Phytochemical Analysis, Antioxidant and Analgesic Activities of Incarvillea compacta Maxim from the Tibetan Plateau
Abstract
:1. Introduction
2. Results
2.1. Total Phenols, Flavonoids and Alkaloids Content
2.2. Chemical Composition of Incarvillea Compacta Maxim
2.2.1. Phenolic Acids
2.2.2. Flavonoids
2.2.3. Alkaloids and Others
2.3. Antioxidant Activity Analysis
2.3.1. Scavenging Effect on DPPH Radicals
2.3.2. Scavenging Effect on ABTS Radicals
2.3.3. Reducing Power Test
2.3.4. β-Carotene Bleaching Test
2.4. Analgesic Test
2.4.1. Formalin Test
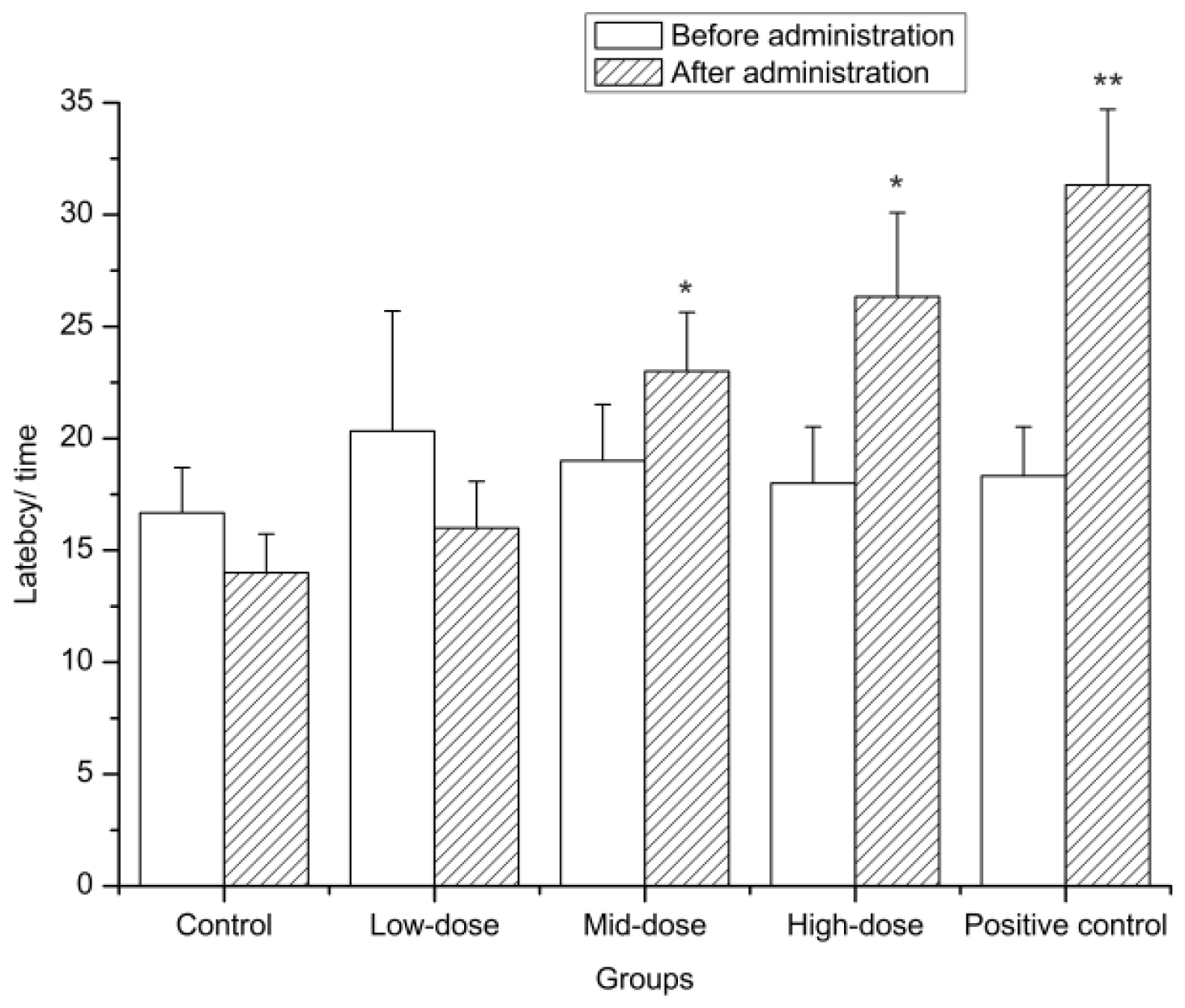
2.4.2. Hot-Plate Test
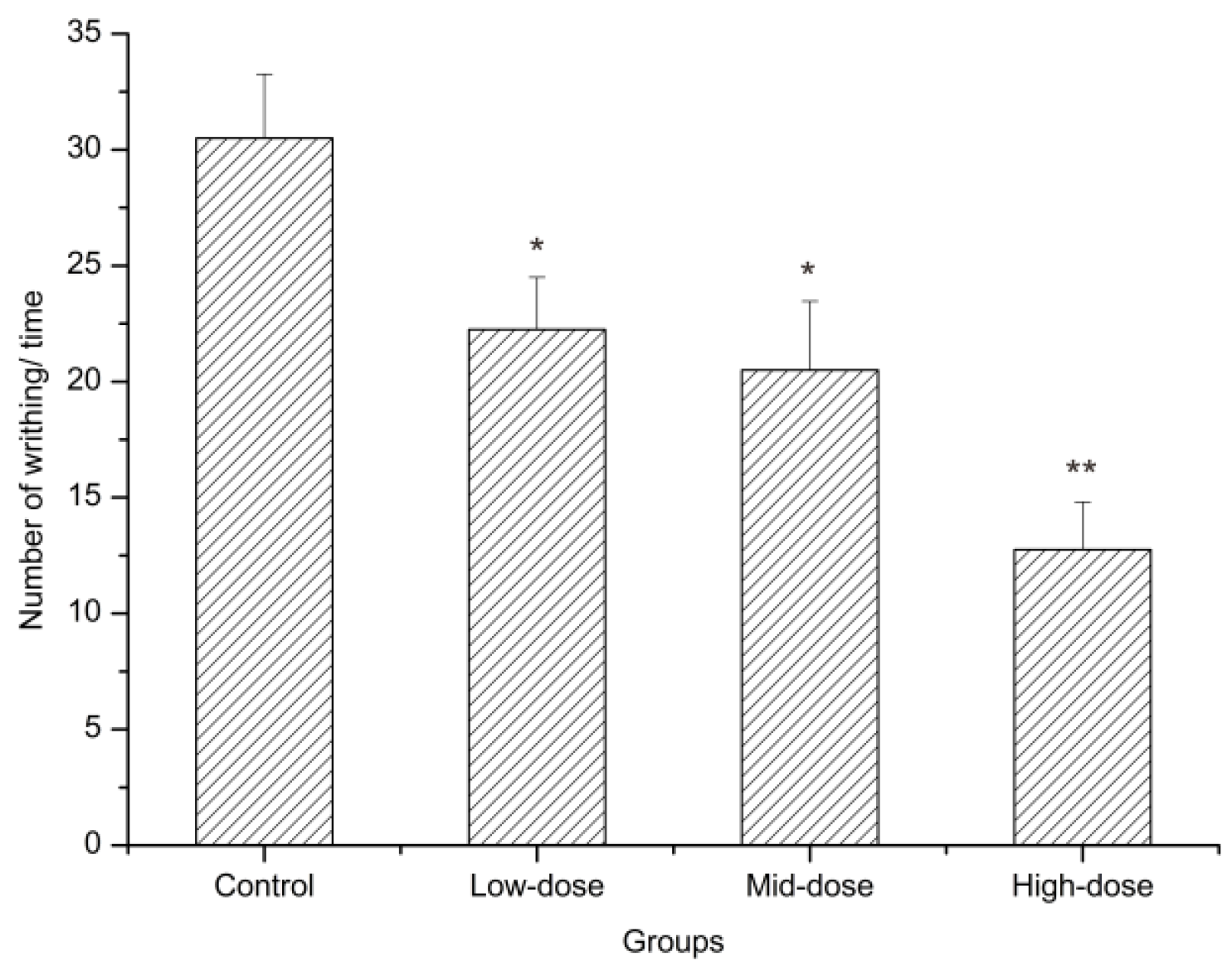
2.4.3. Acetic Acid Writhing Test
3. Discussion
3.1. Total Phenols, Flavonoids, and Alkaloids Content
3.2. Component Analysis
3.3. Antioxidant Activity In Vitro
3.4. Analgesic Effect
4. Materials and Methods
4.1. Chemicals Reagents and Plant Materials
4.2. Sample Preparation and Extractions
4.3. Determination of The Total Alkaloids Content
4.4. Estimation of The Total Flavonoids Content
4.5. Measurement of the Total Phenols Content
4.6. LC-ESI-QTOF-MS/MS Analysis
4.7. In Vitro Antioxidant Activities
4.7.1. Scavenging Effect on DPPH Radicals
4.7.2. Scavenging Effect on ABTS Radicals
4.7.3. Reducing Power Test
4.7.4. β-Carotene Bleaching Test
4.8. Biochemical Assay
4.8.1. Animals
4.8.2. Formalin Test
4.8.3. Hot-Plate Test
4.8.4. Acetic Acid-induced Writhing Test
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chinese Academy of Sciences. Editorial Committee of Flora of China; Science Press: China, Beijing, 2004. [Google Scholar]
- Northwest Institute of Plateau Biology, Chinese Academy of Sciences. Tibetan Medicine Record; Qinghai People’s Publishing Press: Qinghai, China, 1991; pp. 463–464. [Google Scholar]
- Fu, J.-J.; Jin, H.-Z.; Shen, Y.-H.; Qin, J.-J.; Wang, Y.; Huang, Y.; Zeng, Q.; Zhang, W.-D. Chemical Constituents of Plants from the GenusIncarvillea. Chem. Biodivers. 2009, 6, 818–826. [Google Scholar] [CrossRef]
- Su, Y.Q.; Shen, Y.H.; Sheng, L.; Jian, T.; Tian, J.M.; Liu, X.H.; Zhang, W.D. Two new alkaloids from Incarvillea mairei var. grandiflora. Helv. Chim. Acta 2009, 92, 165–170. [Google Scholar] [CrossRef]
- Luo, Y.; Yi, J.; Li, B.; Zhang, G. Novel ceramides and a new glucoceramide from the roots of Incarvillea arguta. Lipids 2004, 39, 907–913. [Google Scholar] [CrossRef] [Green Version]
- Tao, L.; Wei, D.Z.; Yue, H.P.; Zhang, C.; Yun, H.S. A new iridoid from Incarvillea delavayi. Chin. Chem. Lett. 2007, 12, 1512–1514. [Google Scholar]
- Zhao, J.-Q.; Wang, Y.-M.; Dang, J.; Shao, Y.; Mei, L.-J.; Tao, Y.-D. Chemical Constituents of Incarvillea compacta. Chem. Nat. Compd.. 2017, 53, 548–550. [Google Scholar] [CrossRef]
- Shen, T.; Li, X.; Hu, W.; Zhang, L.; Xu, X.; Wu, H.; Ji, L. Hepatoprotective effect of phenylethanoid glycosides from Incarvillea compacta against CCl4-induced cytotoxicity in HepG2 cells. J. Korean Soc. Appl. Boil. Chem. 2015, 58, 617–625. [Google Scholar] [CrossRef]
- Wu, H.-F.; Zhu, Y.-D.; Zhang, L.-J.; Zou, Q.-Y.; Chen, L.; Shen, T.; Wang, X.-F.; Ma, G.-X.; Hu, B.-R.; Hu, W.-C. A new phenylethanoid glycoside from Incarvillea compacta. J. Asian Nat. Prod. Res. 2016, 18, 596–602. [Google Scholar] [CrossRef]
- Wang, M.-L.; Yu, G.; Yi, S.-P.; Zhang, F.-Y.; Wang, Z.-T.; Huang, B.; Su, R.-B.; Jia, Y.-X.; Gong, Z.-H. Antinociceptive effects of incarvillateine, a monoterpene alkaloid from Incarvillea sinensis, and possible involvement of the adenosine system. Sci. Rep. 2015, 5, 16107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.M.; Pu, J.X.; Xiao, W.L.; Sun, H.D. A new phenylethanoid glycoside from Isodon sculponeatus. Chin. Chem. Lett. 2011, 22, 961–963. [Google Scholar] [CrossRef]
- Nakamura, M.; Kido, K.; Kinjo, J.; Nohara, T. Antinociceptive substances from Incarvillea delavayi. Phytochemistry 2000, 53, 253–256. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Phenolic composition and antioxidant potential of grain legume seeds: A review. Food Res. Int. 2017, 101, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ingram, S. Pain: Novel Analgesics from Traditional Chinese Medicines. Curr. Biol. 2014, 24, R114–R116. [Google Scholar] [CrossRef]
- Rajendran, P.; Nandakumar, N.; Rengarajan, T.; Palaniswami, R.; Gnanadhas, E.N.; Lakshminarasaiah, U.; Gopas, J.; Nishigaki, I. Antioxidants and human diseases. Clin. Chim. Acta 2014, 436, 332–347. [Google Scholar] [CrossRef]
- Melov, S.; Ravenscroft, J.; Malik, S.; Gill, M.S.; Walker, D.W.; Clayton, P.E.; Wallace, D.C.; Malfroy, B.; Doctrow, S.R.; Lithgow, G.J. Extension of life-span with superoxide dismutase/catalase mimetics. Science 2000, 289, 1567–1569. [Google Scholar] [CrossRef]
- Verri, W.A., Jr.; Vicentini, F.T.M.C.; Baracat, M.M.; Georgetti, S.R.; Cardoso, R.D.R.; Cunha, T.M.; Ferreira, S.H.; Cunha, F.Q.; Fonseca, M.J.V.; Casagrande, R. Flavonoids as Anti-Inflammatory and Analgesic Drugs: Mechanisms of Action and Perspectives in the Development of Pharmaceutical Forms. Stud. Nat. Prod. Chem. 2012, 36, 297–330. [Google Scholar]
- Yasir, M.; Sultana, B.; Amicucci, M. Biological activities of phenolic compounds extracted from Amaranthaceae plants and their LC/ESI-MS/MS profiling. J. Funct. Foods 2016, 26, 645–656. [Google Scholar] [CrossRef]
- Lin, L.; Yang, Q.; Zhao, K.; Zhao, M. Identification of the free phenolic profile of Adlay bran by UPLC-QTOF-MS/MS and inhibitory mechanisms of phenolic acids against xanthine oxidase. Food Chem. 2018, 253, 108–118. [Google Scholar] [CrossRef]
- Carazzone, C.; Mascherpa, D.; Gazzani, G.; Papetti, A. Identification of phenolic constituents in red chicory salads (Cichorium intybus) by high-performance liquid chromatography with diode array detection and electrospray ionisation tandem mass spectrometry. Food Chem. 2013, 138, 1062–1071. [Google Scholar] [CrossRef]
- Zhuang, B.; Bi, Z.-M.; Wang, Z.-Y.; Duan, L.; Lai, C.-J.-S.; Liu, E.-H. Chemical profiling and quantitation of bioactive compounds in Platycladi Cacumen by UPLC-Q-TOF-MS/MS and UPLC-DAD. J. Pharm. Biomed. Anal. 2018, 154, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Pierson, J.T.; Monteith, G.R.; Roberts-Thomson, S.J.; Dietzgen, R.G.; Gidley, M.J.; Shaw, P.N. Phytochemical extraction, characterisation and comparative distribution across four mango (Mangifera indica L.) fruit varieties. Food Chem. 2014, 149, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Al-Rawahi, A.; Edwards, G.; Al-Sibani, M.; Al-Thani, G.; Al-Harrasi, A.S.; Rahman, M.S. Phenolic Constituents of Pomegranate Peels (Punica granatum L.) Cultivated in Oman. Eur. J. Med. Plants 2014, 4, 315–331. [Google Scholar] [CrossRef]
- Ramirez, J.E.; Zambrano, R.; Sepúlveda, B.; Simirgiotis, M.J. Antioxidant Properties and Hyphenated HPLC-PDA-MS Profiling of Chilean Pica Mango Fruits (Mangifera indica L. Cv. piqueño). Molecules 2013, 19, 438–458. [Google Scholar] [CrossRef] [Green Version]
- Hlila, M.B.; Mosbah, H.; Zanina, N.; Ben Nejma, A.; Ben Jannet, H.; Aouni, M.; Selmi, B. Characterisation of phenolic antioxidants in Scabiosa arenaria flowers by LC-ESI-MS/MS and NMR. J. Pharm. Pharmacol. 2016, 68, 932–940. [Google Scholar] [CrossRef]
- Arimboor, R.; Arumughan, C. HPLC-DAD-MS/MS profiling of antioxidant flavonoid glycosides in sea buckthorn ( Hippophae rhamnoides L.) seeds. Int. J. Food Sci. Nutr. 2012, 63, 730–738. [Google Scholar] [CrossRef] [PubMed]
- Bo, D.; Tingting, Z.; Guorong, F.; Zhanying, H.; Yutian, W. Qualitative and quantitative determination of ten alkaloids in traditional Chinese medicine Corydalis yanhusuo W.T. Wang by LC-MS/MS and LC-DAD. J. Pharmaceut. Biomed. Anal. 2007, 45, 219–226. [Google Scholar]
- Nam, T.-G.; Lee, S.M.; Park, J.-H.; Kim, D.-O.; Baek, N.-I.; Eom, S.H. Flavonoid analysis of buckwheat sprouts. Food Chem. 2015, 170, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Pop, R.M.; Socaciu, C.; Pintea, A.; Buzoianu, A.D.; Sanders, M.G.; Gruppen, H.; Vincken, J.-P.; Vincken, J. UHPLC/PDA-ESI/MS Analysis of the Main Berry and Leaf Flavonol Glycosides from Different CarpathianHippophaë rhamnoidesL. Varieties. Phytochem. Anal. 2013, 24, 484–492. [Google Scholar] [CrossRef]
- Chi, Y.M.; Yan, W.M.; Chen, D.C.; Hiroshi, N.; Yoichi, I.; Ushio, S. A monoterpene alkaloid from Incarvillea sinensis. Phytochemistry 1992, 31, 2930–2932. [Google Scholar] [CrossRef]
- Azimova, S. S, Yunusov M.S. Natural Compounds; Springer: New York, NJ, USA, 2013; pp. 523–524. [Google Scholar]
- Shyaula, S.L.; Choudhary, M.I.; Manandhar, M.D. Megastigmane, iridoid, benzyl alcohol and phenyl propanoid glycosides from the Nepalese sandalwood Osyris wightiana Wall. ex Wight. Mosc. Univ. Chem. 2013, 68, 293–297. [Google Scholar] [CrossRef]
- Hossain, M.B.; Rai, D.K.; Brunton, N.P.; Martin-Diana, A.B.; Barry-Ryan, C. Characterization of Phenolic Composition in Lamiaceae Spices by LC-ESI-MS/MS. J. Agric. Food Chem. 2010, 58, 10576–10581. [Google Scholar] [CrossRef]
- Li, H.; Zhang, D.; Tan, L.-H.; Yu, B.; Zhao, S.-P.; Cao, W.-G. Comparison of the antioxidant properties of various solvent extracts from Dipsacus asperoides and identification of phenolic compounds by LC-ESI-QTOF-MS–MS. South Afr. J. Bot. 2017, 109, 1–8. [Google Scholar] [CrossRef]
- Pereira, O.R.; Silva, A.M.; Domingues, M.R.; Cardoso, S.M.; Domingues, M.R. Identification of phenolic constituents of Cytisus multiflorus. Food Chem. 2012, 131, 652–659. [Google Scholar] [CrossRef] [Green Version]
- Jianping, S.; Feng, L.; Yan, B.; Ping, L.; Changqing, D. Screening non-colored phenolics in red wines using liquid chromatography/ultraviolet and mass spectrometry/mass spectrometry libraries. Molecules 2007, 12, 679–693. [Google Scholar]
- Ajila, C.; Naidu, K.; Bhat, S.; Rao, U. Bioactive compounds and antioxidant potential of mango peel extract. Food Chem. 2007, 105, 982–988. [Google Scholar] [CrossRef]
- Firuzi, O.; Lacanna, A.; Petrucci, R.; Marrosu, G.; Saso, L. Evaluation of the antioxidant activity of flavonoids by “ferric reducing antioxidant power” assay and cyclic voltammetry. Biochim. Biophys. Acta 2005, 1721, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Chinsamy, M.; Finnie, J.; Van Staden, J. Anti-inflammatory, antioxidant, anti-cholinesterase activity and mutagenicity of South African medicinal orchids. South Afr. J. Bot. 2014, 91, 88–98. [Google Scholar] [CrossRef] [Green Version]
- Taiwe, G.S.; Bum, E.N.; Talla, E.; Dimo, T.; Sidiki, N.; Dawe, A.; Nguimbou, R.M.; Dzeufiet, P.D.D.; De Waard, M. Evaluation of antinociceptive effects of Crassocephalum bauchiense Hutch (Asteraceae) leaf extract in rodents. J. Ethnopharmacol. 2012, 141, 234–241. [Google Scholar] [CrossRef] [Green Version]
- Yu-Ming, C.; Motoyuki, N.; Toyokichi, Y.; Xi-Ying, Z.; Wen-Mei, Y.; Fumio, H.; Junei, K.; Toshihiro, N.; Shinobu, S. Pharmacological study on the novel antinociceptive agent, a novel monoterpene alkaloid from Incarvillea sinensis. Biol. Pharm. Bull. 2005, 28, 1989–1991. [Google Scholar]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, andecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef]
- Taghizadeh, S.F.; Rezaee, R.; Davarynejad, G.; Karimi, G.; Nemati, S.H.; Asili, J. Phenolic profile and antioxidant activity of Pistacia vera var. Sarakhs hull and kernel extracts: The influence of different solvents. J. Food Meas. Charact. 2018, 12, 2138–2144. [Google Scholar] [CrossRef]
- Huang, D.S.; Zhang, W.D.; Pei, Y.H.; Peng, X.Y.; Huang, Z.S.; Li, H.L.; Shen, Y.H. Two New Alkaloids from Incarvillea sinensis. Helv. Chim. Acta 2010, 92, 1558–1561. [Google Scholar] [CrossRef]
- Jun, F.J.; Jiang, Q.J.; Qi, Z.; Ying, H.; Dong, Z.W.; Zi, J.H. Two new monoterpene alkaloid derivatives from the roots of Incarvillea arguta. Arch. Pharm. Res. 2011, 34, 199–202. [Google Scholar]
- Liu, J.; Bai, R.; Liu, Y.; Zhang, X.; Kan, J.; Jin, C. Isolation, structural characterization and bioactivities of naturally occurring polysaccharide-polyphenolic conjugates from medicinal plants-A reivew. Int. J. Biol. Macromolecules 2017, S0141813017324303. [Google Scholar] [CrossRef]
- Heleno, S.A.; Martins, A.; Queiroz, M.J.R.; Ferreira, I.C. Bioactivity of phenolic acids: Metabolites versus parent compounds: A review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, Y.H.; Lu, T.; Tang, J.; Liu, R.H.; Li, H.L.; Zhang, W.D. Chemical constituents from Incarvillea delavayi. Chem. Nat. Compd. 2010, 46, 305–307. [Google Scholar] [CrossRef]
- Longhai, L.U.; Ming, Y.; Sheng, L.; Qin, Z.; Zhang, W. Chemical constituents from acetyl acetate extract fraction of Incarvillea delavayi. China J. Chin. Mater. Med. 2009, 34, 1799–1801. [Google Scholar]
- Xing, A.T.; Tian, J.M.; Liu, C.M.; Li, H.L.; Zhang, W.D.; Lei, S. Three New Monoterpene Alkaloids and a New Caffeic Acid Ester from Incarvillea mairei var. multifoliolata. Helv. Chim. Acta 2010, 93, 718–723. [Google Scholar] [CrossRef]
- Martin-Tanguy, J.; Cabanne, F.; Perdrizet, E.; Martin, C. The distribution of hydroxycinnamic acid amides in flowering plants. Phytochemistry 1978, 17, 1927–1928. [Google Scholar] [CrossRef]
- Ruiz, A.; Aguilera, A.; Ercoli, S.; Parada, J.; Winterhalter, P.; Contreras, B.; Cornejo, P. Effect of the frying process on the composition of hydroxycinnamic acid derivatives and antioxidant activity in flesh colored potatoes. Food Chem. 2018, 268, 577–584. [Google Scholar] [CrossRef]
- Khan, S.A.; Chatterjee, S.S.; Kumar, V. Low dose aspirin like analgesic and anti-inflammatory activities of mono-hydroxybenzoic acids in stressed rodents. Life Sci. 2016, 148, 53–62. [Google Scholar] [CrossRef]
- Srinivasulu, C.; Ramgopal, M.; Ramanjaneyulu, G.; Anuradha, C.M.; Suresh Kumar, C. Syringic acid (SA) ‒ A Review of Its Occurrence, Biosynthesis, Pharmacological and Industrial Importance. Biomed. Pharmacother. 2018, 108, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Devi, K.P.; Malar, D.S.; Nabavi, S.F.; Sureda, A.; Xiao, J.; Nabavi, S.M.; Daglia, M. Kaempferol and inflammation: From chemistry to medicine. Pharmacol. Res. 2015, 99, 1–10. [Google Scholar] [CrossRef]
- Dharambir, K.; Ajay, S.; Singh, T.H.; Katrin, S.; Kumar, G.V.; Singh, B.H.; Setzer, W.N.; Gautam, S. Apigenin: A natural bioactive flavone-type molecule with promising therapeutic function. J. Funct. Foods 2018, 48, 457–471. [Google Scholar]
- Wang, W.; Sun, C.; Mao, L.; Ma, P.; Liu, F.; Yang, J.; Gao, Y. The biological activities, chemical stability, metabolism and delivery systems of quercetin: A review. Trends Food Sci. Technol. 2016, 56, 21–38. [Google Scholar] [CrossRef]
- Bae, D.-S.; Kim, Y.-H.; Pan, C.-H.; Nho, C.-W.; Samdan, J.; Yansan, J.; Lee, J.-K. Protopine reduces the inflammatory activity of lipopolysaccharide-stimulated murine macrophages. BMB Rep. 2012, 45, 108–113. [Google Scholar] [CrossRef]
- Yong-Ming, Y.; Min, H.; Mei, Y.; Xiao-Ping, D.; Yong-Xian, C. Terpenoids from Incarvillea arguta. J. Asian Nat. Prod. Res. 2013, 15, 9–14. [Google Scholar]
- Jeong, N.-H.; Yang, E.-J.; Jin, M.; Lee, J.Y.; Choi, Y.-A.; Park, P.-H.; Lee, S.-R.; Kim, S.-U.; Shin, T.-Y.; Kwon, T.K.; et al. Esculetin from Fraxinus rhynchophylla attenuates atopic skin inflammation by inhibiting the expression of inflammatory cytokines. Int. Immunopharmacol. 2018, 59, 209–216. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Li, C.; Ma, Q.; Chen, S. Esculetin inhibits oxidative stress and apoptosis in H9c2 cardiomyocytes following hypoxia/reoxygenation injury. Biochem. Biophys. Commun. 2018, 501, 139–144. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Muniyandi, K.; George, E.; Sathyanarayanan, S.; George, B.P.; Abrahamse, H.; Thamburaj, S.; Thangaraj, P.; Thanagraj, P. Phenolics, tannins, flavonoids and anthocyanins contents influenced antioxidant and anticancer activities of Rubus fruits from Western Ghats, India. Food Sci. Hum. Wellness 2019, 8, 73–81. [Google Scholar] [CrossRef]
- Tiwari, N.; Luqman, S.; Masood, N.; Gupta, M.M. Validated high performance thin layer chromatographic method for simultaneous quantification of major iridoids in Vitex trifolia and their antioxidant studies. J. Pharm. Biomed. Anal. 2012, 61, 207–214. [Google Scholar] [CrossRef]
- Anyasi, T.A.; Jideani, A.I.; Mchau, G.R.; Jideani, A.I.O. Effect of organic acid pretreatment on some physical, functional and antioxidant properties of flour obtained from three unripe banana cultivars. Food Chem. 2015, 172, 515–522. [Google Scholar] [CrossRef]
- Fu, J.J.; Qin, J.J.; Zeng, Q.; Huang, Y.; Jin, H.Z.; Zhang, W.-D. Four new sesquiterpenoids from the roots of Incarvillea arguta and their inhibitory activities against lipopolysaccharide-induced nitric oxide production. Chem. Pharm. Bull. 2011, 42, 1263–1266. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, J.-S.; Yang, B.; Lv, G.-P.; Li, S.-P. Free Radical Scavenging Activity and Characterization of Sesquiterpenoids in Four Species of Curcuma Using a TLC Bioautography Assay and GC-MS Analysis. Molecules 2010, 15, 7547–7557. [Google Scholar] [CrossRef]
- Shi, M.; He, W.; Liu, Y.; Li, X.; Yang, S.; Xu, Q. Protective effect of total phenylethanoid glycosides from Monochasma savatieri Franch on myocardial ischemia injury. Phytomedicine 2013, 20, 1251–1255. [Google Scholar] [CrossRef]
- Ahmad, A.; Gupta, G.; Afzal, M.; Kazmi, I.; Anwar, F. Antiulcer and antioxidant activities of a new steroid from Morus alba. Life Sci. 2013, 92, 202–210. [Google Scholar] [CrossRef]
- Ji, R.-R.; Kohno, T.; A Moore, K.; Woolf, C.J. Central sensitization and LTP: Do pain and memory share similar mechanisms? Trends Neurosci. 2003, 26, 696–705. [Google Scholar] [CrossRef]
- Khan, M.S.A.; Ahmed, N.; Misbah; Arifuddin, M.; Zakaria, Z.A.; Al-Sanea, M.M.; Khundmiri, S.U.K.; Ahmed, I.; Ahmed, S.; Mok, P.L. Anti-nociceptive mechanisms of flavonoids-rich methanolic leaf extract from Terminalia coriacea (Roxb.) Wight & Arn. leaves. Food Chem. Toxicol. 2018, 115, 523–531. [Google Scholar]
- Haigler, H.J.; Spring, D.D. Comparison of the analgesic effects of dimethyl sulfoxide and morphine. Ann. New York Acad. Sci. 2010, 411, 19–27. [Google Scholar] [CrossRef]
- Debprasad, C.; Hemanta, M.; Paromita, B.; Durbadal, O.; Kumar, K.A.; Shanta, D.; Kumar, H.P.; Tapan, C.; Ashoke, S.; Sekhar, C. Inhibition of NO2, PGE2, TNF-α, and iNOS EXpression by Shorea robusta L.: An Ethnomedicine Used for Anti-Inflammatory and Analgesic Activity. Evid.-Based Complement. Altern. Med. 2012, 2012, 1–14. [Google Scholar] [CrossRef]
- Zeng, C.-Q.; Luo, B.-L. Contents mensuration of total alkaloid in Uncaria rhynchophylla by acid dye colorimetry. J. Chin. Med. Mater. 2007, 30, 1021–1024. [Google Scholar]
- Lamaison, J.L.; Carnat, A. Teneurs en principaux flavonoides des fleurs et des feuilles de Crataegus monogyna Jacq. et de Crataegus laevigata (Poiret) DC. en fonction de la periode de vegetation. Plantes médicinales et phytothérapie 1991, 25, 12–16. (In French) [Google Scholar]
- Li, H.-B.; Wong, C.-C.; Cheng, K.-W.; Chen, F. Antioxidant properties in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT-Food Sci. Technol. 2008, 41, 385–390. [Google Scholar] [CrossRef]
- Jeong, C.-H.; Choi, G.N.; Kim, J.H.; Kwak, J.H.; Heo, H.J.; Shim, K.-H.; Cho, B.-R.; Bae, Y.-I.; Choi, J.-S. In vitro Antioxidative Activities and Phenolic Composition of Hot Water Extract from Different Parts of Cudrania tricuspidata. Prev. Nutr. Food Sci. 2009, 14, 283–289. [Google Scholar] [CrossRef]
- Li, N.; Pan, Z.; Zhang, D.; Wang, H.-X.; Yu, B.; Zhao, S.-P.; Guo, J.-J.; Wang, J.-W.; Yao, L.; Cao, W.-G.; et al. Chemical Components, Biological Activities, and Toxicological Evaluation of the Fruit (Aril) of Two Precious Plant Species from Genus Taxus. Chem. Biodivers. 2017, 14, e1700305. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Q.; Wu, Y.; Chen, G.; Yue, W.; Liang, Q. Response Surface Optimized Ultrasonic-Assisted Extraction of Flavonoids from Sparganii Rhizoma and Evaluation of Their in Vitro Antioxidant Activities. Molecules 2012, 17, 6769–6783. [Google Scholar] [CrossRef] [Green Version]
- Tan, L.-H.; Zhang, D.; Wang, G.; Yu, B.; Zhao, S.-P.; Wang, J.-W.; Yao, L.; Cao, W.-G. Comparative analyses of flavonoids compositions and antioxidant activities of Hawk tea from six botanical origins. Ind. Crop. Prod. 2016, 80, 123–130. [Google Scholar] [CrossRef]
- Choi, S.-S.; Lee, J.-K.; Suh, H.-W. Antinociceptive profiles of aspirin and acetaminophen in formalin, substance P and glutamate pain models. Brain Res. 2001, 921, 233–239. [Google Scholar] [CrossRef]
- Akram, E.; Sara, M.K.; Jalal Zarringhalam, M.; Maryam, E.; Shamsali, R. Antinociceptive and anti-inflammatory effects of olive oil (Olea europeae L.) in mice. Pharm. Biol. 2012, 50, 332–337. [Google Scholar]
- Łuszczki, J.J. Dose-response relationship analysis of pregabalin doses and their antinociceptive effects in hot-plate test in mice. Pharmacol. Rep. 2010, 62, 942–948. [Google Scholar] [CrossRef]
- Hajhashemi, V.; Fahmideh, F.; Ghanadian, M. Antinociceptive effect of methanolic extract and alkaloid fractions of Berberis integerrima root in animal models. Avicenna J. Phytomed. 2018, 8, 227–236. [Google Scholar]
Sample Availability: Samples of Incarvillea compacta Maxim are available from the authors. |



| Solvents | Total Phenols (GAE mg/g DW) | Total Flavonoids (RE mg/g DW) | Total Alkaloids (BE mg/g DW) |
|---|---|---|---|
| Water | 22.00 ± 0.80c | 29.24 ± 0.09c | 1.05 ± 0.02c |
| Methanol | 29.24 ± 0.04b | 49.07 ± 0.25b | 5.01 ± 0.07b |
| Acetone | 11.28 ± 0.05a | 9.11 ± 0.08a | 2.43 ± 0.02a |
| Peak No. | Rt (min) | Molecular Formula | [M + H]+ (m/z) | [M − H]− (m/z) | Error (ppm) | MS/MS Fragments | Proposed Compound | Reference | Classification |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 6.88 | C9H10O5 | 197.04555 | −2.1 | 182.024,167.0005 | Syringic acid | [18] | Phenolic acids | |
| 2 | 5.58 | C7H6O3 | 137.02442 | 0.4 | 108.0002, 92.0266 | p-Hydroxybenzoic acid | [19] | Phenolic acids | |
| 3 | 2.02 | C7H12O6 | 191.05611 | −1.1 | 173.0461,127.0410 93.0350, 85.0300, 59.0145 | Quinic acid | [20] | Phenolic acids | |
| 4 | 2.13 | C13H16O9 | 315.0721 | 0.7 | 153.0568, 59.0159 | Protocatechuic acid hexoside | [21] | Phenolic acids | |
| 5 | 12.54 | C27H30O16 | 609.14611 | −0.5 | 301.0347 (47.96) | Rutin | [22] | Flavonoid | |
| 6 | 18.12 | C27H30O15 | 593.15119 | −0.1 | 285.0395 | Kaempferol-3-O-rutinoside (nicotiflorin) | [23] | Flavonoid | |
| 7 | 14.7 | C21H20O12 | 463.0882 | −2 | 301.0345 | Quercetin-3-O-glucoside | [24] | Flavonoid | |
| 8 | 20.43 | C21H20O11 | 447.09329 | −2.4 | 285.0384 | Kaempferol-3-O-glucoside (astragalin) | [24] | Flavonoid | |
| 9 | 21.87 | C21H20O10 | 431.09837 | −5 | 269.0424 | Apigenin-7-O-glucoside (apigetrin) | [25] | Flavonoid | |
| 10 | 2 | C27H30O16 | 609.1461 | −0.5 | 301.0347 | Quercetin-3-glucoside-7-O-rhamnoside | [26] | Flavonoid | |
| 11 | 8.31 | C20H19NO5 | 354.1336 | −1 | 206.0804,188.0694 149.0596 | Protopine | [27] | Alkaloid | |
| 12 | 12.54 | C27H30O16 | 609.1458 | −0.5 | 301.0347,271.0235 | Quercetin-3-O-robinobioside | [28] | Flavonoid | |
| 13 | 19.34 | C16H12O7 | 317.9656 | −0.4 | 317.0645,302.0421 285.0360,274.0463 | Isorhamnetin | [29] | Flavonoid | |
| 14 | 12.59 | C15H10O7 | 303.0497 | −0.7 | 303.0484,285.0411 229.0498,153.0188 | Quercetin | [29] | Flavonoid | |
| 15 | 14.73 | C21H20O12 | 465.1028 | −1.2 | 303.0485 | Quercetin-3-glucoside | [29] | Flavonoid | |
| 16 | 2.37 | C4H6O5 | 133.01425 | 1.2 | 115.0038,71.0155, 51.0191 | Malic Acid | [23] | Organic acid | |
| 17 | 2.61 | C6H8O7 | 303.04993 | 191.01973 | −2 | 111.0088,87.0089, 59.0154 | Citric acid | [22] | Organic acid |
| 18 | 6.36 | C9H6O4 | 177.01933 | −0.7 | 149.0281, 133.0274, 105.0337, 89.0396 | Esculetin | [18] | Coumarins | |
| 19 | 1.69 | C11H21NO | 184.16959 | 0.6 | 184.1691,166.1585,107.0851 | Incarvilline | [30] | Alkaloid | |
| 20 | 2.56 | C10H11NO2 | 178.0863 | 0.7 | 162.0543,132.0797,118.0643,117.0568 | Plantagonine | [31] | Alkaloid | |
| 21 | 10.17 | C16H24O9 | 359.1348 | −0.7 | 197.0810,153.0922 | 8-Epideoxyloganic acid | [32] | Iridoids | |
| 22 | 11.5 | C9H8O3 | 163.04007 | 0.3 | 135.0455, 89.0397 | Coumaric acid | [33] | Phenolic acids | |
| 23 | 3.6 | C7H6O4 | 153.01933 | 0.3 | 109.0295,81.0366, 53.0414 | Protocatechuic acid | [34] | Phenolic acids | |
| 24 | 6.59 | C9H8O4 | 179.03498 | 0.9 | 136.0514 | Caffeic acid | [33] | Phenolic acids | |
| 25 | 38.26 | C15H10O5 | 269.04555 | −3.4 | 117.0351, 151.0046 | Apigenin | [35] | Flavonoid |
| Groups | Analgesic Rate (%) |
|---|---|
| Low-dose | 25.05% |
| Mid-dose | 32.79% |
| High-dose | 58.20% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, J.; Zhang, D.; Yu, C.; Yao, L.; Chen, Z.; Tao, Y.; Cao, W. Phytochemical Analysis, Antioxidant and Analgesic Activities of Incarvillea compacta Maxim from the Tibetan Plateau. Molecules 2019, 24, 1692. https://doi.org/10.3390/molecules24091692
Guo J, Zhang D, Yu C, Yao L, Chen Z, Tao Y, Cao W. Phytochemical Analysis, Antioxidant and Analgesic Activities of Incarvillea compacta Maxim from the Tibetan Plateau. Molecules. 2019; 24(9):1692. https://doi.org/10.3390/molecules24091692
Chicago/Turabian StyleGuo, Jiajia, Dan Zhang, Chao Yu, Ling Yao, Zhuo Chen, Yanduo Tao, and Weiguo Cao. 2019. "Phytochemical Analysis, Antioxidant and Analgesic Activities of Incarvillea compacta Maxim from the Tibetan Plateau" Molecules 24, no. 9: 1692. https://doi.org/10.3390/molecules24091692





