Synthesis and Cytotoxic Activity of New Thiazolopyrimidine Sugar Hydrazones and Their Derived Acyclic Nucleoside Analogues
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Cytotoxic Activity
3. Experimental
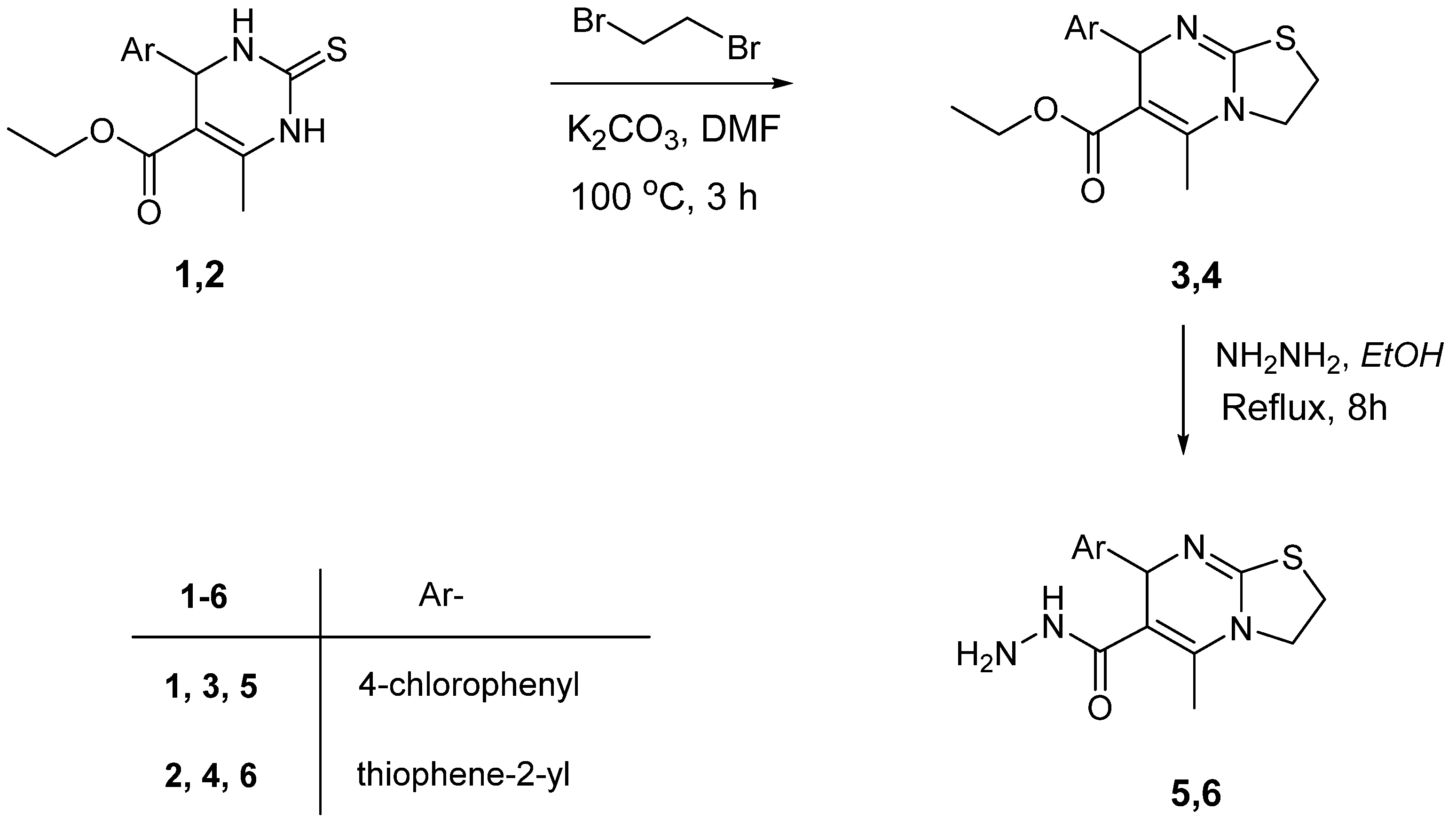
3.1. Synthesis
General Procedures
3.2. Ethyl 7-(Aryl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carboxylate (3, 4)
3.2.1. Ethyl 7-(4-Chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carboxylate (3)
3.2.2. Ethyl 5-Methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carboxylate (4)
3.3. 5-Methyl-7-(aryl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carbohydrazide (5, 6)
3.3.1. 7-(4-Chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carbohydrazide (5)
3.3.2. 5-Methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carbohydrazide (6)
3.4. Sugar-5-(aryl)-7-methyl-2,3-dihydro-5H-thiazolo[3,2-a]pyrimidine-6-carbohydrazone (7–10)
3.4.1. d-Galactose 7-(4-chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carbohydrazone (7)
3.4.2. d-Xylose 7-(4-chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-Carbohydrazone (8)
3.4.3. d-Galactose 5-methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carbohydrazone (9)
3.4.4. d-Xylose 5-methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-Carbohydrazone (10)
3.5. General Procedure for the Preparation of Compounds (11–14)
3.5.1. Penta-O-acetyl-d-galactopentitolyl-7-(4-chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidine-6-carbohydrazone (11)
3.5.2. Tetra-O-acetyl-d-xylotetritolyl-7-(4-chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]- pyrimidine-6-carbohydrazone (12)
3.5.3. Penta-O-acetyl-d-galactopentitolyl-5-methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]- pyrimidine-6-carbohydrazone (13)
3.5.4. Tetra-O-acetyl-d-xylotetritolyl-5-methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]- pyrimidine-6-carbohydrazone (14)
3.6. General Procedure for the Preparation of the Oxadiazoline Substituted Sugar Derivatives (15–18)
3.6.1. 1-(5-(7-(4-Chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidin-6-yl)-2-(penta-O-acetyl-d-galactopentitolyl)-1,3,4-oxadiazol-3(2H)-yl)ethan-1-one (15)
3.6.2. 1-(5-(7-(4-Chlorophenyl)-5-methyl-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidin-6-yl)-2-(tetra-O-acetyl-d-xylotetritolyl)-1,3,4-oxadiazol-3(2H)-yl)ethan-1-one (16)
3.6.3. 1-(5-(5-Methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidin-6-yl)-2-(penta-O-acetyl-d-galactopentitolyl)-1,3,4-oxadiazol-3(2H)-yl)ethan-1-one (17)
3.6.4. 1-(5-(5-Methyl-7-(thiophen-2-yl)-2,3-dihydro-7H-thiazolo[3,2-a]pyrimidin-6-yl)-2-(tetra-O-acetyl-d-xylotetritolyl)-1,3,4-oxadiazol-3(2H)-yl)ethan-1-one (18)
3.7. Materials of the Cell Lines Assay
3.7.1. Cell Culture, Maintenance, and Sub-Culture
3.7.2. Cell Proliferation by MTT Assay
3.7.3. IC50 Measurement
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Martino, J.K.; Boger, D.L. Glycinamide ribonucleotide transformylase (GAR TFase) as a target for cancer therapy. Drug Future 2008, 33, 969–979. [Google Scholar] [CrossRef]
- Curran, W.J. New chemotherapeutic agents: Update of major chemoradiation trials in solid tumors. Oncology 2002, 63, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.M.; Amr, A.E.G.; Alsharari, M.A.; Al-Qalawi, H.R.M.; Germoush, M.O.; Al-Omar, M.A. Anticancer Activities of Some New Synthesized Thiazolo[3,2-a]Pyrido[4,3-d]Pyrimidine Derivatives. Am. J. Biochem. Biotechnol. 2011, 7, 43–54. [Google Scholar] [CrossRef]
- Hammam, A.G.; El-Salam, O.I.A.; Mohamed, A.M.; Hafez, N.A. Novel fluoro substituted benzo[o]pyranwith anti-lung cancer activity. Ind. J. Chem. 2005, 44B, 1887–1893. [Google Scholar]
- Flefel, E.E.; Salama, M.A.; El-Shahat, M.; El-Hashash, M.A.; El-Farargy, A.F. A novel synthesis of some new pyrimidine and thiazolopyrimidine derivatives for anticancer evaluation. Phosphorus Sulfur Silicon Relat. Elem. 2007, 182, 1739–1756. [Google Scholar] [CrossRef]
- Cai, D.; Zhang, Z.-H.; Chen, Y.; Yan, X.-J.; Zou, L.-J.; Wang, Y.-X.; Liu, X.-Q. Synthesis, Antibacterial and Antitubercular Activities of Some 5H-Thiazolo[3,2-a]pyrimidin-5-ones and Sulfonic Acid Derivative. Molecules 2015, 20, 16419–16434. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, J.; Adam, G.; Kolczewski, S.; Mutel, V.; Woltering, T. Structure-activity relationships of substituted 5H-thiazolo[3,2-a]pyrimidines as group 2 metabotropic glutamate receptor antagonists. Bioorg. Med. Chem. Lett. 1999, 9, 1573–1576. [Google Scholar] [CrossRef]
- Al-Omary, F.A.; Hassan, G.S.; El-Messery, S.M.; ElSubbagh, H.I. Substituted thiazoles V. Synthesis and antitumor activity of novel thiazolo[2,3-b]quinazoline and pyrido[4,3-d] thiazolo[3,2-a] pyrimidine analogues. Eur. J. Med. Chem. 2012, 47, 65–72. [Google Scholar] [CrossRef]
- Fatima, S.; Sharma, A.; Saxena, R.; Tripathi, R.; Shukla, S.K.; Pandey, S.K.; Tripathi, R.; Tripathi, R.P. One pot efficient diversity oriented synthesis of polyfunctional styryl thiazolopyrimidines and their bio-evaluation as antimalarial and anti-HIV agents. Eur. J. Med. Chem. 2012, 55, 195–204. [Google Scholar] [CrossRef]
- Yıldırım, A.B.; Mutlu, E.; Yıldırım, M. Cytotoxic Effects of Thiazolo[3,2-C]Pyrimidines Against Mcf-7 And Hepg2/C3a Carcinoma Cell Lines. Hacet. J. Biol. Chem. 2018, 46, 237–246. [Google Scholar] [CrossRef]
- Amr, A.-E.-G.; Maigali, S.S.; Abdulla, M.M. Synthesis, and analgesic and antiparkinsonian activities of thiopyrimidine, pyrane, pyrazoline, and thiazolopyrimidine derivatives from 2-chloro-6-ethoxy-4-acetylpyridine. Mon. Chem. 2008, 139, 1409–1415. [Google Scholar] [CrossRef]
- Branstetter, B.J.; Breitenbucher, J.G.; Lebsack, A.D.; Xiao, W. Thiazolopyrimidine Modulators of TRPV1. U.S. Patent WO 005303, 10 January 2008. [Google Scholar]
- Said, M.; Abouzid, K.; Mouneer, A.; Ahmedy, A.; Osman, A.-M. Synthesis and biological evaluation of new thiazolopyrimidines. Arch. Pharm. Res. 2004, 27, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Ackova, D.G.; Kotur-Stevuljevic, J.; Mishra, C.B.; Luthra, P.M.; Saso, L. Antioxidant Properties of Synthesized Bicyclic Thiazolopyrimidine Derivatives as Possible Therapeutic Agents. Appl. Sci. 2019, 9, 113. [Google Scholar] [CrossRef] [Green Version]
- Youssef, M.M.; Amin, M.A. Microwave Assisted Synthesis of Some New Thiazolopyrimidine, Thiazolodipyrimidine and Thiazolopyrimidothiazolopyrimidine Derivatives with Potential Antioxidant and Antimicrobial Activity. Molecules 2012, 17, 9652–9667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linder, W.; Brandes, W. Pesticidal Thiazolopyrimidine Derivatives. U.S. Patent 4,996,208, 26 February 1991. [Google Scholar]
- Duval, R.; Kolb, S.; Braud, E.; Genest, D.; Garbay, C. Rapid discovery of triazolobenzylidenethiazolopyrimidines (TBTP) as CDC25 phosphatase inhibitors by parallel click chemistry and in situ screening. J. Comb. Chem. 2009, 11, 947–950. [Google Scholar] [CrossRef] [PubMed]
- Kolb, S.; Mondésert, O.; Goddard, M.L.; Jullien, D.; Villoutreix, B.O.; Ducommun, B.; Garbay, C.; Braud, E. Development of novel thiazolopyrimidines as CDC25B phosphatase inhibitors. ChemMedChem 2009, 4, 633–648. [Google Scholar] [CrossRef] [PubMed]
- Mahgoub, M.Y.; Elmaghraby, A.M.; Harb, A.A.; da Silva, J.L.F.; Justino, G.C.; Marques, M.M. Synthesis, Crystal Structure, and Biological Evaluation of Fused Thiazolo[3,2-a]Pyrimidines as New Acetylcholinesterase Inhibitors. Molecules 2019, 24, 2306. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.-J.; Yang, L.; Jin, Z.; Huang, E.F.; Wan, D.C.C.; Lin, H.-Q.; Hu, C. Design, synthesis, and biological evaluation of 7H-thiazolo[3,2-b]- 1,2,4-triazin-7-one derivatives as novel acetylcholinesterase inhibitors. Arkivoc 2009, 10, 333–348. [Google Scholar]
- Rashad, A.E.; Shamroukh, A.H.; Abdel-Megeid, R.E.; El-Sayed, W.A. Synthesis, reactions and antimicrobial evaluation of some polycondensedthieno-pyrimidine derivatives. Synth. Commun. 2010, 40, 1149–1160. [Google Scholar] [CrossRef]
- El-Emary, T.I.; Abdel-Mohsen, S.A. Synthesis and antimicrobial activity of some new 1,3-diphenylpyrazoles bearing pyrimidine, Pyrimidinethione, thiazolopyrimidine, triazolopyrimidine, thio- and alkylthiotriazolopyrimidinone moieties at the 4-position. Phosphorus Sulfur Silicon Relat. Elem. 2006, 181, 2459–2474. [Google Scholar] [CrossRef]
- Maddila, S.; Damu, G.L.V.; Oseghe, E.O.; Abafe, O.A.; Venakata, R.C.; Lavanya, P. Synthesis and biological studies of novel biphenyl-3,5-dihydro-2H-thiazolo-pyrimidines derivatives. J. Korean Chem. Soc. 2012, 56, 334–340. [Google Scholar] [CrossRef]
- Khalilullah, H.; Ahsan, M.J.; Hedaitullah, M.; Khan, S.; Ahmad, B. 1,3,4-Oxadiazole: A Biologically Active Scaffold. Mini-Rev. Med. Chem. 2012, 12, 789–801. [Google Scholar] [CrossRef] [PubMed]
- Kassem, A.F.; Nassar, I.F.; Abdel-Aal, M.T.; Awad, H.M.; El-Sayed, W.A. Synthesis and Anticancer Activity of New ((Furan-2-yl)-1,3,4-thiadiazolyl)-1,3,4-oxadiazole Acyclic Sugar Derivatives. Chem. Pharm. Bull. 2019, 67, 888–895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nassar, I.F.; El kady, D.S.; Awad, H.M.; El-Sayed, W.A. Design, Synthesis, and Anticancer Activity of New Oxadiazolyl-Linked and Thiazolyl-Linked Benzimidazole Arylidines, Thioglycoside, and Acyclic Analogs. J. Heterocycl. Chem. 2019, 56, 1086–1100. [Google Scholar] [CrossRef]
- El-Sayed, W.A.; Khalaf, H.S.; Osman, D.A.A.; Abbas, H.S.; Ali, M.M. Synthesis and Anticancer Activity of New Pyrazolyl and Oxadiazolyl Glycosides Based on Theinopyrimidine Nucleus and Their Acyclic Analogs. Acta Polonea Pharm. Drug Res. 2017, 74, 1739–1751. [Google Scholar]
- Gamal El-Din, M.M.; El-Gamal, M.I.; Abdel-Maksoud, M.S.; Yoo, K.H.; Oh, C.H. Synthesis and in vitro antiproliferative activity of new 1,3,4-oxadiazole derivatives possessing sulfonamide moiety. Eur. J. Med. Chem. 2015, 90, 45–52. [Google Scholar] [CrossRef]
- El-Sadek, M.M.; Hassan, S.Y.; Abd El-Dayem, N.S.; Yacout, G.A. 5-(5-Aryl-1,3,4-oxadiazole-2-carbonyl)furan-3-carboxylate and New Cyclic C-Glycoside Analogues from Carbohydrate Precursors with MAO-B, Antimicrobial and Antifungal Activities. Molecules 2012, 17, 7010–7027. [Google Scholar] [CrossRef] [Green Version]
- Glomb, T.; Szymankiewicz, K.; Swiatek, P. Anti-Cancer Activity of Derivatives of 1,3,4-Oxadiazole. Molecules 2018, 23, 336. [Google Scholar] [CrossRef] [Green Version]
- El Sayed, W.A.; Abbas, H.-A.S.; Ewais, A.M. C-Furyl glycosides, III: Synthesis and antimicrobial evaluation of C-furyl glycosides bearing substituted 1,3,4-oxadiazoles and their sugar hydrazone derivatives. J. Heterocycl. Chem. 2011, 48, 1006–1013. [Google Scholar] [CrossRef]
- Kahl, D.; Hutchings, K.; Lisabeth, E.M.; Larsen, S.D. 5-Aryl-1,3,4-oxadiazol-2-ylthioalkanoic Acids: A Highly Potent New Class of Inhibitors of Rho/Myocardin-Related Transcription Factor (MRTF)/Serum Reponse Factor (SRF)-Mediated Gene Transcription as Potential Antifibrotic Agents for Scleroderma. J. Med. Chem. 2019, 62, 4350–4369. [Google Scholar] [CrossRef]
- El-Sayed, W.A.; El-Sofany, W.I.; Hussein, H.A.; Fathi, N.M. Synthesis and Anticancer Activity of New [(Indolyl)pyrazolyl]-1,3,4-oxadiazole Thioglycosides and Acyclic Nucleoside Analogs. Nucleosides Nucleotides Nucleic Acids 2017, 36, 474–495. [Google Scholar] [CrossRef] [PubMed]
- El Ashry, E.S.H.; El Kilany, Y. Acyclonucleosides: Part 1. Seco-Nucleosides. Adv. Heterocycl. Chem. 1996, 67, 391–438. [Google Scholar]
- El Ashry, E.S.H.; El Kilany, Y. Acyclonucleosides: Part 3. tri-, tetra-, and pentaseco-Nucleosides. Adv. Heterocycl. Chem. 1998, 69, 129–215. [Google Scholar]
- Kassem, A.F.; Abbas, E.M.H.; El-Kady, D.S.; Awad, H.M.; El-Sayed, W.A. Synthesis, Docking Studies and Anticancer Activity of New Tetrazolyl- and (Triazolyl)thiazole Glycosides and Acyclic Analogs. Mini-Rev. Med. Chem. 2019, 19, 933–948. [Google Scholar] [CrossRef]
- Abdel Rahman, A.A.H.; Nassar, I.F.; Shaban, A.K.F.; EL-Kady, D.S.; Awad, H.M.; El Sayed, W.A. Synthesis, Docking Studies into CDK-2 and Anticancer Activity of New Derivatives Based Pyrimidine Scaffold and Their Derived Glycosides. Mini-Rev. Med. Chem. 2019, 19, 1093–1110. [Google Scholar] [CrossRef]
- Abdel-Aal, M.T.; El-Sayed, W.A.; El-Kosy, S.M.; El-Ashry, E.S.H. Synthesis and Antiviral Evaluation of Novel 5-(N-Aryl-aminomethyl-1,3,4-oxadiazol-2-yl)hydrazines and their Sugars, 1,2,4-triazoles, tetrazoles and pyrazolyl Derivatives. Arch. Pharm. Chem. Life Sci. 2008, 341, 307–313. [Google Scholar] [CrossRef]
- Yousif, M.N.M.; El-Sayed, W.A.; Abbas, H.S.; Awad, H.M.; Yousif, N.M. Anticancer Activity of New Substituted Pyrimidines, Their Thioglycosides and Thiazolopyrimidine Derivatives. J. Appl. Pharm. Sci. 2017, 7, 021–032. [Google Scholar]
- Iftikhar, F.; Yaqoob, F.; Tabassum, N.; Jan, M.S.; Sadiq, A.; Tahir, S.; Batool, T.; Niaz, B.; Ansari, F.L.; Choudhary, M.I.; et al. Design, Synthesis, in-Vitro Thymidine Phosphorylase Inhibition, In-Vivo Antiangiogenic and in-Silico Studies of C-6 Substituted Dihydropyrimidines. Bioorg. Chem. 2018, 80, 99–111. [Google Scholar] [CrossRef]
- Flefel, E.M.; El-Sayed, W.A.; El-Sofany, W.; Mohamed, A.M.; Awad, H.M. Synthesis and Anticancer Activity of New 1-Thia-4-azaspiro[4.5]decane, Their Derived Thiazolopyrimidine and 1,3,4-Thiadiazole Thioglycosides. Molecules 2017, 22, 170. [Google Scholar] [CrossRef]
- Fahmy, H.T.Y.; Rostom, S.A.F.; Saudi, M.N.; Zjawiony, J.K.; Robins, D.J. Synthesis and in vitro evaluation of the anticancer activity of novel fluorinated thiazolo[4,5-d]pyrimidines. Arch. Pharm. Pharm. Med. Chem. 2003, 336, 216–225. [Google Scholar] [CrossRef]
- El-Sayed, W.A.; Khalaf, H.S.; Mohamed, S.F.; Hssien, H.A.; Kutkat, O.M.; Amr, A.E.E. Synthesis and Antiviral Activity of 1,2,3-Triazole Glycosides Based Substituted Pyridine Via Click Cycloaddition. Russ. J. Gen. Chem. 2017, 87, 2444–2453. [Google Scholar] [CrossRef]
- Nassar, I.F.; El-Sayed, W.A.; Ragab, T.I.M.; Shalaby, A.S.G.; Mehany, A.B.M. Design, Synthesis of New Pyridine and Pyrimidine Sugar Compounds as Antagonists Targeting the ERα via Structure-Based Virtual Screening. Mini-Rev. Med. Chem. 2019, 19, 395–409. [Google Scholar] [CrossRef] [PubMed]
- Alminderej, F.M.; Elganzory, H.H.; El-Bayaa, M.N.; Awad, H.M.; El-Sayed, W.A. Synthesis and Cytotoxic Activity of New 1,3,4-Thiadiazole Thioglycosides and 1,2,3-Triazolyl-1,3,4-Thiadiazole N-glycosides. Molecules 2019, 24, 3738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdel-Aal, M.T.; El-Sayed, W.A.; El-Ashry, E.S.H. Synthesis and Antiviral Evaluation of Some Sugar Arylglycinoylhydrazones and Their Oxadiazoline Derivatives. Arch. Pharm. Chem. Life Sci. 2006, 339, 656–663. [Google Scholar] [CrossRef]
- Attri, P.; Bhatia, R.; Gaur, J.; Arora, B.; Gupta, A.; Kumar, N.; Choi, E.H. Triethylammonium acetate ionic liquid assisted one-pot synthesis of dihydropyrimidinones and evaluation of their antioxidant and antibacterial activities. Arab. J. Chem. 2017, 10, 206–214. [Google Scholar]
- Chen, L.; Jin, Y.; Fu, W.; Xiao, S.; Feng, C.; Fang, B.; Gu, Y.; Li, C.; Zhao, Y.; Liu, Z.; et al. Design, Synthesis, and Structure–Activity Relationship Analysis of Thiazolo[3,2-a]pyrimidine Derivatives with Anti-inflammatory Activity in Acute Lung Injury. ChemMedChem 2017, 12, 1022–1032. [Google Scholar] [CrossRef]
- Mobinikhaledi, A.; Forughifar, N.; Goodarzi, F. Synthesis of Some Bicyclic Thiazolopyrimidine Derivatives. Phosphorus Sulfur Silicon Relat. Elem. 2003, 178, 2539–2543. [Google Scholar] [CrossRef]
- Abdel Rahman, M.M.; El Ashry, E.S.H.; Abdalla, A.A.; Rashed, N. C-(Polyacetoxy)alkyloxadiazolines and related compounds. Carbhydr. Res. 1979, 73, 103–111. [Google Scholar] [CrossRef]
- Somogyi, L. Structure and reactions of L-rhamnose benzoylhydrazone tetra-acetates. Carbohydr. Res. 1978, 64, 289–292. [Google Scholar] [CrossRef]
- Van Meerloo, J.; Kaspers, G.J.; Cloos, J. Cell sensitivity assays: The MTT assay. J. Methods Mol. Biol. 2011, 731, 237–245. [Google Scholar]
Sample Availability: Samples of the synthesized compounds are available from the authors. |






| Compound | HCT116 Cells | Caco-2 Cells | MDA-MB-231 Cells | MCF7 Cells |
|---|---|---|---|---|
| 1 | 25.28 | 58.31 | 40.78 | ND |
| 4 | 63.61 | 30.84 | 36.55 | ND |
| 8 | 66.75 | 9.63 | 46.99 | 69.90 |
| 9 | 27.95 | ND | ND | ND |
| 10 | 65.89 | 4.79 | 30.58 | 16.85 |
| 11 | 34.80 | 83.01 | 23.35 | 12.47 |
| 13 | 44.04 | 16.82 | 46.30 | 34.83 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basiony, E.A.; Hassan, A.A.; Al-Amshany, Z.M.; Abd-Rabou, A.A.; Abdel-Rahman, A.A.-H.; Hassan, N.A.; El-Sayed, W.A. Synthesis and Cytotoxic Activity of New Thiazolopyrimidine Sugar Hydrazones and Their Derived Acyclic Nucleoside Analogues. Molecules 2020, 25, 399. https://doi.org/10.3390/molecules25020399
Basiony EA, Hassan AA, Al-Amshany ZM, Abd-Rabou AA, Abdel-Rahman AA-H, Hassan NA, El-Sayed WA. Synthesis and Cytotoxic Activity of New Thiazolopyrimidine Sugar Hydrazones and Their Derived Acyclic Nucleoside Analogues. Molecules. 2020; 25(2):399. https://doi.org/10.3390/molecules25020399
Chicago/Turabian StyleBasiony, Ebtesam A., Allam A. Hassan, Zahra M. Al-Amshany, Ahmed A. Abd-Rabou, Adel A.-H. Abdel-Rahman, Nasser A. Hassan, and Wael A. El-Sayed. 2020. "Synthesis and Cytotoxic Activity of New Thiazolopyrimidine Sugar Hydrazones and Their Derived Acyclic Nucleoside Analogues" Molecules 25, no. 2: 399. https://doi.org/10.3390/molecules25020399






