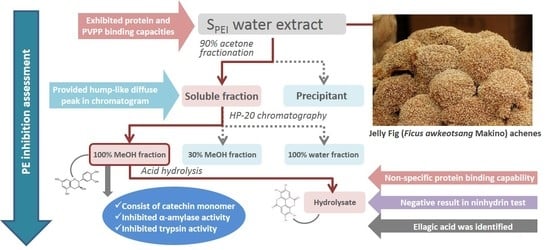
Complex Tannins Isolated from Jelly Fig Achenes Affect Pectin Gelation through Non-Specific Inhibitory Effect on Pectin Methylesterase
Abstract
1. Introduction
2. Results
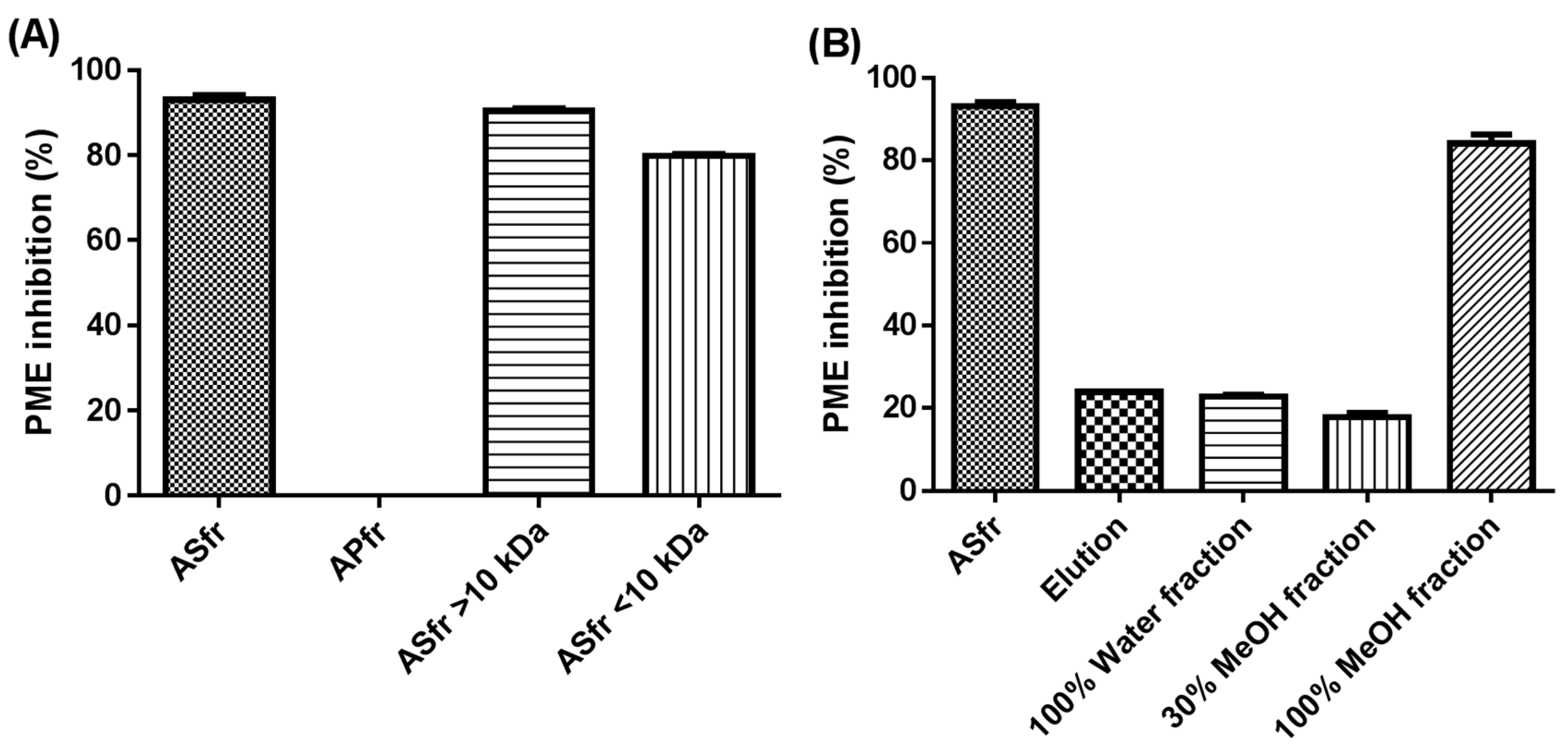
2.1. Effect of Molecular-Weight Fractionation and Polyvinylpolypyrrolidone (PVPP)/Protein Treatments on Inhibition Efficacy of Jelly Fig Achenes (JFA-SPEI)
2.2. Fractionation of Crude JFA-SPEI by Acetone Precipitation
2.3. Further Fractionation of SPEI by HP-20 Macroporous Resin Chromatography
2.4. High-Performance Liquid Chromatography (HPLC) Analysis of Acid Hydrolysate of SPEI Fractions
2.5. Characterization of 100% MeOHfr by Ninhydrin Test
2.6. Identification of JFA-SPEI by HPLC-Ultraviolet (UV) and Mass Spectrometry (MS) Spectrum
2.7. Inhibition of Digestive Enzymes
2.8. Total Tannins Contents
3. Discussion
4. Materials and Methods
4.1. Materials and Chemicals
4.2. Preparation of JFA-SPEI Crude Extract and Pectin Methylesterase (PE) Solution
4.3. Fractionation of SPEI Crude Extract
4.4. Evaluation of PE Inhibitory Activity
4.5. Chemical Characterization of SPEI
4.6. HPLC and MS Analysis
4.7. Digestive Enzyme Inhibition Assay
4.8. Determination of Tannins Level
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Micheli, F. Pectin methylesterases: Cell wall enzymes with important roles in plant physiology. Trends Plant Sci. 2001, 6, 414–419. [Google Scholar] [CrossRef]
- Bosch, M.; Cheung, A.Y.; Hepler, P.K. Pectin methylesterase, a regulator of pollen tube growth. Plant Physiol. 2005, 138, 1334–1346. [Google Scholar] [CrossRef] [PubMed]
- Bonavita, A.; Carratore, V.; Ciardiello, M.A.; Giovane, A.; Servillo, L.; D’Avino, R. Influence of pH on the structure and function of kiwi pectin methylesterase inhibitor. J. Agric. Food. Chem. 2016, 64, 5866–5876. [Google Scholar] [CrossRef] [PubMed]
- Willats, W.G.T.; Orfila, C.; Limberg, G.; Buchholt, H.C.; Van Alebeek, G.J.W.M.; Voragen, A.G.J.; Marcus, S.E.; Christensen, T.M.I.E.; Mikkelsen, C.J.D.; Murray, B.S.; et al. Modulation of the degree and pattern of methyl-esterification of pectic homogalacturonan in plant cell walls: Implications for pectin methyl esterase action, matrix properties, and cell adhesion. J. Biol. Chem. 2001, 276, 19404–19413. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.F.; Kragh, K.M.; Sibbesen, O.; Delcour, J.; Goesaert, H.; Svensson, B.; Tahir, T.A.; Brufau, J.; Perez-Vendrell, A.M.; Bellincampi, D.; et al. Potential role of glycosidase inhibitors in industrial biotechnological applications. Biochim. Biophys. Acta 2004, 1696, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.S.B.; Wu, M.C.; Jiang, C.M.; Hwang, Y.P.; Shen, S.C.; Chang, H.M. Pectinesterase inhibitor from jelly-fig (Ficus awkeotsang Makino) achenes reduces methanol content in carambola wine. J. Agric. Food Chem. 2005, 53, 9506–9511. [Google Scholar] [CrossRef]
- Giovane, A.; Balestrieri, C.; Quagliuolo, L.; Castaldo, D.; Servillo, L. A glycoprotein inhibitor of pectin methylesterase in kiwi fruit. Purification by affinity chromatography and evidence of a ripening-related precursor. Eur. J. Biochem. 1995, 233, 926–929. [Google Scholar] [CrossRef]
- Wolf, S.; Grsic-Rausch, S.; Rausch, T.; Greiner, S. Identification of pollen-expressed pectin methylesterase inhibitors in Arabidopsis. FEBS Lett. 2003, 555, 551–555. [Google Scholar] [CrossRef]
- An, S.H.; Sohn, K.H.; Choi, H.W.; Hwang, I.S.; Lee, S.C.; Hwang, B.K. Pepper pectin methylesterase inhibitor protein CaPMEI1 is required for antifungal activity, basal disease resistance and abiotic stress tolerance. Planta 2008, 228, 61–78. [Google Scholar] [CrossRef]
- Lewis, K.C.; Selzer, T.; Shahar, C.; Udi, Y.; Tworowski, D.; Sagi, I. Inhibition of pectin methyl esterase activity by green tea catechins. Phytochemistry 2008, 69, 2586–2592. [Google Scholar] [CrossRef]
- Jiang, C.M.; Lai, Y.J.; Chang, W.H.; Wu, M.C.; Chang, H.M. Pectinesterase inhibitor in jelly fig (Ficus awkeotsang Makino) achenes. J. Food Sci. 2001, 66, 225–228. [Google Scholar] [CrossRef]
- Jiang, C.M.; Lai, Y.J.; Lee, B.H.; Chang, W.H.; Wu, M.C.; Chang, H.M. Changes in physico-chemical properties of pectin from jelly fig (Ficus awkeotsang Makino) seeds during extraction and gelling. Food Res. Int. 2002, 35, 31–35. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Angulo, I.; Paseiro-Losada, P.; Cruz, J.M. Phenolic profile and antioxidant properties of a crude extract obtained from a brewery waste stream. Food Res. Int. 2013, 51, 663–669. [Google Scholar] [CrossRef]
- Gu, H.F.; Li, C.M.; Xu, Y.J.; Hu, W.F.; Chen, M.H.; Wan, Q.H. Structural features and antioxidant activity of tannin from persimmon pulp. Food Res. Int. 2008, 41, 208–217. [Google Scholar] [CrossRef]
- Mercurio, M.D.; Smith, P.A. Tannin quantification in red grapes and wine: Comparison of polysaccharide- and protein-based tannin precipitation techniques and their ability to model wine astringency. J. Agric. Food Chem. 2008, 56, 5528–5537. [Google Scholar] [CrossRef]
- Es-Safi, N.E.; Guyot, S.; Ducrot, P.H. NMR, ESI/MS, and MALDI-TOF/MS analysis of pear juice polymeric proanthocyanidins with potent free radical scavenging activity. J. Agric. Food Chem. 2006, 54, 6969–6977. [Google Scholar] [CrossRef]
- Reca, I.B.; Lionetti, V.; Camardella, L.; D’Avino, R.; Giardina, T.; Cervone, F.; Bellincampi, D. A functional pectin methylesterase inhibitor protein (SolyPMEI) is expressed during tomato fruit ripening and interacts with PME-1. Plant Mol. Biol. 2012, 79, 429–442. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Feng, J.; Wu, J.; Wang, X.W. BoPMEI1, a pollen-specific pectin methylesterase inhibitor, has an essential role in pollen tube growth. Planta 2010, 231, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Gupta, S.M.; Sane, A.P.; Nath, P. Isolation and characterization of ripening related pectin methylesterase inhibitor gene from banana fruit. Physiol. Mol. Biol. Plants 2012, 18, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Jolie, R.P.; Duvetter, T.; Van Loey, A.M.; Hendrickx, M.E. Pectin methylesterase and its proteinaceous inhibitor: A review. Carbohydr. Res. 2010, 345, 2583–2595. [Google Scholar] [CrossRef]
- Jiang, C.M.; Li, C.P.; Chang, J.C.; Chang, H.M. Characterization of pectinesterase inhibitor in jelly fig (Ficus awkeotsang Makino) achenes. J. Agric. Food Chem. 2002, 50, 4890–4894. [Google Scholar] [CrossRef]
- Chen, C.H.; Wu, M.C.; Hou, C.Y.; Jiang, C.M.; Huang, C.M.; Wang, Y.T. Effect of phenolic acid on antioxidant activity of wine and inhibition of pectin methyl esterase. J. I. Brewing 2009, 115, 328–333. [Google Scholar] [CrossRef]
- Xie, L.; Wehling, R.L.; Ciftci, O.; Zhang, Y. Formation of complexes between tannic acid with bovine serum albumin, egg ovalbumin and bovine beta-lactoglobulin. Food Res. Int. 2017, 102, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Díaz, A.M.; Caldas, G.V.; Blair, M.W. Concentrations of condensed tannins and anthocyanins in common bean seed coats. Food Res. Int. 2010, 43, 595–601. [Google Scholar] [CrossRef]
- Gonçalves, R.; Mateus, N.; Pianet, I.; Laguerre, M.; de Freitas, V. Mechanisms of tannin-induced trypsin inhibition: A molecular approach. Langmuir 2011, 27, 13122–13129. [Google Scholar] [CrossRef]
- Li, P.H.; Lin, Y.W.; Lu, W.C.; Hu, J.M.; Huang, D.W. In vitro hypoglycemic activity of the phenolic compounds in longan fruit (Dimocarpus Longan var. Fen ke) shell against α-glucosidase and β-galactosidase. Int. J. Food Prop. 2016, 19, 1786–1797. [Google Scholar] [CrossRef]
- Apostolidis, E.; Kwon, Y.I.; Shetty, K. Inhibitory potential of herb, fruit, and fungal-enriched cheese against key enzymes linked to type 2 diabetes and hypertension. Innov. Food Sci. Emerg. Technol. 2007, 8, 46–54. [Google Scholar] [CrossRef]
- Hathway, D.E.; Seakins, J.W.T. The influence of tannins on the degradation of pectin by pectinase enzymes. Biochem. J. 1958, 70, 158. [Google Scholar] [CrossRef]
- Wannes, W.A.; Mhamdi, B.; Sriti, J.; Jemia, M.B.; Ouchikh, O.; Hamdaoui, G.; Kchouk, M.E.; Marzouk, B. Antioxidant activities of the essential oils and methanol extracts from myrtle (Myrtus communis var. italica L.) leaf, stem and flower. Food Chem. Toxicol. 2010, 48, 1362–1370. [Google Scholar] [CrossRef]
- Sarin, V.K.; Kent, S.B.; Tam, J.P.; Merrifield, R.B. Quantitative monitoring of solid-phase peptide synthesis by the ninhydrin reaction. Anal. Biochem. 1981, 117, 147–157. [Google Scholar] [CrossRef]
- Amorim, E.L.; Nascimento, J.E.; Monteiro, J.M.; Peixoto Sobrinho, T.J.S.; Araújo, T.A.; Albuquerque, U.P. A simple and accurate procedure for the determination of tannin and flavonoid levels and some applications in ethnobotany and ethnopharmacology. FEC 2008, 2, 88–94. [Google Scholar]
Sample Availability: Not available. |




| Tested Substances | Absorbance at 570 nm | |
|---|---|---|
| Blank | Acid Hydrolysis | |
| Gelatin | 0.36 ± 0.03 | 9.10 ± 0.45 |
| 100% MeOHfr | 0.03 ± 0.01 | 0.03 ± 0.02 |
| Treatments | α-Amylase Relative Activity (%) | Trypsin Relative Activity (%) |
|---|---|---|
| Control without SPEI | 100 a | 100 a |
| ASfr | 76.8 ± 7.8 b | 88.6 ± 0.3 b |
| 100% MeOHfr | 78.4 ± 11.5 b | 70.2 ± 4.2 c |
| Fractions | Tannins | |
|---|---|---|
| Content in Tested Fractions (TAE mg/mL) | Total Tannins Content Divided by Dry Weight of Raw Materials (TAE mg/g D.W.) | |
| Crude extract | 8.48 ± 1.79 a | 1.70 |
| ASfr | 7.05 ± 1.21 ab | 1.41 |
| 100% water fraction | 0.12 ± 0.04 e | 0.02 |
| 30% MeOHfr | 0.16 ± 0.09 e | 0.03 |
| 100% MeOHfr | 6.74 ± 1.93 b | 1.35 |
| APfr | 0.57 ± 0.11 c | 0.12 |
| ASfr acid hydrolysate | 0.32 ± 0.25 cd | 0.06 |
| 100% MeOHfr acid hydrolysate | 0.22 ± 0.16 d | 0.04 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.-T.; Feng, Y.-J.; Lai, Y.-J.; Su, N.-W. Complex Tannins Isolated from Jelly Fig Achenes Affect Pectin Gelation through Non-Specific Inhibitory Effect on Pectin Methylesterase. Molecules 2019, 24, 1601. https://doi.org/10.3390/molecules24081601
Wang S-T, Feng Y-J, Lai Y-J, Su N-W. Complex Tannins Isolated from Jelly Fig Achenes Affect Pectin Gelation through Non-Specific Inhibitory Effect on Pectin Methylesterase. Molecules. 2019; 24(8):1601. https://doi.org/10.3390/molecules24081601
Chicago/Turabian StyleWang, Shang-Ta, You-Jiang Feng, Ying-Jang Lai, and Nan-Wei Su. 2019. "Complex Tannins Isolated from Jelly Fig Achenes Affect Pectin Gelation through Non-Specific Inhibitory Effect on Pectin Methylesterase" Molecules 24, no. 8: 1601. https://doi.org/10.3390/molecules24081601
APA StyleWang, S.-T., Feng, Y.-J., Lai, Y.-J., & Su, N.-W. (2019). Complex Tannins Isolated from Jelly Fig Achenes Affect Pectin Gelation through Non-Specific Inhibitory Effect on Pectin Methylesterase. Molecules, 24(8), 1601. https://doi.org/10.3390/molecules24081601