Suitability and Modification of Different Renewable Materials as Feedstock for Sustainable Flame Retardants
Abstract
1. Introduction
2. Results
2.1. Raw Material Synthesis and Characterization of Flame Retardants
2.2. Thermal Analysis and Fire Behavior
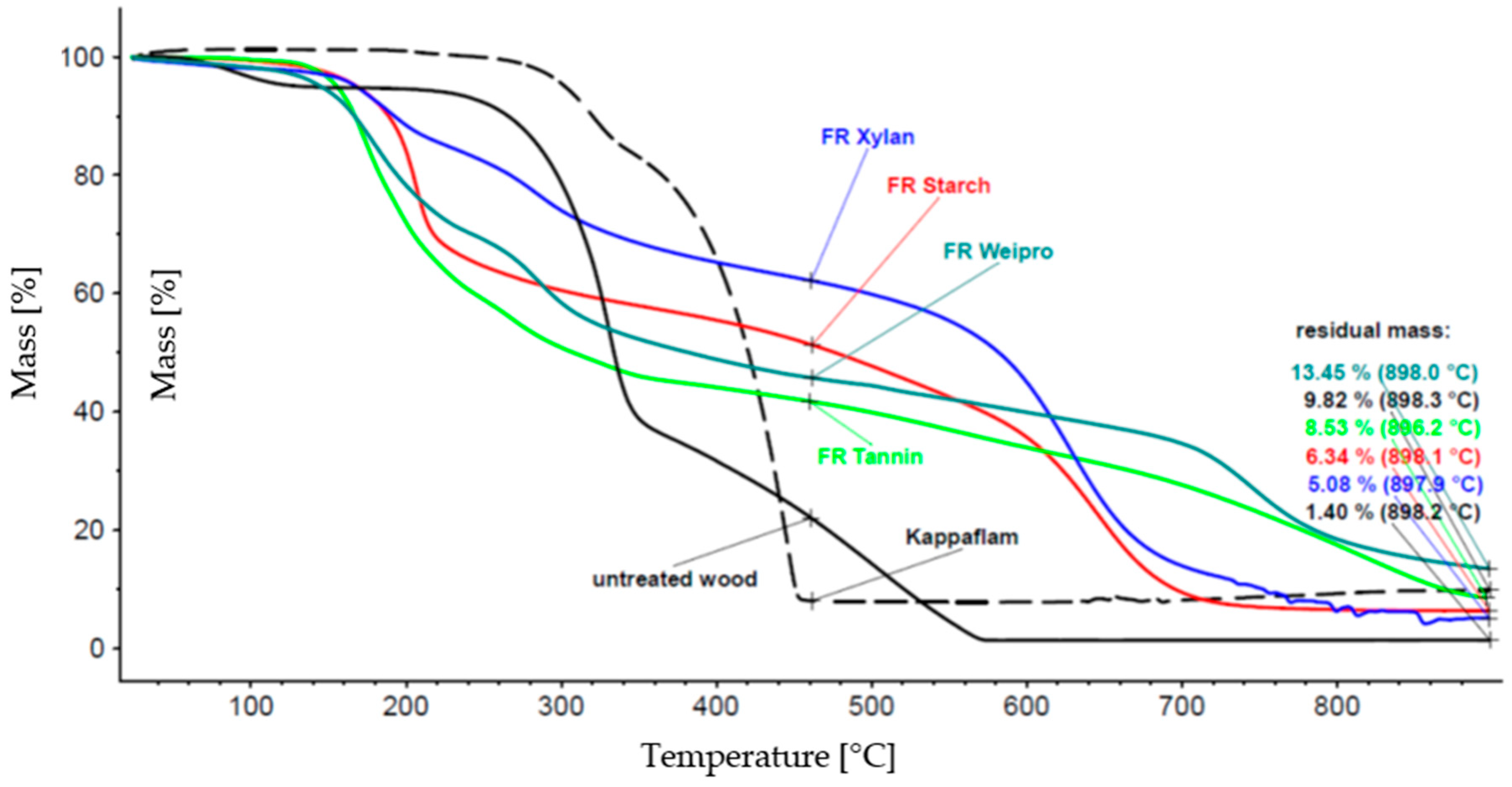
2.2.1. Thermal Analysis
2.2.2. Fire Tests
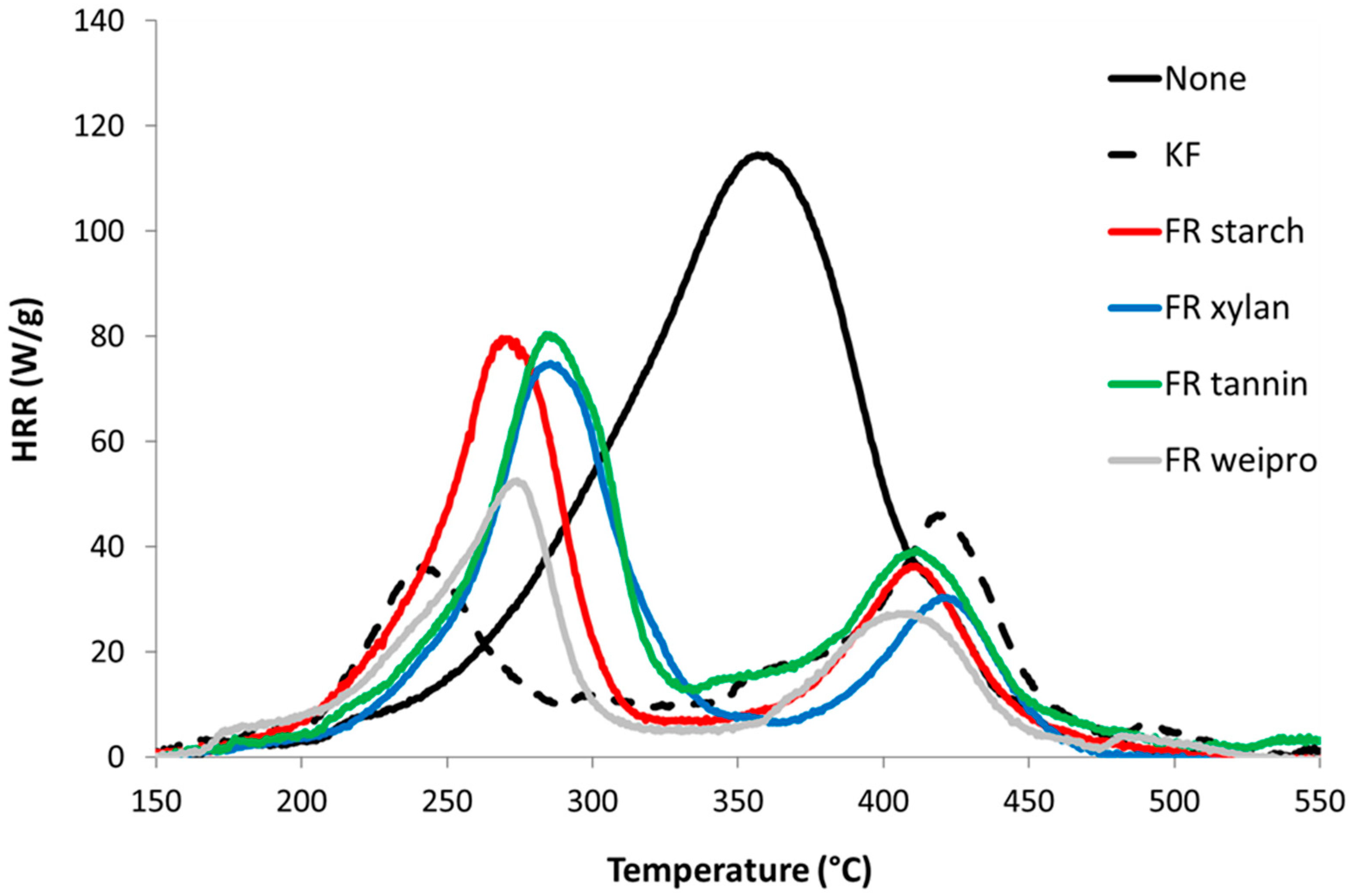
2.2.3. Pyrolysis Combustion Flow Calorimetry
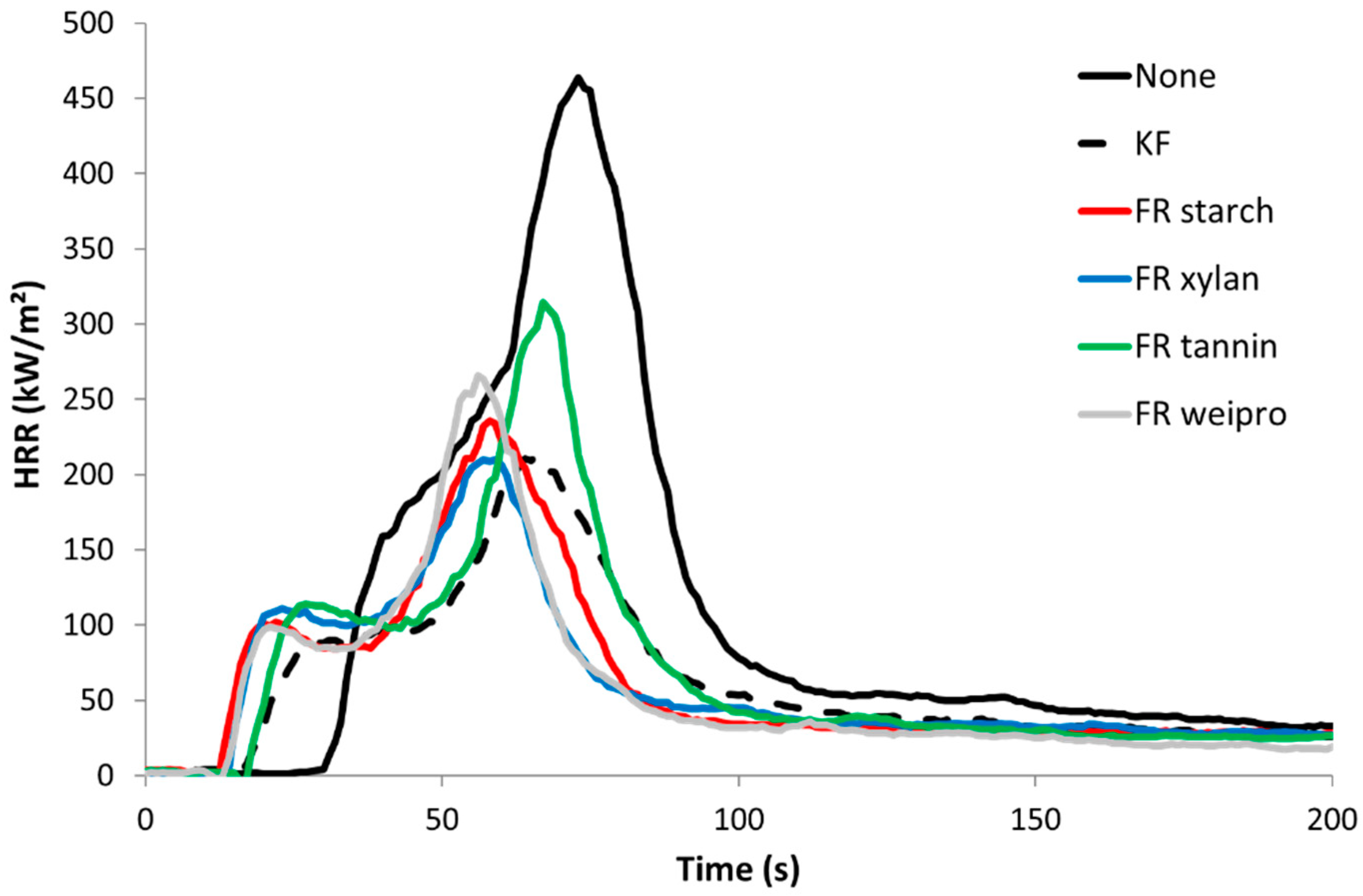
2.2.4. Cone Calorimetry
2.3. Investigation of the Smoldering Behavior
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Synthesis
4.2.2. Phosphate and Nitrogen Content
4.2.3. Thermal Behavior
4.2.4. Application
4.2.5. Small Burner Test (DIN EN ISO 11925-2)
4.2.6. Calorimetry Analyses
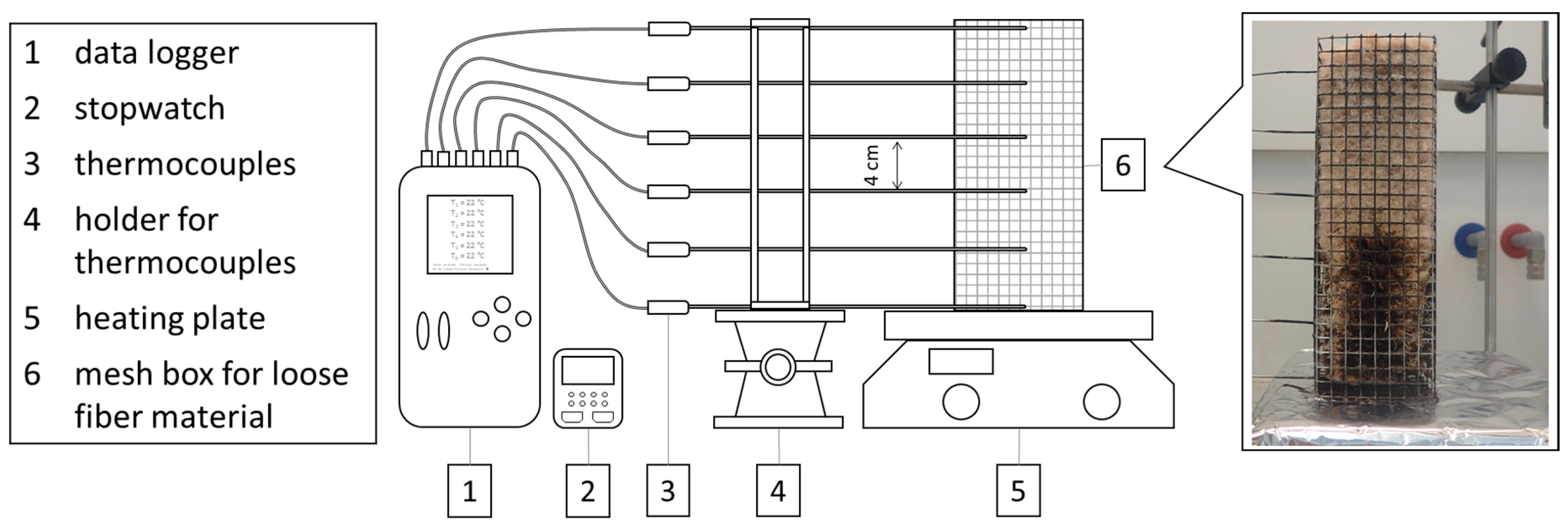
4.2.7. Smoldering Behavior
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shaw, S.D.; Blum, A.; Weber, R.; Kannan, K.; Rich, D.; Lucas, D.; Koshland, C.P.; Dobraca, D.; Hanson, S.; Birnbaum, L.S. Halogenated flame retardants: Do the fire safety benefits justify the risks? Rev. Environ. Health 2010, 25, 261–305. [Google Scholar] [CrossRef] [PubMed]
- Kemmlein, S.S.; Herzke, D.; Law, R.J. Brominated flame retardants in the European chemicals policy of REACH-Regulation and determination in materials. J. Chromatogr. A 2009, 1216, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Alaee, M.M.; Arias, P.P.; Sjödin, A.; Bergman, Å. An overview of commercially used brominated flame retardants, their applications, their use patterns in different countries/regions and possible modes of release. Environ. Int. 2003, 29, 683–689. [Google Scholar] [CrossRef]
- Beard, A. European Initiatives for Selecting Sustainable Flame Retardants. In Chemical Alternatives Assessments; The Royal Society of Chemistry: London, UK, 2013; pp. 44–66. [Google Scholar] [CrossRef]
- Shen, K.K. Review of Recent Advances on the Use of Boron-based Flame Retardants. In Polymer Green Flame Retardants; Elsevier: Amsterdam, The Netherlands, 2014; pp. 367–388. [Google Scholar] [CrossRef]
- ECHA. Member State Committee Draft Support Document for Identification of Boric Acid as a Substance of Very High Concern Because of Its CMR Properties; European Chemicals Agency: Helsinki, Finland, 2010. [Google Scholar]
- The European Commission. COMMISSION REGULATION (EU) 2016/1017 of 23 June 2016 amending Annex XVII to Regulation (EC) No 1907/2006 of the European Parliament and of the Council concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH). Off. J. Eur. Union 2016, L 166, 1–4. [Google Scholar]
- Alongi, J.J.; Han, Z.; Bourbigot, S. Intumescence: Tradition versus novelty. A comprehensive review. Prog. Polym. Sci. 2014, 51, 28–73. [Google Scholar] [CrossRef]
- Costes, L.L.; Laoutid, F.F.; Brohez, S.; Dubois, P. Bio-based flame retardants: When nature meets fire protection. Mater. Sci. Eng. R Rep. 2017, 117, 1–25. [Google Scholar] [CrossRef]
- Bergmeister, E.; Heckmaier, J. Process for Preparation of Phosphate Esters of Polymeric Materials Containing Hydroxyl Groups. U.S. Patent 3250756A, 10 May 1966. [Google Scholar]
- Passauer, L.L.; Fischer, S.S.; Bender, H.H.; Tech, S.; Wagenführ, A. Flame Retardants Comprising Polysaccharide Derivatives Containing Nitrogen and Phosphorus and Use Thereof to Improve the Flame-Retardant Properties of Wood and Wood Composite Materials. Patent WO2013135890A1, 19 September 2013. [Google Scholar]
- Heinze, T.T.; Liebert, T.; Koschella, A. Esterification of Polysaccharides; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar] [CrossRef]
- Passauer, L. Thermal characterization of ammonium starch phosphate carbamates for potential applications as bio-based flame-retardants. Carbohydr. Polym. 2019, 211, 69–74. [Google Scholar] [CrossRef]
- Gebke, S.; Thümmler, K.; Sonnier, R.; Tech, S.; Wagenführ, A.; Fischer, S. Flame Retardancy of Wood Fiber Materials Using Phosphorus-Modified Wheat Starch. Molecules 2020, 25, 335. [Google Scholar] [CrossRef]
- Klein, R.A. SFPE Handbook of Fire Protection Engineering; Springer Science + Business Media: Berlin, Germany, 2002. [Google Scholar]
- Rowell, R.M.; Dietenberger, M.A. Thermal Combustion, and Fire Retardancy of Wood. In Handbook of Wood Chemistry and Wood Composites; Taylor & Francis Group: Oxfordshire, UK, 2013; pp. 127–149. [Google Scholar]
- Shafizadeh, F.; Radbury, A.G.W.B. Smoldering Combustion of Cellulosic Materials. J. Therm. Insul. 1978, 2, 141–152. [Google Scholar] [CrossRef]
- Heinze, U.; Klemm, D.; Unger, E.; Pieschel, F. New starch phosphate carbamides of high swelling ability: Synthesis and characterization. Starch/Staerke 2003, 55, 55–60. [Google Scholar] [CrossRef]
- Gaan, S.; Sun, G.; Hutches, K.; Engelhard, M.H. Effect of nitrogen additives on flame retardant action of tributyl phosphate: Phosphorus–nitrogen synergism. Polym. Degrad. Stab. 2008, 93, 99–108. [Google Scholar] [CrossRef]
- Schartel, B. Phosphorus-based Flame Retardancy Mechanisms—Old Hat or a Starting Point for Future Development? Materials 2010, 3, 4710–4745. [Google Scholar] [CrossRef]
- German Institute for Standardisation. DIN EN ISO 11925-2 Reaction to Fire Tests—Ignitability of Products Subjected to Direct Impingement of Flame—Part 2: Single-Flame Source Test 2011; German Institute for Standardisation: Berlin, German, 2011. [Google Scholar]
- Sonnier, R.; Vahabi, H.; Ferry, L. Pyrolysis-Combustion Flow Calorimetry: A Powerful Tool to Evaluate the Flame Retardancy of Polymers. In Fire and Polymers VI: New Advances in Flame Retardant Chemistry and Science; American Chemical Society: Washington, DC, USA, 2012; pp. 361–390. [Google Scholar] [CrossRef]
- Walters, R.; Safronava, N.; Lyon, R. A microscale combustion calorimeter study of gas phase combustion of polymers. Combust. Flame 2015, 162, 855–863. [Google Scholar] [CrossRef]
- Palumbo, M.; Lacasta, A.M.; Navarro, A.; Giraldo, P.; Lesar, B. Improvement of fire reaction and mould growth resistance of a new bio-based thermal insulation material. Constr. Build. Mater. 2017, 13, 531–539. [Google Scholar] [CrossRef]
- Dorez, G.; Ferry, L.; Sonnier, R.; Taguet, A. Effect of cellulose, hemicellulose and lignin contents on pyrolysis and combustion of natural fibers. J. Anal. Appl. Pyrolysis 2014, 107, 323–331. [Google Scholar] [CrossRef]
- Hajj, R.; El Hage, R.; Sonnier, R.; Otazaghine, B.; Rouif, S.; Nakhl, M.; Lopez-Cuesta, J.-M. Influence of lignocellulosic substrate and phosphorus flame retardant type on grafting yield and flame retardancy. React. Funct. Polym. 2020, 153. [Google Scholar] [CrossRef]
- Moussa, M.; El Hage, R.; Sonnier, R.; Chrusciel, L.; Ziegler-Devin, I.; Brosse, N. Toward the cottonization of hemp fibers by steam explosion. Flame-retardant fibers. Ind. Crops Prod. 2020, 151. [Google Scholar] [CrossRef]
- Schartel, B.; Hull, T.R. Development of fire-retarded materials—Interpretation of cone calorimeter data. Fire Mater. 2007, 31, 327–354. [Google Scholar] [CrossRef]
- Lindholm, J.; Brink, A.; Hupa, M. Cone Calorimeter—A Tool for Measuring Heat. Mater. Sci. 2009. [Google Scholar]
- Lowden, L.A.; Hull, T.R. Flammability behaviour of wood and a review of the methods for its reduction. Fire Sci. Rev. 2013, 2, 1–19. [Google Scholar] [CrossRef]
- Jiang, J.; Li, J.; Hu, J.; Fan, D. Effect of nitrogen phosphorus flame retardants on thermal degradation of wood. Constr. Build. Mater. 2010, 24, 2633–2637. [Google Scholar] [CrossRef]
- Hagen, B.C.; Frette, V.; Kleppe, G.; Arntzen, B.J. Onset of smoldering in cotton: Effects of density. Fire Saf. J. 2011, 46, 73–80. [Google Scholar] [CrossRef]
- Liodakis, S.; Fetsis, I.K.; Agiovlasitis, I.P. The fire-retarding effect of inorganic phosphorus compounds on the combustion of cellulosic materials. J. Therm. Anal. Calorim. 2009, 98, 285–291. [Google Scholar] [CrossRef]
- Steijns, M.; Mars, P. The Adsorption of Sulfur by Microporous Materials. J. Colloid Interface Sci. 1976, 57, 175–180. [Google Scholar] [CrossRef]
- Yang, R.T.; Steinberg, M. Combustion of Carbon. Effect of Sulfur Dioxide. J. Phys. Chem. 1977, 81, 1117–1118. [Google Scholar] [CrossRef]
- Swedish Standards Institute. EN 13805:2002 Foodstuffs—Determination of Trace Elements—Pressure Digestion; German Version 2002; Swedish Standards Institute: Stockholm, Sweden, 2002. [Google Scholar]
- German Institute for Standardisation. ISO 5660-1:2015-03 Reaction-to-Fire Tests—Heat Release, Smoke Production and Mass Loss Rate—Part 1: Heat Release Rate (Cone Calorimeter Method) and Smoke Production Rate (Dynamic Measurement) 2015; German Institute for Standardisation: Berlin, German, 2015. [Google Scholar]





| Modified Biopolymer | Reaction Time [h] | PO43− [%] | Ntot [%] |
|---|---|---|---|
| starch | 6 | 45.9 | 16.5 |
| weipro | 6 | 48.2 | 17.2 |
| xylan | 3 | 45.6 | 15.8 |
| tannin | 6 | 21.5 | 21.8 |
| Additive | Fire Cone Height [cm] | PO43− [%] | Ntot [%] |
|---|---|---|---|
| none | 20.0 | 0 | 0 |
| Native starch | 20.0 | 0 | 0.1 |
| Native weipro | 19.3 | 0 | 1.3 |
| Native xylan | 20.0 | 0 | 0 |
| Native tannin | 17.5 | 0 | 0 |
| Kappaflam | 10.7 | 0.7 | 2.1 |
| FR starch | 13.4 | 4.6 | 1.6 |
| FR weipro | 10.4 | 4.8 | 1.7 |
| FR xylan | 9.3 | 4.6 | 1.6 |
| FR tannin | 15.0 | 2.2 | 2.2 |
| Additive | T1 [°C] | P1HRR [W/g] | T2 [°C] | P2HRR [W/g] | THR [KJ/g] | Residual [%] | HCC [KJ/g] | PO43− [%] | Ntot [%] |
|---|---|---|---|---|---|---|---|---|---|
| none | 363 | 128 | - | - | 13.3 | 13.5 | 15.3 | 0 | 0 |
| KF * | 241 | 35 | 418 | 40 | 5.7 | 30.2 | 8.1 | 0.7 | 2.1 |
| starch | 273 | 77 | 411 | 33 | 6.4 | 29.8 | 9.0 | 4.6 | 1.6 |
| weipro | 271 | 57 | 413 | 27 | 4.8 | 34.0 | 7.3 | 4.8 | 1.7 |
| xylan | 268 | 73 | 412 | 23 | 7.1 | 31.4 | 10.3 | 4.6 | 1.5 |
| tannin | 283 | 76 | 412 | 37 | 8.2 | 26.4 | 11.1 | 2.2 | 2.1 |
| Additive | TTI [s] | PHRR [KW/m²] | THR [KJ/g] | Residual [%] | EHC [KJ/g] | TSP [m2] | PO43− [%] | Ntot [%] |
|---|---|---|---|---|---|---|---|---|
| none | 31 | 486 | 15.9 | 2.5 | 16.3 | 1.3 | ||
| KF | 19 | 209 | 10.3 | 22.9 | 13.3 | 0.36 | 0.7 | 2.1 |
| starch | 16 | 242 | 9.8 | 17.5 | 11.9 | 0.47 | 4.6 | 1.6 |
| weipro | 15 | 266 | 9.8 | 24.4 | 12.9 | 0.42 | 4.8 | 1.7 |
| xylan | 18 | 220 | 10.1 | 20.9 | 12.7 | 0.47 | 4.6 | 1.5 |
| tannin | 21 | 305 | 10.9 | 19.9 | 13.7 | 0.55 | 2.2 | 2.1 |
| Additive | Residue [%] | tS [min] | TSmax [°C] | PO43− [%] |
|---|---|---|---|---|
| none | 32.4 | 47.2 | 597 | 0 |
| Native starch | 36.5 | 36.5 | 600 | 0 |
| Native weipro | 46.0 | 36.2 | 611 | 0 |
| Native xylan | 26.7 | 27.1 | 506 | 0 |
| Native tannin | 22.3 | 51.2 | 572 | 0 |
| Kappaflam | 54.8 | 31.3 | 501 | 0.7 |
| FR starch | 38.4 | 36.2 | 669 | 4.6 |
| FR weipro | 36.3 | 34.9 | 701 | 4.8 |
| FR xylan | 67.4 | 25.7 | 576 | 4.6 |
| FR tannin | 51.6 | 31.0 | 557 | 2.2 |
Sample Availability: Samples of the FRs are available from the corresponding author. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gebke, S.; Thümmler, K.; Sonnier, R.; Tech, S.; Wagenführ, A.; Fischer, S. Suitability and Modification of Different Renewable Materials as Feedstock for Sustainable Flame Retardants. Molecules 2020, 25, 5122. https://doi.org/10.3390/molecules25215122
Gebke S, Thümmler K, Sonnier R, Tech S, Wagenführ A, Fischer S. Suitability and Modification of Different Renewable Materials as Feedstock for Sustainable Flame Retardants. Molecules. 2020; 25(21):5122. https://doi.org/10.3390/molecules25215122
Chicago/Turabian StyleGebke, Stefan, Katrin Thümmler, Rodolphe Sonnier, Sören Tech, Andre Wagenführ, and Steffen Fischer. 2020. "Suitability and Modification of Different Renewable Materials as Feedstock for Sustainable Flame Retardants" Molecules 25, no. 21: 5122. https://doi.org/10.3390/molecules25215122
APA StyleGebke, S., Thümmler, K., Sonnier, R., Tech, S., Wagenführ, A., & Fischer, S. (2020). Suitability and Modification of Different Renewable Materials as Feedstock for Sustainable Flame Retardants. Molecules, 25(21), 5122. https://doi.org/10.3390/molecules25215122







