Chemical Constituents of Vigna luteola and Their Anti-inflammatory Bioactivity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Purification and Characterization
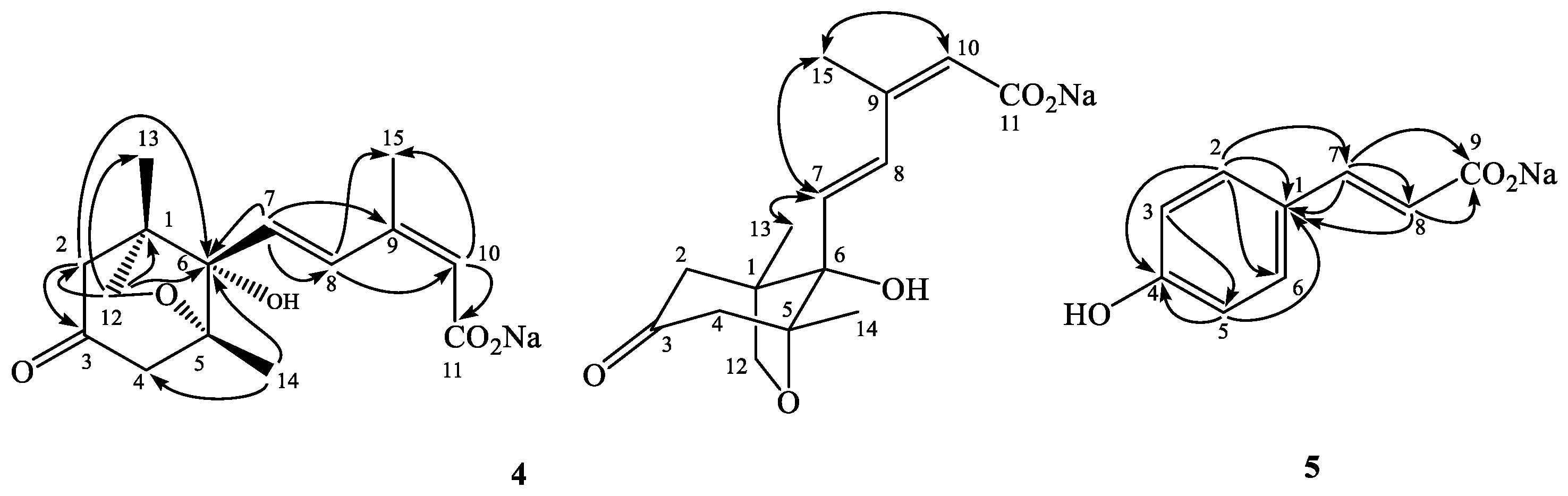
2.2. Structural Elucidations of 1–5
2.3. Anti-Inflammatory Activity
3. Experimental Section
3.1. General
3.2. Plant Materials
3.3. Extraction and Isolation
3.3.1. Viglutin (1)
3.3.2. Viglutoside (2)
3.3.3. Viglutanone (3)
3.3.4. Sodium Phaseate (4)
3.3.5. Sodium p-Coumarate (5)
3.4. Anti-Inflammatory Bioactivity Examination
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Na, H.K.; Kim, E.H.; Jung, J.H.; Lee, H.H.; Hyun, J.W.; Surh, Y.J. (−)-Epigallocatechin gallate induces Nrf 2-mediated antioxidant enzyme expression via activation of PI3K and ERK in human mammary epithelial cells. Arch. Biochem. Biophys. 2008, 476, 171–177. [Google Scholar] [CrossRef]
- Swami, S.; Krishnan, A.V.; Moreno, J.; Bhattacharyya, R.S.; Gardner, C.; Brooks, J.D.; Peehl, D.M.; Feldman, D. Inhibition of prostaglandin synthesis and actions by genistein in human prostate cancer cells and by soy isoflavones in prostate cancer patients. Int. J. Cancer 2009, 124, 2050–2059. [Google Scholar] [CrossRef] [PubMed]
- Bar-Sela, G.; Epelbaum, R.; Schaffer, M. Curcumin as an anti-cancer agent: Review of the gap between basic and clinical applications. Curr. Med. Chem. 2010, 17, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.C.; Yang, C.J.; Lee, Y.C.; Chen, P.C.; Liu, Y.C.; Wu, S.N. The comprehensive electrophysiological study of curcuminoids on delayed-rectifier K+ currents in insulin-secreting cells. Eur. J. Pharmacol. 2018, 819, 233–247. [Google Scholar] [CrossRef]
- Shu, L.; Cheung, K.L.; Khor, T.O.; Chen, C.; Kong, A.N. Phytochemicals: Cancer chemoprevention and suppression of tumor onset and metastasis. Cancer Metastasis Rev. 2010, 29, 483–502. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.M.; Wang, X.; Evers, B.M. Triptolide inhibits proliferation and migration of colon cancer cells by inhibition of cell cycle regulators and cytokine receptors. J. Surg. Res. 2011, 168, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The influence of natural products upon drug discovery. Nat. Prod. Rep. 2000, 17, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Boudjou, S.; Oomah, B.D.; Zaidi, F.; Hosseinian, F. Phenolics content and antioxidant and anti-inflammatory activities of legume fractions. Food Chem. 2013, 138, 1543–1550. [Google Scholar] [CrossRef]
- Lima, A.I.; Mota, J.; Monteiro, S.A.; Ferreira, R.M. Legume seeds and colorectal cancer revisited: Protease inhibitors reduce MMP-9 activity and colon cancer cell migration. Food Chem. 2016, 197, 30–38. [Google Scholar] [CrossRef]
- Hu, X.R.; Chou, G.X.; Zhang, C.G. Flavonoids, alkaloids from the seeds of Crotalaria pallida and their cytotoxicity and anti-inflammatory activities. Phytochemistry 2017, 143, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Malech, H.L.; Gallin, J.I. Neutrophils in human diseases. N. Engl. J. Med. 1987, 317, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Witko-Sarsat, V.; Rieu, P.; Descamps-Latscha, B.; Lesavre, P.; Halbwachs-Mecarelli, L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Investig. 2000, 80, 617–653. [Google Scholar] [CrossRef]
- Okajima, K.; Harada, N.; Uchiba, M. Ranitidine reduces ischemia/reperfusion-induced liver injury in rats by inhibiting neutrophil activation. J. Pharmacol. Exp. Ther. 2002, 301, 1157–1165. [Google Scholar] [CrossRef]
- Ennis, M. Neutrophils in asthma pathophysiology. Curr. Allergy Asthma Rep. 2003, 3, 159–165. [Google Scholar] [CrossRef]
- Vinten-Johansen, J. Involvement of neutrophils in the pathogenesis of lethal myocardial reperfusion injury. Cardiovasc. Res. 2004, 61, 481–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, T.L.; Li, G.L.; Lan, Y.H.; Chia, Y.C.; Hsieh, P.W.; Wu, Y.H.; Wu, Y.C. Potent inhibition of superoxide anion production in activated human neutrophils by isopedicin, a bioactive component of the Chinese medicinal herb Fissistigma Oldhamii. Free Radic. Biol. Med. 2009, 46, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.C.; Ohashi, H. Flora of Taiwan, 2nd ed.; Editorial Committee of Flora of Taiwan: Taipei, Taiwan, 1993; Volume 3, p. 385. [Google Scholar]
- Sreerama, Y.N.; Sashikala, V.B.; Pratape, V.M. Phenolic compounds in cowpea and horse gram flours in comparison to chickpea flour: Evaluation of their antioxidant and enzyme inhibitory properties associated with hyperglycemia and hypertension. Food Chem. 2012, 133, 156–162. [Google Scholar] [CrossRef]
- Silva, L.R.; Pereira, M.J.; Azevedo, J.; Gonçalves, R.F.; Valentão, P.; de Pinho, P.G.; Andrade, P.B. Glycine max (L.) Merr., Vigna radiata L. and Medicago sativa L. sprouts: A natural source of bioactive compounds. Food Res. Int. 2013, 50, 167–175. [Google Scholar] [CrossRef]
- Seneviratne, G.I.; Harborne, J.B. Constitutive flavonoids and induced isoflavonoids as taxonomic makers in the genus Vigna. Biochem. Syst. Ecol. 1992, 20, 459–467. [Google Scholar] [CrossRef]
- Soucek, J.; Skvor, J.; Pouckova, P.; Matousek, J.; Slavik, T.; Matousek, J. Mung bean (Phaseolus aureus) nuclease and its biological and antitumor effects. Neoplasma 2006, 53, 402–409. [Google Scholar]
- Joanitti, G.A.; Azevedo, R.B.; Freitas, S.M. Apoptosis and lysosome membrane permeabilization induction on breast cancer cells by an anticarcinogenic Bowman-Birk protease inhibitor from Vigna unguiculata seeds. Cancer Lett. 2010, 293, 73–81. [Google Scholar] [CrossRef]
- Lattanzio, V.; Arpaia, S.; Cardinali, A.; Venere, D.D.; Linsalata, V. Role of endogenous flavonoids in resistance mechanism of Vigna to aphids. J. Agric. Food Chem. 2000, 48, 5316–5320. [Google Scholar] [CrossRef]
- Franco, O.L.; dos Santos, R.C.; Batista, J.A.N.; Mendes, A.C.M.; de Araújo, M.A.M.; Monnerat, R.G.; Grossi-de-Sá, M.F.; de Freitas, S.M. Effects of black-eyed pea trypsin/chemotrypsin inhibitor on proteolytic activity and on development of Anthonomus grandis. Phytochemistry 2003, 63, 343–349. [Google Scholar] [CrossRef]
- Yao, Y.; Cheng, X.Z.; Wang, L.X.; Wang, S.H.; Ren, G. Major phenolic compounds, antioxidant capacity and antidiabetic potential of rice bean (Vigna umbellata L.) in China. Int. J. Mol. Sci. 2012, 13, 2707. [Google Scholar] [CrossRef]
- Yao, Y.; Cheng, X.; Wang, S.; Wang, L.; Ren, G. Influence of altitudinal variation on the antioxidant and antidiabetic potential of adzuki bean (Vigna angularis). Int. J. Food Sci. Nutr. 2012, 63, 117–124. [Google Scholar] [CrossRef]
- Han, K.H.; Fukushima, M.; Ohba, K.; Shimada, K.I.; Sekikawa, M.; Chiji, H.; Lee, C.H.; Nakano, M. Hepatoprotective effects of the water extract from adzuki bean hulls on acetaminophen-induced damage in rat liver. J. Nutr. Sci. Vitaminol. 2004, 50, 380–383. [Google Scholar] [CrossRef]
- Kojima, M.; Yamashita, S.; Nishi, S.; Saito, Y.; Maeda, R. Antioxidative effect and liver protective action of adzuki polyphenol. Nippon Shokuhin Kagaku Kogaku Kaishi 2006, 53, 386–392. [Google Scholar] [CrossRef]
- Sato, S.; Mukai, Y.; Yamate, J.; Kato, J.; Kurasaki, M.; Hatai, A.; Sagai, M. Effect of polyphenol-containing adzuki bean (Vigna angularis) extract on blood pressure elevation and macrophage infiltration in the heart and kidney of spontaneously hypertensive rats. Clin. Exp. Pharmacol. Physiol. 2008, 35, 43–49. [Google Scholar] [CrossRef]
- Sato, S.; Mukai, Y.; Yamate, J. Beneficial effects of adzuki bean (Vigna angularis) extract: Anti-oxidant, anti-hypertension and treatment for renal damage. Curr. Nutr. Food Sci. 2009, 5, 217–222. [Google Scholar] [CrossRef]
- Franco, O.L.; Murad, A.M.; Leite, J.R.; Mendes, P.A.M.; Prates, M.V.; Jr, C.B. Identification of a cowpea γ-thionin with bactericidal activity. FEBS J. 2006, 273, 3489–3497. [Google Scholar] [CrossRef]
- Thembo, K.M.; Vismer, H.F.; Nyazema, N.Z.; Gelderblom, W.C.A.; Katerere, D.R. Antifungal activity of four weedy plant extracts against selected mycotoxigenic fungi. J. Appl. Microbiol. 2010, 109, 1479–1486. [Google Scholar] [CrossRef] [Green Version]
- Mukai, Y.; Sato, S. Polyphenol-containing adzuki bean (Vigna angularis) extract attenuates blood pressure elevation and modulates nitric oxide synthase and caveolin-1 expressions in rats with hypertension. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 491–497. [Google Scholar] [CrossRef]
- Mukai, Y.; Sato, S. Polyphenol-containing adzuki bean (Vigna angularis) seed coats attenuate vascular oxidative stress and inflammation in spontaneously hypertensive rats. J. Nutr. Biochem. 2011, 22, 16–21. [Google Scholar] [CrossRef]
- Itoh, T.; Furuichi, Y. Lowering serum cholesterol level by feeding a 40% ethanol-eluted fraction from HP-20 resin treated with hot water extract of adzuki beans (Vigna angularis) to rats fed a high-fat cholesterol diet. Nutrition 2009, 25, 318–321. [Google Scholar] [CrossRef]
- Solanki, Y.B.; Jain, S.M. Antihyperlipidemic activity of Clitoria ternatea and Vigna mungo in rats. Pharm. Biol. 2010, 48, 915–923. [Google Scholar] [CrossRef]
- Leu, Y.L.; Hwang, T.L.; Kuo, P.C.; Liou, K.P.; Huang, B.S.; Chen, G.F. Constituents from Vigna vexillata and their anti-inflammatory activity. Int. J. Mol. Sci. 2012, 13, 9754–9768. [Google Scholar] [CrossRef]
- Miyake, Y.; Ebata, M. The structures of a β-galactosidase inhibitor, galactostatin, and its derivatives. Agric. Biol Chem. 1988, 52, 661–666. [Google Scholar]
- Kashman, Y.; Yosief, T.; Carmeli, S. New triterpenoids from the Red Sea sponge Siphonochalina siphonella. J. Nat. Prod. 2001, 64, 175–180. [Google Scholar] [CrossRef]
- Arai, S.; Todoroki, Y.; Ibaraki, S.; Naoe, Y.; Hirai, N.; Ohigashi, H. Synthesis and biological activity of 3′-chloro, -bromo, and-iodoabscisic acids, and biological activity of 3′-fluoro-8′-hydroxyabscisic acid. Phytochemistry 1999, 52, 1185–1193. [Google Scholar] [CrossRef]
- Liao, C.R.; Kuo, Y.H.; Ho, Y.L.; Wang, C.Y.; Yang, C.S.; Lin, C.W.; Chang, Y.S. Studies on cytotoxic constituents from the leaves of Elaeagnus oldhamii Maxim. in non-small cell lung cancer A549 cells. Molecules 2014, 19, 9515–9534. [Google Scholar] [CrossRef]
- Swisłocka, R.; Kowczyk-Sadowy, M.; Kalinowska, M.; Lewandowski, W. Spectroscopic (FT-IR, FT-Raman, 1H and 13C NMR) and theoretical studies of p-coumaric acid and alkali metal p-coumarates. Spectroscopy 2012, 27, 35–48. [Google Scholar] [CrossRef]
- Yu, H.P.; Hsieh, P.W.; Chang, Y.J.; Chung, P.J.; Kuo, L.M.; Hwang, T.L. 2-(2-Fluorobenzamido)benzoate ethyl ester (EFB-1) inhibits superoxide production by human neutrophils and attenuates hemorrhagic shock-induced organ dysfunction in rats. Free Radic. Biol. Med. 2011, 50, 1737–1748. [Google Scholar] [CrossRef]
Sample Availability: Samples of all the isolated compounds are available from the authors. |


| Samples | Inh % a | |
|---|---|---|
| Superoxide Anion Generation | Elastase Release | |
| methanol extract | 51.8 ± 6.8 *** | 108.4 ± 6.9 *** |
| chloroform fraction | 91.2 ± 3.8 *** | 118.0 ± 5.0 *** |
| water fraction | 18.3 ± 6.1 * | 34.6 ± 3.6 *** |
| Compound | Superoxide Anion Generation | Elastase Release | ||
|---|---|---|---|---|
| IC50 (μM) a | Inh % b | IC50 (μM) | Inh % | |
| 26 | 6.1 ± 0.3 | 69.9 ± 4.4 *** | – c | 11.8 ± 2.1 ** |
| 54 | 4.5 ± 0.3 | 93.6 ± 3.3 *** | – | 23.7 ± 1.1 *** |
| 55 | 4.1 ± 0.2 | 99.0 ± 1.9 *** | 3.8 ± 0.1 | 89.4 ± 4.5 *** |
| 57 | 5.0 ± 0.4 | 88.4 ± 5.3 *** | 4.7 ± 0.4 | 89.9 ± 2.2 *** |
| 63 | 9.3 ± 0.3 | 52.5 ± 1.2 *** | 4.9 ± 0.2 | 75.2 ± 3.2 *** |
| 65 | 1.9 ± 0.2 | 89.3 ± 2.9 *** | 6.4 ± 0.7 | 61.3 ± 4.7 *** |
| 66 | – | 27.4 ± 7.5 * | 7.7 ± 0.5 | 60.4 ± 2.3 *** |
| 67 | 3.2 ± 0.1 | 100.0 ± 1.3 *** | 4.1 ± 0.7 | 99.6 ± 7.6 *** |
| 70 | 5.6 ± 0.9 | 85.2 ± 9.7 *** | – | 46.5 ± 2.2 *** |
| LY294002d | 1.0 ± 0.2 | 3.1 ± 0.7 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lam, S.-H.; Li, Y.-C.; Kuo, P.-C.; Hwang, T.-L.; Yang, M.-L.; Wang, C.-C.; Tzen, J.T.C. Chemical Constituents of Vigna luteola and Their Anti-inflammatory Bioactivity. Molecules 2019, 24, 1371. https://doi.org/10.3390/molecules24071371
Lam S-H, Li Y-C, Kuo P-C, Hwang T-L, Yang M-L, Wang C-C, Tzen JTC. Chemical Constituents of Vigna luteola and Their Anti-inflammatory Bioactivity. Molecules. 2019; 24(7):1371. https://doi.org/10.3390/molecules24071371
Chicago/Turabian StyleLam, Sio-Hong, Yue-Chiun Li, Ping-Chung Kuo, Tsong-Long Hwang, Mei-Lin Yang, Chien-Chiao Wang, and Jason T. C. Tzen. 2019. "Chemical Constituents of Vigna luteola and Their Anti-inflammatory Bioactivity" Molecules 24, no. 7: 1371. https://doi.org/10.3390/molecules24071371






