Synthesis of New Derivatives of Benzofuran as Potential Anticancer Agents
Abstract
:1. Introduction
2. Results
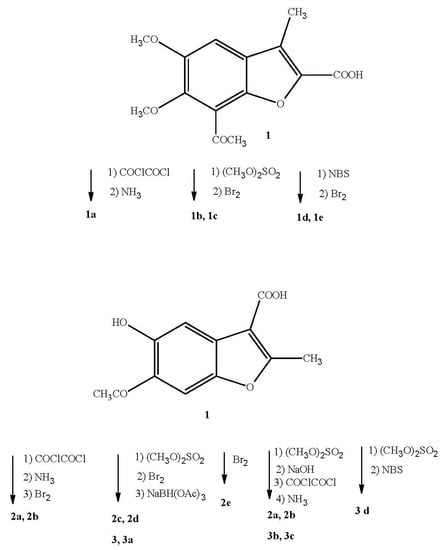
2.1. Synthesis
2.2. MTT Cytotoxicity Studies
2.3. Interaction with DNA
3. Discussion
4. Materials and Methods
4.1. Chemistry
4.2. General Synthetic Procedures
4.2.1. Synthesis of Analogues of Compound VIII
Synthesis of 7-Acetyl-5,6-Dimethoxy-3-Methylbenzofuran-2-Carboxylic Acid (1)
Synthesis of 7-Acetyl-5,6-Dimethoxy-3-Methylbenzofuran-2-Carboxamide (1a)
Synthesis of Methyl 7-Acetyl-5,6-Dimethoxy-3-Methylbenzofuran-2-Carboxylate (1b)
Synthesis of Methyl 7-Acetyl-3-(Bromomethyl)-5,6-Dimethoxybenzofuran-2-Carboxylate (1c)
Synthesis of 1-(2-Bromo-5,6-Dimethoxy-3-Methylbenzofuran-7-yl)ethanone (1d)
Synthesis of 2-Bromo-1-(2-Bromo-5,6-Dimethoxy-3-Methylbenzofuran-7-yl]ethanone (1e)
4.2.2. Synthesis of Analogues of Compound IX
Synthesis of 6-Acetyl-5-Hydroxy-2-Methylbenzofuran-3-Carboxamide (2a)
Synthesis of 6-Acetyl-4-Bromo-5-Hydroxy-2-Methylbenzofuran-3-Carboxamide (2b)
Synthesis of Methyl 6-Acetyl-5-Hydroxy-2-Methylbenzofuran-3-Carboxylate (2c)
Synthesis of Methyl 6-Acetyl-2-(Bromomethyl)-5-Hydroxybenzofuran-3-Carboxylate (2d)
Synthesis of 6-Acetyl-4-Bromo-5-Hydroxy-2-Methylbenzofuran-3-Carboxylic Acid (2e)
4.2.3. Synthesis of Analogues of Compound X
Synthesis of Methyl 6-Acetyl-5-Methoxy-2-Methylbenzofuran-3-Carboxylate (3) and Methyl 6-(Dibromoacetyl)-5-Methoxy-2-Methyl-1-Benzofuran-3-Carboxylate (X)
Synthesis of Methyl 6-(2,2-Dibromo-1-Hydroxyethyl)-5-Methoxy-2-Methylbenzofuran-3-Carboxylate (3a)
Synthesis of 6-Acetyl-5-Methoxy-2-Methylbenzofuran-3-Carboxylic Acid (3b)
Synthesis of 6-Acetyl-5-Methoxy-2-Methylbenzofuran-3-Carboxamide (3c)
Synthesis of Methyl 6-Acetyl-2-(Bromomethyl)-5-Methoxybenzofuran-3-Carboxylate (3d)
4.3. Anticancer Activity
4.3.1. Cells and Cytotoxicity Assay
4.3.2. Induction of Cell Apoptosis Analyzed by Caspase-3/7 Assay
4.3.3. Digestion of Plasmid DNA with BamHI Restriction Nuclease
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Habtemarian, S. Antiinflammatory activity of the antirheumatic herbal drug, gravel root (Eupatorium purpureum): Further biological activities and constituents. Phytoter. Res. 2001, 15, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Pauletti, M.P.; Araujo, A.R.; Young, M.C.; Giesbrecht, A.M.; Bolzani, V.D. nor-Lignans from the leaves of Styrax ferrugineus (Styracaceae) with antibacterial and antifungal activity. Phytochemistry 2000, 55, 597–601. [Google Scholar] [CrossRef]
- Masubuchi, M.; Kawasaki, K.; Ebiike, H.; Ikeda, Y.; Tsujii, S.; Sogabe, S.; Fujii, T.; Sakata, K.; Shiratori, Y.; Aoki, Y.; et al. Design and synthesis of novel benzofurans as new class of antifungal agents targeting fungal N-myristoyltransferase. Part 1. Bioorg. Med. Chem. Lett. 2001, 11, 1833–1837. [Google Scholar] [CrossRef]
- Wróbel, J.E.; Dietrich, A.J.; Antane, M.M. Benzotiophenes, Benzofurans, and Indoles useful in the treatment of insulin resistance and hyperglycemia. U.S. Patent 6,251,936, 26 June 2001. [Google Scholar]
- Kayser, O.; Chen, M.; Kharazmi, A.; Kiderlen, A.F. Aurones Interfere with Leismania major mitochondrial fumarate reductase. Z. Naturforsch. C 2002, 57, 717–720. [Google Scholar] [CrossRef]
- Hayakawa, I.; Shioya, R.; Agatsuma, T.; Furukawa, H.; Naruto, S.; Sugano, Y. 4-Hydroxy-3-methyl-6-phenylbenzofuran-2-carboxylic acid ethyl ester derivatives as potent anti-tumor agents. Bioorg. Med. Chem. Lett. 2004, 14, 455–458. [Google Scholar] [CrossRef] [PubMed]
- Dawood, K.M. Benzofuran derivatives: A patent review. Expert Opin. Ther. Pat. 2013, 23, 1133–1156. [Google Scholar] [CrossRef] [PubMed]
- Kossakowski, J.; Ostrowska, K.; Hejchman, E.; Wolska, I. Synthesis and structural characterization of derivatives of 2- and 3-benzo[b]furan carboxylic acids with potential cytotoxic activity. Il Farmaco 2005, 60, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Graves, A.P.; Brenk, R.; Shoichet, B.K. Decoys for Docking. J. Med. Chem. 2005, 48, 3714–3728. [Google Scholar] [CrossRef]
- Repolles, M.J.; Pubill, C.F.; Cabeza, L.L.; Carbo, B.M.; Cerda, R.J.A.; Negrie, R.C. Benzofuran, dihydrobenzofuran, dihydrobenzopyran and benzopyran derivatives as antidepressant agents. Spanish Patent ES2131020 A1, 1 July 1999. [Google Scholar]
- Kossakowski, J.; Krawiecka, M.; Kuran, B.; Stefańska, J.; Wolska, I. Synthesis and preliminary evaluation of the antimicrobial activity of selected 3-benzofurancarboxylic acid derivatives. Molecules 2010, 15, 4737–4749. [Google Scholar] [CrossRef]
- Kossakowski, J.; Ostrowska, K. Synthesis of new derivatives of 2,3-dihydro-7-benzo[b]furanol with potential pharmacological activity. Acta Pol. Pharm. 2006, 63, 271–275. [Google Scholar]
- Kossakowski, J.; Ostrowska, K.; Struga, M.; Stefańska, J. Synthesis of new derivatives of 2,2-dimethyl-2,3-dihydro-7-benzo[b]furanol with potential antimicrobial activity. Med. Chem. Res. 2009, 18, 555–565. [Google Scholar] [CrossRef]
- Kodama, I.; Kamiya, K.; Toyama, J. Amiodarone: Ionic and cellular mechanisms of action of the most promising class III agent. Am. J. Cardiol. 1999, 84, 20R–28R. [Google Scholar] [CrossRef]
- Courchesne, W.E. Characterization of novel, broad-based fungicidal activity for anti-arrhythmic drug amiodarone. J. Pharmacol. Exp. Ther. 2002, 300, 195–199. [Google Scholar] [CrossRef]
- Courchesne, W.E.; Ozturk, S. Amiodarone induces a caffeine-inhibited, MID1-dependent rise in free cytoplasic calcium in Saccharomyces cerevisiae. Mol. Microbiol. 2003, 47, 223–234. [Google Scholar] [CrossRef]
- Gill, J.; Heel, R.C.; Fitton, A. Amiodarone. An overview of its pharmacological properties, and review of its therapeutic use in cardiac arrhythmias. Drugs 1992, 43, 69–110. [Google Scholar] [CrossRef]
- Pizzichini, M.; Aleo, M.F.; Marcolongo, R.; Marinello, E. The mechanism of benziodarone activity. Quad. Sclavo. Diagn. 1982, 18, 203–208. [Google Scholar]
- Heel, R.C.; Brogden, R.N.; Speight, T.M.; Avery, G.S. Benzbromarone: A review of its pharmacological properties and therapeutic use in gout and hyperuricaemia. Drugs 1977, 14, 349–366. [Google Scholar] [CrossRef]
- Masbernard, A.; Giudicelli, C.P. Ten years’ experience with benzbromarone in the management of gout and hyperuricaemia. S. Afr. Med. J. 1981, 59, 701–706. [Google Scholar]
- Lu, Y.; Shi, T.; Wang, Y.; Yang, H.; Yan, X.; Luo, X.; Jiang, H.; Zhu, W. Halogen Bonding-A Novel Interaction for Rational Drug Design? J. Med. Chem. 2009, 52, 2584–2862. [Google Scholar] [CrossRef]
- Bissantz, C.; Kuhn, B.; Stahl, M. A Medicinal Chemist’s Guide to Molecular Interaction. J. Med. Chem. 2010, 53, 5061–5084. [Google Scholar] [CrossRef]
- Wilcken, R.; Zimmermann, M.O.; Lange, A.; Joerger, A.C.; Boeckler, F.M. Principles and Applications of Halogen Bonding in Medicinal Chemistry and Chemical Biology. J. Med. Chem. 2013, 56, 1363–1388. [Google Scholar] [CrossRef]
- Ford, C.M.; Shing, H.P. Computational Tools To model Halogen Bonds in Medicinal Chemistry. J. Med. Chem. 2016, 59, 1655–1670. [Google Scholar] [CrossRef] [PubMed]
- Krawiecka, M.; Kuran, B.; Kossakowski, J.; Cieślak, M.; Kazmierczak-Barańska, J.; Królewska, K.; Nawrot, B. Synthesis and cytotoxic properties of halogen and aryl-/heteroarylpiperazinyl derivatives of benzofurans. Anti Cancer Agents Med. Chem. 2015, 15, 115–121. [Google Scholar] [CrossRef]
- Kossakowski, J.; Kuran, B.; Kazmierczak-Baranska, J.; Królewska, K.; Nawrot, B.; Krawiecka, M.; Cieslak, M. Halogen derivatives of benzo[b]furans useful as anti-neoplastic or anti-proliferative drugs. Spanish Patent EP 2631232 A1, 9 January 2013. [Google Scholar]
- Królewska-Golińska, K.; Cieślak, M.J.; Sobczak, M.; Dolot, R.; Radzikowska-Cieciura, E.; Napiórkowska, M.; Wybrańska, I.; Nawrot, B. Novel benzo[b]furans with anti-microtubule activity upregulate expression of apoptotic genes and arrest leukemia cells in G2/M phase. Anticancer Agents Med. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zawadowski, T.; Suski, S.; Rump, S.; Borkowska, G. Synthesis of new aminoalkanoles derivatives of benzofurans with an expected β-adrenolityc activity. Acta Polon. Pharm. 1989, XLVI, 201–208. [Google Scholar]
- Fink, S.L.; Cookson, B.T. Apoptosis, Pyroptosis, and Necrosis: Mechanistic Description of Dead and Dying Eukaryotic Cells. Infect. Immun. 2005, 73, 1907–1916. [Google Scholar] [CrossRef] [Green Version]
- Maszewska, M.; Leclaire, J.; Cieslak, M.; Nawrot, B.; Okruszek, A.; Caminade, A.M.; Majoral, J.P. Water-soluble polycationic dendrimers with a phosphoramidothioate backbone: Preliminary studies of cytotoxicity and oligonucleotide/plasmid delivery in human cell culture. Oligonucleotides 2003, 13, 193–205. [Google Scholar] [CrossRef]
Sample Availability: Not available. |









| Compound | clogP |
|---|---|
| VIII | 2.4327 |
| 1 | 1.7254 |
| 1a | 1.3276 |
| 1b | 2.1533 |
| 1c | 2.4745 |
| 1d | 3.0089 |
| 1e | 3.3301 |
| IX | 3.5663 |
| 2a | 1.1219 |
| 2b | 1.8471 |
| 2d | 2.2688 |
| 2e | 2.2449 |
| X | 3.1168 |
| 3a | 2.9350 |
| 3b | 1.7954 |
| 3c | 1.3972 |
| 3d | 2.5445 |
| Compound | HeLa | K562 | MOLT-4 | HUVEC |
|---|---|---|---|---|
| 1c | 50 | 25 | 180 | 30 |
| 1e | 28 | 41 | 70 | 30 |
| 2d | 35 | 20 | 35 | 30 |
| 3a | 80 | 85 | 85 | 67 |
| 3d | 35 | 28 | 55 | 6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Napiórkowska, M.; Cieślak, M.; Kaźmierczak-Barańska, J.; Królewska-Golińska, K.; Nawrot, B. Synthesis of New Derivatives of Benzofuran as Potential Anticancer Agents. Molecules 2019, 24, 1529. https://doi.org/10.3390/molecules24081529
Napiórkowska M, Cieślak M, Kaźmierczak-Barańska J, Królewska-Golińska K, Nawrot B. Synthesis of New Derivatives of Benzofuran as Potential Anticancer Agents. Molecules. 2019; 24(8):1529. https://doi.org/10.3390/molecules24081529
Chicago/Turabian StyleNapiórkowska, Mariola, Marcin Cieślak, Julia Kaźmierczak-Barańska, Karolina Królewska-Golińska, and Barbara Nawrot. 2019. "Synthesis of New Derivatives of Benzofuran as Potential Anticancer Agents" Molecules 24, no. 8: 1529. https://doi.org/10.3390/molecules24081529