struik Publishers: Cape Town, South Africa, 2002. [Google Scholar]
- Wannan, B.S. Analysis of generic relationships in Anacardiaceae. Blumea 2006, 51, 165–195. [Google Scholar] [CrossRef]
- Pell, S.K.; Mitchell, J.D.; Miller, A.J.; Lobova, T.A. Anacardiaceae. In The Families and Genera of Vascular Plants: Flowering Plants Eucodicots: Sapindales, Cucurbitales and Myrtaceae; Kubitzki, K., Ed.; Springer: London, UK, 2011; pp. 7–50. [Google Scholar]
- Marquet, A.; Jansen, P.C.M. Lannea microcarpa Engl. & K. Krause. In Plant Resources of Tropical Africa 3: Dyes and Tannins; Jansen, P.C.M., Cardon, D., Eds.; PROTA Foundation: Wageningen, The Netherlands, 2005; Volume 3, pp. 102–104. [Google Scholar]
- Van Wyk, B.; Gericke, N. People’s Plants: A Guide to Useful Plants of Southern Africa; Briza Publications: Pretoria, South Africa, 2007. [Google Scholar]
- Van Wyk, B.-E.; Van Oudtshoorn, B.; Gericke, N. Medicinal Plants of South Africa; Briza Publishers: Pretoria, South Africa, 2013. [Google Scholar]
- Maroyi, A. Lannea acida: A review of its medicinal uses, phytochemistry and pharmacological properties. Asian J. Pharmaceut. Clin. Res. 2018, 11, 69–74. [Google Scholar] [CrossRef]
- Maroyi, A. Lannea discolor: Its botany, ethnomedicinal uses, phytochemistry and pharmacological properties. Asian J. Pharmaceut. Clin. Res. 2018, 11, 49–54. [Google Scholar] [CrossRef]
- Maroyi, A. Lannea microcarpa: A review of its medicinal uses, phytochemistry and pharmacological properties. J. Pharm. Nutr. Sci. 2018, 8, 168–177. [Google Scholar]
- Neuwinger, H.D. African Traditional Medicine: A Dictionary of Plant Use and Applications; Medpharm Scientific: Stuttgart, Germany, 2000. [Google Scholar]
- Mølgaard, P.; Nielsen, S.B.; Rasmussen, D.E.; Drummond, R.B.; Makaza, N.; Andreassen, J. Anthelmintic screening of Zimbabwean plants traditionally used against schistosomiasis. J. Ethnopharmacol. 2001, 74, 257–264. [Google Scholar] [CrossRef]
- Queiroz, E.F.; Kuhl, C.; Terreaux, C.; Mavi, S.; Hostettmann, K. New dihydroalkylhexenones from Lannea edulis. J. Nat. Prod. 2003, 66, 578–580. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, C.; Maharaj, V.J.; Crouch, N.R.; Grace, O.M.; Pillay, P.; Matsabisa, M.G.; Bhagwandin, N.; Smith, P.J.; Folb, P.I. In vitro antiplasmodial activity of medicinal plants native to or naturalised in South Africa. J. Ethnopharmacol. 2004, 92, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Koné, W.M.; Atindehou, K.K.; Terreaux, C.; Hostettmann, K.; Traoré, D.; Dosso, M. Traditional medicine in north Côte d’Ivoire: Screening of 50 medicinal plants for antibacterial activity. J. Ethnopharmacol. 2004, 93, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Koné, W.M.; Atindehou, K.K.; Dossahoua, T.; Betschart, B. Anthelmintic activity of medicinal plants used in northern Côte d’Ivoire against intestinal helminthiasis. Pharmaceut. Biol. 2005, 43, 72–78. [Google Scholar] [CrossRef]
- Picerno, P.; Mencherini, T.; Della, L.R.; Meloni, M.; Sanogo, R.; Aquino, R.P. An extract of Lannea microcarpa: Composition, activity and evaluation of cutaneous irritation in cell cultures and reconstituted human epidermis. J. Pharm. Pharmacol. 2006, 58, 981–988. [Google Scholar] [CrossRef]
- Lamien-Meda, A.; Lamien, C.E.; Compaore, M.M.Y.; Meda, R.N.T.; Kiendrebeogo, M.; Zeba, B.; Millogo, J.F.; Nacoulma, O.G. Polyphenol content and antioxidant activity of fourteen wild edible fruits from Burkina Faso. Molecules 2008, 13, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Ouattara, L.; Koudou, J.; Karou, D.S.; Giaco, L.; Capelli, G.; Simpore, J.; Fraziano, M.; Colizzi, V.; Traore, A.S. In vitro anti Mycobacterium tuberculosis H37Rv activity of Lannea acida A. Rich from Burkina Faso. Pak. J. Biol. Sci. 2011, 14, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Ouattara, L.; Koudou, J.; Zongo, C.; Barro, N.; Savadogo, A.; Bassole, I.H.N.; Ouattara, A.S.; Traore, A.S. Antioxidant and antibacterial activities of three species of Lannea from Burkina Faso. J. Appl. Sci. 2011, 11, 157–162. [Google Scholar] [CrossRef]
- Mabona, U.; Viljoen, A.; Shikanga, E.; Marston, A.; Van Vuuren, S. Antimicrobial activity of southern African medicinal plants with dermatological relevance: From an ethno pharmacological screening approach, to combination studies and the isolation of a bioactive compound. J. Ethnopharmacol. 2013, 148, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Chakuma, N.; Chipurura, B.; Muchuweti, M.; Chitindingu, K.; Bhebhe, M.; Chagonda, L. Total phenolic content, free radical scavenging and antioxidant potential of Lannea discolor (Sond.) Engl bark and root extracts. J. Biol. Active Prod. Nat. 2015, 5, 71–77. [Google Scholar]
- Kabongo-Kayoka, P.N.; Eloff, J.N.; Obi, C.L.; McGaw, L.J. Antimycobacterial activity and low cytotoxicity of leaf extracts of some African Anacardiaceae tree species. Phytother. Res. 2016, 30, 2001–2011. [Google Scholar] [CrossRef] [PubMed]
- Owusu, G.; Ofori-Amoah, J. Anti-inflammatory and analgesic effects of an aqueous extract of Lannea acida stem bark. Br. J. Pharmaceut. Res. 2017, 16, 1–8. [Google Scholar] [CrossRef]
- Chhabra, S.C.; Mahunnah, R.L.A.; Mshiu, E.N. Plants used in traditional medicine in eastern Tanzania. I. Pteridophytes and Angiosperms (Acanthaceae to Canellaceae). J. Ethnopharmacol. 1987, 21, 253–277. [Google Scholar] [CrossRef]
- Verzar, R.; Petrii, G. Medicinal plants in Mozambique and their popular use. J. Ethnopharmacol. 1987, 19, 67–80. [Google Scholar] [CrossRef]
- Fowler, D.G. Traditional ila plant remedies from Zambia. Kirkia 2002, 18, 35–48. [Google Scholar]
- Tabuti, J.R.S.; Lye, K.A.; Dhillion, S.S. Traditional herbal drugs of Bulamogi, Uganda: Plants, use and administration. J. Ethnopharmacol. 2003, 88, 19–44. [Google Scholar] [CrossRef]
- Maregesi, S.M.; Ngassapa, O.D.; Pieters, L.; Vlietinck, A.J. Ethnopharmacological survey of the Bunda district, Tanzania: Plants used to treat infectious diseases. J. Ethnopharmacol. 2007, 113, 457–470. [Google Scholar] [CrossRef]
- Ribeiro, A.; Romeiras, M.M.; Tavares, J.; Faria, M.T. Ethnobotanical survey in Canhane village, district of Massingir, Mozambique: Medicinal plants and traditional knowledge. J. Ethnobiol. Ethnomed. 2010, 6, 33. [Google Scholar] [CrossRef]
- Gathirwa, J.W.; Rukunga, G.M.; Mwitari, P.G.; Mwikwabe, N.M.; Kimani, C.W.; Muthaura, C.N.; Kiboi, D.M.; Nyangacha, R.M.; Omar, S.A. Traditional herbal antimalarial therapy in Kilifi district, Kenya. J. Ethnopharmacol. 2011, 134, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Wanzala, W.; Syombua, M.; Alwala, J.O. A survey of the applications and use of ethnomedicinal plants and plant products for healthcare from the Ukambani region in Eastern Kenya. Ind. J. Ethnophytopharmaceut. 2016, 2, 6–58. [Google Scholar]
- Kioko, J.; Baker, J.; Shannon, A.; Kiffner, C. Ethnoecological knowledge of ticks and treatment of tick-borne diseases among Maasai people in Northern Tanzania. Vet. World 2015, 8, 755–762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mwale, M.; Bhebhe, E.; Chimonyo, M.; Halimani, T.E. Use of Herbal Plants in Poultry Health Management in the Mushagashe Small-Scale Commercial Farming Area in Zimbabwe. Int. J. Appl. Res. Vet. Med. 2005, 3, 163–170. [Google Scholar]
- Botha, J.; Witkowski, E.T.F.; Shackleton, C.M. An inventory of medicinal plants traded on the western boundary of the Kruger National Park, South Africa. Koedoe 2011, 44, 7–46. [Google Scholar] [CrossRef]
- Hilonga, S.; Otieno, J.N.; Ghorbani, A.; Pereus, D.; Kocyan, A.; de Boer, H. Trade of wild-harvested medicinal plant species in local markets of Tanzania and its implications for conservation. S. Afr. J. Bot. 2018. [Google Scholar] [CrossRef]
- Posthouwer, C.; Veldman, S.; Abihudi, S.; Otieno, J.N.; van Andel, T.R.; de Boer, H.J. Quantitative market survey of non-woody plants sold at Kariakoo Market in Dar es Salaam, Tanzania. J. Ethnopharmacol. 2018, 222, 280–287. [Google Scholar] [CrossRef]
- Hostettmann, K.; Marston, A.; Ndojoko, K.; Wolfender, J. The potential of Africa plants as a source of drug. Curr. Org. Chem. 2000, 4, 973–1010. [Google Scholar] [CrossRef]
- Van Wyk, B.-E. A review of African medicinal and aromatic plants. In Medicinal and Aromatic Plants of the World: Africa; Neffati, M., Najjaa, H., Máthé, Á., Eds.; Springer: Dordrecht, The Netherlands, 2017; Volume 3, pp. 19–60. [Google Scholar]
- Fernandes, R.; Fernandes, A. Anacardiaceae. In Flora Zambesiaca 2; Exell, A.W., Fernandes, A., Wild, H., Eds.; Crown Agents for Overseas Governments and Administrations: London, UK, 1966; Part 2; pp. 550–615. [Google Scholar]
- Kokwaro, J.O. Anacardiaceae. In Flora of Tropical East Africa; Polhill, R.M., Ed.; AA Balkema: Rotterdam, The Netherlands, 1986; pp. 1–59. [Google Scholar]
- Friis, I.B.; Vollensen, K. Flora of the Sudan-Uganda Border Area East of the Nile: 1. Catalogue of Vascular Plants; The Royal Danish Academy of Sciences and Letters: Copenhagen, Denmark, 1998. [Google Scholar]
- Schmidt, E.; Lotter, M.; McCleland, W. Trees and Shrubs of Mpumalanga and Kruger National Park; Jacana Publishers: Johannesburg, South Africa, 2002. [Google Scholar]
- Germishuizen, G.; Meyer, N.L. Plants of Southern Africa: An Annotated Checklist; Strelitzia 14, National Botanical Institute: Pretoria, South Africa, 2003. [Google Scholar]
- Hyde, M.A.; Wursten, B.T.; Ballings, P.; Coates Palgrave, M. Flora of Zimbabwe: Species Information: Lannea schweinfurthii (Engl.) Engl. var. stuhlmannii (Engl.) Kokwaro. Available online: https://www.zimbabweflora.co.zw/speciesdata/species.php?species_id=136560 (accessed on 13 December 2018).
- Hyde, M.A.; Wursten, B.T.; Ballings, P.; Coates Palgrave, M. Flora of Zimbabwe: Species Information: Lannea schweinfurthii (Engl.) Engl. var. tomentosa (Dunkley) Kokwaro. Available online: https://www.zimbabweflora.co.zw/speciesdata/species.php?species_id=136570 (accessed on 13 December 2018).
- Van Wyk, B.; Van Wyk, P. Field Guide to Trees of Southern Africa; Struik Publishers (Pty) Ltd.: Cape Town, South Africa, 1997. [Google Scholar]
- Ruffo, C.K.; Birnie, A.; Tengäs, B. Edible Wild Plants of Tanzania; Regional Land Management Unit (RELMA): Nairobi, Kenya, 2002. [Google Scholar]
- Lovett, J.C.; Ruffo, C.K.; Gereau, R.E.; Taplin, J.R.D. Field Guide to the Moist Forest Trees of Tanzania; Society for Environmental Exploration: London, UK, 2006. [Google Scholar]
- Venter, F.; Venter, J.-A. Making the Most of Indigenous Trees; Briza Publications: Pretoria, South Africa, 1996. [Google Scholar]
- Kindt, R.; van Breugel, P.; Lillesø, J.; Bingham, M.; Demissew, S.; Dudley, C.; Friis, I.; Gachathi, F.; Kalema, J.; Mbago, F. Potential Natural Vegetation of Eastern Africa: Forest and Landscape; University of Copenhagen: Copenhagen, Denmark, 2011; pp. 1–62. [Google Scholar]
- Darbyshire, I.; Kordofani, M.; Farag, I.; Candiga, R.; Pickering, H. The Plants of Sudan and South Sudan; Kew publishing, Royal Botanic Gardens, Kew: London, UK, 2015. [Google Scholar]
- World Health Organization (WHO). The Health of the People: What Works: The African Regional Health Report 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Seebaluck-Sandoram, R.; Mahomoodally, F.M. Management of infectious diseases in Africa. In Medicinal Spices and Vegetables from Africa: Therapeutic Potential Against Metabolic, Inflammatory, Infectious and Systemic Diseases; Kuete, V., Ed.; Academic Press: London, UK, 2017; pp. 133–151. [Google Scholar]
- Pauw, E.; Eloff, J.N. Which tree orders in southern Africa have the highest antimicrobial activity and selectivity against bacterial and fungal pathogens of animals? BMC Complement. Altern. Med. 2014, 14, 317. [Google Scholar] [CrossRef] [PubMed]
- Moshi, M.J.; Cosam, J.C.; Mbwambo, Z.H.; Kapingu, M.; Nkunya, M.H.H. Testing beyond ethnomedical claims: Brine shrimp lethality of some Tanzanian plants. Pharmaceut. Biol. 2004, 42, 547–551. [Google Scholar] [CrossRef]
- Chinsembu, K.C.; Hedimbi, M. An ethnobotanical survey of plants used to manage HIV/AIDS opportunistic infections in Katima Mulilo, Caprivi region, Namibia. J. Ethnobiol. Ethnomed. 2010, 6, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagata, J.M.; Jew, A.R.; Kimeu, J.M.; Salmen, C.R.; Bukusi, E.A.; Cohen, C.R. Medical pluralism on Mfangano Island: Use of medicinal plants among persons living with HIV/AIDS in Suba district, Kenya. J. Ethnopharmacol. 2011, 135, 501–509. [Google Scholar] [CrossRef]
- Chinsembu, K.C. Ethnobotanical study of medicinal flora utilised by traditional healers in the management of sexually transmitted infections in Sesheke district, western province, Zambia. Revista Brasileira de Farmacognosia 2016, 26, 268–274. [Google Scholar] [CrossRef]
- Chinsembu, K.C. Ethnobotanical study of plants used in the management of HIV/AIDS-related diseases in Livingstone, southern province, Zambia. Evid. Based Complement. Altern. Med. 2016, 2016. [Google Scholar] [CrossRef]
- Chinsembu, K.C.; Syakalima, M.; Semenya, S.S. Ethnomedicinal plants used by traditional healers in the management of HIV/AIDS opportunistic diseases in Lusaka, Zambia. S. Afr. J. Bot. 2018. [Google Scholar] [CrossRef]
- Maroyi, A. Traditional and medicinal uses of essential oil producing tree Sclerocarya birrea in south-central Zimbabwe. J. Essent. Oil Bearing Plants 2014, 17, 776–786. [Google Scholar] [CrossRef]
- Maroyi, A. Treatment of diarrhoea using traditional medicines: Contemporary research in South Africa and Zimbabwe. Afr. J. Trad. Complement. Altern. Med. 2016, 13, 5–10. [Google Scholar] [CrossRef]
- Eloff, J.N.; McGaw, LJ. Plant extracts used to manage bacterial, fungal and parasitic infections in southern Africa. In Modern Phytomedicine: Turning Medicinal Plants into Drugs; Ahmad, I., Aqil, F., Owais, M., Eds.; Wiley-VCH: Weinheim, Germany, 2014; pp. 97–121. [Google Scholar]
- Eloff, J.N.; McGaw, L.J. Using African plant biodiversity to combat microbial infections. In Novel Plant Bioresources: Applications in Food Medicine and Cosmetics; Gurib-Fakim, A., Ed.; John Wiley: Chichester, UK, 2014; pp. 163–173. [Google Scholar]
- Palombo, E.A. Traditional plants and herbal remedies used in the treatment of diarrheal disease: Mode of action, quality, efficacy and safety considerations. In Modern Phytomedicine: Turning Medicinal Plants into Drugs; Ahmad, I., Aqil, F., Owais, M., Eds.; Wiley-VCH: Weinheim, Germany, 2014; pp. 247–269. [Google Scholar]
- Zahin, M.; Aqil, F.; Khan, M.S.A.; Ahmad, I. Ethnomedicinal plants derived antibacterials and their prospects. In Ethnomedicine: A Source of Complementary Therapeutics; Chattopadhyay, D., Ed.; Research Signpost: Kerala, India, 2010; pp. 149–178. [Google Scholar]
- Pakia, M.; Cooke, J.A. The ethnobotany of the Midzichenda tribes of the coastal forest areas in Kenya: 2. Medicinal plant uses. S. Afr. J. Bot. 2003, 69, 382–395. [Google Scholar] [CrossRef]
- Kaingu, C.K.; Oduma, J.A.; Kanui, T.I. Practices of traditional birth attendants in Machakos district, Kenya. J. Ethnopharmacol. 2011, 137, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Danley, K. Letters of the Bush: A Case Study of Traditional Setswana Herbal Medicine. 2006. Independent Study Project (ISP) Collection, Paper 270. Available online: https://pdfs.semanticscholar.org/f8a7/daace1c771f718bd97d2b666195e71c7535a.pdf (accessed on 12 December 2018).
- Gelfand, M. Medicine and Magic of the Mashona; Juta: Cape Town, South Africa, 1956. [Google Scholar]
- Gelfand, M.; Mavi, S.; Drummond, R.B.; Ndemera, B. The Traditional Medical Practitioners in Zimbabwe: His Principles of Practice and Pharmacopoeia; Mambo Press: Gweru, Zimbabwe, 1985. [Google Scholar]
- Johns, T.; Kokwaro, J.O.; Kimanani, E.K. Herbal remedies of the Luo of Siaya district, Kenya: Establishing quantitative criteria for consensus. Econ. Bot. 1990, 44, 369–381. [Google Scholar] [CrossRef]
- Hutchings, A.; Scott, A.H.; Lewis, G.; Cunningham, A. Zulu Medicinal Plants: An Inventory; University of Natal Press: Scottsville, South Africa, 1996. [Google Scholar]
- Geissler, P.W.; Harris, S.A.; Prince, R.J.; Olsen, A.; Odhiambo, R.A.; Oketch-Rabah, H.; Madiega, P.A.; Andersen, A.; Mølgaard, P. Medicinal plants used by Luo mothers and children in Bondo district, Kenya. J. Ethnopharmacol. 2002, 83, 39–54. [Google Scholar] [CrossRef]
- Arwa, S.P.; Nyunja, R.O.; Onyango, J.C. Plant species in the folk medicine of Kit Mikayi region, western Kenya. Ethnobot. Leaflets 2010, 14, 836–840. [Google Scholar]
- Muniafu, M.M.; Kipkore, K.W.; Maima, A.O.; Kwena, M.O.; Kahindi, J.H.P. Survey of medicinal plants used by the Giriama of Basi, Kenya. Pharmaceut. J. Kenya 2014, 21, 7–15. [Google Scholar]
- Bruschi, P.; Morganti, M.; Mancini, M.; Signorini, M.A. Traditional healers and laypeople: A qualitative and quantitative approach to local knowledge on medicinal plants in Muda (Mozambique). J. Ethnopharmacol. 2011, 138, 543–563. [Google Scholar] [CrossRef]
- Luoga, E.J.; Witkowski, E.T.F.; Balkwill, K. Differential utilization and ethnobotany of trees in Kitulanghalo forest reserve and surrounding communal lands, eastern Tanzania. Econ. Bot. 2000, 54, 328–343. [Google Scholar] [CrossRef]
- Long, C. Swaziland’s Flora: siSwati Names and Uses; Swaziland National Trust Commission: Mbambane, Swaziland, 2005; Available online: http://www.sntc.org.sz/index.asp (accessed on 19 October 2018).
- Tshikalange, T.E.; Mophuting, B.C.; Mahore, J.; Winterboer, S.; Lall, N. An ethnobotanical study of medicinal plants used in villages under Jongilanga tribal council, Mpumalanga, South Africa. Afr. J. Trad. Complement. Altern. Med. 2016, 13, 83–89. [Google Scholar] [CrossRef]
- Mabogo, E.E.N. The Ethnobotany of the Vhavenda. Master’s Thesis, University of Pretoria, Pretoria, South Africa, 1990. [Google Scholar]
- Arnold, H.-J.; Gulumian, M. Pharmacopoeia of traditional medicine in Venda. J. Ethnopharmacol. 1984, 12, 35–74. [Google Scholar] [CrossRef]
- Watt, J.M.; Breyer-Brandwijk, M.G. The Medicinal and Poisonous Plants of Southern and Eastern Africa; E. and S. Livingstone: London, UK, 1962. [Google Scholar]
- Chhabra, S.C.; Uiso, F.C.; Mshiu, E.N. Phytoehemical screening of Tanzanian medicinal plants. I. J. Ethnopharmacol. 1984, 11, 157–179. [Google Scholar] [CrossRef]
- Oyugi, J.O. Analysis of Proximate, Micronutrients and Determination of Phytochemicals in Selected Medicinal Plants in Mbita-Homabay County. Master’s Thesis, Kenyatta University, Nairobi, Kenya, 2016. [Google Scholar]
- Chaves, S.K.M.; Feitosa, C.M.; Araújo, L.S. Alkaloids pharmacological activities: Prospects for the development of phytopharmaceuticals for neurodegenerative diseases. Curr. Pharm. Biotechnol. 2016, 17, 629–635. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Ericson-Neilsen, W.; Kaye, A.D. Steroids: Pharmacology, complications and practice delivery issues. Ochsner. J. 2014, 14, 203–207. [Google Scholar] [PubMed]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Proanthocyanidins and hydrolysable tannins: Occurrence, dietary intake and pharmacological effects. Brit. J. Pharmacol. 2017, 174, 1244–1262. [Google Scholar] [CrossRef]
- Adewusi, E.A.; Steenkamp, V. In vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from southern Africa. Asian Pac. J. Trop. Med. 2011, 4, 829–835. [Google Scholar] [CrossRef] [Green Version]
- Seoposengwe, K.; van Tonder, J.J.; Steenkamp, V. In vitro neuroprotective potential of four medicinal plants against rotenone-induced toxicity in SH-SY5Y neuroblastoma cells. BMC Complement. Altern. Med. 2013, 13, 353. [Google Scholar] [CrossRef]
- Maregesi, S.M.; Pieters, L.; Ngassapa, O.D.; Apers, S.; Vingerhoets, R.; Cos, P.; Vanden Berghe, D.A.; Vlietinck, A.J. Screening of some Tanzanian medicinal plants from Bunda district for antibacterial, antifungal and antiviral activities. J. Ethnopharmacol. 2008, 119, 58–66. [Google Scholar] [CrossRef]
- Lall, N.; de Canha, M.N.; Reid, A.; Oosthuizen, C.B.; Langhansova, L.; Mahore, J.; Winterboer, S.; Hamilton, C.; Kumar, V.; Gasa, N.; Twilley, D. Antibacterial and anticancer activity of ethnomedicinal plants used in the Jongilanga community, Mpumalanga. Int. J. Pharmacog. Phytochem. Res. 2018, 10, 25–37. [Google Scholar]
- Maregesi, S.; Van Miert, S.; Pannecouque, C.; Haddad, M.H.F.; Hermans, N.; Wright, C.W.; Vlietinck, A.J.; Apers, S.; Pieters, L. Screening of Tanzanian medicinal plants against Plasmodium falciparum and human immunodeficiency virus. Planta Med. 2010, 76, 195–201. [Google Scholar] [CrossRef]
- Johns, T.; Faubert, G.M.; Kokwaro, J.O.; Mahunnah, R.L.A.; Kimanani, E.K. Anti-giardial activity of gastrointestinal remedies of the Luo of East-Africa. J. Ethnopharmacol. 1995, 46, 17–23. [Google Scholar] [CrossRef]
- Lawal, F.; Bapela, M.J.; Adebayo, S.S.; Malterud, K.E.; Tshikalange, T.E. Anti-inflammatory potential of South African medicinal plants used in the treatment of sexually transmitted infections. S. Afr. J. Bot. 2018. [Google Scholar] [CrossRef]
- Yaouba, S.; Koch, A.; Guantai, E.M.; Derese, S.; Irungu, B.; Heydenreich, M.; Yenesew, A. Alkenyl cyclohexanone derivatives from Lannea rivae and Lannea schweinfurthii. Phytochem. Lett. 2018, 23, 141–148. [Google Scholar] [CrossRef]
- Sobeh, M.; Mahmoud, M.F.; Hasan, R.A.; Abdelfattah, M.A.O.; Sabry, O.M.; Ghareeb, M.A.; El-Shazly, A.M.; Wink, M. Tannin-rich extracts from Lannea stuhlmannii and Lannea humilis (Anacardiaceae) exhibit hepatoprotective activities in vivo via enhancement of the anti-apoptotic protein Bcl-2. Sci. Rep. 2018, 8, 9343. [Google Scholar] [CrossRef] [PubMed]
- Gathirwa, J.W.; Rukunga, G.M.; Njagi, E.N.M.; Omar, S.A.; Mwitari, P.G.; Guantai, A.N.; Tolo, F.M.; Kimani, C.W.; Muthaura, C.N.; Kirira, P.G.; et al. The in vitro anti-plasmodial and in vivo antimalarial efficacy of combinations of some medicinal plants used traditionally for treatment of malaria by the Meru community in Kenya. J. Ethnopharmacol. 2008, 115, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Muthaura, C.N.; Keriko, J.M.; Mutai, C.; Yenesew, A.; Gathirwa, J.W.; Irungu, B.N.; Nyangacha, R.; Mungai, G.M.; Derese, S. Antiplasmodial potential of traditional phytotherapy of some remedies used in treatment of malaria in Meru—Tharaka Nithi county of Kenya. J. Ethnopharmacol. 2015, 175, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Nibret, E.; Ashour, M.L.; Rubanza, C.D.; Wink, M. Screening of some Tanzanian medicinal plants for their trypanocidal and cytotoxic activities. Phytother. Res. 2010, 24, 945–947. [Google Scholar] [CrossRef] [PubMed]
- Adewusi, E.A.; Fouche, G.; Steenkamp, V. Effect of four medicinal plants on amyloid-β induced neurotoxicity in SH-SY5Y cells. Afr. J. Trad. Comp. Altern. Med. 2013, 10, 6–11. [Google Scholar]
- Gunter, T.E.; Gerstner, B.; Gunter, K.K. Manganese transport via the transferring mechanism. Neuotoxicology 2013, 34, 118–127. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Assessing Quality of Herbal Medicines with Reference to Contaminants and Residues; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- World Health Organization (WHO). Quality Control Methods for Medicinal Plant Materials; World Health Organization: Geneva, Switzerland, 1998. [Google Scholar]
- Mtengeti, E.J.; Mhelela, A. Screening of potential indigenous browse species in semi-arid central Tanzania: A case of Gairo division. Livest. Res. Rural Dev. 2006, 18, 9. [Google Scholar]
- Muithya, J.N. Phytochemical and in Vitro Anti-microbial Screening of Eechinops hispidus Fresen. and Grewia similis K. Schum. Ph.D. Thesis, School of Pure and Applied Science, Kenyatta University, Nairobi, Kenya, 2010. [Google Scholar]
- World Health Organization (WHO). Guidelines for Laboratory and Field testing of Mosquito larvicides World Health Organization Communicable Disease Control, Prevention and Eradication; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]


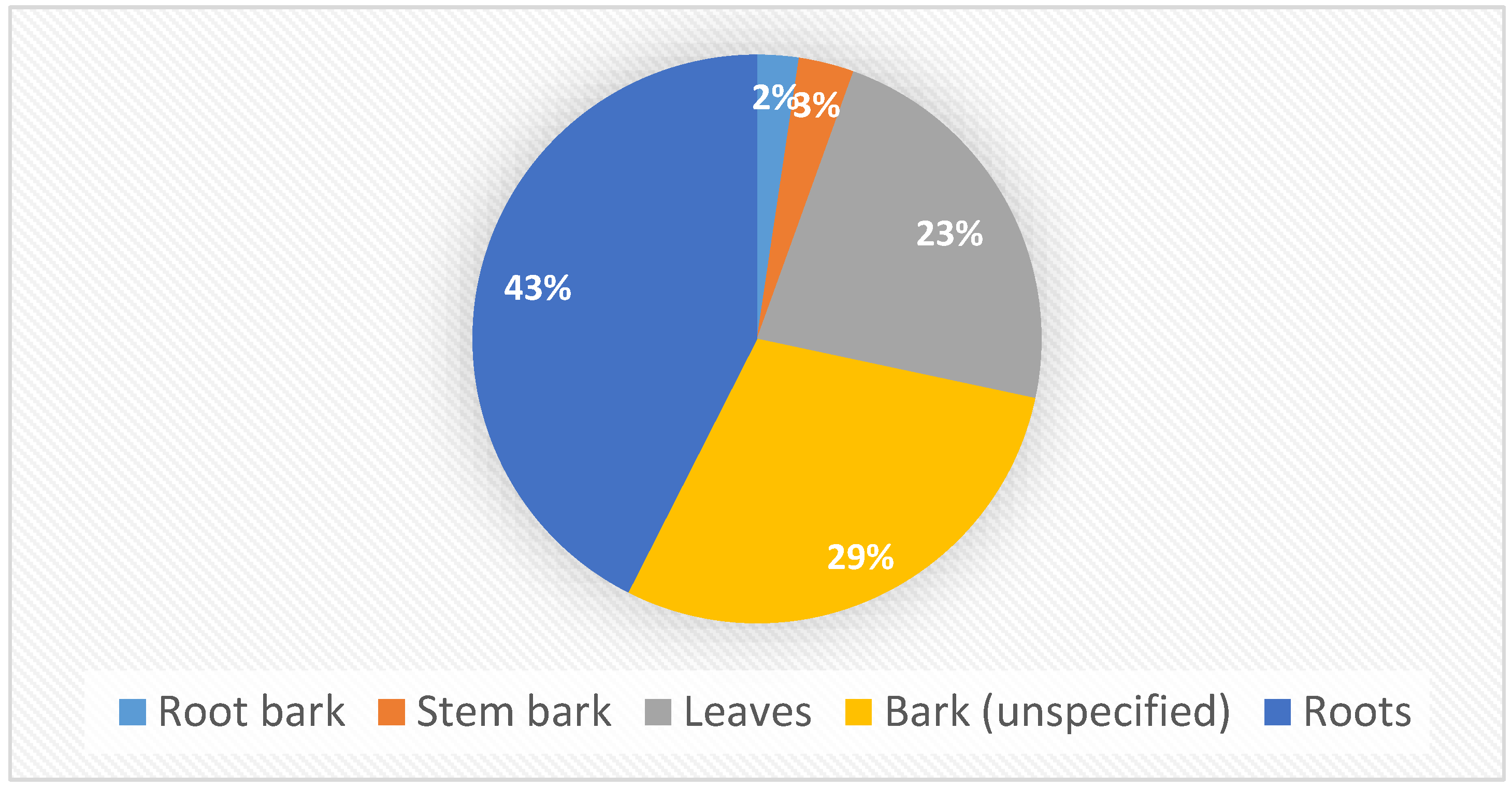
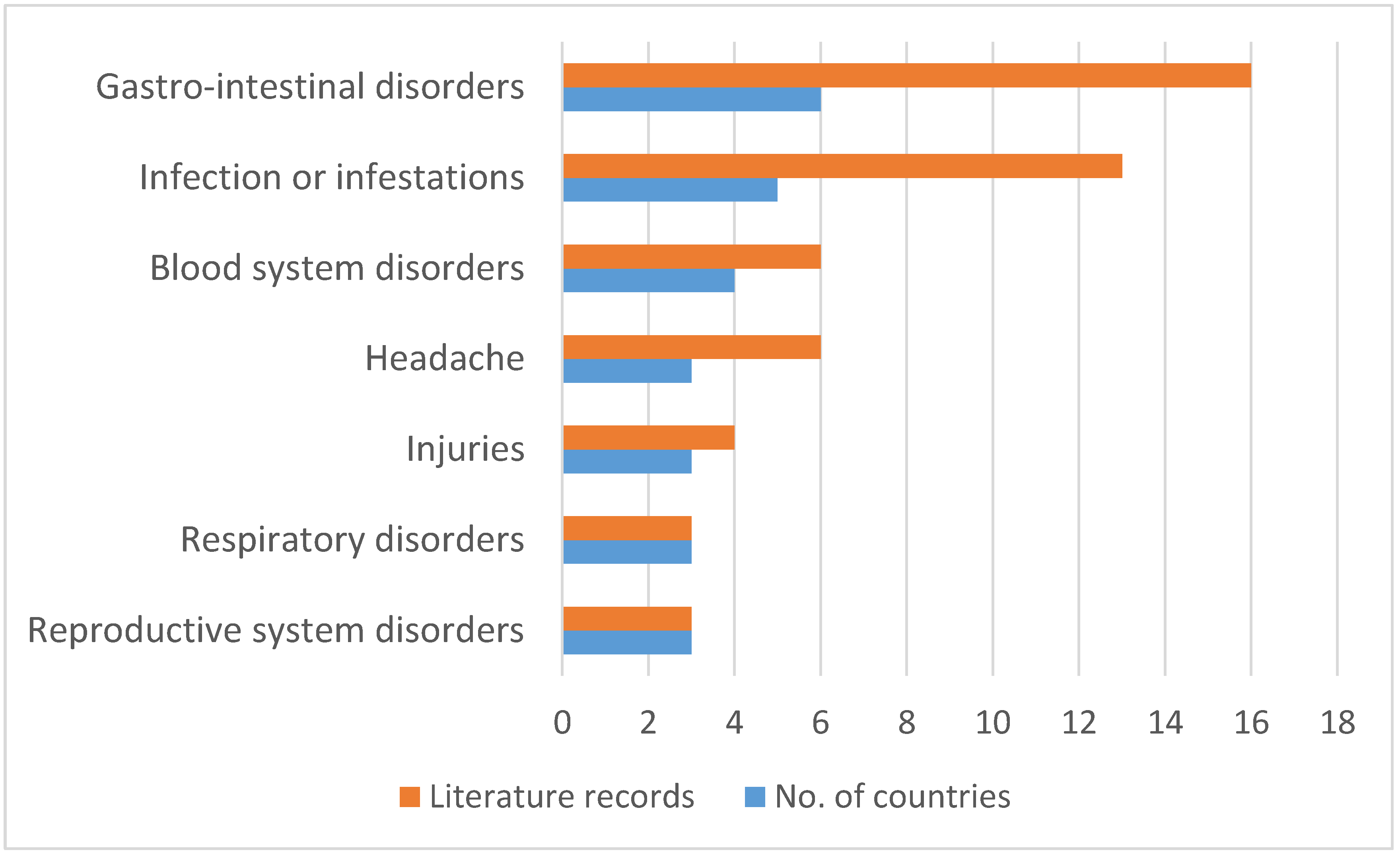
| Medicinal Use | Parts of the Plant Used | Country | References |
|---|---|---|---|
| Animal diseases (coccidiosis, corridor disease and prophylactic measure against poultry diseases) | Roots | Tanzania and Zimbabwe | [35,36] |
| Birth-related disorders (induce labour, pre, intra and post-partum, post-partum haemorrhage, pregnancy anaemia and retained after birth) | Bark, leaves and roots | Kenya and Tanzania | [27,70,71] |
| Blood pressure and diarrhoea | Bark mixed with roots of Plectranthus barbatus Andrews and Solanum incanum L. | Kenya | [34] |
| Blood system disorders (anaemia and blood pressure) | Bark and roots | Botswana, Kenya, Mozambique and Tanzania | [27,32,34,38,50,72] |
| Fever and malaria | Bark and roots | Tanzania | [27,58] |
| Fits | Bark and leaves | Zambia | [29] |
| Gastro-intestinal disorders (amoebic dysentery, diarrhoea, dysentery, stomach problems and stomachache) | Stems, leaves, bark and roots | Kenya, Mozambique, South Africa, Tanzania, Zambia and Zimbabwe | [9,27,28,29,32,33,50,51,60,73,74,75,76,77,78,79] |
| Headache | Leaves, bark and roots | Kenya, South Africa and Tanzania | [27,50,51,75,76,78] |
| Infections or infestations (abscesses, boils, carbuncles, cellulitis, gonorrhoea, herpes simplex, herpes zoster, HIV/AIDS, oral candidiasis, skin infections, skin rash, smallpox, syphilis and venereal diseases) | Stems, roots, leaves and bark | Kenya, Mozambique, Namibia, Tanzania and Zambia | [27,28,29,31,34,59,60,61,62,63,70,75,80] |
| Injuries (sores and wounds) | Bark and roots | South Africa, Swaziland and Tanzania | [9,27,58,81,82] |
| Mental disorder | Leaves | Mozambique | [28] |
| Pain (abdominal and body pains) | Leaves and roots | South Africa and Tanzania | [27,83] |
| Protective charm | Roots | Kenya and South Africa | [8,70,76,84] |
| Reproductive system disorders (abortifacient and sterility) | Leaves | Kenya, Tanzania and Uganda | [27,30,79] |
| Respiratory disorders (asthma, cough, tuberculosis and tussis) | Bark, leaves and roots | Kenya, Mozambique and Tanzania | [27,28,75] |
| Sedative | Roots | South Africa and Swaziland | [8,82,85] |
| Snake bite | Leaves and root bark | Kenya and South Africa | [8,34,86] |
| Stomach ulcers | Leaves and roots | Kenya and Tanzania | [79,81] |
| Swellings | Bark, leaves and roots | Kenya | [75] |
| Nutritional Composition | Values | Plant Parts | Reference |
|---|---|---|---|
| Acid detergent fibre (%) | 14.4 | Leaves | [109] |
| Alkaloids (%) | 1.8–4.7 | Leaves and roots | [88] |
| Ash (%) | 5.0–11.9 | Leaves and roots | [88,109] |
| Calcium (%) | 1.1 | Leaves | [109] |
| Carbohydrates (%) | 43.4–52.2 | Leaves and roots | [88] |
| Copper (ppm) | 0.9 | Leaves and roots | [88] |
| Dry matter (%) | 34.5 | Leaves | [109] |
| Fibre (%) | 0.5–1.0 | Leaves and roots | [88] |
| In vitro dry matter digestibility (%) | 61.3 | Leaves | [109] |
| Iron (ppm) | 1.4 | Leaves and roots | [88] |
| Lead (ppm) | 1.0–1.1 | Leaves and roots | [88] |
| Lipid (%) | 5.6–9.0 | Leaves and roots | [88] |
| Manganese (ppm) | 1.7–2.0 | Leaves and roots | [88] |
| Moisture (%) | 13.7–14.5 | Leaves and roots | [88] |
| Neutral detergent fibre (%) | 47.5 | Leaves | [109] |
| Phosphorus (%) | 0.3 | Leaves | [109] |
| Protein (%) | 15.4–22.0 | Leaves and roots | [88,109] |
| Saponins (%) | 10.2–16.5 | Leaves and roots | [88] |
| Total flavonol (mg quercetin/g of extract) | 17.3 | Roots | [93] |
| Total flavonoid (mg quercetin/g of extract) | 13.6 | Leaves and roots | [88,93] |
| Total phenol (mg tannic acid/g of extract) | 101.3 | Roots | [93] |
| Total phenolic content (mg gallic acid equivalent/g of extract) | 336 | Bark, leaves and roots | [88,101] |
| Zinc (ppm) | 1.2–2.6 | Leaves and roots | [88] |
| No. | Compound | Molecular Formula | Plant Part | References |
|---|---|---|---|---|
| Cardanol | ||||
| 1 | 3-[heneicos-16′(Z),18′(E)-dienyl] phenol | C27H44O | Leaves, roots and stems | [2] |
| 2 | 3-[nonadec-14′(Z),16′(E)-dienyl] phenol | C25H40O | Leaves, roots and stems | [2] |
| 3 | 3-[heptadecyl] phenol | C24H42O | Leaves, roots and stems | [2] |
| 4 | 3-[heptadec-12′(Z),14′(E)-dienyl] phenol | C23H36O | Leaves, roots and stems | [2] |
| 5 | 3-[tridecyl] phenol | C23H42O | Leaves, roots and stems | [2] |
| 6 | 3-((E)-nonadec-16′-enyl) phenol | C25H42O | Roots | [100] |
| Cyclohexenones | ||||
| 7 | 5-hydroxy-5-[tridecyl] cyclohex-2-enone | C19H34O | Leaves, roots and stems | [2] |
| 8 | 5-hydroxy-5-[pentadecyl] cyclohex-2-enone | C21H38O | Leaves, roots and stems | [2] |
| 9 | 5-hydroxy-5-[heptadecyl] cyclohex-2-enone | C23H42O | Leaves, roots and stems | [2] |
| 10 | 5-hydroxy-5-[pentadec-12′(E)-enyl] cyclohex-2-enone | C25H46O | Leaves, roots and stems | [2] |
| 11 | 5-hydroxy-5-[heptadec-14′(E)-enyl] cyclohex-2-enone | C23H40O | Leaves, roots and stems | [2] |
| Cyclohexenols | ||||
| 12 | 1-((E)-heptadec-14′-enyl)cyclohex-4-ene-1,3-diol | C23H42O2 | Roots | [100] |
| 13 | 1-[tridecyl] cyclohex-4-en-1,3-diol | C19H36O2 | Leaves, roots and stems | [2] |
| 14 | 1-[nonadecyl] cyclohex-4-en-1,3-diol | C25H48O2 | Leaves, roots and stems | [2] |
| 15 | 1-[heneicosyl] cyclohex-4-en-1,3-diol | C27H52O2 | Leaves, roots and stems | [2] |
| 16 | 1-[tricosyl] cyclohex-4-en-1,3-diol | C29H56O2 | Leaves, roots and stems | [2] |
| 17 | 1-[pentadec-12′(E)-enyl] cyclohex-4-en-1,3-diol | C21H38O2 | Leaves, roots and stems | [2,100] |
| 18 | 1-[nonadec-14′(Z),16′(E)-dienyl] cyclohex-4-en-1,3-diol | C25H44O2 | Leaves, roots and stems | [2] |
| 19 | 1-[heneicosen-16′(Z),18′(E)-dienyl] cyclohex-4-en-1,3 diol | C27H48O2 | Leaves, roots and stems | [2] |
| 20 | 1-[tridecyl] cyclohex-3-en-1,2,5-triol | C19H36O3 | Leaves, roots and stems | [2] |
| 21 | 1-[heptadecyl] cyclohex-3-en-1,2,5-triol | C23H44O3 | Leaves, roots and stems | [2] |
| Cyclitol | ||||
| 22 | Quinic acid | C7H12O6 | Bark | [101] |
| Dicarboxylic acid | ||||
| 23 | Malic acid | C4H6O5 | Bark | [101] |
| Flavonoids | ||||
| 24 | Caffeoylquinic acid | C16H18O9 | Bark | [101] |
| 25 | Catechin | C15H14O6 | Leaves, roots and stems | [2,100] |
| 26 | Epicatechin | C15H14O6 | Leaves, roots and stems | [2,101] |
| 27 | Epicatechin gallate | C22H18O10 | Bark, leaves, roots and stems | [2,101] |
| 28 | Feruloylquinic acid | C17H20O9 | Bark | [101] |
| 29 | Ligustroside | C4H6O5 | Bark | [101] |
| 30 | Procyanidin dimer mono gallate | C37H30O16 | Bark | [101] |
| 31 | Rutin | C27H30O16 | Leaves, roots and stems | [2] |
| Terpene | ||||
| 32 | Lupenone | C30H48O | Leaves, roots and stems | [2] |
| 33 | Sitosterol | C29H50O | Leaves, roots and stems | [2] |
| 34 | Sitosterol glucoside | C35H60O6 | Leaves, roots and stems | [2] |
| 35 | Taraxerol | C30H50O | Roots | [110] |
| 36 | Taraxerone | C30H48O | Roots | [110] |
| Activity Tested | Extract | Plant Part | Model | Effect | Reference |
|---|---|---|---|---|---|
| Acetylcholinesterase inhibitory | Ethyl acetate | Roots | Ellman’s colorimetric | Dose dependent activity recorded with IC50 = 0.0003 mg/mL | [93] |
| Anti-apoptotic | Ethyl acetate | Root bark | Cell death by apoptosis | Exhibited activities with LC50 = 36.0 μg/mL | [94] |
| Anti-apoptotic | Methanol | Root bark | Cell death by apoptosis | Exhibited activities with LC50 = 78.9 μg/mL | [94] |
| Antibacterial | Aqueous | Stem bark | Micro-dilution assay | Exhibited activities against Bacillus cereus with minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) value of 1000 μg/mL | [95] |
| Antibacterial | Methanol | Stem bark | Micro-dilution assay | Exhibited activities against Bacillus cereus with MIC and MBC value of 1000 μg/mL | [95] |
| Antibacterial | Hexane | Root | Disc diffusion method | Exhibited activities against both Enterococcus faecalis and Enterococcus faecium with 10 mm zone of inhibition | [2] |
| Antibacterial | Methanol | Root | Disc diffusion method | Exhibited activities against both Psudomonus aeruginosa and Salmonella typhimurium with 8 mm zone of inhibition, Enterococcus faecalis (13 mm), Enterococcus faecium (15 mm) and Staphylococcus aureus (15 mm) | [2] |
| Antibacterial | Methanol | Stem | Disc diffusion method | Exhibited activities against both Psudomonus aeruginosa and Salmonella typhimurium with 8 mm zone of inhibition, Enterococcus faecium (12 mm), Staphylococcus aureus (12 mm) and Enterococcus faecalis (14 mm) | [2] |
| Antibacterial | Ethanol | Root bark | Micro-dilution technique | Exhibited activities with MIC values of >1000 µg/mL and 125 µg/mL against Mycobacterium smegmatis and Propionibacterium acnes, respectively | [96] |
| Antiviral | Methanol | Stem bark | 50% end point titration technique | Exhibited activities with reduction factor (RF) values of 101 and 103 at concentrations of 25 µg/mL and 50 µg/mL, respectively against Semliki Forest virus A7 | [95] |
| Antiviral | Methanol | Stem bark | 50% end point titration technique | Exhibited activities with reduction factor (RF) values of 101 and 103 at concentrations of 25 µg/mL and 50 µg/mL, respectively against Semliki Forest virus A7 | [97] |
| Antiviral | Aqueous | Stem bark | Micro-dilution technique | Exhibited activities with IC50 value of 53.2 µg/mL and 89.4 µg/mL against human immunodeficiency virus type 1 (HIV-1, IIIB strain) and type 2 (HIV-2, ROD strain), respectively | [97] |
| Antiviral | 80% methanol | Stem bark | Micro-dilution technique | Exhibited activities with IC50 value of 7.1 µg/mL and 9.9 µg/mL against (HIV-1, IIIB strain and HIV-2, ROD strain, respectively | [97] |
| Anti-giardial | Methanol | Root bark | Growth inhibition of Giardia lamblia trophozoites | Extract was lethal or inhibited growth of Giardia lamblia trophozoites at 1000 ppm | [98] |
| Anti-inflammatory | Acetone | Roots | Lipoxygenase (15-LOX) inhibitory assay | Extract exhibited activities with IC50 value of 43 μg/mL | [99] |
| Anti-inflammatory | Methanol | Root | Carrageenan-induced rat paw edema method | Extracts showed moderate activities at 60 min and 120 min post-carrageenan administration | [100] |
| Antioxidant | Methanol | Root | 2,2´-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging assays | Exhibited activities with IC50 values of 0.004 mg/mL | [93] |
| Antioxidant | Methanol | Roots | 2,2-diphenyl-1-picryl hydrazyl (DPPH) radical scavenging assays | Exhibited activities with IC50 values of 0.01 mg/mL | [93] |
| Antioxidant | Methanol | Roots | DPPH radical scavenging assay | Exhibited activities with IC50 value of 22.8 µg/mL | [2] |
| Antioxidant | Methanol | Bark | DPPH radical scavenging assay | Exhibited activities with half maximal effective concentration (EC50) value of 5.6 µg/mL | [101] |
| Antioxidant | Methanol | Bark | Ferric reducing antioxidant power (FRAP) assays | Exhibited activities with 18.3 mM FeSO4 equivalent/mg sample | [101] |
| Antioxidant | Methanol | Bark | Antioxidant capacity (TAC) in Wistar rats | The extract increased TAC of liver tissues | [101] |
| Antiplasmodial | Aqueous | Stem bark | [G-3H] hypoxanthine incorporation assay | Extract exhibited activities with IC50 values of 10.6 µg/mL and 75.9 µg/mL, against Plasmodium falciparum chloroquine sensitive (D6) and chloroquine resistant (W2), respectively | [102] |
| Antiplasmodial | Methanol | Stem bark | [G-3H] hypoxanthine incorporation assay | Extract exhibited activities with IC50 values of 11.4 µg/mL and 36.3 µg/mL, against D6 and W2, respectively | [102] |
| Antiplasmodial | Aqueous | Stem bark | In vivo four-day suppressive test | Extract caused chemo-suppression of 83.1% | [102] |
| Antiplasmodial | Methanol | Stem bark | In vivo four-day suppressive test | Extract caused chemo-suppression of 91.4% | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Turraea robusta | In vitro drug interactions | Synergism and additive behaviour observed with sum of fractional inhibition concentration (SFIC) values of 0.6–1.7 | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Searsia natalensis | In vitro drug interactions | Additive behaviour observed with SFIC values of 1.0–1.4 | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Boscia salicifolia | In vitro drug interactions | Additive and synergistic behaviour observed with SFIC values of 0.9–1.3 | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Sclerocarya birrea | In vitro drug interactions | Additive and antagonistic behaviour observed with SFIC values of 1.2–2.2 | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Turraea robusta | In vivo drug interactions | Antagonistic behaviour observed with SFIC value 0.6, chemo-suppression of 26.8% and 57.5% after oral and interperitonial injection (i.p.) administration, respectively | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Searsia natalensis | In vivo drug interactions | Additive behaviour observed with SFIC value of 1.0, chemo-suppression of 50.7% and 71.9% after oral and i.p. administration, respectively | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Boscia salicifolia | In vivo drug interactions | Synergistic behaviour observed with SFIC value of 0.9, chemo-suppression of 61.0% and 90.6% after oral and i.p. administration, respectively | [102] |
| Antiplasmodial | Aqueous | Stem bark combined with Sclerocarya birrea | In vivo drug interactions | Synergistic behaviour observed with SFIC value of 1.2, chemo-suppression of 57.2% and 39.0% after oral and i.p. administration, respectively | [102] |
| Antiplasmodial | 80% methanol | Stem bark | Twofold serial dilutions | Extract exhibited with IC50 values ranging from 62.3 µg/mL to 125 µg/mL and MIC value of 125 µg/mL | [97] |
| Antiplasmodial | Methanol | Leaves | [G-3H] hypoxanthine incorporation assay | Extract exhibited activities with IC50 values of 38.9 µg/mL and 54.2 µg/mL, against D6 and W2, respectively | [33] |
| Antiplasmodial | Methanol | Leaves combined with Searsia natalensis | In vitro drug interactions | Synergistic behaviour observed with SFIC values of 0.4 - <1.0 | [33] |
| Antiplasmodial | Methanol | Leaves | In vivo four-day suppressive test | Extract caused chemo-suppression of 83.5% | [33] |
| Antiplasmodial | Methanol | Leaves combined with Searsia natalensis | In vivo four-day suppressive test | Extract caused chemo-suppression of 87.7% | [33] |
| Antiplasmodial | Aqueous | Stem bark | [G-3H] hypoxanthine incorporation assay | Extract exhibited activities with IC50 values of 10.6 µg/mL and 75.8 µg/mL, against D6 and W2, respectively | [103] |
| Antiplasmodial | Methanol | Stem bark | [G-3H] hypoxanthine incorporation assay | Extract exhibited activities with IC50 values of 11.4 µg/mL and 36.3 µg/mL, against D6 and W2, respectively | [103] |
| Antitrypanosomal | Dichloromethane | Roots | Colorimetric assay via the oxidation-reduction indicator resazurin | Extract exhibited activities with IC50 value of 22.2 µg/mL | [104] |
| Antitrypanosomal | Methanol | Roots | Colorimetric assay via the oxidation-reduction indicator resazurin | Extract exhibited activities with IC50 value of 44.2 µg/mL | [104] |
| Hepatoprotective | Aqueous | Stem bark | Biochemical and histopathological changes in Wistar rats | Extract reduced levels of serum aspartate aminotransferase (AST) and total bilirubin and significantly attenuated deleterious histopathologic changes in liver | [101] |
| Larvicidal | Ethyl acetate | Leaves, roots and stems | Larval mortality of Anopheles gambiae | Extracts exhibited activities with leaves exhibiting the median lethal dose (LC50) value of 47.9 µg/mL, stems (59.7 µg/mL) and roots (73.7 µg/mL) | [2] |
| Larvicidal | Hexane | Leaves, roots and stems | Larval mortality of Anopheles gambiae | Extracts exhibited activities with stem exhibiting LC50 value of 46.0 µg/mL, leaves (51.6 µg/mL) and roots (73.4 µg/mL) | [2] |
| Larvicidal | Methanol | Leaves, roots and stems | Larval mortality of Anopheles gambiae | Extracts exhibited activities with stems exhibiting LC50 value of 139.1 µg/mL, roots (147.1 µg/mL) and leaves (240.4 µg/mL) | [2] |
| Cytotoxicity | Methanol | Roots | Plaque reduction assay | Extract showed weak activities on Vero cells with CC50 value of 225.3 µg/mL and selectivity index of 6.2 and 198.8 against W2 and D6, respectively | [102] |
| Cytotoxicity | Dichloromethane | Roots | Colorimetric assay via the oxidation-reduction indicator resazurin | Extract exhibited activities with IC50 value of 27.2 µg/mL and selectivity index of 1.2 | [104] |
| Cytotoxicity | Methanol | Roots | Colorimetric assay via the oxidation-reduction indicator resazurin | Extract exhibited activities with IC50 value of 83.3 µg/mL and selectivity index of 1.9 | [104] |
| Cytotoxicity | Aqueous | Stem bark | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) calorimetric assay | Extract exhibited activities with CC50 values of >125 µg/mL | [97] |
| Cytotoxicity | 80% methanol | Stem bark | MTT calorimetric assay | Extract exhibited activities with CC50 values of 72.3 µg/mL | [97] |
| Cytotoxicity | Aqueous | Leaves | Growth inhibition of Vero E6 cells | Extract exhibited mild activities with CC50 values of 76 µg/mL and a selectivity index of 1.4 | [33] |
| Cytotoxicity | Methanol | Roots | Cytotoxicity determined against SH-SY5Y (human neuroblastoma) cells | Extract resulted in a concentration-dependent decrease in cell survival | [105] |
| Cytotoxicity | Methanol | Roots | MTT assay | Extract exhibited activities with IC50 values >100 µg/mL | [105] |
| Cytotoxicity | Methanol | Roots | Neutral red uptake assay | Extract exhibited activities with IC50 values >100 µg/mL | [105] |
| Cytotoxicity | Methanol | Roots | MTT assay | Extract exhibited activities with CC50 of 7.4 μg/mL and 74.0 μg/mL, on Vero cell lines and DU-145 prostate cancer cell lines, respectively | [100] |
| Toxicity | Methanol | Stems | Brine shrimp lethality test | Extract exhibited activities with LC50 value of 67.9 μg/mL | [58] |
| Toxicity | Methanol | Roots | in vivo acute toxicity activities | No deaths were observed at the highest concentration tested and LD50 values for both extracts was above 5000 mg/kg body weight | [102] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maroyi, A. Review of Ethnomedicinal, Phytochemical and Pharmacological Properties of Lannea schweinfurthii (Engl.) Engl. Molecules 2019, 24, 732. https://doi.org/10.3390/molecules24040732
Maroyi A. Review of Ethnomedicinal, Phytochemical and Pharmacological Properties of Lannea schweinfurthii (Engl.) Engl. Molecules. 2019; 24(4):732. https://doi.org/10.3390/molecules24040732
Chicago/Turabian StyleMaroyi, Alfred. 2019. "Review of Ethnomedicinal, Phytochemical and Pharmacological Properties of Lannea schweinfurthii (Engl.) Engl." Molecules 24, no. 4: 732. https://doi.org/10.3390/molecules24040732






