Understanding the Deactivation Phenomena of Small-Pore Mo/H-SSZ-13 during Methane Dehydroaromatisation
Abstract
1. Introduction
2. Results
2.1. Study of the Calcination Process
2.2. Operando X-Ray Absorption Spectroscopy
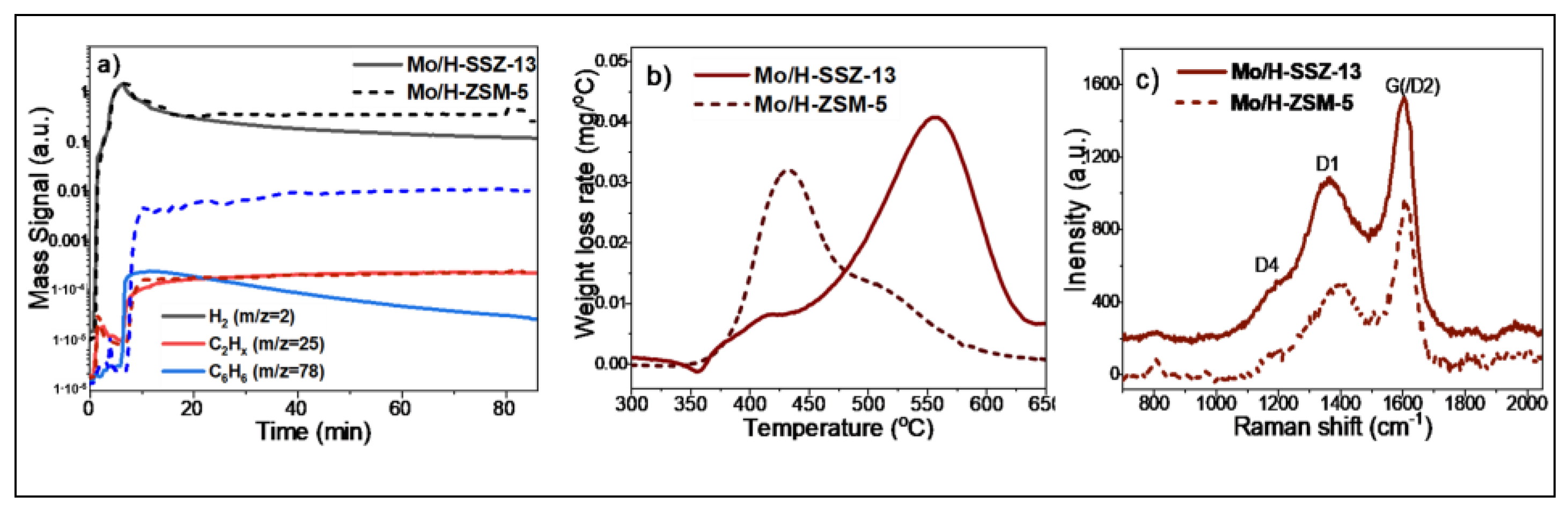
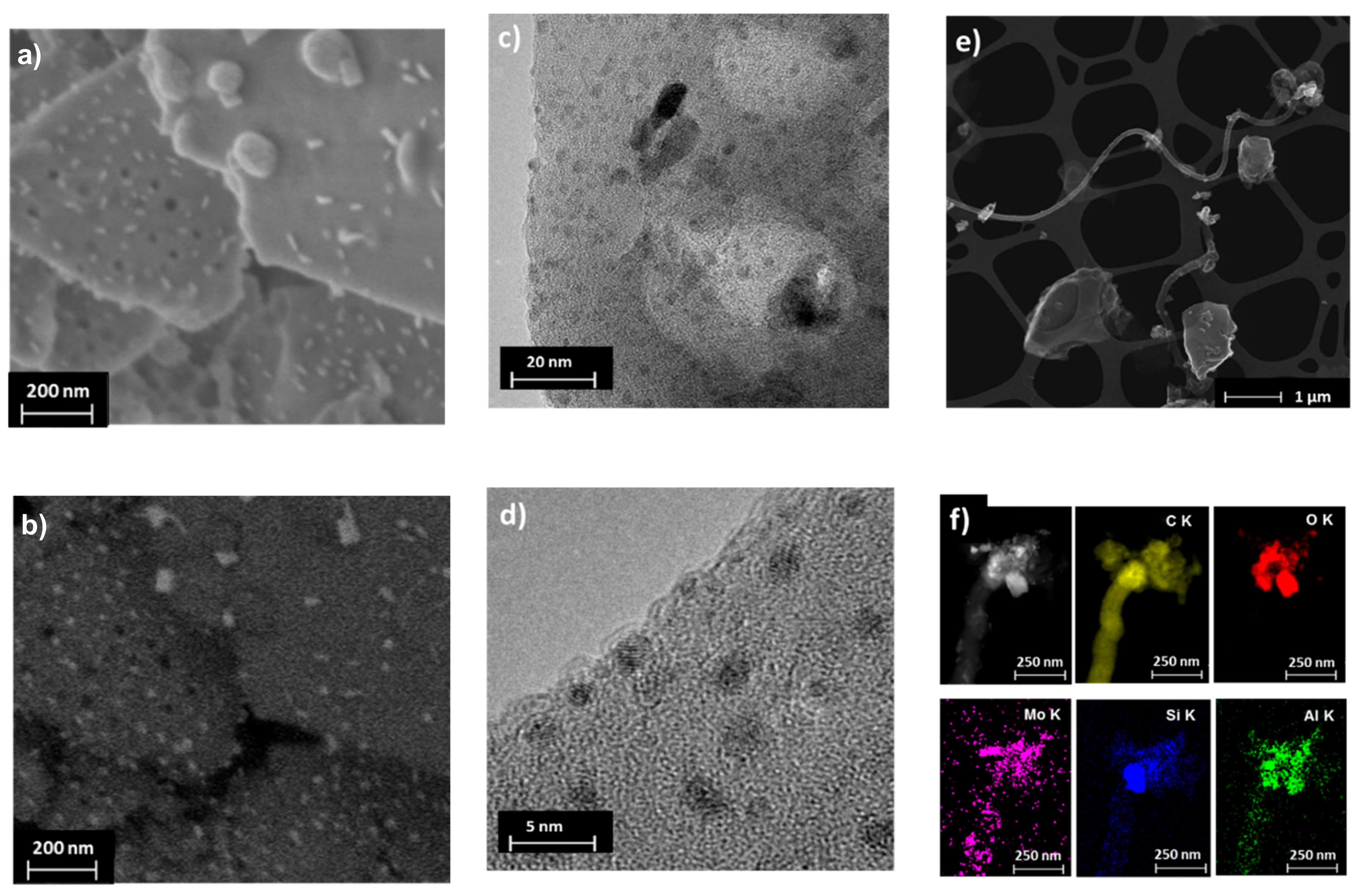
2.3. Laboratory MDA Investigations
3. Summary and Conclusions
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, L.; Tao, L.; Xie, M.; Xu, G.; Huang, J.; Xu, Y. Dehydrogenation and aromatization of methane under non-oxidizing conditions. Catal. Lett. 1993, 21, 35–41. [Google Scholar]
- Spivey, J.J.; Hutchings, G. Catalytic aromatization of methane. Chem. Soc. Rev. 2014, 43, 792–803. [Google Scholar]
- Xu, Y.; Liu, S.; Wang, L.; Xie, M.; Guo, X. Methane activation without using oxidants over Mo/HZSM-5 zeolite catalysts. Catal. Lett. 1995, 30, 135–149. [Google Scholar]
- Fournier, M.; Louis, C.; Che, M.; Chaquin, P.; Masure, D. Polyoxometallates as models for oxide catalysts Part I. An UV-visible reflectance study of polyoxomolybdates: Influence of polyhedra arrangement on the electronic transitions and comparison with supported molybdenum catalysts. J. Catal. 1989, 119, 400–414. [Google Scholar] [CrossRef]
- Vollmer, I.; Li, G.; Yarulina, I.; Kosinov, N.; Hensen, E.J.; Houben, K.; Mance, D.; Baldus, M.; Gascon, J.; Kapteijn, F. Relevance of the Mo-precursor state in H-ZSM-5 for methane dehydroaromatization. Catal. Sci. Technol. 2018, 8, 916–922. [Google Scholar] [CrossRef]
- Rahman, M.; Sridhar, A.; Khatib, S.J. Impact of the presence of Mo carbide species prepared ex situ in Mo/HZSM-5 on the catalytic properties in methane aromatization. Appl. Catal. A Gen. 2018, 558, 67–80. [Google Scholar] [CrossRef]
- Karakaya, C.; Zhu, H.; Kee, R.J. Kinetic modeling of methane dehydroaromatization chemistry on Mo/Zeolite catalysts in packed-bed reactors. Chem. Eng. Sci. 2015, 123, 474–486. [Google Scholar] [CrossRef]
- Wong, K.S.; Thybaut, J.W.; Tangstad, E.; Stöcker, M.W.; Marin, G.B. Methane aromatisation based upon elementary steps: Kinetic and catalyst descriptors. Microporous Mesoporous Mater. 2012, 164, 302–312. [Google Scholar]
- Yin, F.; Li, M.R.; Wang, G.C. Periodic density functional theory analysis of direct methane conversion into ethylene and aromatic hydrocarbons catalyzed by Mo4C2/ZSM-5. Phys. Chem. Chem. Phys. 2017, 19, 22243–22255. [Google Scholar]
- Li, G.; Vollmer, I.; Liu, C.; Gascon, J.; Pidko, E.A. Structure and Reactivity of the Mo/ZSM-5 Dehydroaromatization Catalyst: An Operando Computational Study. ACS Catal. 2019, 9, 8731–8737. [Google Scholar]
- Kosinov, N.; Coumans, F.J.A.G.; Uslamin, E.; Kapteijn, F.; Hensen, E.J.M. Selective Coke Combustion by Oxygen Pulsing During Mo/ZSM-5-Catalyzed Methane Dehydroaromatization. Angew. Chem. 2016, 128, 15310–15314. [Google Scholar]
- Morejudo, S.H.; Zanón, R.; Escolástico, S.; Yuste-Tirados, I.; Malerød-Fjeld, H.; Vestre, P.K.; Coors, W.G.; Martínez, A.; Norby, T.; Serra, J.M.; et al. Direct conversion of methane to aromatics in a catalytic co-ionic membrane reactor. Science 2016, 353, 563–566. [Google Scholar] [CrossRef]
- Kosinov, N.; Uslamin, E.A.; Meng, L.; Parastaev, A.; Liu, Y.; Hensen, E.J.M. Reversible Nature of Coke Formation on Mo/ZSM-5 Methane Dehydroaromatization Catalysts. Angew. Chem. Int. Ed. 2019, 58, 7068–7072. [Google Scholar]
- Gao, J.; Zheng, Y.; Fitzgerald, G.B.; de Joannis, J.; Tang, Y.; Wachs, I.E.; Podkolzin, S.G. Structure of Mo2Cxand Mo4Cx Molybdenum Carbide Nanoparticles and Their Anchoring Sites on ZSM-5 Zeolites. J. Phys. Chem. C 2014, 118, 4670–4679. [Google Scholar]
- Lezcano-González, I.; Oord, R.; Rovezzi, M.; Glatzel, P.; Botchway, S.W.; Weckhuysen, B.M.; Beale, A.M. Molybdenum Speciation and its Impact on Catalytic Activity during Methane Dehydroaromatization in Zeolite ZSM-5 as Revealed by Operando X-Ray Methods. Angew. Chem. Int. Ed. 2016, 55, 5215–5219. [Google Scholar]
- Gao, J.; Zheng, Y.; Jehng, J.-M.; Tang, Y.; Wachs, I.E.; Podkolzin, S.G. Identification of molybdenum oxide nanostructures on zeolites for natural gas conversion. Science 2015, 348, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Kosinov, N.; Wijpekma, S.G.A.; Uslamin, E.; Rohling, R.; Coumans, J.A.G.F.; Mezari, B.; Parastaev, A.; Poryvaev, S.A.; Fedin, V.M.; Evgeny, A.P.; et al. Confined carbon mediates dehydroaromatization of methane over Mo/ZSM-5. Angew. Chem. Int. Ed. 2018, 57, 1016–1020. [Google Scholar]
- Rahman, M.; Infantes-Molina, A.; Hoffman, A.S.; Bare, S.R.; Emerson, K.L.; Khatib, S.J. Effect of Si/Al ratio of ZSM-5 support on structure and activity of Mo species in methane dehydroaromatization. Fuel 2020, 278, 118290. [Google Scholar]
- Dubray, F.; Moldovan, S.; Kouvatas, C.; Grand, J.; Aquino, C.; Barrier, N.; Gilson, J.P.; Nesterenko, N.; Minoux, D.; Mintova, S. Direct Evidence for Single Molybdenum Atoms Incorporated in the Framework of MFI Zeolite Nanocrystals. J. Am. Chem. Soc. 2019, 141, 8689–8693. [Google Scholar] [CrossRef]
- Agote-Arán, M.; Kroner, A.B.; Islam, H.U.; Sławiński, W.A.; Wragg, D.S.; Lezcano-González, I.; Beale, A.M. Determination of Molybdenum Species Evolution during Non-Oxidative Dehydroaromatization of Methane and its Implications for Catalytic Performance. Chem. Catal. Chem. 2018, 11, 473–480. [Google Scholar] [CrossRef]
- Agote-Arán, M.; Fletcher, R.E.; Briceno, M.; Kroner, A.B.; Sazanovich, I.V.; Slater, B.; Rivas, M.E.; Smith, A.W.J.; Collier, P.; Lezcano-González, I.; et al. Implications of the Molybdenum Coordination Environment in MFI Zeolites on Methane Dehydroaromatisation Performance. Chem. Catal. Chem. 2019, 12, 294–304. [Google Scholar] [CrossRef]
- Kosinov, N.; Uslamin, E.A.; Coumans, F.J.A.G.; Wijpkema, A.S.G.; Rohling, R.Y.; Hensen, E.J.M. Structure and Evolution of Confined Carbon Species during Methane Dehydroaromatization over Mo/ZSM-5. ACS Catal. 2018, 8, 8459–8467. [Google Scholar] [CrossRef]
- Vollmer, I.; Kosinov, N.; Szécsényi, Á.; Li, G.; Yarulina, I.; Abou-Hamad, E.; Gurinov, A.; Ould-Chikh, S.; Aguilar-Tapia, A.; Hazemann, J.-L.; et al. A site-sensitive quasi-in situ strategy to characterize Mo/HZSM-5 during activation. J. Catal. 2019, 370, 321–331. [Google Scholar] [CrossRef]
- Vollmer, I.; Van Der Linden, B.; Ould-Chikh, S.; Tapia, A.A.; Yarulina, I.; Abou-Hamad, E.; Sneider, Y.G.; Suarez, A.I.O.; Hazemann, J.-L.; Kapteijn, F.; et al. On the dynamic nature of Mo sites for methane dehydroaromatization. Chem. Sci. 2018, 9, 4801–4807. [Google Scholar] [CrossRef] [PubMed]
- Borry, R.W.; Kim, Y.H.; Huffsmith, A.; Reimer, A.J.A.; Iglesia, E. Structure and Density of Mo and Acid Sites in Mo-Exchanged H-ZSM5 Catalysts for Nonoxidative Methane Conversion. J. Phys. Chem. B 1999, 103, 5787–5796. [Google Scholar] [CrossRef]
- Li, W.; Meitzner, G.D.; Borry, R.W.; Iglesia, E. Raman and X-Ray Absorption Studies of Mo Species in Mo/H-ZSM5 Catalysts for Non-Oxidative CH4 Reactions. J. Catal. 2000, 191, 373–383. [Google Scholar] [CrossRef]
- Ding, W.; Li, S.; Meitzner, G.D.; Iglesia, E. Methane Conversion to Aromatics on Mo/H-ZSM5: Structure of Molybdenum Species in Working Catalysts. J. Phys. Chem. B 2001, 105, 506–513. [Google Scholar] [CrossRef]
- Matus, E.; Ismagilov, I.Z.; Sukhova, O.B.; Zaikovskii, V.I.; Tsikoza, L.T.; Ismagilov, Z.R.; Moulijn, J.A. Study of Methane Dehydroaromatization on Impregnated Mo/ZSM-5 Catalysts and Characterization of Nanostructured Molybdenum Phases and Carbonaceous Deposits. Ind. Eng. Chem. Res. 2007, 46, 4063–4074. [Google Scholar] [CrossRef]
- Ma, S.; Guo, X.; Zhao, L.; Scott, S.; Bao, X. Recent progress in methane dehydroaromatization: From laboratory curiosities to promising technology. J. Energy Chem. 2013, 22, 1–20. [Google Scholar] [CrossRef]
- Guo, X.; Fang, G.; Li, G.; Ma, C.; Fan, H.; Yu, L.; Wu, X.; Deng, D.; Wei, M.; Tan, D.; et al. Direct, Nonoxidative Conversion of Methane to Ethylene, Aromatics, and Hydrogen. Science 2014, 344, 616–619. [Google Scholar] [CrossRef]
- Tempelman, C.H.; Hensen, E.J. On the deactivation of Mo/HZSM-5 in the methane dehydroaromatization reaction. Appl. Catal. B Environ. 2015, 176–177, 731–739. [Google Scholar] [CrossRef]
- Sachtler, W.M.H. Transition Metal Clusters and Isolated Atoms in Zeolite Cages; Springer Science and Business Media LLC: Amsterdam, The Netherlands, 1990; pp. 69–85. [Google Scholar]
- Zhang, C.; Li, S.; Yuan, Y.; Zhang, W.; Wu, T.; Lin, L. Aromatization of methane in the absence of oxygen over Mo-based catalysts supported on different types of zeolites. Catal. Lett. 1998, 56, 207–213. [Google Scholar] [CrossRef]
- Kosinov, N.; Coumans, F.J.A.G.; Uslamin, E.A.; Wijpkema, A.S.G.; Mezari, B.; Hensen, E.J.M. Methane Dehydroaromatization by Mo/HZSM-5: Mono- or Bifunctional Catalysis? ACS Catal. 2017, 7, 520–529. [Google Scholar] [CrossRef]
- Olson, D.H.; Kokotailo, G.T.; Lawton, S.L.; Meier, W.M. Crystal structure and structure-related properties of ZSM-5. J. Phys. Chem. 1981, 85, 2238–2243. [Google Scholar] [CrossRef]
- MFI. Framework Type. Available online: http://europe.iza-structure.org/IZA-SC/framework.php?STC=MFI (accessed on 1 November 2017).
- Harding, B.M.; Kariuki, M.M. Framework Type CHA. Acta Crystallogr. 1994, 34, 852–854. [Google Scholar]
- CHA. Framework Type. Available online: http://europe.iza-structure.org/IZA-SC/framework.php?STC=CHA (accessed on 1 November 2017).
- Webster, C.E.; Drago, R.S.; Zerner, M.C. Molecular dimensions for adsorptives. J. Am. Chem. Soc. 1998, 120, 5509–5516. [Google Scholar] [CrossRef]
- Baertsch, C.D.; Funke, H.H.; Falconer, A.J.L.; Noble, R.D. Permeation of Aromatic Hydrocarbon Vapors through Silicalite−Zeolite Membranes. J. Phys. Chem. 1996, 100, 7676–7679. [Google Scholar] [CrossRef]
- Ma, D.; Shu, Y.; Han, X.; Liu, X.; Xu, A.Y.; Bao, X. Mo/HMCM-22 Catalysts for Methane Dehydroaromatization: A Multinuclear MAS NMR Study. J. Phys. Chem. B 2001, 105, 1786–1793. [Google Scholar] [CrossRef]
- Wragg, D.S.; Akporiaye, D.; Fjellvåg, H. Direct observation of catalyst behaviour under real working conditions with X-ray diffraction: Comparing SAPO-18 and SAPO-34 methanol to olefin catalysts. J. Catal. 2011, 279, 397–402. [Google Scholar] [CrossRef]
- Knözinger, H.; Jezlorowski, H. Scopus—Document Details; Volume 82; Issue 18; 1978. Available online: https://www-scopus-com.libproxy.ucl.ac.uk/record/display.uri?eid=2-s2.0-33947093887&origin=resultslist&sort=plf-f&src=s&st1=jezlorowski&st2=&sid=5bba066550cfcf9c834519eb9dc477d3&sot=b&sdt=b&sl=62&s=AUTHOR-NAME%28jezlorowski%29+AND+PUBYEAR+%3E+1974+AND+PUB (accessed on 20 October 2017).
- Kosinov, N.N.; Coumans, F.J.; Li, G.G.; Uslamin, E.; Mezari, B.B.; Wijpkema, A.S.; Pidko, E.A.; Hensen, E.J. Stable Mo/HZSM-5 methane dehydroaromatization catalysts optimized for high-temperature calcination-regeneration. J. Catal. 2017, 346, 125–133. [Google Scholar] [CrossRef]
- Dippel, B.; Janderb, H.; Heintzenberga, J. NIR FT Raman spectroscopic study of flame soot. Phys. Chem. Chem. Phys. 1999, 1, 4707–4712. [Google Scholar] [CrossRef]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Hoekstra, J.; Beale, A.M.; Soulimani, F.; Versluijs-Helder, M.; Geus, J.W.; Jenneskens, L.W. Base Metal Catalyzed Graphitization of Cellulose: A Combined Raman Spectroscopy, Temperature-Dependent X-ray Diffraction and High-Resolution Transmission Electron Microscopy Study. J. Phys. Chem. 2015, 119, 10653–10661. [Google Scholar] [CrossRef]
- Beale, A.M.; Lezcano-Gonzalez, I.; Slawinksi, W.A.; Wragg, D.S. Correlation between Cu ion migration behaviour and deNO x activity in Cu-SSZ-13 for the standard NH3-SCR reaction. Chem. Commun. 2016, 52, 6170–6173. [Google Scholar] [CrossRef] [PubMed]
- Moliner, M.; Franch, C.; Palomares, E.; Grill, M.; Corma, A. Cu–SSZ-39, an active and hydrothermally stable catalyst for the selective catalytic reduction of NOx. Chem. Commun. 2012, 48, 8264–8266. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Cabañas, M.-J.; Barrett, P.A.; Camblor, M.A. Synthesis and structure of pure SiO2 chabazite: The SiO2 polymorph with the lowest framework density. Chem. Commun. 1998, 17, 1881–1882. [Google Scholar] [CrossRef]
- BM01 ESRF Swiss Norwegian Beamline. Available online: http://www.esrf.eu/UsersAndScience/Experiments/CRG/BM01/bm01-a (accessed on 2 October 2017).
- Norbyb, P. In-Situ Time Resolved Synchrotron Powder Diffraction Studies of Syntheses and Chemical Reactions. Mater. Sci. Forum 1996, 228, 147–152. [Google Scholar] [CrossRef]
- Hammersley, A.P.; Svensson, S.O.; Hanfland, M.; Fitch, A.N.; Hausermann, D. Two-dimensional detector software: From real detector to idealised image or two-theta scan. High Press. Res. 1996, 14, 235–248. [Google Scholar] [CrossRef]
- SNBL Software. Available online: http://www.esrf.eu/home/UsersAndScience/Experiments/CRG/BM01/bm01/image.htm/snbl-tool-box.html (accessed on 29 October 2020).
- McCusker, L.B.; Von Dreele, R.B.; Cox, D.E.; Louër, D.; Scardi, P. Rietveld refinement guidelines. J. Appl. Crystallogr. 1999, 32, 36–50. [Google Scholar] [CrossRef]
- Stinton, G.W.; Evans, J.S.O. Parametric Rietveld refinement. J. Appl. Crystallogr. 2007, 40, 87–95. [Google Scholar] [CrossRef]
- Dent, A.J.; Cibin, G.; Ramos, S.; Smith, A.D.; Scott, S.M.; Varandas, L.; Jones, C.P.; Pearson, M.R.; Krumpa, N.A. R B18: A core XAS spectroscopy beamline for Diamond. J. Phys. Conf. Ser. 2009, 190, 012039. [Google Scholar] [CrossRef]
- Kroner, A.B.; Mohammed, K.M.H.; Gilbert, M.; Duller, G.; Cahill, L.; Leicester, P.; Woolliscroft, R.; Shotton, E.J. A Flexible Gas Flow Reaction Cell for in Situ X-Ray Absorption Spectroscopy Studies. AIP Conf. Proc. 2016, 1741, 030014. [Google Scholar]
- Ravel, B.; Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 2005, 12, 537–541. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |




| Experiment | Stage of the Cycle | Oxidation Estate | Edge Position (eV) | Edge Step |
|---|---|---|---|---|
| Exp. 1 | As-prepared | 5.9 | 20,014.6 | 0.360 |
| Calcined | 5.8 | 20,014.3 | 0.158 | |
| MDA 700 °C | 2.1 | 20,007.4 | 0.161 | |
| Exp. 2 | As-prepared | 5.8 | 20,014.4 | 0.410 |
| Cycle 1, calc. | 5.8 | 20,014.5 | 0.194 | |
| Cycle 1, MDA 650 °C | 2.4 | 20,008.1 | 0.197 | |
| Cycle 2, calc. | 5.8 | 20,014.3 | 0.180 | |
| Cycle 2, MDA 650 °C | 2.5 | 20,008.2 | 0.178 | |
| Cycle 3, calc. | 5.8 | 20,014.3 | 0.158 | |
| Cycle 3, MDA 780 °C | 1.8 | 20,006.9 | 0.157 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agote-Arán, M.; Kroner, A.B.; Wragg, D.S.; Sławiński, W.A.; Briceno, M.; Islam, H.U.; Sazanovich, I.V.; Rivas, M.E.; Smith, A.W.J.; Collier, P.; et al. Understanding the Deactivation Phenomena of Small-Pore Mo/H-SSZ-13 during Methane Dehydroaromatisation. Molecules 2020, 25, 5048. https://doi.org/10.3390/molecules25215048
Agote-Arán M, Kroner AB, Wragg DS, Sławiński WA, Briceno M, Islam HU, Sazanovich IV, Rivas ME, Smith AWJ, Collier P, et al. Understanding the Deactivation Phenomena of Small-Pore Mo/H-SSZ-13 during Methane Dehydroaromatisation. Molecules. 2020; 25(21):5048. https://doi.org/10.3390/molecules25215048
Chicago/Turabian StyleAgote-Arán, Miren, Anna B. Kroner, David S. Wragg, Wojciech A. Sławiński, Martha Briceno, Husn U. Islam, Igor V. Sazanovich, María E. Rivas, Andrew W. J. Smith, Paul Collier, and et al. 2020. "Understanding the Deactivation Phenomena of Small-Pore Mo/H-SSZ-13 during Methane Dehydroaromatisation" Molecules 25, no. 21: 5048. https://doi.org/10.3390/molecules25215048
APA StyleAgote-Arán, M., Kroner, A. B., Wragg, D. S., Sławiński, W. A., Briceno, M., Islam, H. U., Sazanovich, I. V., Rivas, M. E., Smith, A. W. J., Collier, P., Lezcano-González, I., & Beale, A. M. (2020). Understanding the Deactivation Phenomena of Small-Pore Mo/H-SSZ-13 during Methane Dehydroaromatisation. Molecules, 25(21), 5048. https://doi.org/10.3390/molecules25215048







