3.1. Chemistry
3.1.1. General Information
The commercially obtained chemicals were used directly without further purification. Solvents were purified and distilled following the standard procedures. All the reactions were monitored by thin-layer chromatography (TLC). The NMR spectra were taken on a Bruker AV-400 MHz spectrometer (400 MHz for 1H and 101 MHz for 13C) and chemical shifts were expressed in ppm downfield using tetramethylsilane as the internal standard. HRMSwere performed on a VG ZAB-HS mass spectrometer under electron spray ionization (ESI). All the derivatives for testing bioactivity were purified to ≥ 95% purity which was determined by HPLC analysis on a Shimadzu Prominence-i LC-2030C 3D system (column, InertSustain C18, 4.6 mm × 250 mm, 5 μM; mobile phase, gradient elution of methanol/H2O (90:10); low rate, 1.0 mL/min; UV wavelength, 190−800 nm; temperature, 40 °C; injection volume, 10 μL).
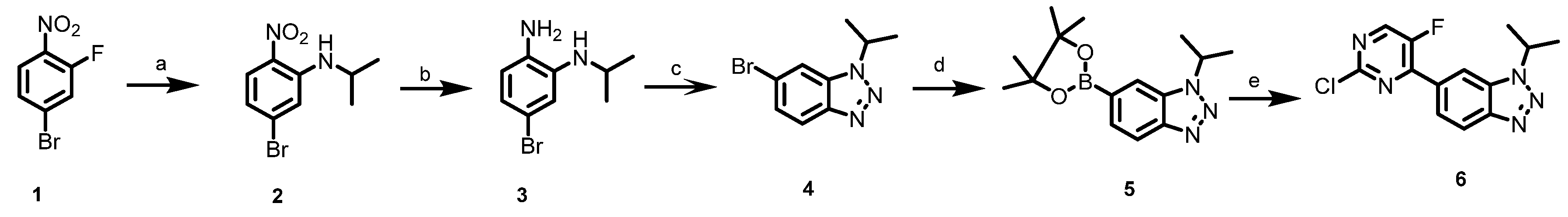
3.1.2. Synthesis of Intermediate Compound 6
5-bromo-N-isopropyl-2-nitroaniline(2). An orange mixture of 4-bromo-2-fluoro-1 -nitrobenzene (4 g, 18.18 mmol), isopropylamine (1.7 mL, 20 mmol) and K2CO3 (2.51 g, 36.36 mmol) in DMF (40 mL) was stirred at room temperature overnight. The resulting mixture was diluted with water and the mixture was extracted by ethyl acetate, and the combined organic layers were washed by water and brine, and dried by anhydrous magnesium sulfate. The solvent was evaporated, and the residue was purified by silica gel column chromatography to obtain 2 (4.52 g, 96%) as a bright orange solid. 1H NMR (400 MHz, DMSO-d6) δ 8.08–7.77 (m, 2H), 7.20 (d, J = 2.5 Hz, 1H), 6.76 (dd, J = 9.1, 2.3 Hz, 1H), 4.08–3.77 (m, 1H), 1.23 (dd, J = 6.4, 2.3 Hz, 6H). 13C NMR (101 MHz, DMSO-d6) δ 145.12, 131.60, 130.47, 128.55, 118.48, 117.14, 44.09, 22.51.
5-bromo-N1-isopropylbenzene-1,2-diamine(3). Compound 2 (4 g, 15.4 mmol) was dissolved in AcOH (100 mL) and Fe powder (8.6 g, 154 mmol, 10 equivalents) was added. The mixture was stirred at RT for 0.5 h and then heated at 70 °C for 3 h. The suspension was basified with aqNaHCO3, diluted with CH2Cl2 (150 mL), and filtered through celite. The filtrate was extracted with CH2Cl2 (2 × 100 mL) and the combined organic phases were dried (K2CO3) and evaporated to furnish 3 (3.2 g, 91%) as a dark solid. The crude product 3 was not subjected to further purification and was taken directly.
6-bromo-1-isopropyl-1H-benzo[d](1,2,3)triazole(4). To a 0 °C mixture of the crude product 3 (3.2 g, 14 mmol) in conc. HCl (40 mL) was added NaNO2(1.1g,15.4 mmol) in H2O (10 mL). The mixture was allowed to warm to room temperature and was stirred for 1 h. After recooling to 0 °C, the mixture was treated with 6N NaOH until basic, the precipitate was then filtered, rinsed with H2O and dried to afford 4 (2.62 g, 78%). 1H NMR (400 MHz, DMSO-d6) δ 8.27 (d, J = 2.4 Hz, 1H), 7.98 (dd, J = 8.9, 2.3 Hz, 1H), 7.48 (dq, J = 8.8, 1.6 Hz, 1H), 5.21 (td, J = 6.7, 2.3 Hz, 1H), 1.59 (dd, J = 6.8, 2.4 Hz, 6H). 13C NMR (101 MHz, DMSO-d6) δ 147.32, 132.32, 129.26, 119.05, 117.79, 84.53, 83.27, 51.07, 25.27, 25.14, 22.75.
1-isopropyl-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H benzo[d](1,2,3) triazole(5). To a suspension of compound (4) (3 g, 12.5 mmol) in 50 mL 1,4-dioxane the following substances were added: bis(pinacolato)diboron (3.81 g, 15 mmol 1.2 equiv), PdCl2(pddf) (457 mg, 0.625 mmol, 0.05 equiv) and KOAc (4.3 g, 43.8 mmol, 3.5 equiv) and the flask was purged with N2. Then the flask was sealed and the mixture was heated for 12 h at 95℃. The reaction was cooled to RT, the solvent was removed under reduced pressure, and the residue was purified by silica gel column chromatography to obtain 5 (2.4 g, 67%).1H NMR (400 MHz, DMSO-d6) δ 8.17 (s, 1H), 8.01 (d, J = 8.3 Hz, 1H), 7.64 (d, J = 8.4 Hz, 1H), 5.33 (p, J = 6.7 Hz, 1H), 1.61 (d, J = 6.7 Hz, 6H), 1.32 (s, 12H).13C NMR (101 MHz, DMSO-d6) δ 147.32, 132.32, 129.26, 119.05, 117.79, 83.27, 51.07, 25.14, 22.75.
6-(2-chloro-5-fluoropyrimidin-4-yl)-1-isopropyl-1H-benzo[d](1,2,3) triazole(6). To a suspension of compound (5) (2 g, 7 mmol) in 50 mL 1,4-dioxane and 5 mL H2O, the following substances were added: 2,4-dichloro-5-fluoropyrimidine (2.3 g, 14 mmol, 2 equiv), PdCl2(pddf) (512 mg, 0.7 mmol, 0.1 equiv) and NaHCO3 (1.8 g, 21 mmol, 3 equiv) and the flask was purged with N2. Then the flask was sealed and the mixture was heated for 12 h at 100 °C. The reaction was cooled to RT, the solvent was removed under reduced pressure, and the residue was purified by silica gel column chromatography to obtain 6 (1.2 g, 59%).1H NMR (400 MHz, DMSO-d6) δ 8.98 (d, J = 3.0 Hz, 1H), 8.46 (s, 1H), 8.16 (d, J = 8.7 Hz, 1H), 8.03–7.82 (m, 1H), 5.34 (p, J = 6.7 Hz, 1H), 1.64 (d, J = 6.6 Hz, 6H). 13C NMR (101 MHz, DMSO-d6) δ 155.49 (d, J = 265.4 Hz), 154.70 (d, J = 3.6 Hz), 153.95 (d, J = 10.1 Hz), 150.48 (d, J = 26.8 Hz), 146.76, 132.36, 130.17 (d, J = 5.1 Hz), 124.57 (d, J = 5.8 Hz), 120.16, 112.80 (d, J = 7.3 Hz), 51.58, 22.57.
3.1.3. General Procedure for the Preparation of 8A-P
To a stirred solution of cyclopropane-1,1-dicarboxylic acid (7) (13.01 g, 100 mmol) in isopropyl acetate (150 mL) at 0 °C, thionyl chloride (12.5 g, 105 mmol) was added dropwise. After addition, the resulting mixture was stirred at room temperature and stirred for 6 h. The resulting mixture was then treated with a solution of aniline or substituted aniline (110 mmol) and triethylamine (110 mmol) in isopropyl acetate (40 mL) over 1 h. After stirring for 2 h, the resulting mixture was added to ethyl acetate (500 mL). The solvent was washed by 1 N HCl solution and brine. The organic phase was dried over MgSO4, evaporated and the residue treated with heptane (200 mL). The product slurry was stirred for 0.5 h, filtered and dried under vacuum to obtain product.
1-((3-fluorophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8A). White solid (76%), 1H NMR (400 MHz, DMSO-d6) δ 13.12 (s, 1H), 10.73 (s, 1H), 7.61 (dt, J = 11.3, 2.0 Hz, 1H), 7.41–7.27 (m, 2H), 6.88 (ddt, J = 9.1, 6.7, 2.7 Hz, 1H), 1.41 (s, 4H).
1-((4-fluorophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8B). White solid (67%), 1H NMR (400 MHz, DMSO-d6) δ 13.11 (s, 1H), 10.81–10.50 (m, 1H), 7.63 (dd, J = 8.9, 5.0 Hz, 2H), 7.13 (tt, J = 8.9, 3.0 Hz, 2H), 1.54–1.30 (m, 4H).
1-((3-chlorophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8C). White solid (81%), 1H NMR (400 MHz, DMSO-d6) δ 13.12 (s, 1H), 10.70 (s, 1H), 7.83 (t, J = 1.9 Hz, 1H), 7.46 (d, J = 8.2 Hz, 1H), 7.32 (td, J = 8.1, 1.5 Hz, 1H), 7.10 (dd, J = 7.9, 2.1 Hz, 1H), 1.42 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 173.52, 167.03, 140.20, 133.04, 130.33, 123.03, 118.79, 117.69, 28.87, 16.98.
1-((4-chlorophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8D). White solid (55%), 1H NMR (400 MHz, DMSO-d6) δ 13.10 (s, 1H), 10.65 (s, 1H), 7.71–7.54 (m, 2H), 7.49–7.23 (m, 2H), 1.41 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 173.60, 166.80, 137.75, 128.56, 126.87, 120.85, 28.80, 16.95.
1-((3-bromophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8E). White solid (76%), 1H NMR (400 MHz, DMSO-d6) δ 13.12 (s, 1H), 10.69 (s, 1H), 7.97 (t, J = 2.0 Hz, 1H), 7.50 (dt, J = 7.5, 1.9 Hz, 1H), 7.39–7.15 (m, 2H), 1.42 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 174.03, 167.51, 140.83, 131.13, 126.43, 122.14, 122.01, 118.57, 29.38, 17.50.
1-((4-bromophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8F). White solid (84%), 1H NMR (400 MHz, DMSO-d6) δ 13.13 (s, 1H), 10.70 (s, 1H), 7.64–7.55 (m, 2H), 7.51–7.41 (m, 2H), 1.43 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 174.22, 167.32, 138.61, 131.95, 121.73, 115.43, 29.15, 17.70.
1-((3-methoxyphenyl)carbamoyl)cyclopropane-1-carboxylic acid (8G). White solid (65%), 1H NMR (400 MHz, DMSO-d6) δ 13.09 (s, 1H), 10.62 (s, 1H), 7.33 (t, J = 2.1 Hz, 1H), 7.20 (t, J = 8.1 Hz, 1H), 7.13 (dt, J = 8.2, 1.3 Hz, 1H), 6.63 (ddd, J = 8.1, 2.5, 1.0 Hz, 1H), 3.72 (s, 3H), 1.43 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 174.40, 167.20, 159.99, 140.42, 129.95, 112.05, 109.40, 105.50, 55.40, 29.01, 17.77.
1-((4-methoxyphenyl)carbamoyl)cyclopropane-1-carboxylic acid (8H). Off-white solid (70%), 1H NMR (400 MHz, DMSO-d6) δ 10.49 (s, 1H), 7.60–7.31 (m, 2H), 7.12–6.68 (m, 2H), 3.71 (s, 3H), 1.42 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 174.56, 166.77, 155.78, 132.36, 121.41, 114.27, 55.57, 28.59, 17.81.
1-(m-tolylcarbamoyl)cyclopropane-1-carboxylic acid (8I). White solid (75%), 1H NMR (400 MHz, DMSO-d6) δ 13.14 (s, 1H), 10.58 (s, 1H), 7.52–7.31 (m, 2H), 7.17 (t, J = 7.8 Hz, 1H), 6.87 (d, J = 7.4 Hz, 1H), 2.27 (s, 3H), 1.42 (s, 4H).
1-(p-tolylcarbamoyl)cyclopropane-1-carboxylic acid (8J). White solid (79%), 1H NMR (400 MHz, DMSO-d6) δ 13.11 (s, 1H), 10.54 (s, 1H), 7.60–7.36 (m, 2H), 7.20–6.99 (m, 2H), 2.25 (s, 3H), 1.42 (s, 4H).
1-((3-cyanophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8K). White solid (79%), 1H NMR (400 MHz, DMSO-d6) δ 13.15 (s, 1H), 10.84 (s, 1H), 8.11 (q, J = 1.3 Hz, 1H), 7.84 (ddd, J = 6.0, 3.3, 2.2 Hz, 1H), 7.69 – 7.39 (m, 2H), 1.43 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 173.88, 167.82, 140.06, 130.65, 127.37, 124.38, 122.49, 119.12, 112.03, 29.46, 17.44.
1-((4-cyanophenyl)carbamoyl)cyclopropane-1-carboxylic acid (8L). Yellow solid (79%), 1H NMR (400 MHz, DMSO-d6) δ 13.13 (s, 1H), 10.90 (s, 1H), 7.90–7.62 (m, 4H), 1.42 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 173.76, 167.90, 143.48, 133.70, 119.78, 119.49, 105.53, 29.85, 17.29.
1-((3-(trifluoromethyl)phenyl)carbamoyl)cyclopropane-1-carboxylic acid (8M). White solid (81%), 1H NMR (400 MHz, DMSO-d6) δ 13.13 (s, 1H), 10.83 (s, 1H), 8.13 (t, J = 1.9 Hz, 1H), 7.95–7.72 (m, 1H), 7.53 (t, J = 8.0 Hz, 1H), 7.46–7.30 (m, 1H), 1.43 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 173.92, 167.75, 140.04, 130.39, 130.34, 130.08, 129.77, 129.45, 128.60, 125.89, 123.33, 123.18, 120.13, 120.09, 115.89, 115.85, 29.49, 17.34.
1-((4-(trifluoromethyl)phenyl)carbamoyl)cyclopropane-1-carboxylic acid (8N). Yellow solid (82%), 1H NMR (400 MHz, DMSO-d6) δ 12.95 (s, 1H), 10.91 (s, 1H), 7.82 (d, J = 8.5 Hz, 2H), 7.65 (d, J = 8.5 Hz, 2H), 1.44 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 174.04, 167.78, 142.80, 126.48, 126.45, 126.41, 126.13, 124.03, 123.71, 123.43, 119.65, 29.44, 17.55.
1-(phenylcarbamoyl)cyclopropane-1-carboxylic acid (8O). White solid (66%), 1H NMR (400 MHz, DMSO-d6) δ 13.10 (s, 1H), 10.60 (s, 1H), 7.63–7.54 (m, 2H), 7.40–7.27 (m, 2H), 7.11–6.99 (m, 1H), 1.42 (s, 4H).
1-((4-chloro-3-(trifluoromethyl)phenyl)carbamoyl)cyclopropane-1-carboxylic acid (8P). White solid (67%), 1H NMR (400 MHz, DMSO-d6) δ 13.13 (s, 1H), 10.88 (s, 1H), 8.20 (d, J = 2.3 Hz, 1H), 7.85 (d, J = 8.8 Hz, 1H), 7.71–7.43 (m, 1H), 1.42 (s, 4H); 13C NMR (101 MHz, DMSO-d6) δ 173.24, 167.32, 138.23, 131.95, 131.91, 126.82, 126.51, 124.01, 121.28, 117.97, 117.91, 29.02, 16.75.
3.1.4. General Procedure for the Preparation of 10A−P
To a two-necked flask, compound 8A–P (30 mmol), 4-(aminomethyl)aniline dihydrochloride (30 mmol), EDCI (45 mmol), HOBt (36 mmol), DIEA (120 mmol) and DMF (30 mL) were charged. The mixture was stirred at room temperature for 12 h, then quenched by water. The mixture was extracted by ethyl acetate, and the combined organic layers were washed by saturated aqueous NaHCO3 solution, water and brine, dried by anhydrous magnesium sulfate. The solvent was evaporated, and the residue was purified by silica gel column chromatography.
N-(4-(aminomethyl)phenyl)-N-(3-fluorophenyl)cyclopropane-1,1-dicarboxamide(10A). Brown solid (69%), 1H NMR (400 MHz, DMSO-d6) δ 10.91 (s, 1H), 8.27 (s, 1H), 7.63 (d, J = 11.6 Hz, 1H), 7.32 (d, J = 4.4 Hz, 2H), 6.91 (dd, J = 21.6, 5.2 Hz, 3H), 6.51 (d, J = 8.1 Hz, 2H), 4.96 (s, 2H), 4.15 (d, J = 5.8 Hz, 2H), 1.39 (d, J = 3.1 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.18, 168.48, 162.05 (d, J = 240.9 Hz), 147.48, 140.40 (d, J = 11.0 Hz), 130.14 (d, J = 9.6 Hz), 128.15, 126.10, 115.54, 113.68, 109.85 (d, J = 20.9 Hz), 106.67 (d, J = 26.2 Hz), 42.35, 29.52, 15.92.
N-(4-(aminomethyl)phenyl)-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide (10B). Brown solid (78%),1H NMR (400 MHz, DMSO-d6) δ 10.69 (s, 1H), 8.31 (d, J = 5.9 Hz, 1H), 7.61 (dd, J = 8.8, 5.0 Hz, 2H), 7.13 (t, J = 8.7 Hz, 2H), 6.94 (d, J = 8.0 Hz, 2H), 6.51 (d, J = 8.0 Hz, 2H), 4.95 (s, 2H), 4.16 (d, J = 5.8 Hz, 2H), 1.39 (d, J = 6.0 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.24, 168.24, 158.15 (d, J = 240.5 Hz), 147.50, 135.02, 128.16, 126.10, 121.81 (d, J = 7.7 Hz), 115.10 (d, J = 21.9 Hz), 113.69, 42.35, 29.24, 15.84.
N-(4-(aminomethyl)phenyl)-N-(3-chlorophenyl)cyclopropane-1,1-dicarboxamide (10C). Brown solid (65%),1H NMR (400 MHz, DMSO-d6) δ 10.83 (s, 1H), 8.28 (t, J = 5.8 Hz, 1H), 7.85 (t, J = 2.1 Hz, 1H), 7.45 (ddd, J = 8.2, 2.1, 1.0 Hz, 1H), 7.32 (t, J = 8.1 Hz, 1H), 7.11 (ddd, J = 7.9, 2.1, 1.0 Hz, 1H), 6.95–6.88 (m, 2H), 6.52–6.46 (m, 2H), 4.95 (s, 2H), 4.15 (d, J = 5.8 Hz, 2H), 1.38 (d, J = 2.3 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.52, 169.00, 148.00, 140.64, 133.42, 130.71, 128.66, 126.57, 123.60, 119.92, 118.72, 114.15, 42.85, 30.14, 16.35.
N-(4-(aminomethyl)phenyl)-N-(4-chlorophenyl)cyclopropane-1,1-dicarboxamide(10D). Brown solid (70%),1H NMR (400 MHz, DMSO-d6) δ 10.79 (s, 1H), 8.27 (d, J = 5.9 Hz, 1H), 7.63 (d, J = 8.4 Hz, 2H), 7.35 (d, J = 8.4 Hz, 2H), 6.93 (d, J = 8.0 Hz, 2H), 6.50 (d, J = 7.9 Hz, 2H), 4.96 (s, 2H), 4.15 (d, J = 5.8 Hz, 2H), 1.38 (d, J = 4.6 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.17, 168.34, 147.47, 137.62, 128.45, 128.15, 127.02, 126.08, 121.46, 113.68, 42.35, 29.43, 15.88.
N-(4-(aminomethyl)phenyl)-N-(3-bromophenyl)cyclopropane-1,1-dicarboxamide (10E). Brown solid (50%), 1H NMR (400 MHz, DMSO-d6) δ 10.80 (s, 1H), 8.28 (t, J = 5.8 Hz, 1H), 7.99 (s, 1H), 7.49 (d, J = 6.8 Hz, 1H), 7.25 (d, J = 6.5 Hz, 2H), 6.93 (d, J = 8.0 Hz, 2H), 6.50 (d, J = 7.9 Hz, 2H), 4.96 (s, 2H), 4.14 (d, J = 5.7 Hz, 2H), 1.37 (s, 4H). 13C NMR (101 MHz, DMSO-d6) δ 169.99, 168.50, 147.46, 140.27, 130.52, 128.15, 126.11, 126.00, 122.29, 121.39, 118.62, 113.67, 42.36, 29.65, 15.83.
N-(4-(aminomethyl)phenyl)-N-(4-bromophenyl)cyclopropane-1,1-dicarboxamide (10F). Brown solid (55%),1H NMR (400 MHz, DMSO-d6) δ 10.80 (s, 1H), 8.28 (t, J = 5.8 Hz, 1H), 7.58 (d, J = 8.6 Hz, 2H), 7.47 (d, J = 8.5 Hz, 2H), 6.93 (d, J = 7.9 Hz, 2H), 6.50 (d, J = 7.9 Hz, 2H), 4.95 (s, 2H), 4.15 (d, J = 5.6 Hz, 2H), 1.38 (d, J = 4.7 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.18, 168.35, 147.49, 138.03, 131.36, 128.16, 126.07, 121.82, 115.05, 113.67, 42.35, 29.44, 15.92.
N-(4-(aminomethyl)phenyl)-N-(3-methoxyphenyl)cyclopropane-1,1-dicarboxamide(10G). Brown solid (71%),1H NMR (400 MHz, DMSO-d6) δ 10.76 (s, 1H), 8.25 (t, J = 5.9 Hz, 1H), 7.29 (s, 1H), 7.19 (t, J = 8.1 Hz, 1H), 7.11 (d, J = 8.1 Hz, 1H), 6.92 (d, J = 8.0 Hz, 2H), 6.64 (dd, J = 8.1, 2.4 Hz, 1H), 6.50 (d, J = 8.0 Hz, 2H), 4.98 (s, 2H), 4.14 (d, J = 5.7 Hz, 2H), 3.72 (s, 3H), 1.38 (d, J = 7.8 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.48, 168.17, 159.45, 147.42, 139.78, 129.38, 128.13, 126.10, 113.70, 112.05, 109.13, 105.45, 54.97, 42.30, 29.20, 15.98.
N-(4-(aminomethyl)phenyl)-N-(4-methoxyphenyl)cyclopropane-1,1-dicarboxamide (10H). Brown solid (74%),1H NMR (400 MHz, DMSO-d6) δ 10.54 (s, 1H), 8.36 (t, J = 5.4 Hz, 1H), 7.48 (d, J = 8.6 Hz, 2H), 6.93 (d, J = 8.0 Hz, 2H), 6.87 (d, J = 8.6 Hz, 2H), 6.50 (d, J = 8.0 Hz, 2H), 4.97 (s, 2H), 4.14 (d, J = 5.7 Hz, 2H), 3.71 (s, 3H), 1.38 (d, J = 7.7 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.48, 167.98,155.42, 147.51, 131.66, 128.15, 126.08, 121.61, 113.68, 55.12, 42.30, 28.88, 15.90.
N-(4-(aminomethyl)phenyl)-N-(m-tolyl)cyclopropane-1,1-dicarboxamide (10I). Brown solid (62%),1H NMR (400 MHz, DMSO-d6) δ 10.73 (s, 1H), 8.28 (s, 1H), 7.49–7.25 (m, 2H), 7.17 (t, J = 7.7 Hz, 1H), 6.51 (d, J = 7.9 Hz, 2H), 4.96 (s, 2H), 4.15 (d, J = 5.7 Hz, 2H), 2.27 (s, 3H), 1.40 (d, J = 8.3 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.59, 168.12, 147.51, 138.47, 137.85, 128.46, 128.16, 126.06, 124.19,120.41, 117.01, 113.69, 42.32, 28.97, 21.07, 16.07.
N-(4-(aminomethyl)phenyl)-N-(p-tolyl)cyclopropane-1,1-dicarboxamide (10J). Brown solid (69%),1H NMR (400 MHz, DMSO-d6) δ 10.67 (s, 1H), 8.29 (d, J = 5.9 Hz, 1H), 7.46 (dd, J = 8.2, 2.1 Hz, 2H), 7.10 (d, J = 7.9 Hz, 2H), 6.94 (d, J = 7.8 Hz, 2H), 6.52 (dd, J = 8.2, 2.2 Hz, 2H), 4.97 (s, 2H), 4.16 (d, J = 5.5 Hz, 2H), 2.25 (s, 3H), 1.40 (dd, J = 10.4, 2.7 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.56, 168.04, 147.48, 136.05, 132.45, 129.00, 128.16, 126.09, 119.92, 113.71, 42.32, 28.94, 20.41, 16.01.
N-(4-(aminomethyl)phenyl)-N-(3-cyanophenyl)cyclopropane-1,1-dicarboxamide (10K). Brown solid (59%),1H NMR (400 MHz, DMSO-d6) δ 10.94 (s, 1H), 8.29 (s, 1H), 8.13 (s, 1H), 7.85 – 7.68 (m, 1H), 7.51 (d, J = 4.7 Hz, 2H), 6.92 (d, J = 7.8 Hz, 2H), 6.50 (d, J = 7.9 Hz, 2H), 4.98 (s, 2H), 4.15 (d, J = 5.8 Hz, 2H), 1.39 (s, 4H). 13C NMR (101 MHz, DMSO-d6) δ 169.87, 168.77, 147.49, 139.51, 130.01, 128.16, 126.92, 126.08, 124.48, 122.73, 118.67, 113.66, 111.37, 42.37, 29.72, 15.85.
N-(4-(aminomethyl)phenyl)-N-(4-cyanophenyl)cyclopropane-1,1-dicarboxamide (10L). Brown solid (63%),1H NMR (400 MHz, DMSO-d6) δ 11.12 (s, 1H), 8.29 (s, 1H), 7.81 (d, J = 8.9 Hz, 2H), 7.76 (d, J = 8.9 Hz, 2H), 6.93 (d, J = 8.4 Hz, 2H), 6.60–6.34 (m, 2H), 5.01 (s, 2H), 4.15 (d, J = 5.7 Hz, 2H), 1.40 (s, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.40, 169.29, 147.96, 143.47, 133.56, 128.66, 126.56, 120.28, 119.54, 114.16, 105.54, 42.86, 30.46, 16.44.
N-(4-(aminomethyl)phenyl)-N-(3-(trifluoromethyl)phenyl)cyclopropane-1,1-dicarboxamide (10M). Brown solid (76%), 1H NMR (400 MHz, DMSO-d6) δ 10.91 (s, 1H), 8.31 (t, J = 5.8 Hz, 1H), 8.15 (s, 1H), 7.79 (d, J = 8.2 Hz, 1H), 7.53 (t, J = 8.0 Hz, 1H), 7.40 (d, J = 7.7 Hz, 1H), 6.93 (d, J = 7.9 Hz, 2H), 6.50 (d, J = 7.9 Hz, 2H), 4.95 (s, 2H), 4.16 (d, J = 5.6 Hz, 2H), 1.39 (d, J = 4.6 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 169.87, 168.74, 147.49, 139.51, 129.71, 129.30 (d, J = 31.7 Hz), 128.15, 126.11, 124.10 (d, J = 272.2 Hz), 123.41, 119.68 (d, J = 3.8 Hz), 116.05 (d, J = 4.4 Hz), 113.65, 42.38, 29.80, 15.75.
N-(4-(aminomethyl)phenyl)-N-(4-(trifluoromethyl)phenyl)cyclopropane-1,1-dicarboxamide (10N). Brown solid (70%), 1H NMR (400 MHz, DMSO-d6) δ 11.08 (s, 1H), 8.28 (d, J = 5.9 Hz, 1H), 7.83 (d, J = 8.4 Hz, 2H), 7.66 (d, J = 8.4 Hz, 2H), 6.93 (d, J = 7.9 Hz, 2H), 6.51 (d, J = 7.9 Hz, 2H), 4.95 (s, 2H), 4.16 (d, J = 5.6 Hz, 2H), 1.41 (d, J = 3.2 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.12, 168.71, 147.50, 142.27, 128.16, 126.06, 125.84 (d, J = 3.6 Hz), 124.62 (d, J = 213.2 Hz), 123.12 (d, J = 26.2 Hz), 119.68, 113.66, 42.37, 29.63, 16.02.
N-(4-(aminomethyl)phenyl)-N-phenylcyclopropane-1,1-dicarboxamide (10O). Brown solid (78%), 1H NMR (400 MHz, DMSO-d6) δ 10.75 (s, 1H), 8.28 (t, J = 5.9 Hz, 1H), 7.58 (d, J = 8.0 Hz, 2H), 7.30 (t, J = 7.8 Hz, 2H), 7.06 (t, J = 7.4 Hz, 1H), 6.93 (d, J = 7.9 Hz, 2H), 6.51 (d, J = 7.9 Hz, 2H), 4.96 (s, 2H), 4.15 (d, J = 5.7 Hz, 2H), 1.40 (d, J = 8.7 Hz, 4H). 13C NMR (101 MHz, DMSO-d6) δ 170.47, 168.19, 147.50, 138.59, 128.61, 128.15, 126.07, 123.49, 119.89, 113.68, 42.33, 29.14, 15.98.
N-(4-(aminomethyl)phenyl)-N-(4-chloro-3-(trifluoromethyl)phenyl)cyclopropane-1,1-dicarboxamide (10P). Brown solid (80%),1H NMR (400 MHz, DMSO-d6) δ 10.94 (s, 1H), 8.31 (t, J = 5.9 Hz, 1H), 8.25 (d, J = 2.3 Hz, 1H), 7.85 (dd, J = 8.8, 2.3 Hz, 1H), 7.63 (d, J = 8.7 Hz, 1H), 6.93 (d, J = 7.9 Hz, 2H), 6.50 (d, J = 7.9 Hz, 2H), 4.95 (s, 2H), 4.15 (d, J = 5.7 Hz, 2H), 1.38 (s, 4H).13C NMR (101 MHz, DMSO-d6) δ 169.59, 168.83, 147.49, 138.28, 131.79, 128.15, 126.52 (d, J = 30.6 Hz), 126.09, 124.62, 123.99, 122.73 (d, J = 272.8 Hz), 118.63 (d, J = 5.7 Hz), 113.64, 42.39, 30.03, 15.65.
3.1.5. General Procedure for the Preparation of 12A-P
To a suspension of 6-(2-chloro-5-fluoropyrimidin-4-yl)-1-isopropyl-1H-benzo[d](1,2,3)riazole (6) (583.4 mg, 2 mmol) in 20 mL 1,4-dioxane, the following compounds were added: 10A–P (2 mmol), Pd(OAc)2 (11 mg, 0.05 mmol), BINAP (62 mg, 0.1 mmol) and Cs2CO3 (978 mg, 3 mmol) and the flask was purged with Ar. Then, the flask was sealed and the mixture was heated for 12 h at 100℃. The reaction was cooled to RT, the solvent was removed under reduced pressure, and the residue was purified by silica gel column chromatography to obtain 12A–P.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(3-fluorophenyl)cyclopropane-1,1-dicarboxamide(12A). Light yellow solid; 43% yield; mp 104.9 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.90 (s, 1H), 9.85 (s, 1H), 8.68 (d, J = 3.3 Hz, 1H), 8.51 (s, 1H), 8.44 (t, J = 5.9 Hz, 1H), 8.21 (d, J = 8.8 Hz, 1H), 8.02 (d, J = 8.8 Hz, 1H), 7.74 (d, J = 8.5 Hz, 2H), 7.63 (d, J = 11.5 Hz, 1H), 7.34—7.26 (m, 2H), 7.22 (d, J = 8.4 Hz, 2H), 6.90–6.83 (m, 1H), 5.31 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.7 Hz, 2H), 1.67 (d, J = 6.7 Hz, 6H), 1.41 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.88, 168.87, 162.53 (d, J = 240.9 Hz), 156.97 (d, J = 2.7 Hz), 151.79, 151.03 (d, J = 9.6 Hz), 149.28, 148.26 (d, J = 25.6 Hz), 146.63, 140.93 (d, J = 11.0 Hz), 139.65, 132.87, 132.36, 132.30, 130.63 (d, J = 9.5 Hz), 127.95, 124.69 (d, J = 5.6 Hz), 119.98, 119.07, 116.02 (d, J = 2.5 Hz), 112.23 (d, J = 6.6 Hz), 110.34 (d, J = 21.1 Hz), 107.15 (d, J = 26.2 Hz), 51.76, 42.75, 30.17, 22.47, 16.44. 19F NMR (376 MHz, DMSO-d6) δ -107.43(s, 1F), -145.82 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H29F2N8O2+ 583.2376, found 583.2367 [M + H]+. HPLC purity 99%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide (12B).Light yellow solid; 47% yield; mp 109.5 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.67 (s, 1H), 9.84 (s, 1H), 8.68 (d, J = 3.4 Hz, 1H), 8.51 (s, 1H), 8.45 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.03 (d, J = 8.9 Hz, 1H), 7.76–7.72 (m, 2H), 7.65–7.56 (m, 2H), 7.22 (d, J = 8.3 Hz, 2H), 7.12 (t, J = 8.9 Hz, 2H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.41 (d, J = 2.4 Hz, 4H). 13C NMR (101 MHz, DMSO) δ 170.45, 168.14, 158.14 (d, J = 239.8 Hz), 156.89–156.32 (m), 151.30, 150.54 (d, J = 8.7 Hz), 148.79, 147.76 (d, J = 26.1 Hz), 146.14, 139.15, 135.02 (d, J = 2.1 Hz), 132.40, 131.87, 127.45, 124.19 (d, J = 5.5 Hz), 121.80 (d, J = 7.7 Hz), 119.49, 118.61, 115.20, 114.97, 111.73 (d, J = 6.5 Hz), 51.27, 42.25, 29.38, 21.97, 15.85. 19F NMR (376 MHz, DMSO-d6) δ −114.18(s, 1F), −145.83 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H29F2N8O2+ 583.2376, found 583.2377 [M + H]+. HPLC purity 98%.
N-(3-chlorophenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12C). Light yellow solid; 56% yield; mp 119.4 °C.1H NMR (400 MHz, DMSO-d6) δ 10.83 (s, 1H), 9.85 (s, 1H), 8.68 (d, J = 3.4 Hz, 1H), 8.51 (s, 1H), 8.45 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.02 (d, J = 8.8 Hz, 1H), 7.85 (s, 1H), 7.74 (d, J = 8.5 Hz, 2H), 7.46 (d, J = 8.3 Hz, 1H), 7.30 (t, J = 8.1 Hz, 1H), 7.22 (d, J = 8.3 Hz, 2H), 7.09 (dd, J = 7.8, 2.2 Hz, 1H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.7 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.40 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.75, 168.92, 157.07–156.90 (m), 151.79, 151.04 (d, J = 9.7 Hz), 149.28, 148.26 (d, J = 26.1 Hz), 146.63, 140.65, 139.65, 133.41, 132.88, 132.36, 132.30, 130.69, 127.96, 124.70 (d, J = 5.9 Hz), 123.60, 119.99, 119.92, 119.07, 118.71, 112.23 (d, J = 6.6 Hz), 51.77, 42.77, 30.26, 22.48, 16.38. 19F NMR (376 MHz, DMSO-d6) δ −145.8 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H29ClFN8O2+ 599.2081, found 599.2077 [M + H]+. HPLC purity 99%.
N-(4-chlorophenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12D). Light yellow solid; 52% yield; mp118 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.79 (s, 1H), 9.84 (s, 1H), 8.67 (d, J = 3.4 Hz, 1H), 8.51–8.49 (m, 1H), 8.44 (t, J = 5.9 Hz, 1H), 8.21 (d, J = 8.8 Hz, 1H), 8.02 (d, J = 8.8 Hz, 1H), 7.73 (d, J = 8.6 Hz, 2H), 7.63 (d, J = 8.9 Hz, 2H), 7.32 (d, J = 8.9 Hz, 2H), 7.21 (d, J = 8.6 Hz, 2H), 5.31 (p, J = 6.7 Hz, 1H), 4.28 (d, J = 5.8 Hz, 2H), 1.67 (d, J = 6.7 Hz, 6H), 1.47–1.32 (m, 4H). 13C NMR (101 MHz, DMSO) δ 170.91, 168.73, 156.98 (d, J = 2.4 Hz), 151.79, 151.04 (d, J = 9.2 Hz), 149.28, 148.25 (d, J = 25.5 Hz), 146.63, 139.65, 138.13, 132.87, 132.36, 132.30, 128.93, 127.96, 127.51, 124.70 (d, J = 6.0 Hz), 121.94, 119.98, 119.09, 112.23 (d, J = 6.7 Hz), 51.76, 42.75, 30.06, 22.47, 16.42. 19F NMR (376 MHz, DMSO-d6) δ −145.81 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H29ClFN8O2+ 599.2081, found 599.2072 [M + H]+. HPLC purity 96%.
N-(3-bromophenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12E). Light yellow solid; 44% yield; mp 114.6 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.80 (s, 1H), 9.85 (s, 1H), 8.68 (d, J = 3.4 Hz, 1H), 8.51 (s, 1H), 8.45 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.08-7.96 (m, 2H), 7.74 (d, J = 8.4 Hz, 2H), 7.49 (d, J = 2.2 Hz, 1H), 7.27–7.16 (m, 4H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.40 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.72, 168.91, 156.98 (d, J = 3.0 Hz), 151.79, 151.03 (d, J = 9.3 Hz), 149.28, 148.26 (d, J = 25.9 Hz), 146.63, 140.79, 139.65, 132.88, 132.36, 132.31, 131.00, 127.96, 126.50, 124.70 (d, J = 5.9 Hz), 122.78, 121.88, 119.99, 119.10, 119.08, 112.23 (d, J = 6.7 Hz), 51.77, 42.77, 30.29, 22.48, 16.36. 19F NMR (376 MHz, DMSO-d6) δ −145.81 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H29BrFN8O2+ 643.1575, found 643.1572 [M + H]+. HPLC purity 97%.
N-(4-bromophenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12F). Light yellow solid; 42% yield; mp 214 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.79 (s, 1H), 9.84 (s, 1H), 8.68 (d, J = 3.5 Hz, 1H), 8.51 (s, 1H), 8.44 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.7 Hz, 1H), 8.02 (d, J = 8.8 Hz, 1H), 7.74 (d, J = 8.2 Hz, 2H), 7.58 (d, J = 9.0 Hz, 2H), 7.46 (d, J = 8.9 Hz, 2H), 7.21 (d, J = 8.3 Hz, 2H), 5.32 (p, J = 6.8 Hz, 1H), 4.29 (d, J = 5.7 Hz, 2H), 1.68 (d, J = 6.8 Hz, 6H), 1.41 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.40, 168.25, 156.48 (d, J = 2.4 Hz), 151.30, 150.56 (d, J = 9.4 Hz), 148.79, 147.77 (d, J = 26.1 Hz), 146.13, 139.15, 138.06, 132.37, 131.87, 131.81, 131.35, 127.46, 124.20 (d, J = 5.6 Hz), 121.81, 119.50, 118.60, 115.04, 111.74 (d, J = 6.8 Hz), 51.27, 42.26, 29.60, 21.99, 15.93. 19F NMR (376 MHz, DMSO-d6) δ −145.81 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H29BrFN8O2+ 643.1575, found 643.1576 [M + H]+. HPLC purity 95%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(3-methoxyphenyl)cyclopropane-1,1-dicarboxamide (12G).Light yellow solid; 47% yield; mp 101 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.76 (s, 1H), 9.84 (s, 1H), 8.68 (d, J = 3.3 Hz, 1H), 8.51 (s, 1H), 8.42 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.9 Hz, 1H), 8.03 (dd, J = 8.8, 1.4 Hz, 1H), 7.81–7.64 (m, 2H), 7.31 (d, J = 2.3 Hz, 1H), 7.26–7.09 (m, 4H), 6.63 (ddd, J = 8.1, 2.7, 1.2 Hz, 1H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.7 Hz, 2H), 3.71 (s, 3H), 1.68 (d, J = 6.7 Hz, 6H), 1.42 (d, J = 3.9 Hz, 4H). 13C NMR (101 MHz, DMSO) δ 170.71, 168.08, 159.44, 156.48 (d, J = 2.3 Hz), 151.30, 150.54 (d, J = 8.9 Hz), 148.79, 147.76 (d, J = 25.8 Hz), 146.14, 139.80, 139.16, 132.37, 131.84 (d, J = 5.4 Hz), 129.36, 128.12, 127.44, 124.19 (d, J = 5.6 Hz), 119.49, 118.60, 113.67, 112.06, 111.73 (d, J = 6.6 Hz), 109.10, 105.49, 54.95, 51.28, 42.23, 29.33, 21.97, 16.03. 19F NMR (376 MHz, DMSO-d6) δ -145.79 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H32FN8O3+ 595.2576, found 595.2578 [M + H]+. HPLC purity 99%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(4-methoxyphenyl)cyclopropane-1,1-dicarboxamide (12H). Light yellow solid; 55% yield; mp 181 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.50 (s, 1H), 9.84 (s, 1H), 8.68 (d, J = 3.3 Hz, 1H), 8.51 (s, 1H), 8.47 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.7 Hz, 1H), 8.03 (d, J = 8.8 Hz, 1H), 7.74 (d, J = 8.6 Hz, 2H), 7.49 (d, J = 9.0 Hz, 2H), 7.22 (d, J = 8.6 Hz, 2H), 6.86 (d, J = 9.0 Hz, 2H), 5.31 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 6.0 Hz, 2H), 3.70 (s, 3H), 1.68 (d, J = 6.5 Hz, 6H), 1.40 (d, J = 6.4 Hz, 4H). 13C NMR (101 MHz, DMSO) δ 170.68, 167.84, 156.48 (d, J = 2.4 Hz), 155.42, 151.30, 150.53 (d, J = 9.1 Hz), 148.79, 147.78 (d, J = 25.9 Hz), 146.14, 139.16, 132.39, 131.87, 131.81, 131.69, 127.45, 124.20 (d, J = 6.0 Hz), 121.59, 119.49, 118.61, 113.67, 111.73 (d, J = 6.8 Hz), 55.10, 51.28, 42.23, 29.04, 21.97, 15.90. 19F NMR (376 MHz, DMSO-d6) δ −145.79 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H32FN8O3+ 595.2576, found 595.2576 [M + H]+. HPLC purity 97%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(m-tolyl)cyclopropane-1,1-dicarboxamide (12I). Light yellow solid; 61% yield; mp 85 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.72 (s, 1H), 9.85 (s, 1H), 8.68 (d, J = 3.4 Hz, 1H), 8.52 (s, 1H), 8.44 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.03 (dt, J = 8.8, 1.4 Hz, 1H), 7.83–7.72 (m, 2H), 7.46–7.36 (m, 2H), 7.22 (d, J = 8.5 Hz, 2H), 7.16 (t, J = 7.8 Hz, 1H), 6.86 (d, J = 7.5 Hz, 1H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 2.25 (s, 3H), 1.68 (d, J = 6.7 Hz, 6H), 1.42 (d, J = 3.8 Hz, 4H). 13C NMR (101 MHz, DMSO) δ 171.31, 168.52, 156.98 (d, J = 2.1 Hz), 151.80, 151.03 (d, J = 9.4 Hz), 149.29, 148.27 (d, J = 25.8 Hz), 146.64, 139.67, 138.99, 138.32, 132.86, 132.37, 132.32, 128.93, 127.95, 124.70 (d, J = 5.3 Hz), 120.90, 119.99, 119.10, 117.50, 112.23 (d, J = 6.6 Hz), 51.77, 42.73, 29.60, 22.46, 21.55, 16.60. 19F NMR (376 MHz, DMSO-d6) δ −145.81 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H32FN8O2+ 579.2627, found 579.2626 [M + H]+. HPLC purity98%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(p-tolyl)cyclopropane-1,1-dicarboxamide (12J). Light yellow solid; 57% yield; mp 190 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.65 (s, 1H), 9.84 (s, 1H), 8.67 (d, J = 3.3 Hz, 1H), 8.51 (s, 1H), 8.45 (t, J = 5.9 Hz, 1H), 8.21 (d, J = 8.8 Hz, 1H), 8.02 (d, J = 8.8 Hz, 1H), 7.75 (d, J = 8.3 Hz, 2H), 7.46 (d, J = 8.1 Hz, 2H), 7.22 (d, J = 8.3 Hz, 2H), 7.07 (d, J = 8.1 Hz, 2H), 5.31 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 2.22 (s, 3H), 1.67 (d, J = 6.7 Hz, 6H), 1.48–1.33 (m, 4H). 13C NMR (101 MHz, DMSO) δ 171.29, 168.44, 156.98 (d, J = 2.8 Hz), 151.80, 150.99 (d, J = 8.7 Hz), 149.29, 148.26 (d, J = 25.8 Hz), 146.64, 139.67, 136.56, 132.93, 132.86, 132.36, 132.32, 129.46, 127.94, 124.69 (d, J = 5.8 Hz), 120.41, 119.98, 119.11, 112.21 (d, J = 6.6 Hz), 51.77, 42.73, 29.56, 22.46, 20.88, 16.54. 19F NMR (376 MHz, DMSO-d6) δ −145.76 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H32FN8O2+ 579.2627, found 579.2623 [M + H]+. HPLC purity 95%.
N-(3-cyanophenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12K). Light yellow solid; 63% yield; mp 108.4 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.94 (s, 1H), 9.84 (s, 1H), 8.67 (d, J = 3.3 Hz, 1H), 8.51 (s, 1H), 8.46 (t, J = 5.9 Hz, 1H), 8.21 (d, J = 8.8 Hz, 1H), 8.14 (s, 1H), 8.02 (d, J = 8.8 Hz, 1H), 7.83 (ddd, J = 5.2, 4.1, 2.2 Hz, 1H), 7.74 (d, J = 8.6 Hz, 2H), 7.53–7.47 (m, 2H), 7.21 (d, J = 8.6 Hz, 2H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.41 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.59, 169.18, 156.97 (d, J = 2.2 Hz), 151.79, 151.05 (d, J = 9.4 Hz), 149.28, 148.25 (d, J = 26.1 Hz), 146.63, 140.02, 139.65, 132.87, 132.36, 132.30, 130.50, 127.96, 127.42, 124.96, 124.70 (d, J = 5.9 Hz), 123.21, 119.99, 119.15, 119.08, 112.23 (d, J = 7.0 Hz), 111.86, 51.76, 42.78, 30.37, 22.48, 16.36. 19F NMR (376 MHz, DMSO-d6) δ −145.81 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H29FN9O2+ 590.2423, found 590.2418 [M + H]+. HPLC purity 99%.
N-(4-cyanophenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12L). Light yellow solid; 59% yield; mp 241 °C. 1H NMR (400 MHz, DMSO-d6) δ 11.10 (s, 1H), 9.83 (s, 1H), 8.67 (d, J = 3.4 Hz, 1H), 8.51 (s, 1H), 8.43 (t, J = 5.9 Hz, 1H), 8.21 (d, J = 8.8 Hz, 1H), 8.02 (d, J = 8.9 Hz, 1H), 7.81 (d, J = 8.9 Hz, 2H), 7.77–7.71 (m, 4H), 7.21 (d, J = 8.6 Hz, 2H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.7 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.42 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.64, 169.21, 156.98 (d, J = 2.3 Hz), 151.80, 151.05 (d, J = 9.4 Hz), 149.29, 148.24 (d, J = 25.6 Hz), 146.63, 143.47, 139.65, 133.54, 132.85, 132.36, 132.30, 127.96, 124.69 (d, J = 6.0 Hz), 120.30, 119.99, 119.50, 119.09, 112.23 (d, J = 6.6 Hz), 105.57, 51.77, 42.78, 30.57, 22.48, 16.46. 19F NMR (376 MHz, DMSO-d6) δ −145.82 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H29FN9O2+ 590.2423, found 590.2420 [M + H]+. HPLC purity 96%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(3-(trifluoromethyl)phenyl)cyclopropane-1,1-dicarboxamide (12M). Light yellow solid; 46% yield; mp 189 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.90 (s, 1H), 9.84 (s, 1H), 8.68 (d, J = 3.4 Hz, 1H), 8.51 (d, J = 1.3 Hz, 1H), 8.47 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.15 (s, 1H), 8.02 (d, J = 8.9 Hz, 1H), 7.79 (d, J = 8.4 Hz, 1H), 7.74 (d, J = 8.6 Hz, 2H), 7.52 (t, J = 8.0 Hz, 1H), 7.39 (d, J = 7.6 Hz, 1H), 7.22 (d, J = 8.7 Hz, 2H), 5.32 (p, J = 6.7 Hz, 1H), 4.30 (d, J = 5.8 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.41 (d, J = 1.9 Hz, 4H). 13C NMR (101 MHz, DMSO) δ 170.58, 169.16, 156.99 (d, J = 2.7 Hz), 151.79, 151.06 (d, J = 9.1 Hz), 149.29, 148.25 (d, J = 25.3 Hz), 146.64, 140.02, 139.65, 132.91, 132.36, 132.32, 130.21, 129.78 (d, J = 31.4 Hz), 127.96, 125.95, 124.69 (d, J = 6.0 Hz), 123.91, 120.20 (d, J = 4.2 Hz), 119.99, 119.09, 116.58 (d, J = 7.1 Hz), 112.23 (d, J = 6.6 Hz), 51.76, 42.79, 30.44, 22.46, 16.27. 19F NMR (376 MHz, DMSO-d6) δ -56.5(s, 3F), −145.86 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H29F4N8O2+ 633.2344, found 633.2347 [M + H]+. HPLC purity 99%.
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-(4-(trifluoromethyl)phenyl)cyclopropane-1,1-dicarboxamide (12N). Light yellow solid; 48% yield; mp 219 °C. 1H NMR (400 MHz, DMSO-d6) δ 11.06 (s, 1H), 9.84 (s, 1H), 8.68 (d, J = 3.3 Hz, 1H), 8.51 (d, J = 1.3 Hz, 1H), 8.44 (t, J = 5.8 Hz, 1H), 8.22 (dd, J = 8.7, 0.8 Hz, 1H), 8.02 (dt, J = 8.8, 1.3 Hz, 1H), 7.83 (d, J = 8.4 Hz, 2H), 7.78 – 7.71 (m, 2H), 7.64 (d, J = 8.6 Hz, 2H), 7.26–7.17 (m, 2H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.43 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.82, 169.11, 156.99 (d, J = 2.6 Hz), 151.79, 151.06 (d, J = 9.7 Hz), 149.29, 148.25 (d, J = 25.6 Hz), 146.64, 142.78, 139.66, 132.86, 132.36, 132.30, 127.96, 126.34 (d, J = 4.5 Hz), 126.17, 124.69 (d, J = 6.0 Hz), 124.12–123.42 (m), 120.19, 119.99, 119.09, 112.23 (d, J = 6.6 Hz), 51.76, 42.77, 30.30, 22.47, 16.51. 19F NMR (376 MHz, DMSO-d6) δ −55.52(s, 3F), −145.85 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H29F4N8O2+ 633.2344, found 633.2343 [M + H]+. HPLC purity 98%.
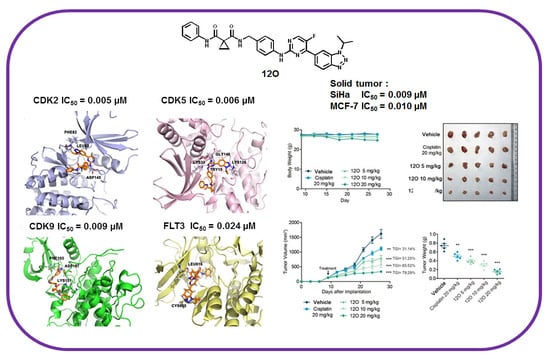
N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)-N-phenylcyclopropane-1,1-dicarboxamide (12O). Light yellow solid; 41% yield; mp 103 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.74 (s, 1H), 9.85 (s, 1H), 8.68 (dd, J = 3.4, 1.0 Hz, 1H), 8.51 (d, J = 1.3 Hz, 1H), 8.44 (t, J = 5.9 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.02 (dt, J = 8.8, 1.3 Hz, 1H), 7.84–7.71 (m, 2H), 7.65–7.54 (m, 2H), 7.28 (dd, J = 8.5, 7.3 Hz, 2H), 7.24–7.18 (m, 2H), 7.09–6.97 (m, 1H), 5.31 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 1.67 (d, J = 6.7 Hz, 6H), 1.45–1.35 (m, 4H). 13C NMR (101 MHz, DMSO) δ 171.20, 168.59, 156.98 (d, J = 2.0 Hz), 151.79, 151.03 (d, J = 9.8 Hz), 149.28, 148.28 (d, J = 26.2 Hz), 146.63, 139.65, 139.11, 132.87, 132.36, 132.31, 129.08, 127.95, 124.70 (d, J = 5.8 Hz), 123.98, 120.38, 119.99, 119.09, 112.24 (d, J = 6.3 Hz), 51.77, 42.73, 29.77, 22.47, 16.52. 19F NMR (376 MHz, DMSO-d6) δ −145.81 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C31H30FN8O2+ 565.2470, found 565.2468 [M + H]+. HPLC purity 99%.
N-(4-chloro-3-(trifluoromethyl)phenyl)-N-(4-((5-fluoro-4-(1-isopropyl-1H-benzo[d](1,2,3)triazol-6-yl)pyrimidin-2-yl)amino)benzyl)cyclopropane-1,1-dicarboxamide (12P). Light yellow solid; 44% yield; mp 108.1 °C. 1H NMR (400 MHz, DMSO-d6) δ 10.93 (s, 1H), 9.84 (s, 1H), 8.67 (d, J = 3.3 Hz, 1H), 8.51 (d, J = 1.3 Hz, 1H), 8.48 (t, J = 5.9 Hz, 1H), 8.25 (d, J = 2.6 Hz, 1H), 8.22 (dd, J = 8.8, 0.7 Hz, 1H), 8.02 (dt, J = 8.8, 1.4 Hz, 1H), 7.85 (dd, J = 8.8, 2.6 Hz, 1H), 7.73 (d, J = 8.6 Hz, 2H), 7.62 (d, J = 8.8 Hz, 1H), 7.21 (d, J = 8.6 Hz, 2H), 5.32 (p, J = 6.7 Hz, 1H), 4.29 (d, J = 5.8 Hz, 2H), 1.68 (d, J = 6.7 Hz, 6H), 1.40 (s, 4H). 13C NMR (101 MHz, DMSO) δ 170.29, 169.23, 156.97 (d, J = 2.2 Hz), 151.79, 151.06 (d, J = 8.3 Hz), 149.28, 148.24 (d, J = 26.2 Hz), 146.63, 139.65, 138.80, 132.89, 132.36, 132.30, 127.97, 126.99 (d, J = 31.0 Hz), 125.11, 124.68 (d, J = 5.8 Hz), 124.53 (d, J = 8.3 Hz), 121.85, 119.98, 119.14, 119.07, 112.22 (d, J = 6.3 Hz), 51.76, 42.80, 30.69, 22.47, 16.15. 19F NMR (376 MHz, DMSO-d6) δ −56.74(s, 3F), −145.88 (s, 1F). ESI-HRMS m/z calcd. for chemical formula: C32H28ClF4N8O2+ 667.1954, found 667.1953 [M + H]+. HPLC purity 96%.