Encapsulation of Risperidone by Methylated β-Cyclodextrins: Physicochemical and Molecular Modeling Studies
Abstract
:1. Introduction
2. Results and Discussion
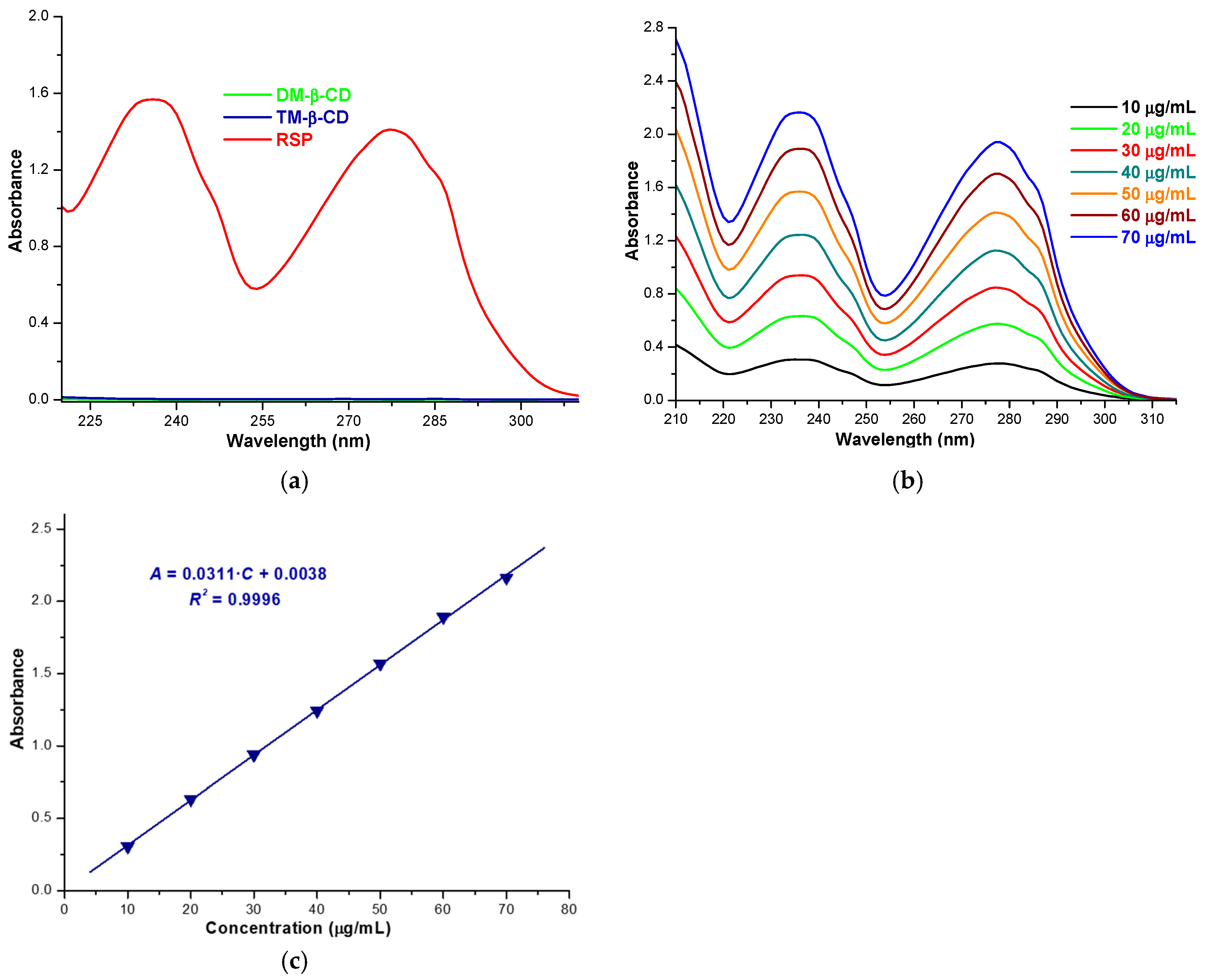
2.1. Stoichiometry Determination of RSP/CD Inclusion Complexes
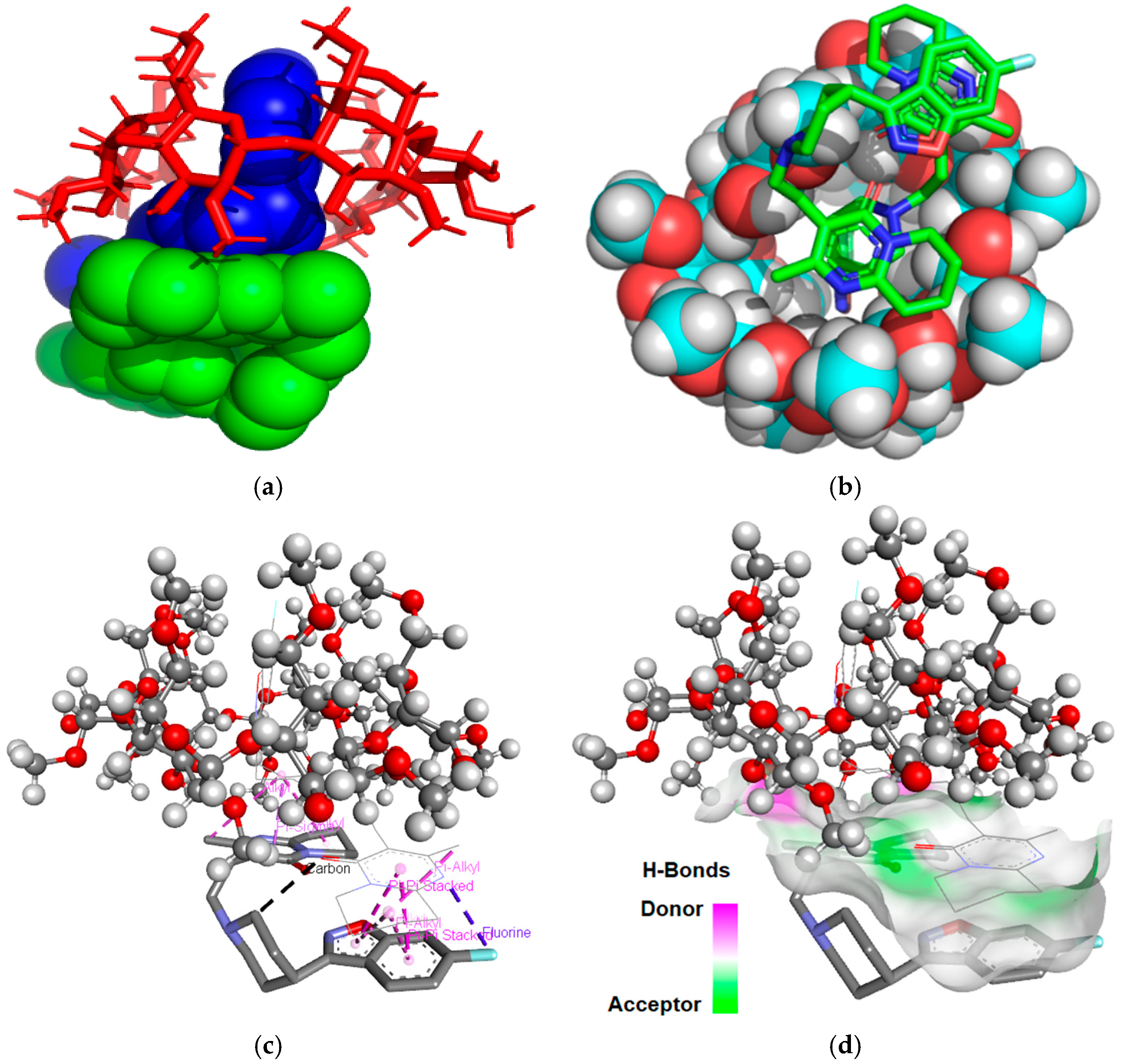
2.2. Molecular Modeling Studies
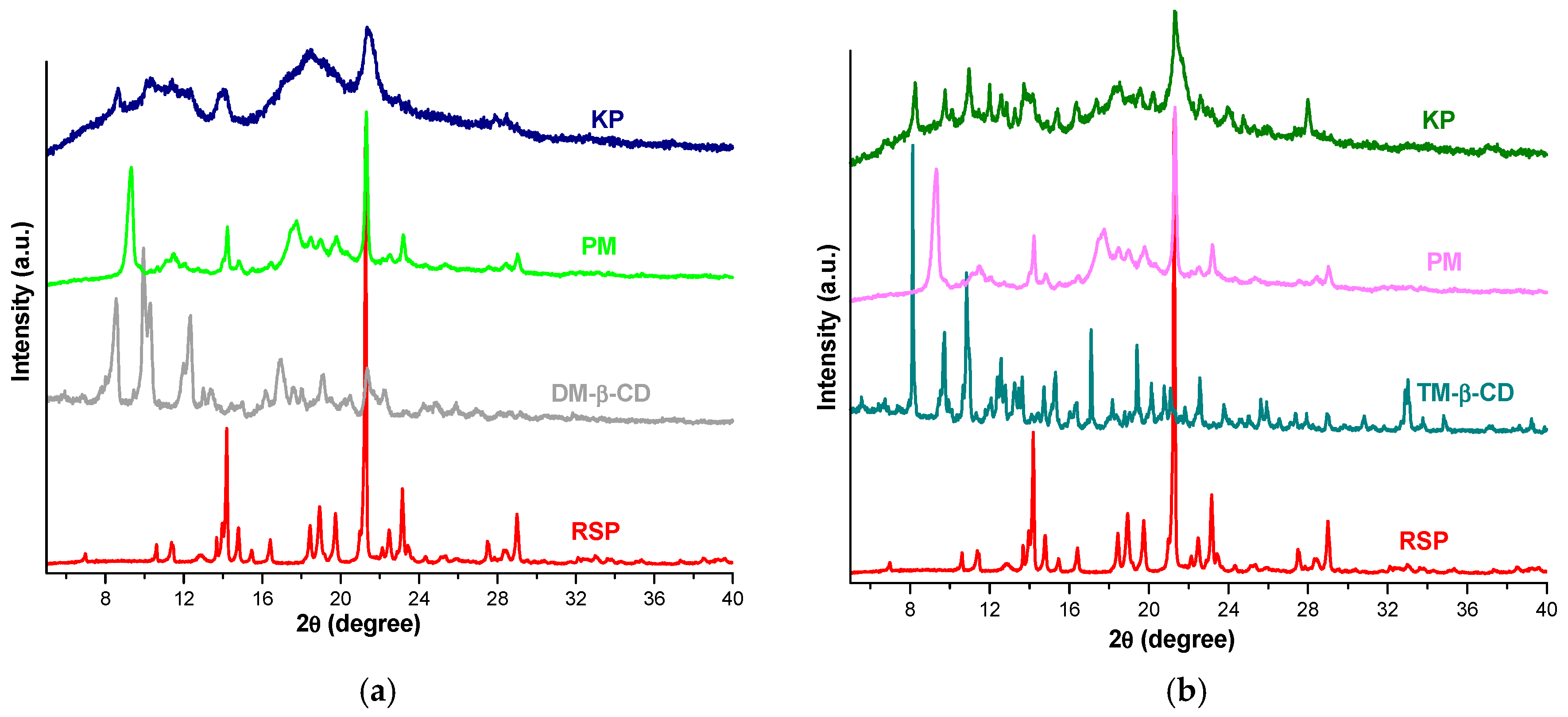
2.3. X-ray Diffraction Studies
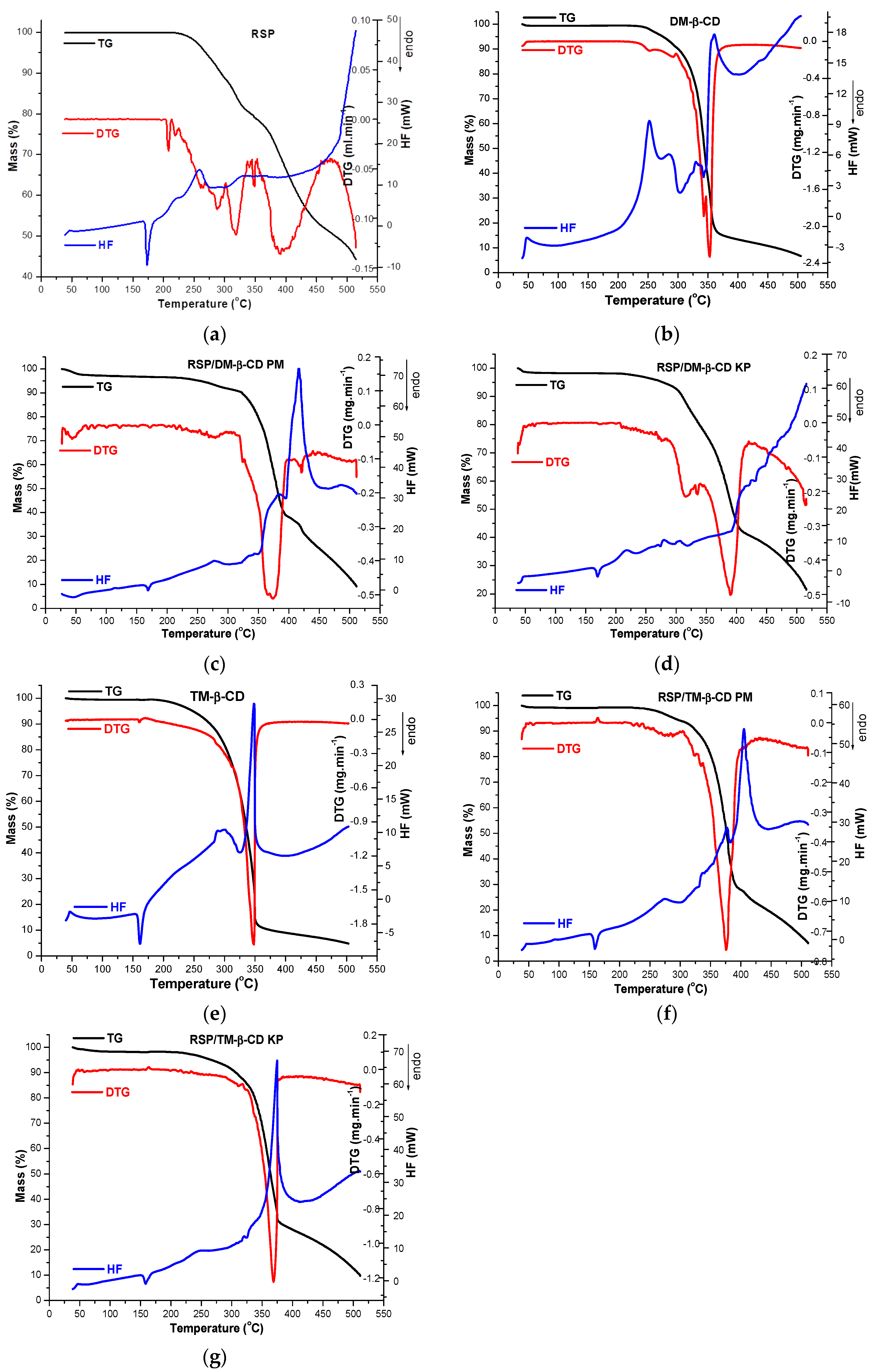
2.4. Thermal Metods
2.5. FTIR Spectroscopy
2.6. Solubility Profile of RSP/CD Kneaded Products
3. Materials and Methods
3.1. Materials
3.2. Stoichiometry Determination of RSP/CD Inclusion Complexes
3.3. Molecular Modeling Studies
3.4. Preparation of the Solid Inclusion Complexes and Physical Mixtures
3.5. X-ray Diffraction Studies
3.6. Thermal Metods
3.7. FTIR Spectroscopy
3.8. Solubility Profile of RSP/CD Kneaded Products
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Carneiro, S.B.; Duarte, F.Í.C.; Heimfarth, L.; Quintans, J.D.S.S.; Quintans-Júnior, L.J.; Júnior, V.F.D.V.; De Lima, Á.A.N. Cyclodextrin-drug inclusion complexes: In vivo and in vitro approaches. Int. J. Mol. Sci. 2019, 20, 642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crini, G.; Fourmentin, S.; Fenyvesi, É.; Torri, G.; Fourmentin, M.; Morin-Crini, N. Cyclodextrins, from molecules to applications. Environ. Chem. Lett. 2018, 16, 1361–1375. [Google Scholar] [CrossRef]
- Han, D.; Han, Z.; Liu, L.; Wang, Y.; Xin, S.; Zhang, H.; Yu, Z. Solubility enhancement of myricetin by inclusion complexation with heptakis-o-(2-hydroxypropyl)-β-cyclodextrin: A joint experimental and theoretical study. Int. J. Mol. Sci. 2020, 21, 766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, W.; Liu, Y. Characterization and stability of beta-acids/hydroxypropyl-β-cyclodextrin inclusion complex. J. Mol. Struct. 2020, 1201, 127159. [Google Scholar] [CrossRef]
- Usacheva, T.; Kabirov, D.; Beregova, D.; Gamov, G.; Sharnin, V.; Biondi, M.; Mayol, L.; D’Aria, F.; Giancola, C. Thermodynamics of complex formation between hydroxypropyl-β-cyclodextrin and quercetin in water–ethanol solvents at T = 298.15 K. J. Therm. Anal. Calorim. 2019, 138, 417–424. [Google Scholar] [CrossRef]
- Li, S.; Yuan, L.; Chen, Y.; Zhou, W.; Wang, X. Studies on the inclusion complexes of daidzein with β-cyclodextrin and derivatives. Molecules 2017, 22, 2183. [Google Scholar] [CrossRef] [Green Version]
- He, J.; Zheng, Z.P.; Zhu, Q.; Guo, F.; Chen, J. Encapsulation mechanism of oxyresveratrol by β-cyclodextrin and hydroxypropyl-β-cyclodextrin and computational analysis. Molecules 2017, 22, 1801. [Google Scholar] [CrossRef] [Green Version]
- Simsek, T.; Rasulev, B.; Mayer, C.; Simsek, S. Preparation and characterization of inclusion complexes of β-cyclodextrin and phenolics from wheat bran by combination of experimental and computational techniques. Molecules 2020, 25, 4275. [Google Scholar] [CrossRef]
- Du, F.; Pan, T.; Ji, X.; Hu, J.; Ren, T. Study on the preparation of geranyl acetone and β-cyclodextrin inclusion complex and its application in cigarette flavoring. Sci. Rep. 2020, 10, 12375. [Google Scholar] [CrossRef]
- Tănase, I.M.; Sbârcea, L.; Ledeți, A.; Vlase, G.; Barvinschi, P.; Văruţ, R.M.; Dragomirescu, A.; Axente, C.; Ledeți, I. Physicochemical characterization and molecular modeling study of host–guest systems of aripiprazole and functionalized cyclodextrins. J. Therm. Anal. Calorim. 2020, 141, 1027–1039. [Google Scholar] [CrossRef]
- Tănase, I.M.; Sbârcea, L.; Ledeţi, A.; Barvinschi, P.; Cîrcioban, D.; Vlase, G.; Văruţ, R.M.; Ledeţi, I. Compatibility studies with pharmaceutical excipients for aripiprazole–heptakis (2,6-di-O-methyl)-β-cyclodextrin supramolecular adduct. J. Therm. Anal. Calorim. 2020, 142, 1963–1976. [Google Scholar] [CrossRef]
- Sbârcea, L.; Udrescu, L.; Ledeţi, I.; Szabadai, Z.; Fuliaş, A.; Sbârcea, C. β-Cyclodextrin inclusion complexes of lisinopril and zofenopril: Physicochemical characterization and compatibility study of lisinopril-β-cyclodextrin with lactose. J. Therm. Anal. Calorim. 2016, 123, 2377–2390. [Google Scholar] [CrossRef]
- Sbârcea, L.; Ledeţi, I.; Drăgan, L.; Kurunczi, L.; Fuliaş, A.; Udrescu, L. Fosinopril sodium-hydroxypropyl-β-cyclodextrin inclusion complex: Thermal decomposition kinetics and compatibility studies. J. Therm. Anal. Calorim. 2015, 120, 981–990. [Google Scholar] [CrossRef]
- Saokham, P.; Muankaew, C.; Jansook, P.; Loftsson, T. Solubility of cyclodextrins and drug/cyclodextrin complexes. Molecules 2018, 23, 1161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García, A.; Leonardi, D.; Vasconi, M.D.; Hinrichsen, L.I.; Lamas, M.C. Characterization of albendazole-randomly methylated-β-cyclodextrin inclusion complex and in vivo evaluation of its antihelmitic activity in a murine model of trichinellosis. PLoS ONE 2014, 9, e113296. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, E.B.; da Silva Júnior, W.F.; de Oliveira Pinheiro, J.G.; da Fonseca, A.G.; Moura Lemos, T.M.A.; de Oliveira Rocha, H.A.; de Azevedo, E.P.; Mendonça Junior, F.J.B.; De Lima, Á.A.N. Characterization and antiproliferative activity of a novel 2-aminothiophene derivative-β-cyclodextrin binary system. Molecules 2018, 23, 3130. [Google Scholar] [CrossRef] [Green Version]
- Szente, L.; Szejtli, J. Highly soluble cyclodextrin derivatives: Chemistry, properties, and trends in development. Adv. Drug Deliv. Rev. 1999, 36, 17–28. [Google Scholar] [CrossRef]
- Brewster, M.E.; Loftsson, T. Cyclodextrins as pharmaceutical solubilizers. Adv. Drug Deliv. Rev. 2007, 59, 645–666. [Google Scholar] [CrossRef]
- Germann, D.; Kurylo, N.; Han, F. Risperidone. In Profiles of Drug Substances, Excipients and Related Methodology; Academic Press Elsevier: San Diego, CA, USA, 2012; Volume 37, pp. 313–361. ISBN 9780123972200. [Google Scholar]
- Yunusa, I.; El Helou, M.L. The Use of Risperidone in Behavioral and Psychological Symptoms of Dementia: A Review of Pharmacology, Clinical Evidence, Regulatory Approvals, and Off-Label Use. Front. Pharmacol. 2020, 11, 596. [Google Scholar] [CrossRef]
- Love, R.C.; Nelson, M.W. Pharmacology and clinical experience with risperidone. Expert Opin. Pharmacother. 2000, 1, 1441–1453. [Google Scholar] [CrossRef]
- Rahman, Z.; Zidan, A.S.; Khan, M.A. Risperidone solid dispersion for orally disintegrating tablet: Its formulation design and non-destructive methods of evaluation. Int. J. Pharm. 2010, 400, 49–58. [Google Scholar] [CrossRef]
- Censi, R.; Di Martino, P. Polymorph impact on the bioavailability and stability of poorly soluble drugs. Molecules 2015, 20, 18759–18776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, D.; Chakraborty, S.; Singh, S.; Mishra, B. Preparation and in-vitro characterization of Risperidone-cyclodextrin inclusion complexes as a potential injectable product. Daru 2009, 17, 226–235. [Google Scholar]
- Jug, M.; Kos, I.; Bećirević-Laćan, M. The pH-dependent complexation between risperidone and hydroxypropyl-β- cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2009, 64, 163–171. [Google Scholar] [CrossRef]
- El-Barghouthi, M.I.; Masoud, N.A.; Al-Kafawein, J.K.; Zughul, M.B.; Badwan, A.A. Host-guest interactions of risperidone with natural and modified cyclodextrins: Phase solubility, thermodynamics and molecular modeling studies. J. Incl. Phenom. 2005, 53, 15–22. [Google Scholar] [CrossRef]
- Savjani, K.T.; Gajjar, A.K.; Savjani, J.K. Drug Solubility: Importance and Enhancement Techniques. ISRN Pharm. 2012, 2012, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Job, P. Formation and stability of inorganic complexes in solution. Ann. Chim. 1928, 9, 113–203. [Google Scholar]
- Sbârcea, L.; Ledeţi, A.; Udrescu, L.; Văruţ, R.M.; Barvinschi, P.; Vlase, G.; Ledeţi, I. Betulonic acid—Cyclodextrins inclusion complexes. J. Therm. Anal. Calorim. 2019, 138, 2787–2797. [Google Scholar] [CrossRef]
- Morris, G.M.; Ruth, H.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. Software news and updates AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [Green Version]
- Huey, R.; Morris, G.M.; Olson, A.J.; Goodsell, D.S. A Semiempirical Free Energy Force Field with Charge-Based Desolvation. J. Comput. Chem. 2007, 28, 1145–1152. [Google Scholar] [CrossRef]
- DeLano, W.L. PyMOL; DeLano Scientific: San Carlos, CA, USA, 2002. [Google Scholar]
- Sbârcea, L.; Udrescu, L.; Drǎgan, L.; Trandafirescu, C.; Szabadai, Z.; Bojiţǎ, M. Fosinopril-cyclodextrin inclusion complexes: Phase solubility and physicochemical analysis. Pharmazie 2011, 66, 584–589. [Google Scholar] [CrossRef]
- Sbarcea, L.; Udrescu, L.; Dragan, L.; Trandafirescu, C.; Sasca, V.; Barvinschi, P.; Bojita, M. Characterization of fosinopril natrium-hydroxypropyl-β-cyclodextrin inclusion complex. Rev. Chim. 2011, 62, 349–351. [Google Scholar]
- Daniel, J.S.P.; Veronez, I.P.; Rodrigues, L.L.; Trevisan, M.G.; Garcia, J.S. Risperidone—Solid-state characterization and pharmaceutical compatibility using thermal and non-thermal techniques. Thermochim. Acta 2013, 568, 148–155. [Google Scholar] [CrossRef]
- Circioban, D.; Ledeti, A.; Vlase, G.; Coricovac, D.; Moaca, A.; Farcas, C.; Vlase, T.; Ledeti, I.; Dehelean, C. Guest–host interactions and complex formation for artemisinin with cyclodextrins: Instrumental analysis and evaluation of biological activity. J. Therm. Anal. Calorim. 2018, 134, 1375–1384. [Google Scholar] [CrossRef]
- Doile, M.M.; Fortunato, K.A.; Schmücker, I.C.; Schucko, S.K.; Silva, M.A.S.; Rodrigues, P.O. Physicochemical properties and dissolution studies of dexamethasone acetate-β-cyclodextrin inclusion complexes produced by different methods. AAPS PharmSciTech 2008, 9, 314–321. [Google Scholar] [CrossRef] [Green Version]
- Circioban, D.; Ledeti, I.; Suta, L.M.; Vlase, G.; Ledeti, A.; Vlase, T.; Varut, R.; Sbarcea, L.; Trandafirescu, C.; Dehelean, C. Instrumental analysis and molecular modelling of inclusion complexes containing artesunate. J. Therm. Anal. Calorim. 2020, 142, 1951–1961. [Google Scholar] [CrossRef]
- Mennini, N.; Maestrelli, F.; Cirri, M.; Mura, P. Analysis of physicochemical properties of ternary systems of oxaprozin with randomly methylated-ß-cyclodextrin and L-arginine aimed to improve the drug solubility. J. Pharm. Biomed. Anal. 2016, 129, 350–358. [Google Scholar] [CrossRef]
- Lavorgna, M.; Iacovino, R.; Russo, C.; Di Donato, C.; Piscitelli, C.; Isidori, M. A new approach for improving the antibacterial and tumor cytotoxic activities of pipemidic acid by including it in trimethyl-β-cyclodextrin. Int. J. Mol. Sci. 2019, 20, 416. [Google Scholar] [CrossRef] [Green Version]
- Baka, E.; Comer, J.E.A.; Takács-Novák, K. Study of equilibrium solubility measurement by saturation shake-flask method using hydrochlorothiazide as model compound. J. Pharm. Biomed. Anal. 2008, 46, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Protein Data Bank. Available online: http://www.pdb.org/pdb/home/home.do (accessed on 15 October 2020).




Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sbârcea, L.; Tănase, I.-M.; Ledeți, A.; Cîrcioban, D.; Vlase, G.; Barvinschi, P.; Miclău, M.; Văruţ, R.-M.; Trandafirescu, C.; Ledeți, I. Encapsulation of Risperidone by Methylated β-Cyclodextrins: Physicochemical and Molecular Modeling Studies. Molecules 2020, 25, 5694. https://doi.org/10.3390/molecules25235694
Sbârcea L, Tănase I-M, Ledeți A, Cîrcioban D, Vlase G, Barvinschi P, Miclău M, Văruţ R-M, Trandafirescu C, Ledeți I. Encapsulation of Risperidone by Methylated β-Cyclodextrins: Physicochemical and Molecular Modeling Studies. Molecules. 2020; 25(23):5694. https://doi.org/10.3390/molecules25235694
Chicago/Turabian StyleSbârcea, Laura, Ionuț-Mihai Tănase, Adriana Ledeți, Denisa Cîrcioban, Gabriela Vlase, Paul Barvinschi, Marinela Miclău, Renata-Maria Văruţ, Cristina Trandafirescu, and Ionuț Ledeți. 2020. "Encapsulation of Risperidone by Methylated β-Cyclodextrins: Physicochemical and Molecular Modeling Studies" Molecules 25, no. 23: 5694. https://doi.org/10.3390/molecules25235694








