Microsphere Polymers in Molecular Imprinting: Current and Future Perspectives
Abstract
1. Introduction
2. Microsphere Polymers
3. Preparation of Molecularly Imprinted Microspheres
3.1. Precipitation Polymerisation
3.2. Controlled/‘Living’ Radical Precipitation Polymerisation (CRPP)
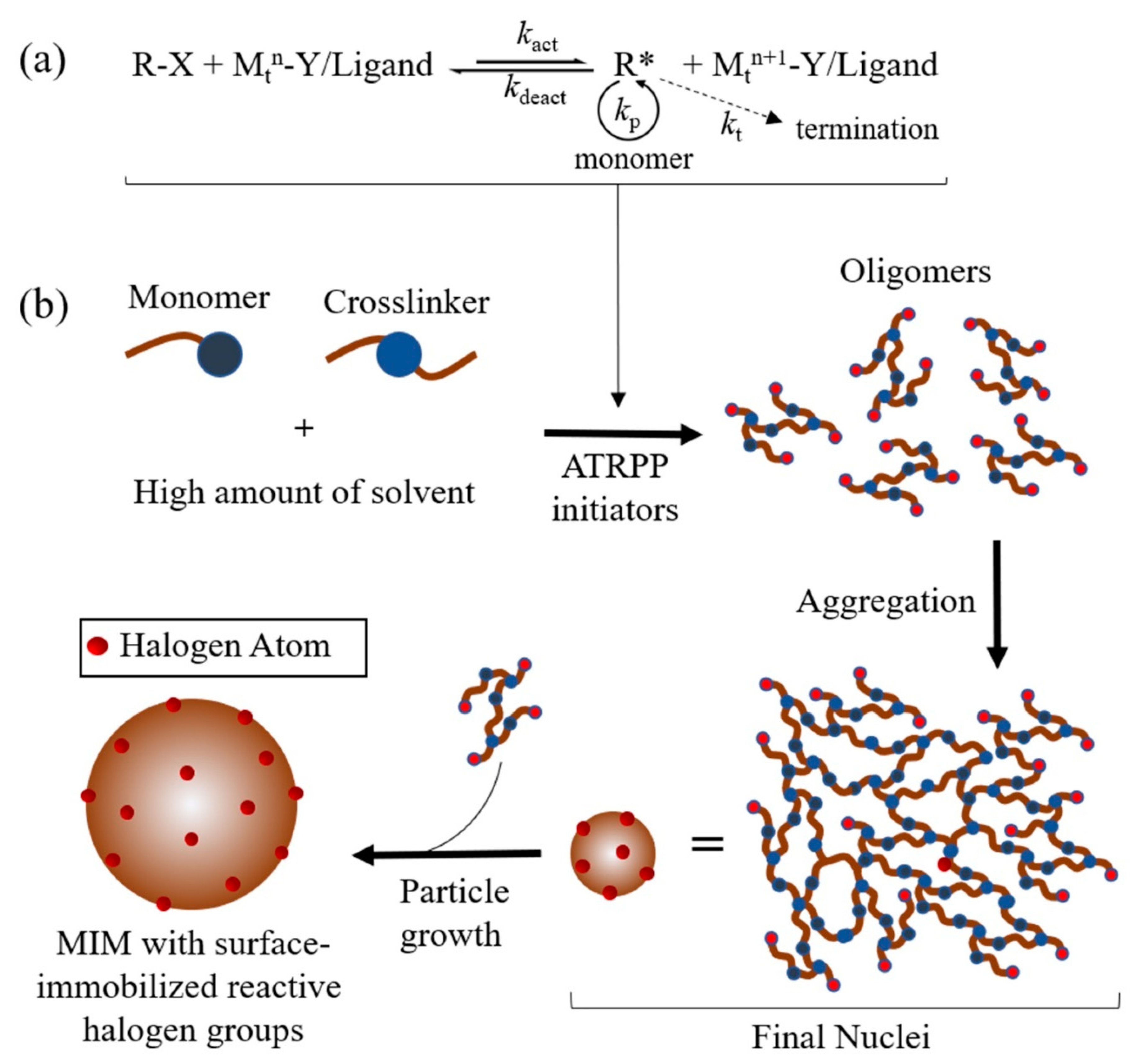
3.2.1. Atom Transfer Radical Precipitation Polymerisation (ATRPP)
3.2.2. Iniferter-induced ‘Living’ Radical Precipitation Polymerisation (ILRPP)
3.2.3. Reversible Addition-Fragmentation Chain Transfer Precipitation Polymerisation (RAFTPP)
3.3. Pickering Emulsion Polymerisation
3.4. Suspension Polymerisation
4. Green Aspects in Molecularly Imprinted Microspheres
5. Conclusions and Future Perspectives
- Comparison study. Future investigations to conclude which technique is better still need to be done with the same ratio composition of template:monomer:crosslinker in the same volume and type of solvent.
- Applicability of MIMs as drug delivery systems. MIMs have excellent potential as drug delivery systems because of their selective binding characteristics and their ability to release the template from the matrix. MIMs can also be used as targeting systems for the recognition of large molecules in gene therapy.
- Multiple stimuli-responsive MIMs. As there are thermo-, photo-, and pH-responsive MIMs, it is highly possible to develop multiple stimuli-responsive MIMs. Another environmental variable that can be used as a stimulus is biomolecule-responsive, which is the ability to undergo conformational change in response to signal biomolecules.
- New CRPP methods. Another CRPP could be developed, for example developing nitroxide-mediated precipitation polymerisation (NMPP) by substituting the conventional initiator with a nitroxide compound.
- The Pickering emulsion method can be an option for developing MIMs with both hydrophobic and hydrophilic properties. A double emulsion Pickering method can be developed for compound separation to achieve a greater degree of selectivity and affinity.
- Green strategies. Due to the numerous advantages of MIMs and rapid awareness of the importance of green chemistry, challenges such as how to conduct research and developments with a greener approach were provided for researchers to accomplish.
Funding
Conflicts of Interest
References
- Zu, B.; Pan, G.; Guo, X.; Zhang, Y.; Zhang, H. Preparation of molecularly imprinted polymer microspheres via atom transfer radical precipitation polymerization. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 3257–3270. [Google Scholar] [CrossRef]
- Beltran, A.; Marcé, R.M.; Cormack, P.A.G.; Borrull, F. Synthesis by precipitation polymerisation of molecularly imprinted polymer microspheres for the selective extraction of carbamazepine and oxcarbazepine from human urine. J. Chromatogr. A 2009, 1216, 2248–2253. [Google Scholar] [CrossRef] [PubMed]
- Alexander, C.; Andersson, H.S.; Andersson, L.I.; Ansell, R.J.; Kirsch, N.; Nicholls, I.A.; O’Mahony, J.; Whitcombe, M.J. Molecular imprinting science and technology: A survey of the literature for the years up to and including 2003. J. Mol. Recognit. 2006, 19, 106–180. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Ho Row, K. Characteristic and Synthetic Approach of Molecularly Imprinted Polymer. Int. J. Mol. Sci. 2006, 7, 155–178. [Google Scholar] [CrossRef]
- Pan, G.; Zu, B.; Guo, X.; Zhang, Y.; Li, C.; Zhang, H. Preparation of molecularly imprinted polymer microspheres via reversible addition-fragmentation chain transfer precipitation polymerization. Polymer 2009, 50, 2819–2825. [Google Scholar] [CrossRef]
- Wang, J.; Cormack, P.A.G.; Sherrington, D.C.; Khoshdel, E. Monodisperse, Molecularly Imprinted Polymer Microspheres Prepared by Precipitation Polymerization for Affinity Separation Applications. Angew. Chem. Int. Ed. 2003, 42, 5336–5338. [Google Scholar] [CrossRef]
- Zhang, H.; Ye, L.; Mosbach, K. Non-covalent molecular imprinting with emphasis on its application in separation and drug development. J. Mol. Recognit. 2006, 19, 248–259. [Google Scholar] [CrossRef]
- Pan, G.; Zhang, Y.; Ma, Y.; Li, C.; Zhang, H. Efficient one-pot synthesis of water-compatible molecularly imprinted polymer microspheres by facile RAFT precipitation polymerization. Angew. Chem. Int. Ed. 2011, 50, 11731–11734. [Google Scholar] [CrossRef] [PubMed]
- Hasanah, A.N.; Soni, D.; Pratiwi, R.; Rahayu, D.; Megantara, S. Synthesis of Diazepam-Imprinted Polymers with Two Functional Monomers in Chloroform Using a Bulk Polymerization Method. J. Chem. 2020, 2020, 1–8. [Google Scholar] [CrossRef]
- Turiel, E.; Martín-Esteban, A. Molecularly imprinted polymers-based microextraction techniques. TrAC Trends Anal. Chem. 2019, 118, 574–586. [Google Scholar] [CrossRef]
- Kubo, T.; Otsuka, K. Recent progress in molecularly imprinted media by new preparation concepts and methodological approaches for selective separation of targeting compounds. TrAC Trends Anal. Chem. 2016, 81, 102–109. [Google Scholar] [CrossRef]
- Sarafraz-Yazdi, A.; Razavi, N. Application of molecularly-imprinted polymers in solid-phase microextraction techniques. TrAC Trends Anal. Chem. 2015, 73, 81–90. [Google Scholar] [CrossRef]
- Keçili, R.; Hussain, C.M. Recent Progress of Imprinted Nanomaterials in Analytical Chemistry. Int. J. Anal. Chem. 2018, 2018, 8503853. [Google Scholar] [CrossRef]
- Kryscio, D.R.; Peppas, N.A. Critical review and perspective of macromolecularly imprinted polymers. Acta Biomater. 2012, 8, 461–473. [Google Scholar] [CrossRef]
- Dinc, M.; Esen, C.; Mizaikoff, B. Recent advances on core–shell magnetic molecularly imprinted polymers for biomacromolecules. TrAC Trends Anal. Chem. 2019, 114, 202–217. [Google Scholar] [CrossRef]
- Piletsky, S.; Canfarotta, F.; Poma, A.; Bossi, A.M.; Piletsky, S. Molecularly Imprinted Polymers for Cell Recognition. Trends Biotechnol. 2020, 38, 368–387. [Google Scholar] [CrossRef] [PubMed]
- Muratsugu, S.; Shirai, S.; Tada, M. Recent progress in molecularly imprinted approach for catalysis. Tetrahedron Lett. 2020, 61, 151603. [Google Scholar] [CrossRef]
- Mokhtari, P.; Ghaedi, M. Water compatible molecularly imprinted polymer for controlled release of riboflavin as drug delivery system. Eur. Polym. J. 2019, 118, 614–618. [Google Scholar] [CrossRef]
- Luliński, P. Molecularly imprinted polymers based drug delivery devices: A way to application in modern pharmacotherapy. A review. Mater. Sci. Eng. C 2017, 76, 1344–1353. [Google Scholar] [CrossRef]
- Yang, Y.; Yan, W.; Guo, C.; Zhang, J.; Yu, L.; Zhang, G.; Wang, X.; Fang, G.; Sun, D. Magnetic molecularly imprinted electrochemical sensors: A review. Anal. Chim. Acta 2020, 1106, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Beluomini, M.A.; da Silva, J.L.; de Sá, A.C.; Buffon, E.; Pereira, T.C.; Stradiotto, N.R. Electrochemical sensors based on molecularly imprinted polymer on nanostructured carbon materials: A review. J. Electroanal. Chem. 2019, 840, 343–366. [Google Scholar] [CrossRef]
- Yang, B.; Fu, C.; Li, J.; Xu, G. Frontiers in highly sensitive molecularly imprinted electrochemical sensors: Challenges and strategies. TrAC Trends Anal. Chem. 2018, 105, 52–67. [Google Scholar] [CrossRef]
- Wackerlig, J.; Lieberzeit, P.A. Molecularly imprinted polymer nanoparticles in chemical sensing—Synthesis, characterisation and application. Sensors Actuators B Chem. 2015, 207, 144–157. [Google Scholar] [CrossRef]
- Gui, R.; Jin, H. Recent advances in synthetic methods and applications of photo-luminescent molecularly imprinted polymers. J. Photochem. Photobiol. C Photochem. Rev. 2019, 41, 100315. [Google Scholar] [CrossRef]
- Qu, Y.; Qin, L.; Liu, X.; Yang, Y. Reasonable design and sifting of microporous carbon nanosphere-based surface molecularly imprinted polymer for selective removal of phenol from wastewater. Chemosphere 2020, 251, 126376. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Feng, T.; Xu, J.; Xue, C. Recent advances of molecularly imprinted polymer-based sensors in the detection of food safety hazard factors. Biosens. Bioelectron. 2019, 141, 111447. [Google Scholar] [CrossRef]
- Monier, M.; Youssef, I.; Abdel-Latif, D.A. Synthesis of imprinted styrene-maleic acid functionalized resin for enantio-selective extraction of R-amphetamine. Chem. Eng. J. 2019, 356, 693–701. [Google Scholar] [CrossRef]
- Monier, M.; Shafik, A.L.; Abdel-Latif, D.A. Surface molecularly imprinted amino-functionalized alginate microspheres for enantio-selective extraction of L-ascorbic acid. Carbohydr. Polym. 2018, 195, 652–661. [Google Scholar] [CrossRef]
- Voros, V.; Drioli, E.; Fonte, C.; Szekely, G. Process Intensification via Continuous and Simultaneous Isolation of Antioxidants: An Upcycling Approach for Olive Leaf Waste. ACS Sustain. Chem. Eng. 2019, 7, 18444–18452. [Google Scholar] [CrossRef]
- Didaskalou, C.; Buyuktiryaki, S.; Kecili, R.; Forte, C.P.; Szekely, G. Valorisation of agricultural waste with adsorption/nanofiltration hybrid process: From materials to sustainable process design. Green Chem. 2017, 19, 3116–3125. [Google Scholar] [CrossRef]
- Keçili, R.; Dolak, İ.; Ziyadanoğulları, B.; Ersöz, A.; Say, R. Ion imprinted cryogel-based supermacroporous traps for selective separation of cerium(III) in real samples. J. Rare Earths 2018, 36, 857–862. [Google Scholar] [CrossRef]
- Hasanah, A.N.; Dwi Utari, T.N.; Pratiwi, R. Synthesis of Atenolol-Imprinted Polymers with Methyl Methacrylate as Functional Monomer in Propanol Using Bulk and Precipitation Polymerization Method. J. Anal. Methods Chem. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Pratiwi, R.; Megantara, S.; Rahayu, D.; Pitaloka, I.; Hasanah, A.N. Comparison of Bulk and Precipitation Polymerization Method of Synthesis Molecular Imprinted Solid Phase Extraction for Atenolol using Methacrylic Acid. J. Young Pharm. 2019, 11, 12–16. [Google Scholar] [CrossRef]
- Pérez-Moral, N.; Mayes, A.G. Comparative study of imprinted polymer particles prepared by different polymerisation methods. Anal. Chim. Acta 2004, 504, 15–21. [Google Scholar] [CrossRef]
- Wei, M.; Geng, X.; Liu, Y.; Long, H.; Du, J. A novel electrochemical sensor based on electropolymerized molecularly imprinted polymer for determination of luteolin. J. Electroanal. Chem. 2019, 842, 184–192. [Google Scholar] [CrossRef]
- He, Y.; Wang, J.; Khan, M.; He, Y.; Hong, S.; Wang, M.; Wang, J.; She, Y.; Abd El-Aty, A.M.; Abd El-Aty, A.M.; et al. Development of fluorescent lateral flow test strips based on an electrospun molecularly imprinted membrane for detection of triazophos residues in tap water. New J. Chem. 2020, 44, 6026–6036. [Google Scholar] [CrossRef]
- Wu, X.; Wu, Y.; Chen, L.; Yan, L.; Zhou, S.; Zhang, Q.; Li, C.; Yan, Y.; Li, H. Bioinspired synthesis of pDA@GO-based molecularly imprinted nanocomposite membranes assembled with dendrites-like Ag microspheres for high-selective adsorption and separation of ibuprofen. J. Memb. Sci. 2018, 553, 151–162. [Google Scholar] [CrossRef]
- Wu, Y.; Lu, J.; Xing, W.; Ma, F.; Gao, J.; Lin, X.; Yu, C.; Yan, M. Double-layer-based molecularly imprinted membranes for template-dependent recognition and separation: An imitated core-shell-based synergistic integration design. Chem. Eng. J. 2020, 397, 125371. [Google Scholar] [CrossRef]
- Bai, J.; Zhang, Y.; Zhang, W.; Ma, X.; Zhu, Y.; Zhao, X.; Fu, Y. Synthesis and characterization of molecularly imprinted polymer microspheres functionalized with POSS. Appl. Surf. Sci. 2020, 511, 145506. [Google Scholar] [CrossRef]
- Ye, L.; Weiss, R.; Mosbach, K. Synthesis and characterization of molecularly imprinted microspheres. Macromolecules 2000, 33, 8239–8245. [Google Scholar] [CrossRef]
- Ye, L.; Cormack, P.A.G.; Mosbach, K. Molecularly imprinted monodisperse microspheres for competitive radioassay. Anal. Commun. 1999, 36, 35–38. [Google Scholar] [CrossRef]
- Zaidi, S.A. Molecular imprinted polymers as drug delivery vehicles. Drug Deliv. 2016, 23, 2262–2271. [Google Scholar] [CrossRef] [PubMed]
- Saralidze, K.; Koole, L.H.; Knetsch, M.L.W. Polymeric microspheres for medical applications. Materials 2010, 3, 3537–3564. [Google Scholar] [CrossRef]
- Kawaguchi, H. Functional polymer microspheres. Prog. Polym. Sci. 2000, 25, 1171–1210. [Google Scholar] [CrossRef]
- Zhu, W.; Ma, W.; Li, C.; Pan, J.; Dai, X.; Gan, M.; Qu, Q.; Zhang, Y. Magnetic molecularly imprinted microspheres via yeast stabilized Pickering emulsion polymerization for selective recognition of λ-cyhalothrin. Colloids Surfaces A Physicochem. Eng. Asp. 2014, 453, 27–36. [Google Scholar] [CrossRef]
- Gan, M.; Pan, J.; Zhang, Y.; Dai, X.; Yin, Y.; Qu, Q.; Yan, Y. Molecularly imprinted polymers derived from lignin-based Pickering emulsions and their selectively adsorption of lambda-cyhalothrin. Chem. Eng. J. 2014, 257, 317–327. [Google Scholar] [CrossRef]
- Zhu, Y.; Jiang, D.; Sun, D.; Yan, Y.; Li, C. Fabrication of magnetic imprinted sorbents prepared by Pickering emulsion polymerization for adsorption of erythromycin from aqueous solution. J. Environ. Chem. Eng. 2016, 4, 3570–3579. [Google Scholar] [CrossRef]
- Liang, W.; Hu, H.; Guo, P.; Ma, Y.; Li, P.; Zheng, W.; Zhang, M. Combining pickering emulsion polymerization with molecular imprinting to prepare polymer microspheres for selective solid-phase extraction of Malachite Green. Polymers 2017, 9, 344. [Google Scholar] [CrossRef]
- Gong, Y.; Niu, Y.; Gong, X.; Ma, M.; Ren, X.; Zhu, W.; Luo, R.; Gong, B. Preparation of 17β-estradiol-imprinted material by surface-initiated atom transfer radical polymerization and its application. J. Sep. Sci. 2015, 38, 1254–1261. [Google Scholar] [CrossRef]
- Zhou, T.; Shen, X.; Chaudhary, S.; Ye, L. Molecularly imprinted polymer beads prepared by pickering emulsion polymerization for steroid recognition. J. Appl. Polym. Sci. 2013, 131, 39606–39607. [Google Scholar] [CrossRef]
- Renkecz, T.; László, K.; Horváth, V. Molecularly imprinted microspheres prepared by precipitation polymerization at high monomer concentrations. Mol. Impr. 2014, 2, 1–17. [Google Scholar] [CrossRef]
- Jin, Y.; Jiang, M.; Shi, Y.; Lin, Y.; Peng, Y.; Dai, K.; Lu, B. Narrowly dispersed molecularly imprinted microspheres prepared by a modified precipitation polymerization method. Anal. Chim. Acta 2008, 612, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Horváth, V.; Lorántfy, B.; Tóth, B.; Bognár, J.; László, K.; Horvai, G. Preparation of terbutylazine imprinted polymer microspheres using viscous polymerization solvents. J. Sep. Sci. 2009, 32, 3347–3358. [Google Scholar] [CrossRef] [PubMed]
- Renkecz, T.; Lászlõ, K.; Horváth, V. In situ synthesis of molecularly imprinted nanoparticles in porous support membranes using high-viscosity polymerization solvents. J. Mol. Recognit. 2012, 25, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Benito-Peña, E.; Navarro-Villoslada, F.; Carrasco, S.; Jockusch, S.; Ottaviani, M.F.; Moreno-Bondi, M.C. Experimental Mixture Design as a Tool for the Synthesis of Antimicrobial Selective Molecularly Imprinted Monodisperse Microbeads. ACS Appl. Mater. Interfaces 2015, 7, 10966–10976. [Google Scholar] [CrossRef]
- Sambe, H.; Hoshina, K.; Moaddel, R.; Wainer, I.W.; Haginaka, J. Uniformly-sized, molecularly imprinted polymers for nicotine by precipitation polymerization. J. Chromatogr. A 2006, 1134, 88–94. [Google Scholar] [CrossRef]
- Zhou, T.; Jørgensen, L.; Mattebjerg, M.A.; Chronakis, I.S.; Ye, L. Molecularly imprinted polymer beads for nicotine recognition prepared by RAFT precipitation polymerization: A step forward towards multi-functionalities. RSC Adv. 2014, 4, 30292–30299. [Google Scholar] [CrossRef]
- Jiang, Y.; Tong, A.J. Synthesis of molecularly imprinted microspheres for recognition of trans-aconitic acid. J. Appl. Polym. Sci. 2004, 94, 542–547. [Google Scholar] [CrossRef]
- Li, P.; Rong, F.; Yuan, C. Morphologies and binding characteristics of molecularly imprinted polymers prepared by precipitation polymerization. Polym. Int. 2003, 52, 1799–1806. [Google Scholar] [CrossRef]
- Liu, Y.; Hoshina, K.; Haginaka, J. Monodispersed, molecularly imprinted polymers for cinchonidine by precipitation polymerization. Talanta 2010, 80, 1713–1718. [Google Scholar] [CrossRef]
- Ho, K.C.; Yeh, W.M.; Tung, T.S.; Liao, J.Y. Amperometric detection of morphine based on poly(3,4- ethylenedioxythiophene) immobilized molecularly imprinted polymer particles prepared by precipitation polymerization. Anal. Chim. Acta 2005, 542, 90–96. [Google Scholar] [CrossRef]
- Chow, A.L.J.; Bhawani, S.A. Synthesis and Characterization of Molecular Imprinting Polymer Microspheres of Cinnamic Acid: Extraction of Cinnamic Acid from Spiked Blood Plasma. Int. J. Polym. Sci. 2016, 2016, 2418915. [Google Scholar] [CrossRef]
- Roland, R.M.; Bhawani, S.A. Synthesis and Characterization of Molecular Imprinting Polymer Microspheres of Piperine: Extraction of Piperine from Spiked Urine. J. Anal. Methods Chem. 2016, 2016, 5671507. [Google Scholar] [CrossRef]
- Tan, L.; Li, Y.; Pan, X.; Marina, M.L.; Jiang, Z. Boronate affinity glycosyl molecularly imprinted polymer microspheres for the determination of teicoplanin using ultra-high performance liquid chromatography coupled with tandem mass spectrometry. J. Chromatogr. A 2019, 15, 460776. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Xie, L.F.; Dong, Q.; Liu, H.L.; Huang, Y.P.; Liu, Z.S. Synthesis of monodisperse molecularly imprinted microspheres with multi-recognition ability via precipitation polymerization for the selective extraction of cyromazine, melamine, triamterene and trimethoprim. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 1007, 127–131. [Google Scholar] [CrossRef]
- Dai, J.; Zou, Y.; Zhou, Z.; Dai, X.; Pan, J.; Yu, P.; Zou, T.; Yan, Y.; Li, C. Narrowly dispersed imprinted microspheres with hydrophilic polymer brushes for the selective removal of sulfamethazine. RSC Adv. 2014, 4, 1965–1973. [Google Scholar] [CrossRef]
- Song, R.; Hu, X.; Guan, P.; Li, J.; Qian, L.; Wang, Q. Synthesis of glutathione imprinted polymer particles via controlled living radical precipitation polymerization. Chin. J. Polym. Sci. 2015, 33, 404–415. [Google Scholar] [CrossRef]
- Gao, X.; Hu, X.; Guan, P.; Du, C.; Ding, S.; Zhang, X.; Li, B.; Wei, X.; Song, R. Synthesis of core-shell imprinting polymers with uniform thin imprinting layer: Via iniferter-induced radical polymerization for the selective recognition of thymopentin in aqueous solution. RSC Adv. 2016, 6, 110019–110031. [Google Scholar] [CrossRef]
- Su, L.; Guo, X.; Han, S. Preparation and evaluation of vanillin molecularly imprinted polymer microspheres by reversible addition-fragmentation chain transfer precipitation polymerization. Anal. Methods 2014, 6, 2512–2517. [Google Scholar] [CrossRef]
- Yoshimatsu, K.; Reimhult, K.; Krozer, A.; Mosbach, K.; Sode, K.; Ye, L. Uniform molecularly imprinted microspheres and nanoparticles prepared by precipitation polymerization: The control of particle size suitable for different analytical applications. Anal. Chim. Acta 2007, 584, 112–121. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Zhao, M.; Guo, X.; Zhang, H. Narrowly Dispersed Molecularly Imprinted Polymer Microspheres with Photo- and Thermo-Responsive Template Binding Properties in Pure Aqueous Media by RAFT Polymerization. Mol. Impr. 2012, 1, 3–16. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Zhao, M.; Guo, X.; Zhang, H. Efficient synthesis of narrowly dispersed molecularly imprinted polymer microspheres with multiple stimuli-responsive template binding properties in aqueous media. Chem. Commun. 2012, 48, 6217–6219. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Zheng, C.; Zhang, H. Improvement of surface hydrophilicity and biological sample-compatibility of molecularly imprinted polymer microspheres by facile surface modification with α-cyclodextrin. Eur. Polym. J. 2019, 115, 12–21. [Google Scholar] [CrossRef]
- Ma, Y.; Gao, J.; Zheng, C.; Zhang, H. Well-defined biological sample-compatible molecularly imprinted polymer microspheres by combining RAFT polymerization and thiol-epoxy coupling chemistry. J. Mater. Chem. B 2019, 7, 2474–2483. [Google Scholar] [CrossRef]
- Niu, H.; Yang, Y.; Zhang, H. Efficient one-pot synthesis of hydrophilic and fluorescent molecularly imprinted polymer nanoparticles for direct drug quantification in real biological samples. Biosens. Bioelectron. 2015, 74, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Li, Y.; Han, J.; Dong, X. Synthesis of tetracycline-imprinted polymer microspheres by reversible addition- fragmentation chain-transfer precipitation polymerization using polyethylene glycol as a coporogen. J. Sep. Sci. 2014, 37, 1118–1125. [Google Scholar] [CrossRef]
- Su, L.; Li, J.; Han, S. Controllable Preparation and Application of Quercetin Molecularly Imprinted Polymer. Russ. J. Appl. Chem. 2019, 92, 972–981. [Google Scholar] [CrossRef]
- Xiao, Y.; Xiao, R.; Tang, J.; Zhu, Q.; Li, X.; Xiong, Y.; Wu, X. Preparation and adsorption properties of molecularly imprinted polymer via RAFT precipitation polymerization for selective removal of aristolochic acid I. Talanta 2017, 162, 415–422. [Google Scholar] [CrossRef]
- Ayari, M.G.; Kadhirvel, P.; Favetta, P.; Plano, B.; Dejous, C.; Carbonnier, B.; Agrofoglio, L.A. Synthesis of imprinted hydrogel microbeads by inverse Pickering emulsion to controlled release of adenosine 5′-monophosphate. Mater. Sci. Eng. C 2019, 101, 254–263. [Google Scholar] [CrossRef]
- Geng, L.; Kou, X.; Lei, J.; Su, H.; Ma, G.; Su, Z. Preparation, characterization and adsorption performance of molecularly imprinted microspheres for erythromycin using suspension polymerization. J. Chem. Technol. Biotechnol. 2012, 87, 635–642. [Google Scholar] [CrossRef]
- Rachkov, A.; Minoura, N. Recognition of oxytocin and oxytocin-related peptides in aqueous media using a molecularly imprinted polymer synthesized by the epitope approach. J. Chromatogr. A 2000, 889, 111–118. [Google Scholar] [CrossRef]
- Bai, J.; Zhang, Y.; Chen, L.; Yan, H.; Zhang, C.; Liu, L.; Xu, X. Synthesis and characterization of paclitaxel-imprinted microparticles for controlled release of an anticancer drug. Mater. Sci. Eng. C 2018, 92, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Mao, C.; Xie, X.; Liu, X.; Cui, Z.; Yang, X.; Yeung, K.W.K.; Pan, H.; Chu, P.K.; Wu, S. The controlled drug release by pH-sensitive molecularly imprinted nanospheres for enhanced antibacterial activity. Mater. Sci. Eng. C 2017, 77, 84–91. [Google Scholar] [CrossRef]
- Parisi, O.I.; Ruffo, M.; Malivindi, R.; Vattimo, A.F.; Pezzi, V.; Puoci, F. Molecularly imprinted polymers (MIPs) as theranostic systems for sunitinib controlled release and self-monitoring in cancer therapy. Pharmaceutics 2020, 12, 41. [Google Scholar] [CrossRef] [PubMed]
- Parisi, O.I.; Morelli, C.; Puoci, F.; Saturnino, C.; Caruso, A.; Sisci, D.; Trombino, G.E.; Picci, N.; Sinicropi, M.S. Magnetic molecularly imprinted polymers (MMIPs) for carbazole derivative release in targeted cancer therapy. J. Mater. Chem. B 2014, 2, 6619–6625. [Google Scholar] [CrossRef]
- Kupai, J.; Rojik, E.; Huszthy, P.; Szekely, G. Role of chirality and macroring in imprinted polymers with enantiodiscriminative power. ACS Appl. Mater. Interfaces 2015, 7, 9516–9525. [Google Scholar] [CrossRef]
- Yang, J.; Li, Y.; Wang, J.; Sun, X.; Cao, R.; Sun, H.; Huang, C.; Chen, J. Molecularly imprinted polymer microspheres prepared by Pickering emulsion polymerization for selective solid-phase extraction of eight bisphenols from human urine samples. Anal. Chim. Acta 2015, 872, 35–45. [Google Scholar] [CrossRef]
- Jiang, M.; Shi, Y.; Zhang, R.L.; Shi, C.H.; Peng, Y.; Huang, Z.; Lu, B. Selective molecularly imprinted stationary phases for Bisphenol A analysis prepared by modified precipitation polymerization. J. Sep. Sci. 2009, 32, 3265–3273. [Google Scholar] [CrossRef]
- Wang, Z.; Qiu, T.; Guo, L.; Ye, J.; He, L.; Li, X. Polymerization induced shaping of Pickering emulsion droplets: From simple hollow microspheres to molecularly imprinted multicore microrattles. Chem. Eng. J. 2018, 332, 409–418. [Google Scholar] [CrossRef]
- Yan, S.; Fang, Y.; Yap, W.; Gao, Z. Characterization and Quality Assessment of Binding Properties of the Monocrotophos Molecularly Imprinted Microspheres Prepared by Precipitation Polymerization in Toluene. Polym. Eng. Sci. 2007, 47, 1302–1308. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Liu, X.X.; Li, R.H. Preparation and characterization of hydrophilic molecularly imprinted microspheres for difenoconazole. Chem. Pap. 2018, 72, 1277–1286. [Google Scholar] [CrossRef]
- Liu, X.; Wang, Y.; Li, L.; Li, R. Synthesis and characterization of azoxystrobin hydrophilic molecularly imprinted microspheres. J. Macromol. Sci. Part. A Pure Appl. Chem. 2019, 56, 907–917. [Google Scholar] [CrossRef]
- Yao, W.; Fang, Y.; Li, G.; Gao, Z.; Cheng, Y. Adsorption of carbaryl using molecularly imprinted microspheres prepared by precipitation polymerization. Polym. Adv. Technol. 2008, 19, 812–816. [Google Scholar] [CrossRef]
- Lai, J.P.; Yang, M.L.; Niessner, R.; Knopp, D. Molecularly imprinted microspheres and nanospheres for di(2-ethylhexyl) phthalate prepared by precipitation polymerization. Anal. Bioanal. Chem. 2007, 389, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Xu, Z.; Yuan, Q.; Tang, Y.; Zuo, X.; Lai, J. Recognition characteristics of molecularly imprinted microspheres for triazine herbicides using hydrogen-bond array strategy and their analytical applications for corn and soil samples. J. Chromatogr. A 2011, 1218, 1340–1346. [Google Scholar] [CrossRef]
- Gao, L.; Li, X.; Zhang, Q.; Dai, J.; Wei, X.; Song, Z.; Yan, Y.; Li, C. Molecularly imprinted polymer microspheres for optical measurement of ultra trace nonfluorescent cyhalothrin in honey. Food Chem. 2014, 156, 1–6. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, H.; Xu, Y.; Zhang, F.; Zhao, J.; Wang, L.; Hou, J.; Ding, H.; Li, Y.; Jin, H.; et al. Determination triazine pesticides in cereal samples based on single-hole hollow molecularly imprinted microspheres. J. Chromatogr. A 2015, 1376, 26–34. [Google Scholar] [CrossRef]
- Lu, X.; Yang, Y.; Zeng, Y.; Li, L.; Wu, X. Rapid and reliable determination of p-nitroaniline in wastewater by molecularly imprinted fluorescent polymeric ionic liquid microspheres. Biosens. Bioelectron. 2018, 99, 47–55. [Google Scholar] [CrossRef]
- Dai, C.M.; Geissen, S.U.; Zhang, Y.L.; Zhang, Y.J.; Zhou, X.F. Selective removal of diclofenac from contaminated water using molecularly imprinted polymer microspheres. Environ. Pollut. 2011, 159, 1660–1666. [Google Scholar] [CrossRef]
- Fang, L.; Chen, S.; Guo, X.; Zhang, Y.; Zhang, H. Azobenzene-containing molecularly imprinted polymer microspheres with photo- and thermoresponsive template binding properties in pure aqueous media by atom transfer radical polymerization. Langmuir 2012, 28, 9767–9777. [Google Scholar] [CrossRef]
- Li, J.; Zu, B.; Zhang, Y.; Guo, X.; Zhang, H. One-Pot Synthesis of Surface-Functionalized Molecularly Imprinted Polymer Microspheres by Iniferter-Induced ‘“Living”’ Radical Precipitation Polymerization. J. Polym. Sci. Part. A Polym. Chem. 2010, 48, 3217–3228. [Google Scholar] [CrossRef]
- Pan, G.; Zhang, Y.; Guo, X.; Li, C.; Zhang, H. An efficient approach to obtaining water-compatible and stimuli-responsive molecularly imprinted polymers by the facile surface-grafting of functional polymer brushes via RAFT polymerization. Biosens. Bioelectron. 2010, 26, 976–982. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Ma, Y.; Zhang, Y.; Guo, X.; Li, C.; Zhang, H. Controlled synthesis of water-compatible molecularly imprinted polymer microspheres with ultrathin hydrophilic polymer shells via surface-initiated reversible addition-fragmentation chain transfer polymerization. Soft Matter 2011, 7, 8428–8439. [Google Scholar] [CrossRef]
- Ma, Y.; Pan, G.; Zhang, Y.; Guo, X.; Zhang, H. Narrowly dispersed hydrophilic molecularly imprinted polymer nanoparticles for efficient molecular recognition in real aqueous samples including river water, milk, and bovine serum. Angew. Chem. Int. Ed. 2013, 125, 1511–1514. [Google Scholar] [CrossRef]
- Yang, L.; Fan, Z.; Wang, T.; Cai, W.; Yang, M.; Jiang, P.; Zhang, M.; Dong, X. Preparation of a Pyrazosulfuron-Ethyl Imprinted Polymer with Hydrophilic External Layers by Reversible Addition-Fragmentation Chain Transfer Precipitation and Grafting Polymerization. Anal. Lett. 2011, 44, 2617–2632. [Google Scholar] [CrossRef]
- Yang, M.; Zhang, Y.; Lin, S.; Yang, X.; Fan, Z.; Yang, L.; Dong, X. Preparation of a bifunctional pyrazosulfuron-ethyl imprinted polymer with hydrophilic external layers by reversible addition-fragmentation chain transfer polymerization and its application in the sulfonylurea residue analysis. Talanta 2013, 114, 143–151. [Google Scholar] [CrossRef]
- Zhao-Hui, Z.; Yu-Mei, L.; Ying-Ju, L.; Shou-Zhuo, Y. Microspheres Sensor Based on Molecularly Imprinted Polymer Synthesized by Precipitation Polymerization. Chin. J. Chem. 2003, 21, 550–555. [Google Scholar] [CrossRef]
- Carrasco, S.; Benito-Peña, E.; Walt, D.R.; Moreno-Bondi, M.C. Fiber-optic array using molecularly imprinted microspheres for antibiotic analysis. Chem. Sci. 2015, 6, 3139–3147. [Google Scholar] [CrossRef]
- Shi, Y.; Peng, D.D.; Shi, C.H.; Zhang, X.; Xie, Y.T.; Lu, B. Selective determination of trace 17β-estradiol in dairy and meat samples by molecularly imprinted solid-phase extraction and HPLC. Food Chem. 2011, 126, 1916–1925. [Google Scholar] [CrossRef]
- Lai, J.P.; Lu, X.Y.; Lu, C.Y.; Ju, H.F.; He, X.W. Preparation and evaluation of molecularly imprinted polymeric microspheres by aqueous suspension polymerization for use as a high-performance liquid chromatography stationary phase. Anal. Chim. Acta 2001, 442, 105–111. [Google Scholar] [CrossRef]
- Kitabatake, T.; Tabo, H.; Matsunaga, H.; Haginaka, J. Preparation of monodisperse curcumin-imprinted polymer by precipitation polymerization and its application for the extraction of curcuminoids from Curcuma longa L. Anal. Bioanal. Chem. 2013, 405, 6555–6561. [Google Scholar] [CrossRef]
- Du, J.J.; Gao, R.X.; Yu, H.; Li, X.J.; Mu, H. Selective extraction of dimethoate from cucumber samples by use of molecularly imprinted microspheres. J. Pharm. Anal. 2015, 5, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Pardeshi, S.; Dhodapkar, R.; Kumar, A. Molecularly imprinted microspheres and nanoparticles prepared using precipitation polymerisation method for selective extraction of gallic acid from Emblica officinalis. Food Chem. 2014, 146, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Valero-Navarro, Á.; Gómez-Romero, M.; Fernández-Sánchez, J.F.; Cormack, P.A.G.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Synthesis of caffeic acid molecularly imprinted polymer microspheres and high-performance liquid chromatography evaluation of their sorption properties. J. Chromatogr. A 2011, 1218, 7289–7296. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Masumoto, S.; Matsunaga, H.; Haginaka, J. Molecularly imprinted polymer for glutathione by modified precipitation polymerization and its application to determination of glutathione in supplements. J. Pharm. Biomed. Anal. 2017, 144, 230–235. [Google Scholar] [CrossRef]
- Xu, S.; Li, J.; Chen, L. Molecularly imprinted polymers by reversible addition-fragmentation chain transfer precipitation polymerization for preconcentration of atrazine in food matrices. Talanta 2011, 85, 282–289. [Google Scholar] [CrossRef]
- Yin, R.; Chen, L.; Ma, L. Extraction of matrine from soil with matrix solid-phase dispersion by molecularly imprinted polymers derived from lignin-based Pickering emulsions. J. Sep. Sci. 2019, 42, 3563–3570. [Google Scholar] [CrossRef]
- Yang, H.H.; Zhou, W.H.; Guo, X.C.; Chen, F.R.; Zhao, H.Q.; Lin, L.M.; Wang, X.R. Molecularly imprinted polymer as SPE sorbent for selective extraction of melamine in dairy products. Talanta 2009, 80, 821–825. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Yang, H.; Wang, J.; Deng, A. Preparation and characterization of molecularly imprinted microspheres for selective extraction of trace melamine from milk samples. Microchim. Acta 2011, 174, 191. [Google Scholar] [CrossRef]
- Shi, X.; Wu, A.; Zheng, S.; Li, R.; Zhang, D. Molecularly imprinted polymer microspheres for solid-phase extraction of chloramphenicol residues in foods. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 850, 24–30. [Google Scholar] [CrossRef]
- Li, K.; Stöver, H.D.H. Synthesis of monodisperse poly(divinylbenzene) microspheres. J. Polym. Sci. Part. A Polym. Chem. 1993, 31, 3257–3263. [Google Scholar] [CrossRef]
- Xia, Q.; Yun, Y.; Li, Q.; Huang, Z.; Liang, Z. Preparation and characterization of monodisperse molecularly imprinted polymer microspheres by precipitation polymerization for kaempferol. Des. Monomers Polym. 2017, 20, 201–209. [Google Scholar] [CrossRef]
- Matyjaszewski, K. Comparison and classification of controlled/living radical polymerizations. ACS Symp. Ser. 2000, 768, 2–26. [Google Scholar] [CrossRef]
- Braunecker, W.A.; Matyjaszewski, K. Controlled/living radical polymerization: Features, developments, and perspectives. Prog. Polym. Sci. 2007, 32, 93–146. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Xia, J. Atom transfer radical polymerization. Chem. Rev. 2001, 101, 2921–2990. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhang, Y.; Guo, X.; Zhang, H. Narrow or Monodisperse, Highly cross-linked, and living polymer micrompheres by ATRP polymerization. Macromolecules 2011, 44, 5893–5904. [Google Scholar] [CrossRef]
- Jiang, J.; Zhang, Y.; Guo, X.; Zhang, H. Ambient temperature synthesis of narrow or monodisperse, highly cross-linked, and “living” polymer microspheres by atom transfer radical precipitation polymerization. RSC Adv. 2012, 2, 5651–5662. [Google Scholar] [CrossRef]
- Puoci, F.; Iemma, F.; Picci, N. Stimuli-Responsive Molecularly Imprinted Polymers for Drug Delivery: A Review. Curr. Drug Deliv. 2008, 5, 85–96. [Google Scholar] [CrossRef]
- Jakubowski, W.; Matyjaszewski, K. Activators regenerated by electron transfer for atom-transfer radical polymerization of (meth)acrylates and related block copolymers. Angew. Chem. Int. Ed. 2006, 45, 4482–4486. [Google Scholar] [CrossRef]
- Otsu, T.; Yoshida, M. Role of initiator-transfer agent-terminator (iniferter) in radical polymerizations: Polymer design by organic disulfides as iniferters. Makromol. Chem. Rapid Commun 1982, 3, 127–132. [Google Scholar] [CrossRef]
- Otsu, T. Iniferter concept and living radical polymerization. J. Polym. Sci. Part. A Polym. Chem. 2000, 38, 2121–2136. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, M.; Zhang, H.; Ma, F.; Zhang, Y.; Guo, X. Efficient synthesis of monodisperse, highly crosslinked, and “living” functional polymer microspheres by the ambient temperature iniferter-induced “living” radical precipitation polymerization. J. Polym. Sci. Part. A Polym. Chem. 2013, 51, 1983–1998. [Google Scholar] [CrossRef]
- MacKová, H.; Horák, D. Effects of the reaction parameters on the properties of thermosensitive poly(N-isopropylacrylamide) microspheres prepared by precipitation and dispersion polymerization. J. Polym. Sci. Part. A Polym. Chem. 2006, 44, 968–982. [Google Scholar] [CrossRef]
- Moad, G.; Chiefari, J.; Chong, Y.K.; Krstina, J.; Mayadunne, R.T.; Postma, A.; Rizzardo, E.; Thang, S.H. Living free radical polymerization with reversible addition-fragmentation chain transfer (the life of RAFT). Polym. Int. 2000, 49, 993–1001. [Google Scholar] [CrossRef]
- Moad, G.; Rizzardo, E.; Thang, S.H. Radical addition-fragmentation chemistry in polymer synthesis. Polymer 2008, 49, 1079–1131. [Google Scholar] [CrossRef]
- Dinsmore, A.D.; Hsu, M.F.; Nikolaides, M.G.; Marquez, M.; Bausch, A.R.; Weitz, D.A. Colloidosomes: Selectively Permeable Capsules Composed of Colloidal Particles. Science 2002, 298, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Thompson, K.L.; Armes, S.P.; Howse, J.R.; Ebbens, S.; Ahmad, I.; Zaidi, J.H.; York, D.W.; Burdis, J.A. Covalently cross-linked colloidosomes. Macromolecules 2010, 43, 10466–10474. [Google Scholar] [CrossRef]
- Shen, X.; Ye, L. Interfacial molecular imprinting in nanoparticle-stabilized emulsions. Macromolecules 2011, 44, 5631–5637. [Google Scholar] [CrossRef]
- Binks, B.P.; Lumsdon, S.O. Transitional phase inversion of solid-stabilized emulsions using particle mixtures. Langmuir 2000, 16, 3748–3756. [Google Scholar] [CrossRef]
- Kim, K.; Kim, D. High-performance liquid chromatography separation characteristics of molecular-imprinted poly(methacrylic acid) microparticles prepared by suspension polymerization. J. Appl. Polym. Sci. 2005, 96, 200–212. [Google Scholar] [CrossRef]
- Liu, P.; Liu, L.; Zhang, L.; Jiang, N.; Liu, Z.; Wang, Y. Synthesis and characterization of molecularly imprinted polymers for recognition of ciprofloxacin. Front. Chem. China 2008, 3, 378–383. [Google Scholar] [CrossRef]
- Barros, L.A.; Custodio, R.; Rath, S. Design of a new molecularly imprinted polymer selective for hydrochlorothiazide based on theoretical predictions using Gibbs free energy. J. Braz. Chem. Soc. 2016, 27, 2300–2311. [Google Scholar] [CrossRef]
- Wei, S.; Molinelli, A.; Mizaikoff, B. Molecularly imprinted micro and nanospheres for the selective recognition of 17β-estradiol. Biosens. Bioelectron. 2006, 21, 1943–1951. [Google Scholar] [CrossRef]
- Kupai, J.; Razali, M.; Buyuktiryaki, S.; Kecili, R.; Szekely, G. Long-term stability and reusability of molecularly imprinted polymers. Polym. Chem. 2017, 8, 666–673. [Google Scholar] [CrossRef]
- Keçili, R.; Yilmaz, E.; Ersöz, A.; Say, R. Imprinted Materials: From Green Chemistry to Sustainable Engineering. In Sustainable Nanoscale Engineering: From Materials Design to Chemical Processing; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 317–350. ISBN 9780128146828. [Google Scholar] [CrossRef]
- Madikizela, L.M.; Tavengwa, N.T.; Tutu, H.; Chimuka, L. Green aspects in molecular imprinting technology: From design to environmental applications. Trends Environ. Anal. Chem. 2018, 17, 14–22. [Google Scholar] [CrossRef]
- Viveiros, R.; Rebocho, S.; Casimiro, T. Green strategies for molecularly imprinted polymer development. Polymers 2018, 10, 306. [Google Scholar] [CrossRef] [PubMed]





| Application | Template | Monomer and Crosslinker | Polymerisation Technique | References |
|---|---|---|---|---|
| Selective Drug and Metabolite Recognition | 17β-estradiol | MAA, EGDMA, TRIM | PP | [40,41] |
| 4-VP, AM, TRIM | ATRPP | [49] | ||
| MAA, EGDMA | PE | [50] | ||
| Theophylline | MAA, EGDMA, TRIM | PP | [40,41] | |
| MAA, DVB | [6] | |||
| Caffeine | MAA, EGDMA, TRIM | PP | [40] | |
| Naproxen, Diclofenac, Toltrazuril | 4-VP, MAA, HEMA, MAAm, EGDMA, TRIM, DVB | PP | [51] | |
| Estradiol (E2) | 4-VP, EGDMA | PP | [52] | |
| Kaempferol | 4-VP, EGDMA | PP | [5] | |
| Terbutylazine | MAA, EGDMA | PP | [53,54] | |
| Enrofloxacine | MAA, HEMA, DVB, EGDMA, TRIM | PP | [55] | |
| Nicotine | MAA, TFMAA, DVB | PP | [56] | |
| MAA, EDGMA | RAFTPP | [57] | ||
| Trans-aconitic acid | MAA, TRIM | PP | [58] | |
| l-2-chloromandelic acid | AM, BDDA | PP | [59] | |
| Cinchonidine | MAA, HEMA, DVB | PP | [60] | |
| Morphine | MAA, TRIM | PP | [61] | |
| Cinnamic Acid | AA, DVB | PP | [62] | |
| Piperine | AA, EGDMA | PP | [63] | |
| Mannose-tryptophan | MMA, DVB | PP | [64] | |
| 2,4-diamino-6-methyl-1,3,5-triazine, cyromazine, trimethoprim | MAA, DVB | PP | [65] | |
| Sulfamethazine | 4-VP, HEMA, EGDMA | ATRPP | [66] | |
| Glutathione | 4-VP, EGDMA | ILRPP | [67] | |
| Thymopentin | p-CMS, VI, EGDMA | ILRPP | [68] | |
| Vanillin | MAA, EDGMA | RAFTPP | [69] | |
| Propranolol | MAA, DVB | PP | [70] | |
| MPABA, NIPAAm, EGDMA | RAFTPP | [71] | ||
| 4-VP, NIPAAm, MPABA, DMAEMA, EGDMA | [72] | |||
| MAA, EGDMA | [73] | |||
| MAA, HEMA, EGDMA | [74] | |||
| Tetracycline | MAA, HEMA, AnHEMA, EGDMA | RAFTPP | [75] | |
| MAA, EGDMA | [76] | |||
| Quercetin | 2-VP, EGDMA | RAFTPP | [77] | |
| Aristolochic Acid I | AA, EGDMA | RAFTPP | [78] | |
| Isopropylaminopropanediol | MAA, EGDMA, AIBN | PE | [79] | |
| Erythromycin | MAA, EGDMA | SP | [80] | |
| YPLG | MAA, EGDMA | SP | [81] | |
| Controlled Drug Release | Paclitaxel | MAA, EGDMA | RAFTPP | [82] |
| Adhenosine 5′-monophosphate | DMAEM, NIPAAm, MBAM | PE | [79] | |
| Vancomycin | HEMA, DEAEMA | PP | [83] | |
| Sunitib | MAA | PP | [84] | |
| 1,4-dimethyl-6-hydroxy-9H-carbazole (CAB1) | MAA, EGDMA | PP | [85] | |
| 1-(1-naphthyl)ethylamine | Macrocyclic | SP | [86] | |
| Environmental Contaminants | Tebuconazole | 4-VP, EGDMA | PP | [52] |
| Bisphenol A | 4-VP, EGDMA | PP | [52] | |
| ATRPP | [1] | |||
| PE | [87] | |||
| 4-VP, MAA, EGDMA, TRIM | PP | [88] | ||
| 4-VP, DVB | PE | [89] | ||
| Monocrotophos | MAA, EGDMA | PP | [90] | |
| Difenoconazole | HPMA, EGDMA | PP | [91] | |
| Azoxystrobin | HPMA, EGDMA | PP | [92] | |
| Carbaryl | MAA, EGDMA | PP | [93] | |
| Di(2-ethylhexyl)phthalate | MAA, EGDMA, TRIM | PP | [94] | |
| Simetryne | ABA, DVB | PP | [95] | |
| Cyhalothrin | AM, EGDMA | PP | [96] | |
| Polystyrene | MAA, DVB | PP | [97] | |
| p-nitroaniline | IL, EGDMA | PP | [98] | |
| Diclofenac | 2-VP, EGDMA | PP | [99] | |
| 2,4-Dichlorophenoxy- acetic acid | MAzoPy, EGDMA | ATRPP | [100] | |
| 4-VP, NIPAAm, EGDMA | ILRPP | [101] | ||
| 4-VP, EGDMA | RAFTPP | [5,8] | ||
| 4-VP, NIPAAm, EGDMA | [102] | |||
| 4-VP, HEMA, EGDMA | [103,104] | |||
| Pyrazosulfuron- ethyl | MAA, 4-VP, AA, EGDMA, DVB | RAFTPP | [105] | |
| MAA, EGDMA | [106] | |||
| Sensor | Dipyridamole | MAA, EGDMA | PP | [107] |
| Enrofloxacine | MAA, HEMA, DVB, EGDMA | PP | [108] | |
| MISPE | 17β-estradiol | 4-VP, EGDMA | PP | [109] |
| 4-aminopyridine | MAA, EGDMA | SP | [110] | |
| Extraction From Natural Ingredients or Food | Curcumin | 4-VP, MAA, MAM, DVB | PP | [111] |
| Dimethoate | MAA, MMA, AM, EGDMA | PP | [112] | |
| Gallic Acid | AA, EGDMA | PP | [113] | |
| Caffeic Acid | 4-VP, DVB | PP | [114] | |
| Glutathione | MAA, DVB | PP | [115] | |
| Atrazine | MAA, EGDMA | RAFTPP | [116] | |
| Matrine | MAA, EGDMA | PE | [117] | |
| Melamine | MAA, EGDMA | SP | [118,119] | |
| Chloramphenicol | DEAEM, EGDMA | SP | [120] |
| Polymerisation Technique | Advantages | Diadvantages |
|---|---|---|
| Precipitation Polymerisation |
|
|
| Controlled/‘living’ radical precipitation polymerisation (CRPP) 2 |
| |
| Atom Transfer Radical Precipitation Polymerisation (ATRPP) |
|
|
| Iniferter-induced ‘Living’ Radical Precipitation Polymerisation (ILRPP) |
|
|
| Reversible Addition-Fragmentation Chain Transfer Precipitation Polymerisation (RAFTPP) |
|
|
| Pickering Emulsion |
|
|
| Suspension Polymerisation |
|
|
| Template | Monomer | PP | ATRPP | RAFTPP | PE | ||||
|---|---|---|---|---|---|---|---|---|---|
| Binding Affinity | IF | Binding Affinity | IF | Binding Affinity | IF | Binding Affinity | IF | ||
| 17β-estradiol | MAA | More than 50% analyte [40,41] | 2.3 [40] 4.0 [41] | - | - | - | - | 30–60% analyte [50] | 3.0 [50] |
| 4-VP | 0.75 mg/g [109] | 4.55 [109] | 180.65 mg/g [49] | 6.67–7.38 [49] | - | - | - | - | |
| Nicotine | MAA | Not mention in the article [56] | 10.5 [56] | - | - | Not mention in the article | 3.33 [57] | - | - |
| Bisphenol A | 4-VP | Not mention in the article | 3.91 [52] 4.83 [88] | Not mention in the article | 10 [1] | - | - | 1.32 mg/g [87] | 6.5 [87] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orowitz, T.E.; Ana Sombo, P.P.A.A.; Rahayu, D.; Hasanah, A.N. Microsphere Polymers in Molecular Imprinting: Current and Future Perspectives. Molecules 2020, 25, 3256. https://doi.org/10.3390/molecules25143256
Orowitz TE, Ana Sombo PPAA, Rahayu D, Hasanah AN. Microsphere Polymers in Molecular Imprinting: Current and Future Perspectives. Molecules. 2020; 25(14):3256. https://doi.org/10.3390/molecules25143256
Chicago/Turabian StyleOrowitz, Tirza Ecclesia, Patria Pari Agnes Ago Ana Sombo, Driyanti Rahayu, and Aliya Nur Hasanah. 2020. "Microsphere Polymers in Molecular Imprinting: Current and Future Perspectives" Molecules 25, no. 14: 3256. https://doi.org/10.3390/molecules25143256
APA StyleOrowitz, T. E., Ana Sombo, P. P. A. A., Rahayu, D., & Hasanah, A. N. (2020). Microsphere Polymers in Molecular Imprinting: Current and Future Perspectives. Molecules, 25(14), 3256. https://doi.org/10.3390/molecules25143256







