Enhancement of an In Vivo Anti-Inflammatory Activity of Oleanolic Acid through Glycosylation Occurring Naturally in Stauntonia hexaphylla
Abstract
:1. Introduction
2. Results
2.1. Structure Identification of Isolated Compounds
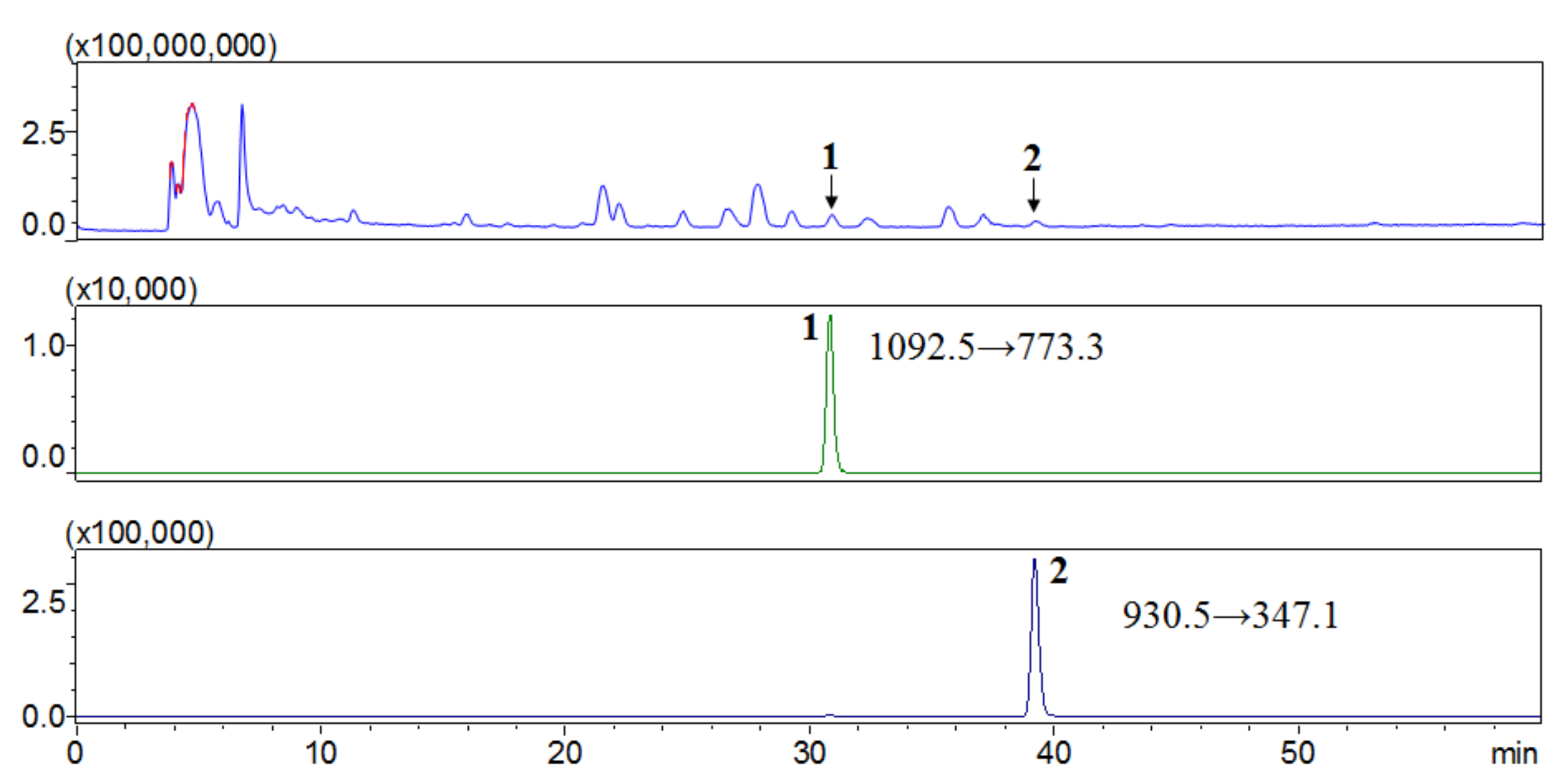
2.2. Quantitative Analysis of Compounds 1, and 2 in the EtOH Extract of S. hexaphylla Leaves
2.3. Immunoregulatory Activities of the Isolated Compounds
2.3.1. Cytotoxic Properties of Compounds 1 and 2
2.3.2. Inhibition of the Activation of the RAW 264.7 Cell Line by the Oleanolic Acid Saponins In Vitro
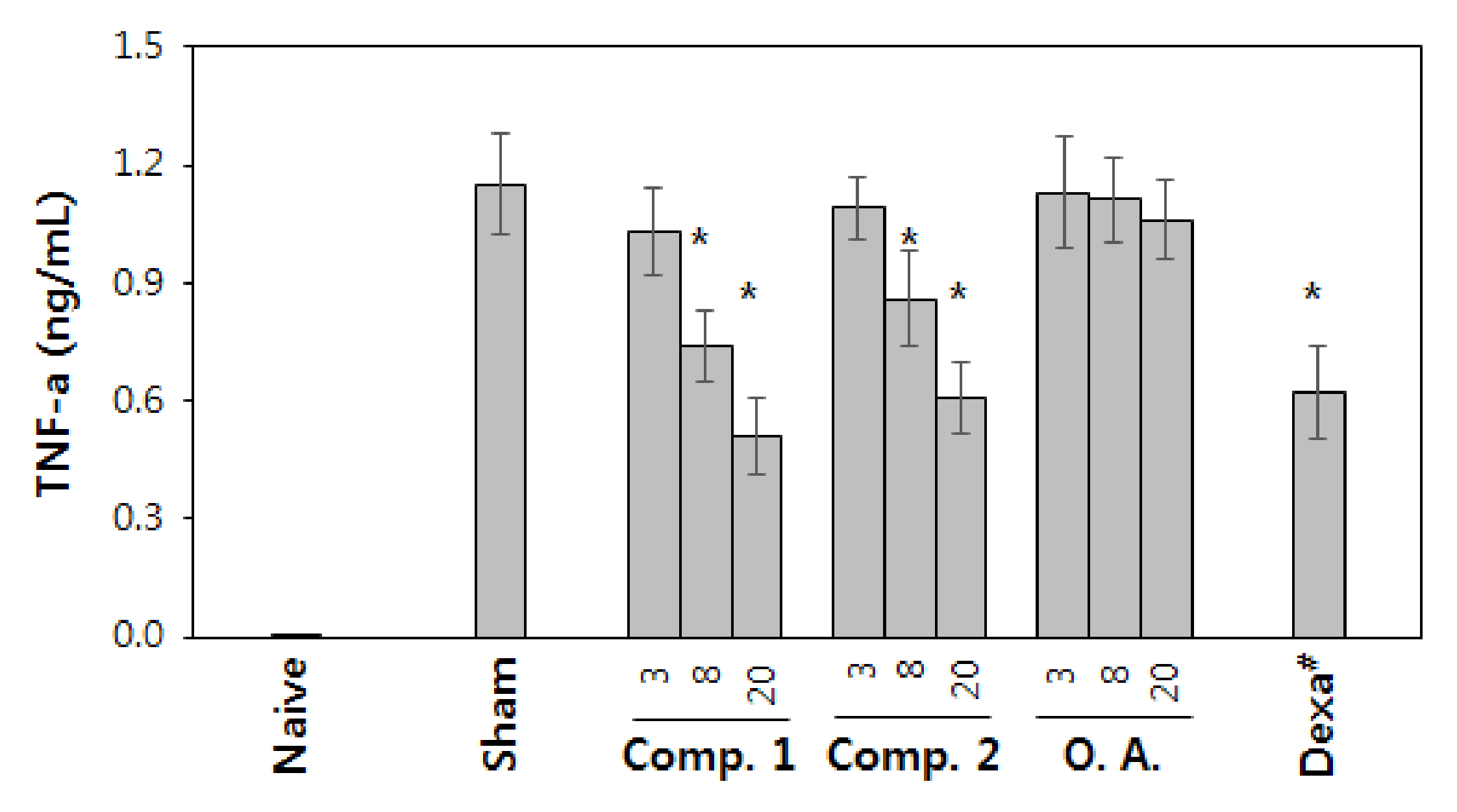
2.3.3. Inhibition of the Activation of Macrophage by Orally Administered Oleanolic Acid Saponins In Vivo
3. Discussion
4. Materials and Methods
4.1. Chemical and Reagents
4.2. Plant Material
4.3. Biodirected Isolation of the Active Compounds
4.4. Acid Hydrolysis and Sugar Identification
4.5. LC-MS/MS Analysis of the Isolated Compounds
4.6. Biological Studies
4.6.1. Cytotoxicity Assay Using MTT
4.6.2. Nitric Oxide Production Assay with the RAW 264.7 Macrophage Cell Line
4.6.3. LPS-Induced Sepsis and Measurement of TNF-α in the Blood
4.6.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Delves, P.; Martin, S.; Burton, D. Roitt’s Essential Immunology Wiley, 12th ed.; Wiley-Blackwell: London, UK, 2011. [Google Scholar]
- Wang, C.Y.; Lee, S.; Jang, H.-J.; Su, X.D.; Wang, H.-S.; Kim, Y.H.; Yang, S.Y. Inhibition potential of phenolic constituents from the aerial parts of Tetrastigma hemsleyanum against soluble epoxide hydrolase and nitric oxide synthase. J. Enzym. Inhib. Med. Chem. 2019, 34, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Tang, Y.; Hua, S. Immunological approaches towards cancer and inflammation: A cross talk. Front. Immunol. 2018, 9, 563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Todoric, J.; Antonucci, L.; Karin, M. Targeting inflammation in cancer prevention and therapy. Cancer Prev. Res. 2016, 9, 895–905. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Kim, H.; Choi, H.; Jo, A.; Kang, H.; Yun, H.; Choi, C.; Im, S. Anti-inflammatory effects of a Stauntonia hexaphylla fruit extract in lipopolysaccharide-activated RAW-264.7 macrophages and rats by carrageenan-induced hind paw swelling. Nutrients 2018, 10, 110. [Google Scholar]
- Wang, H.-B.; Yu, D.-Q.; Liang, X.-T.; Watanabe, N.; Tamai, M.; Omura, S. Yemuoside YM7, YM11, YM13, and YM14: Four nortriterpenoid saponins from Stauntonia chinensis. Planta Med. 1989, 55, 303–306. [Google Scholar] [CrossRef]
- Ikuta, A.; Itokawa, H. The triterpenes from Stauntonia hexaphylla callus tissues and their biosynthetic significance. J. Nat. Prod. 1989, 52, 623–628. [Google Scholar] [CrossRef]
- Ikuta, A.; Morikawa, A.; Kubota, K. A saponin from callus tissue of Stauntonia hexaphylla. Phytochemistry 1991, 30, 2425–2427. [Google Scholar] [CrossRef]
- Wei, Y.; Ma, C.-M.; Chen, D.-Y.; Hattori, M. Anti-HIV-1 protease triterpenoids from Stauntonia obovatifoliola Hayata subsp. intermedia. Phytochemistry 2008, 69, 1875–1879. [Google Scholar] [CrossRef]
- Liu, D.; Wang, D.; Yang, W.; Meng, D. Potential anti-gout constituents as xanthine oxidase inhibitor from the fruits of Stauntonia brachyanthera. Bioorg. Med. Chem. 2017, 25, 3562–3566. [Google Scholar] [CrossRef]
- Liu, X.-L.; Li, S.; Meng, D.-L. Anti-gout nor-oleanane triterpenoids from the leaves of Stauntonia brachyanthera. Bioorg. Med. Chem. Lett. 2016, 26, 2874–2879. [Google Scholar] [CrossRef]
- Hwang, S.H.; Kwon, S.H.; Kim, S.B.; Lim, S.S. Inhibitory activities of Stauntonia hexaphylla leaf constituents on rat lens aldose reductase and formation of advanced glycation end products and antioxidant. Biomed. Res. Int. 2017, 2, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheon, Y.-H.; Baek, J.M.; Park, S.-H.; Ahn, S.-J.; Lee, M.S.; Oh, J.; Kim, J.-Y. Stauntonia hexaphylla (Lardizabalaceae) leaf methanol extract inhibits osteoclastogenesis and bone resorption activity via proteasome-mediated degradation of c-Fos protein and suppression of NFATc1 expression. BMC Complem. Altern. Med. 2015, 15, 1–9. [Google Scholar]
- Vinh, L.B.; Jang, H.-J.; Phong, N.V.; Cho, K.W.; Park, S.S.; Kang, J.S.; Kim, Y.H.; Yang, S.Y. Isolation, structural elucidation, and insights into the anti-inflammatory effects of triterpene saponins from the leaves of Stauntonia hexaphylla. Bioorg. Med. Chem. Lett. 2019, 29, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Vinh, L.B.; Jo, S.J.; Nguyen Viet, P.; Gao, D.; Cho, K.W.; Koh, E.-J.; Park, S.S.; Kim, Y.H.; Yang, S.Y. The chemical constituents of ethanolic extract from Stauntonia hexaphylla leaves and their anti-inflammatory effects. Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Su, X.-D.; Jang, H.-J.; Wang, C.-Y.; Lee, S.W.; Rho, M.-C.; Kim, Y.H.; Yang, S.Y. Anti-inflammatory potential of saponins from Aster tataricus via NF-κB/MAPK activation. J. Nat. Prod. 2019, 82, 1139–1148. [Google Scholar] [CrossRef]
- Quang, T.H.; Ngan, N.T.; Minh, C.V.; Kiem, P.V.; Nhiem, N.X.; Tai, B.H.; Thao, N.P.; Tung, N.H.; Song, S.B.; Kim, Y.H. Anti-inflammatory triterpenoid saponins from the stem bark of Kalopanax pictus. J. Nat. Prod. 2011, 74, 1908–1915. [Google Scholar] [CrossRef]
- Vinh, L.B.; Jang, H.-J.; Phong, N.V.; Dan, G.; Cho, K.W.; Kim, Y.H.; Yang, S.Y. Bioactive triterpene glycosides from the fruit of Stauntonia hexaphylla and insights into the molecular mechanism of its inflammatory effects. Bioorg. Med. Chem. Lett. 2019, 29, 2085–2089. [Google Scholar] [CrossRef]
- Kawai, H.; Kuroyanagi, M.; Umehara, K.; Ueno, A.; Satake, M. Studies on the saponins of Lonicera japonica Thunb. Chem. Pharm. Bull. 1988, 36, 4769–4775. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Ma, N.; He, C.; Hu, Y.; Li, P.; Chen, M.; Su, H.; Wan, J.-B. Qualitative and quantitative analysis of the saponins in Panax notoginseng leaves using ultra-performance liquid chromatography coupled with time-of-flight tandem mass spectrometry and high performance liquid chromatography coupled with UV detector. J. Ginseng. Res. 2018, 42, 149–157. [Google Scholar] [CrossRef]
- Vinh, L.B.; Lee, Y.; Han, Y.K.; Kang, J.S.; Park, J.U.; Kim, Y.R.; Yang, S.Y.; Kim, Y.H. Two new dammarane-type triterpene saponins from Korean red ginseng and their anti-inflammatory effects. Bioorg. Med. Chem. Lett. 2017, 27, 5149–5153. [Google Scholar] [CrossRef]
- Yang, C.-R.; Zhang, Y.; Jacob, M.R.; Khan, S.I.; Zhang, Y.-J.; Li, X.-C. Antifungal activity of C-27 steroidal saponins. Antimicrob. Agents Chemother. 2006, 50, 1710–1714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandal, P.; Babu, S.S.; Mandal, N. Antimicrobial activity of saponins from Acacia auriculiformis. Fitoterapia 2005, 76, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Vinh, L.B.; Nguyet, N.T.M.; Yang, S.Y.; Kim, J.H.; Thanh, N.V.; Cuong, N.X.; Nam, N.H.; Minh, C.V.; Hwang, I.; Kim, Y.H. Cytotoxic triterpene saponins from the mangrove Aegiceras corniculatum. Nat. Prod. Res. 2017, 33, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Pham, H.T.T.; Hoang, M.C.; Ha, T.K.Q.; Dang, L.H.; Tran, V.O.; Nguyen, T.B.T.; Lee, C.H.; Oh, W.K. Discrimination of different geographic varieties of Gymnema sylvestre, an anti-sweet plant used for the treatment of type 2 diabetes. Phytochemistry 2018, 150, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Vinh, L.B.; Park, J.U.; Nguyet, N.T.M.; Yang, S.Y.; Kim, Y.R.; Kim, Y.H. Ginsenosides from Korean red ginseng modulate T cell function via the regulation of NF-AT-mediated IL-2 production. Food Sci. Biotechnol. 2019, 28, 237–242. [Google Scholar] [CrossRef]
- Lee, C.H.; Kim, J.-H. A review on the medicinal potentials of ginseng and ginsenosides on cardiovascular diseases. J. Ginseng Res. 2014, 38, 161–166. [Google Scholar] [CrossRef] [Green Version]
- Cheok, C.Y.; Salman, H.A.K.; Sulaiman, R. Extraction and quantification of saponins: A review. Food Res. Int. 2014, 59, 16–40. [Google Scholar] [CrossRef]
- Moses, T.; Papadopoulou, K.K.; Osbourn, A. Metabolic and functional diversity of saponins, biosynthetic intermediates and semi-synthetic derivatives. Crit. Rev. Biochem. Mol. Biol. 2014, 49, 439–462. [Google Scholar] [CrossRef] [Green Version]
- Hadziabdic, J.; Elezovic, A.; Rahic, O.; Mujezin, I.; Vranic, E. Stability of suspensions: Theoretical and practical considerations before compounding. Int. J. Pharm. Compd. 2015, 19, 78–85. [Google Scholar]
- Oka, T.; Kalesnikoff, J.; Starkl, P.; Tsai, M.; Galli, S.J. Evidence questioning cromolyn’s effectiveness and selectivity as a ‘mast cell stabilizer’ in mice. Lab. Investig. 2012, 92, 1472–1482. [Google Scholar] [CrossRef]
Sample Availability: Sample of the active compounds (1−2) available from the authors. |




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vinh, L.B.; Nguyet, N.T.M.; Ye, L.; Dan, G.; Phong, N.V.; Anh, H.L.T.; Kim, Y.H.; Kang, J.S.; Yang, S.Y.; Hwang, I. Enhancement of an In Vivo Anti-Inflammatory Activity of Oleanolic Acid through Glycosylation Occurring Naturally in Stauntonia hexaphylla. Molecules 2020, 25, 3699. https://doi.org/10.3390/molecules25163699
Vinh LB, Nguyet NTM, Ye L, Dan G, Phong NV, Anh HLT, Kim YH, Kang JS, Yang SY, Hwang I. Enhancement of an In Vivo Anti-Inflammatory Activity of Oleanolic Acid through Glycosylation Occurring Naturally in Stauntonia hexaphylla. Molecules. 2020; 25(16):3699. https://doi.org/10.3390/molecules25163699
Chicago/Turabian StyleVinh, Le Ba, Nguyen Thi Minh Nguyet, Liu Ye, Gao Dan, Nguyen Viet Phong, Hoang Le Tuan Anh, Young Ho Kim, Jong Seong Kang, Seo Young Yang, and Inkyu Hwang. 2020. "Enhancement of an In Vivo Anti-Inflammatory Activity of Oleanolic Acid through Glycosylation Occurring Naturally in Stauntonia hexaphylla" Molecules 25, no. 16: 3699. https://doi.org/10.3390/molecules25163699







