Solubility and Thermal Degradation of Quercetin in CO2-Expanded Liquids
Abstract
:1. Introduction
2. Results and Discussion
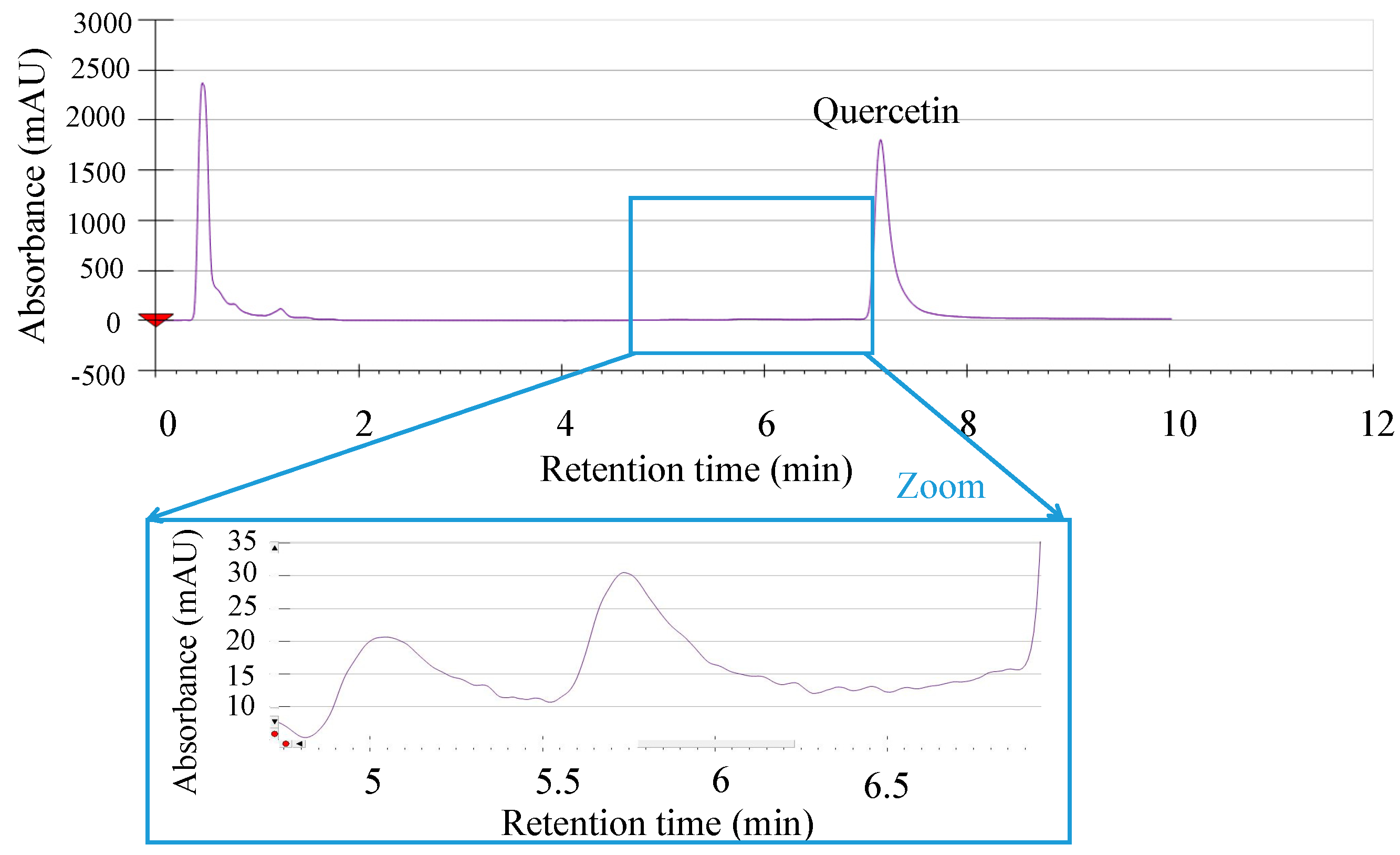
2.1. Thermal Degradation Measurements
2.2. Solubility Measurements
3. Materials and Methods
3.1. Chemicals
3.2. Equipment Setup
3.3. Thermal Degradation Measurements
3.4. UHPLC-QTOF/MS Analysis
3.5. Solubility Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Darband, S.G.; Kaviani, M.; Yousefi, B.; Sadighparvar, S.; Pakdel, F.G.; Attari, J.A.; Mohebbi, I.; Naderi, S.; Majidinia, M. Quercetin: A functional dietary flavonoid with potential chemo-preventive properties in colorectal cancer. J. Cell. Phys. 2018, 233, 6544–6560. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Song, Y.; Liang, Y.N.; Li, R. Quercetin treatment improves renal function and protects the kidney in a rat model of adenine-induced chronic kidney disease. Med. Sci. Mon. 2018, 24, 4760–4766. [Google Scholar] [CrossRef] [PubMed]
- Ben Yakoub, A.R.; Abdehedi, O.; Jridi, M.; Elfalleh, W.; Nasri, M.; Ferchichi, A. Flavonoids, phenols, antioxidant, and antimicrobial activities in various extracts from Tossa jute leave (Corchorus olitorus L.). Ind. Crop. Prod. 2018, 118, 206–213. [Google Scholar] [CrossRef]
- Vetrova, E.V.; Maksimenko, E.V.; Borisenko, S.N.; Lekar, A.V.; Borisenko, N.I.; Minkin, V.I. Extraction of Rutin and Quercetin Antioxidants from the Buds of Sophora Japonica (Sophora japonica L.) by Subcritical Water. Russ. J. Phys. Chem. B 2017, 11, 1202–1206. [Google Scholar] [CrossRef]
- Lichtenthaler, R.N. Gerd Brunner: Gas Extraction—An Introduction to Fundamentals of Supercritical Fluids and the Application to Separation Processes. Topics in Physical Chemistry, Vol. 4, eds. H. Baumgärtel, E. U. Franck, W. Grünbein. Steinkopff, Darmstadt/Springer, New York, 1994, 387 S., DM 64,—. Berichte Bunsenges. Phys. Chem. 1996, 100, 1090–1091. [Google Scholar]
- Musialik, M.; Kuzmicz, R.; Pawlowski, T.S.; Litwinienko, G. Acidity of hydroxyl groups: An overlooked influence on antiradical properties of flavonoids. J. Org. Chem. 2009, 74, 2699–2709. [Google Scholar] [CrossRef]
- Turturică, M.; Stănciuc, N.; Bahrim, G.; Râpeanu, G. Effect of thermal treatment on phenolic compounds from plum (prunus domestica) extracts—A kinetic study. J. Food Eng. 2016, 171, 200–207. [Google Scholar] [CrossRef]
- Ramešová, Š.; Sokolová, R.; Degano, I.; Bulíčková, J.; Žabka, J.; Gál, M. On the stability of the bioactive flavonoids quercetin and luteolin under oxygen-free conditions. Anal. Bioanal. Chem. 2012, 402, 975–982. [Google Scholar] [CrossRef]
- Dall’Acqua, S.; Miolo, G.; Innocenti, G.; Caffieri, S. The photodegradation of quercetin: Relation to oxidation. Molecules 2012, 17, 8898–8907. [Google Scholar] [CrossRef] [Green Version]
- Srinivas, K.; King, J.W.; Howard, L.R.; Monrad, J.K. Solubility and solution thermodynamic properties of quercetin and quercetin dihydrate in subcritical water. J. Food Eng. 2010, 100, 208–218. [Google Scholar] [CrossRef]
- Razmara, R.S.; Daneshfar, A.; Sahraei, R. Solubility of quercetin in water + methanol and water + ethanol from (292.8 to 333.8) K. J. Chem. Eng. Data 2010, 55, 3934–3936. [Google Scholar] [CrossRef]
- Chebil, L.; Humeau, C.; Anthony, J.; Dehez, F.; Engasser, J.M.; Ghoul, M. Solubility of flavonoids in organic solvents. J. Chem. Eng. Data 2007, 52, 1552–1556. [Google Scholar] [CrossRef]
- Chafer, A.; Fornari, T.; Berna, A.; Stateva, R.P. Solubility of quercetin in supercritical CO2+ ethanol as a modifier: Measurements and thermodynamic modelling. J. Supercrit. Fluids 2004, 32, 89–96. [Google Scholar] [CrossRef]
- Abraham, M.H.; Acree Jr, W.E. On the solubility of quercetin. J. Mol. Liq. 2014, 197, 157–159. [Google Scholar] [CrossRef]
- Seal, T. Quantitative HPLC analysis of phenolic acids, flavonoids and ascorbic acid in four different solvent extracts of two wild edible leaves, Sonchus arvensis and Oenanthe linearis of North-Eastern region in India. J. Appl. Pharm. Sci. 2016, 6, 157–166. [Google Scholar] [CrossRef] [Green Version]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Effects of solvent type on phenolics and flavonoids content and antioxidant activities in two varieties of young ginger (Zingiber officinale Roscoe) extracts. J. Med. Plants Res. 2011, 5, 1147–1154. [Google Scholar]
- Subramaniam, B.; Chaudhari, R.V.; Chaudhari, A.S.; Akien, G.R.; Xie, Z. Supercritical fluids and gas-expanded liquids as tunable media for multiphase catalytic reactions. Chem. Eng. Sci. 2014, 115, 3–18. [Google Scholar] [CrossRef]
- Nunes, A.V.M.; Duarte, C.M.M. Dense CO₂ as a Solute, Co-Solute or Co-Solvent in Particle Formation Processes: A Review. Materials 2011, 4, 2017–2041. [Google Scholar] [CrossRef] [Green Version]
- Al-Hamimi, S.; Abellan Mayoral, A.; Cunico, L.P.; Turner, C. Carbon Dioxide Expanded Ethanol Extraction: Solubility and Extraction Kinetics of α-Pinene and cis-Verbenol. Anal. Chem. 2016, 88, 4336–4345. [Google Scholar] [CrossRef] [Green Version]
- Clarke, C.J.; Tu, W.C.; Levers, O.; Bröhl, A.; Hallett, J.P. Green and Sustainable Solvents in Chemical Processes. Chem. Rev. 2018, 118, 747–800. [Google Scholar] [CrossRef]
- Cunico, L.P.; Acosta, M.C.; Turner, C. Experimental measurements and modeling of curcumin solubility in CO2-expanded ethanol. J. Supercrit. Fluids 2017, 130, 381–388. [Google Scholar] [CrossRef]
- Abou-Shehada, S.; Clark, J.H.; Paggiola, G.; Sherwood, J. Tunable solvents: Shades of green. Chem. Eng. Proc. Proc. Intens. 2016, 99, 88–96. [Google Scholar] [CrossRef]
- Barnes, J.S.; Foss, F.W., Jr.; Schug, K.A. Thermally accelerated oxidative degradation of quercetin using continuous flow kinetic electrospray-ion trap-time of flight mass spectrometry. J. Am. Soc. Mass Spec. 2013, 24, 1513–1522. [Google Scholar] [CrossRef] [PubMed]
- Buchner, N.; Krumbein, A.; Rohn, S.; Kroh, L.W. Effect of thermal processing on the flavonols rutin and quercetin. Rap. Comm. Mass. Spec. 2006, 20, 3229–3235. [Google Scholar] [CrossRef]
- Boots, A.W.; Kubben, N.; Haenen, G.R.M.M.; Bast, A. Oxidized quercetin reacts with thiols rather than with ascorbate: Implication for quercetin supplementation. Biochem. Biophys. Res. Comm. 2003, 308, 560–565. [Google Scholar] [CrossRef]
- Wianowska, D.; Dawidowicz, A.L.; Bernacik, K.; Typek, R. Determining the true content of quercetin and its derivatives in plants employing SSDM and LC–MS analysis. Eur. Food Res. Technol. 2017, 243, 27–40. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Zhao, X.H. Degradation kinetics of fisetin and quercetin in solutions affected by medium pH, temperature and co-existing proteins. J. Serb. Chem. Soc. 2016, 81, 243–253. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Sandahl, M.; Sjöberg, P.J.R.; Turner, C. Pressurised hot water extraction in continuous flow mode for thermolabile compounds: Extraction of polyphenols in red onions. Anal. Bioanal. Chem. 2014, 406, 441–445. [Google Scholar] [CrossRef] [Green Version]
- Jessop, P.G.; Jessop, D.A.; Fu, D.; Phan, L. Solvatochromic parameters for solvents of interest in green chemistry. Green Chem. 2012, 14, 1245–1259. [Google Scholar] [CrossRef]
- Zvezdanović, J.B.; Marković, D.Z.; Cvetković, D.J.; Stanojević, J.S. UV-induced change in the antioxidant activity of quercetin toward benzophenone-initiated lipid peroxidation. J. Serb. Chem. Soc. 2012, 77, 1571–1588. [Google Scholar] [CrossRef]


| CO2 (% mol) | Temperature (K) | k (min −1) 10−4 | |
|---|---|---|---|
| CO2-Expanded Ethanol | CO2-Expanded Ethyl Lactate | ||
| 10 | 308 | Not observed | Not observed |
| 30 | Not observed | Not observed | |
| 50 | Not observed | Not observed | |
| 10 | 323 | 0.44 | 7.05 |
| 30 | Not observed | 2.94 | |
| 50 | Not observed | Not observed | |
| 10 | 343 | 4.83 | 9.33 |
| 30 | Not observed | 6.83 | |
| 50 | Not observed | 4.09 | |
| CO2 (% mol) | Temperature (K) | Solubility (Molar Fraction) 10−4 | |
|---|---|---|---|
| CO2-Expanded Ethanol | CO2-Expanded Ethyl Lactate | ||
| 10 | 308 | 10.61 ± 0.16 | 43.22 ± 0.16 |
| 30 | 7.74 ± 0.06 | 19.24 ± 0.07 | |
| 50 | 5.53 ± 0.06 | 8.93 ± 0.12 | |
| 10 | 323 | 10.67 ± 0.69 (calc. 10.80) | 41.71 ± 0.12 (50.68) |
| 30 | 13.07 ± 0.09 | 27.74 ± 0.09 (30.09) | |
| 50 | 6.13 ± 0.08 | 11.16 ± 0.02 | |
| 10 | 343 | 11.87 ± 0.50 (calc. 13.56) | 37.49 ± 0.06 (48.51) |
| 30 | 14.00 ± 0.15 | 21.98 ± 0.26 (26.55) | |
| 50 | 8.06 ± 0.03 | 10.18 ± 0.01 (11.40) | |
Sample Availability: Not available. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cunico, L.P.; Cobo, A.M.; Al-Hamimi, S.; Turner, C. Solubility and Thermal Degradation of Quercetin in CO2-Expanded Liquids. Molecules 2020, 25, 5582. https://doi.org/10.3390/molecules25235582
Cunico LP, Cobo AM, Al-Hamimi S, Turner C. Solubility and Thermal Degradation of Quercetin in CO2-Expanded Liquids. Molecules. 2020; 25(23):5582. https://doi.org/10.3390/molecules25235582
Chicago/Turabian StyleCunico, Larissa P., Andrés Medina Cobo, Said Al-Hamimi, and Charlotta Turner. 2020. "Solubility and Thermal Degradation of Quercetin in CO2-Expanded Liquids" Molecules 25, no. 23: 5582. https://doi.org/10.3390/molecules25235582






