1. Introduction
Vegetable oils are valuable natural source of triacylglycerols (TAG), which are composed of fatty acids (FAs) and glycerol. The chain length of FA can vary between C:8 and C:24, wherein the most prevalent are those with C:16 and C:18 [
1]. The characteristics of edible oils and fats are subject to several processing steps, mainly during extraction [
2].
The chemical composition also has an influence on oil quality, which shall be fully determined herein, considering the involved industrial process, together with its resulting oxidative stability [
3]. Besides, aspects of color, values of acidity, peroxides, and others shall be analyzed, complementing and proving its composition as an oil to be considered adequate for human consumption [
4].
There is growing evidence that FAs play a crucial role in human nutrition [
5], including the therapeutic and prophylactic prevention of diseases [
6]. Therefore, it is necessary to investigate the physicochemical properties of edible oils, to know their characterization, and the behavior of their compounds, so that their pathway of action in the organism is understood [
7].
FAs have distinct effects on cell stress, and evidence indicates that excess consumption of saturated fatty acid (SFA) has detrimental effects on health, favoring the inflammatory process [
8], compared to monounsaturated fatty acids (MUFAs) and polyunsaturated (PUFAs) [
9]. The most abundant FAs in the organism, considering both the adipose tissue reservoirs and the dietary fat intake, are oleic (MUFA) and palmitic (SFA) free fatty acids. [
10].
The oleic and palmitic FAs are predominant components of olive oil [
11], commonly utilized in its extra virgin form as the primary source of fat in the Mediterranean diet, for its high content of MUFAs and polyphenols [
12,
13]. Their ingestion acts as an effective method to modulate factors related to oxidative stress and inflammation through the biomarkers CRP and IL-6 [
14], besides improving the lipidic profile reducing hyperlipidemia in the vascular system [
15].
Among the main oils rich in MUFAs is soybean oil. This oil is highly produced and heavily used oil in cooking, processed foods, margarines, and is the oil of choice in many restaurants [
16]. In a study by Deol et al. (2015), mice fed a high soybean oil diet showed obesogenic and diabetogenic effects when compared to other lipid sources [
17].
Although not being one of the main traditional oil crops, such as soybean, canola, sunflower and [
18], another vegetable oil with a high content of MUFAs is
C. brasiliense, with 54.28% oleic FA [
19,
20]. It is from a typical Brazilian fruit known as pequi, with a yellow-orangish colour and peculiar odor, reported as being rich in antioxidant compounds, such as phenolics, carotenoids, tocopherols, phytosterols [
21], lycopene, and beta-carotene [
22], besides showing anti-inflammatory and cardioprotective effect [
23].
Hildebrand et al. (2017) highlight the importance of new studies that evaluate foods with protective effects on human health, especially those that have anti-inflammatory action [
24]. Therefore, the main objective of our study was to analyze the physical and chemical characteristics of the pulp oil of
Caryocar brasiliense Cambess., to evaluate the effects of supplementing this oil and other lipid sources, such as soybean oil and olive oil in a model animal.
2. Results and Discussion
The indices of acidity, peroxides, saponification, and iodine are some of the main parameters that indicate oil quality [
25] and together are also correlated to product stability [
9]. The acidity index indicates the presence of free fatty acids that, in significant quantities, turn the oil more liable to rancification [
25]. In our study, the acidity content found in the
C. brasiliense oil (
Table 1) is within the maximum limit allowed for crude oils (<4 mg KOH/g) [
4]. Similarly to our study, values within the recommended acidity standard were obtained (0.68 mg KOH/g
−1) in another species,
Caryocar coriaceum Wittm. [
26], and also in
Acrocomia aculeata oil (0.97 mg KOH/g
−1), composed of a profile of FAs similar to
C. brasiliense oil [
27].
The peroxide index is an indicator of the initial stages of rancification and a measure of primary products of the lipidic oxidation [
25]. The peroxide index presented a high value (13.63 mEq O
2 kg
−1) that can be explained by the oil unsaturation degree, which may indicate the start of the oxidation process. However, the index is following the maximum standard for crude oils (<15 mEq O
2 kg
−1), demonstrating adequacy for consumption [
4]. Differing from the index of
C. brasiliense, another study showed a result under 4.40 mEq O
2 kg
−1, found in
C. coriaceum oil [
28].
The iodine index is a measure of the unsaturation of fats and oils and consequentially the susceptibility to oxidation [
29]. The obtained iodine index (76.7 I
2/100
−1 g) is close to values found in analyses of 10 oils of different cultivars of olive (
Olea europaea L.), between 80 and 89 I
2/100
−1 g [
30]. Its counterpart, the oil of the fruit of
Byrsonima cydoniifolia A. Juss., also native to South America, has an iodine index with significantly higher unsaturation degree (120.84 I
2/100
−1 g) [
31], compared with
C. brasiliense oil. This indicates higher stability of
C. brasiliense compared with the others cited; the the higher the unsaturation, the lower its oxidative stability [
25].
The saponification index is related to its molecular weight or chain length of triglycerides that compose the oil [
32]. The reported result presented a low value (136.5 mg KOH g
−1), near the recommended level for olive oil (184–196 mg KOH/g) and palm oil (190–209 mg KOH/g) [
4,
33], with characteristics similar to
C. brasiliense. This indicates that the oil does not contain many fatty acids with low molecular weight.
Another parameter for oil characterization is it coloration, one of the initial factors pointing to possible lipidic oxidation and consequent degradation, as color alteration is caused by the degradation of essential FAs and others compounds [
2]. The fruit of
C. brasiliense is considered a source of carotenoids [
23]; the liposoluble pigments responsible for the orange and red coloration [
34].
The analyzed
C. brasiliense oil evidenced the presence of carotenoids, due to the positive values of a* and b* (
Table 1), showing that most pigments are yellowish, followed by red, which is correlated with the total content of carotenoids detected in the samples. The high value of C* indicates a high-intensity color of the oil, thus considered dark. High contents of carotenoids are found in a limited number of edible oils, when compared with
Mauritia flexuosa oil and palm oil, considered rich in this compound, with concentrations of 1722.87 mg kg
−1 [
35] and 1385 mg kg
−1, respectively [
36].
C. brasiliense oil stands out, with a content of 2.39 µg/g of total carotenoids.
In
Figure 1 it is possible to observe the analysis of UV–Vis absorption and in
Figure 2 the fluorescence of
C. brasiliense oil at the concentration of 5 × 10
−3 g mL
−1 (a) and the result of fluorescence in pure oil (b). We verified the presence of natural antioxidants, such as tocopherols, and tocotrienols, named vitamin E [
37] by the absorption band approximately in 313 nm (
Figure 1) and the emission band in 327 nm, excited in 290 nm (
Figure 2a).
With regard to the fluorescence analysis directly in undiluted vegetable oil (2b), we observed the presence of an emission band centered at 530 nm when excited at 470 nm. That fluorescence band can be attributed to the carotenoids [
38]. The presence of chloropylls is usually emitted in the range of 650 to 750 nm [
7,
37], and was slightly ranked in the analysis of undiluted oil (
Figure 2b). That datum is correlated with the value of total carotenoids found (
Table 1), demonstrating the low concentration of carotenoids and higher presence of tocopherols
C. brasiliense oil, with α-tocopherol standing out.
The thermal degradation (
Figure 3) of
C. brasiliense oil was observed mainly in the range of 195 °C to 457 °C, later stabilizing its mass. The peak occurred at 388 °C, with a loss of total mass of 99.8%. The residues were 0.1%, below possible analytical errors. The loss of initial mass at ~195 °C can be attributed to moisture loss of the oil and the volatilization of compounds such as aldehydes and short-chain fatty acids, a common factor in vegetable oils, as the oil composition influences the total mass loss [
39].
The chemical composition of
C. brasiliense oil includes antioxidant and oxidizable compounds that influence its oxidative stability [
40]. The role of α-tocopherol is not yet totally defined, but it is known that its presence improves the oxidative stability in vegetable oils [
3]. Moreover, it is considered one of the best phenolic antioxidants as it rapidly reacts with the alkyl peroxyl radical, forming more stable adducts and protecting lipids from peroxidation [
41].
There is no minimum period of induction recommended for good quality edible oils; nevertheless, we consider that the
C. brasiliense oil reached a long induction period of 8.6 h and consequent excellent oxidative stability (
Figure 4), mainly because of the high presence of SFA and MFA and other minor components. This means
C. brasiliense oil has a suitable shelf life. Similarly, the
C. brasiliense pulp and nut oils evaluated by Torres et al. [
21] differed in time of oxidative induction between 7.33 and 15.91 h. Other oils considered very stable present induction values of 12 h (soybean), 9.96 h (maize), and 8.63 h (canola) [
3].
Moreover, we can consider that the prevention mechanism of oxidation through antioxidants occurred in the
C. brasiliense pulp oil as its content of unsaturated FAs is higher than saturated (
Table 2), which make the oil more susceptible to degradation [
42,
43]. The characterization of the profile of the FAs of
C. brasiliense oil revealed a high content of monounsaturated fatty acids (MUFA), especially the oleic FA (56.61%), followed by the saturated FA palmitic (37.78%) and polyunsaturated linoleic acid (3.9%). The values we found are similar to those reported by Nascimento-Silva et al. (2019) [
23]: 55.87%, 35.17%, and 1.53%, respectively, except linoleic FA that has a higher percentage in their study. Similar content (1.36%) was also reported by Roll et al. (2018) [
19].
The oxidative stability, the parameters of quality within recommendations for edible vegetable oils, and the prevalence of unsaturated FAs are the factors that determine whether an oil is adequate for human consumption. Moreover, oils with a high level of MUFAs and PUFAs can improve the levels of serum lipids [
44]. After 90 days of supplementation, we observed better levels of total cholesterol, LDL-c, and non-HDL-c (
Table 3) in the groups supplemented with
C. brasiliense oil as a lipidic source. The presence of PUFAs can explain the reduced plasmatic levels of total cholesterol as they act as a protective factor in the homeostasis of cholesterol due to the high number of unsaturations and thus less phospholipid–cholesterol interaction [
45].
Another study states that diets rich in MUFAs can reduce the levels of total cholesterol total and LDL-c [
46]. This could be one of the factors contributing to the low levels of LDL-c and non-HDL-c we found, despite high levels of SFA associated with the high plasmatic levels of LDL-c [
47]. The group supplemented with olive oil in higher dosage (2000 mg/kg) also showed values significantly reduced, possibly due to the higher concentration of MUFAs present in this oil, as well as in C.
brasiliense oil, which are mainly composed of oleic acid, followed by palmitic and linoleic acid [
48]. We point out that the supplementation with
C. brasiliense oil (2000 mg/kg) reached a better response in the parameters that are commonly associated with atherosclerosis (LDL-c and non-HDL-c) [
49] when compared with groups CG and OO1, supplemented with soybean and olive oils, respectively.
Regard to body weight gain and adiposity index (
Table 4), we noticed that animals kept a pattern of weight not differing statistically (
p ≤ 0.05). Nevertheless, the group receiving higher doses of MUFA (OO +
C. brasiliense oil) presented the lowest mean weight. Similar results we observed in adipose tissue weight (
Table 4), the group supplemented with olive oil plus
C. brasiliense oil had lower weight of the principal visceral fats, including epididymal adipose tissue; in mice, this is one of the main deposit areas of visceral fat [
50]. Other relevant sites, such as mesenteric and retroperitoneal fat [
51], also diminished compared with the control group.
This effect can be associated with the presence of tocopherols and mainly of tocotrienols that are present in both
C. brasiliense oil, as demonstrated in our study, and olive oil [
52]. Another work pointed out that the ingestion of gamma-tocotrienol (60 mg/kg/day) was capable of reducing the fat mass induced by different doses of glucocorticoids. Uto-Kondo et al. (2009) [
53] evaluated the effect of a palm oil fraction rich in tocotrienol on the differentiation of adipocyte into 3T3-L1 cells and found that this antioxidant suppressed the differentiation of pre-adipocytes into adipocytes, potentially reducing weight gain.
Among the possible alterations in the liver (
Table 5), we did not identify statistical difference regarding the presence of hepatic steatosis (
p = 0.17) and microvesicular steatosis (
p = 0.45). However, a build-up of free FAs occurred in the liver, except group OO + CO that showed only microvesicular steatosis, demonstrating that the synergic effect of the mix of olive oil with
C. brasiliense oil may have played a slight protective role, due to a higher concentration of antioxidants. These are known for beneficial action in biological systems and protection against oxidative damages [
54] as the oxidative stress is one of the causes of hepatocellular lesions [
51].
We detected significant differences between groups regarding the presence of Mallory Hyaline, and in post-test it was significantly more present in animals of all groups compared with GC, as well as group OO + CO compared with OO1. Apoptosis was significantly more prevalent in group CO1 compared with CG and OO1, and no presence was recorded in OO1. Palmeira et al. (2015) [
55] reported that the administration of
C. brasiliense oil at 400 mg/kg in mice induced with diethylnitrosamine 10 μg/g reduced the development of preneoplastic lesions and hepatic adenoma. Another study on
C. brasiliense nut oil found that it can attenuate the biochemical markers of hepatic lesion and inflammation [
21].
Among the data obtained in the histological analyses of the pancreas (
Table 6), we did not find an association between the presence of Langerhans Islets (
p = 0.93) or inflammatory cells in all groups (
p = 0.38). None of the samples analyzed in our study showed alterations in pancreatic acini.
The amount of consumed SFA influences the accumulation of free FAs in the liver and activation of inflammatory markers [
56]. For the inflammatory response to start it needs proinflammatory cytokines and chemokines such as TNF-α, IL-6, and MCP-1 [
57].
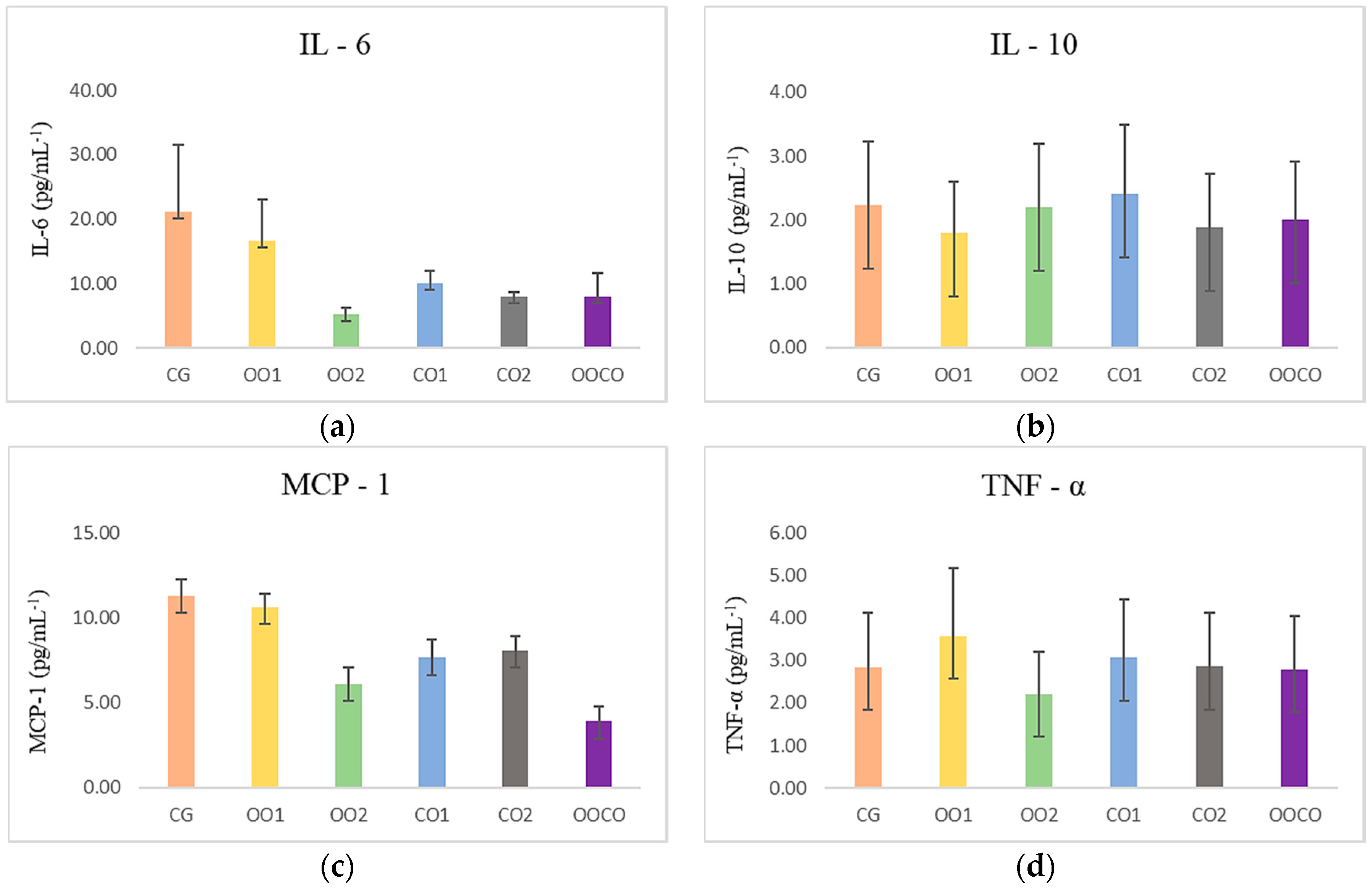
Figure 5 shows the levels of circulating inflammatory cytokines IL-6 (
p = 0.944), MCP-1 6 (
p = 0.640), TNF-α (
p = 0.834), and anti-inflammatory IL-10 (
p = 0.709), without significant difference between treatments. Recent studies reported that phytochemicals present in plants could inhibit the inflammation, reducing the production of macrophages, proinflammatory factors and also blocking inflammatory pathways that liberate cytokines [
58,
59]. In a study on rats utilizing supplementation with
C. brasiliense nut oil at a concentration of 6 mL/kg, the results suggest that it attenuates the acute inflammatory response when induced by CCl
4, modulating the circulating levels of leptin, IL-6, LTB4, and LTB-5 positively [
21].
The tested dosages of CO did not demonstrate a protective effect on those parameters when compared with the group supplemented with soybean oil (CG) and both doses of olive oil (OO1; OO2). The cytokine IL-10 is necessary to inhibit the synthesis of proinflammatory cytokines [
56]. It can exert anti-inflammatory effects through signal transducer pathway of the Janus kinase (JAK) of activation 3 (JAK-STAT3), binding IL-10 to the receptor in the targeted cell membrane—tyrosine kinase 2—leading to the activation of the signal transducer and activator of transcription 3 (STAT3). In our study, IL-10 was more active in the group supplemented with olive oil at the lower dose (1000 mg/kg). Nevertheless, more research is necessary to elucidate the molecular action mechanisms of oleic acid [
21], which is the major component in CO and OO, and the phytochemicals present in
C. brasiliense oil, such as phenolics, carotenoids, tocopherols, and phytosterols, that constitute possible mediators of these effects.