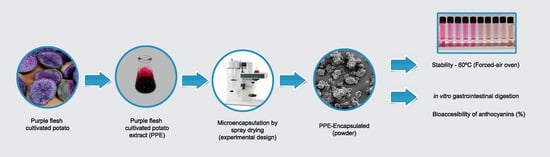
Microencapsulation of Anthocyanin Extracted from Purple Flesh Cultivated Potatoes by Spray Drying and Its Effects on In Vitro Gastrointestinal Digestion
Abstract
:1. Introduction
2. Results
2.1. Characterization of Raw Material and PPE
2.2. The Encapsulation of Anthocyanins from PPE
2.3. Characterization of the PPE-MD System Obtained Under Optimal Conditions
2.4. Stability of Microencapsulated PPE during Storage
2.5. In Vitro Bioaccessibility of Anthocyanins
3. Materials and Methods
3.1. Raw Materials
3.2. Preparation of the Purple Potato Extract (PPE)
3.2.1. Characterization of PPE
3.2.2. AT content and HPLC Profile
3.3. Microencapsulation of PPE
3.3.1. Preparation and Characterization of PPE Microparticles
3.3.2. Morphology
3.3.3. Particle size
3.3.4. Encapsulation efficiency of the AT and the yield of the process
3.3.5. Accelerated Storage Stability Test of the Microparticle Powders
3.3.6. In vitro digestion model
Mouth digestion
Gastric digestion
Gut digestion
3.3.7. Experimental Design
3.3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abreviations
| ANOVA | analysis of variance |
| AT | antohocyanins |
| aw | water activity |
| BA | bioaccessibility |
| CCD | central composite design |
| Cy-3-glu | cyanidin-3-glucoside |
| ΔΕ | color difference values |
| DF | desirability function |
| EE | encapsulation efficiency |
| HPLC-DAD | high-performance liquid chromatography-photo diose array detector |
| MD | maltodextrin |
| PP | purple flesh cultivated potato |
| PPE | purple flesh cultivated potato extract |
| RSM | response surface methodology |
| SD | spray drying |
| SEM | scanning electron microscopy |
| TE | trolox equivalent |
References
- Tsuda, S.; Murakarni, M.; Kano, K.; Taniguchi, K.; Sasaki, Y.F. DNA damage induced by red food dyes orally administered to pregnant and male mice. Toxicol. Sci. 2001, 61, 92–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stevens, L.J.; Kuczek, T.; Burgess, J.R.; Stochelski, M.A.; Arnold, L.E.; Galland, L. Mechanisms of behavioral, atopic, and other reactions to artificial food colors in children. Nutr. Rev. 2013, 71, 268–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robertson, T.; Alzaabi, A.; Robertson, M.; Fielding, B. Starchy carbohydrates in a healthy diet: The role of the humble potato. Nutrients 2018, 10, 1764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zaheer, K.; Akhtar, M.H. Potato Production, Usage, and Nutrition-A Review. Crit. Rev. Food Sci. 2016, 56, 711–721. [Google Scholar] [CrossRef]
- Nielsen, T.S.; Bendiks, Z.; Thomsen, B.; Wright, M.E.; Theil, P.K.; Scherer, B.L.; Marco, M.L. High-Amylose Maize, Potato, and Butyrylated Starch Modulate Large Intestinal Fermentation, Microbial Composition, and Oncogenic miRNA Expression in Rats Fed A High-Protein Meat Diet. Int. J. Mol. Sci. 2019, 20, 2137. [Google Scholar] [CrossRef] [Green Version]
- Marthandam Asokan, S.; Wang, T.; Su, W.T.; Lin, W.T. Antidiabetic Effects of a Short Peptide of Potato Protein Hydrolysate in STZ-Induced Diabetic Mice. Nutrients 2019, 11, 779. [Google Scholar] [CrossRef] [Green Version]
- Clifford, M.N. Review Anthocyanins—Nature, occurrence and dietary burden. J. Sci. Food Agr. 2000, 80, 1063–1072. [Google Scholar] [CrossRef]
- Fernandez-Lopez, J.A.; Angosto, J.; Gimenez, P.; Leon, G. Thermal stability of selected natural red extracts used as food colorants. Plant Foods Hum. Nutr. 2013, 68, 11–17. [Google Scholar] [CrossRef]
- Guo, J.; Giusti, M.M.; Kaletunç, G. Encapsulation of purple corn and blueberry extracts in alginate-pectin hydrogel particles: Impact of processing and storage parameters on encapsulation efficiency. Food Res. Int. 2018, 107, 414–422. [Google Scholar] [CrossRef]
- He, J.; Giusti, M.M. Anthocyanins: Natural colorants with health-promoting properties. Annu. Rev. Food Sci. 2010, 1, 163–187. [Google Scholar] [CrossRef]
- Reyes-Farias, M.; Vasquez, K.; Fuentes, F.; Ovalle-Marin, A.; Parra-Ruiz, C.; Zamora, O.; Pino, M.T.; Quitral, V.; Jimenez, P.; Garcia, L.; et al. Extracts of Chilean native fruits inhibit oxidative stress, inflammation and insulin-resistance linked to the pathogenic interaction between adipocytes and macrophages. J. Funct. Foods 2016, 27, 69–83. [Google Scholar] [CrossRef]
- Hayashi, K.; Hibasami, H.; Murakami, T.; Terahara, N.; Mori, M.; Tsukui, A. Induction of apoptosis in cultured human stomach cancer cells by potato anthocyanins and its inhibitory effects on growth of stomach cancer in mice. Food Sci. Technol. Res. 2006, 12, 22–26. [Google Scholar] [CrossRef] [Green Version]
- Charepalli, V.; Reddivari, L.; Radhakrishnan, S.; Vadde, R.; Agarwal, R.; Vanamala, J.K.P. Anthocyanin-containing purple-fleshed potatoes suppress colon tumorigenesis via elimination of colon cancer stem cells. J. Nutr. Biochem. 2015, 26, 1641–1649. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Kano, M.; Takayanagi, T.; Yamakawa, O.; Ishikawa, F. Absorption of acylated anthocyanins in rats and humans after ingesting an extract of Ipomoea batatas purple sweet potato tuber. Biosci. Biotech. 2004, 68, 1500–1507. [Google Scholar] [CrossRef] [PubMed]
- Leri, F.; Innocenti, M.; Andrenelli, L.; Vecchio, V.; Mulinacci, N. Rapid HPLC/DAD/MS method to determine phenolic acids, glycoalkaloids and anthocyanins in pigmented potatoes (Solanum tuberosum L.) and correlations with variety and geographical origin. Food Chem. 2011, 125, 750–759. [Google Scholar]
- Oertel, A.; Matros, A.; Hartmann, A.; Arapitsas, P.; Dehmer, K.J.; Martens, S.; Mock, H.P. Metabolite profiling of red and blue potatoes revealed cultivar and tissue specific patterns for anthocyanins and other polyphenols. Planta 2017, 246, 281–297. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, S.A.; Jafari, S.M.; Ghorbani, M.; Assadpoor, E. Spray-drying microencapsulation of anthocyanins by natural biopolymers: A review. Dry technol. 2014, 32, 509–518. [Google Scholar] [CrossRef]
- Fredes, C.; Becerra, C.; Parada, J.; Robert, P. The microencapsulation of maqui (Aristotelia chilensis (Mol.) Stuntz) juice by spray-drying and freeze-drying produces powders with similar anthocyanin stability and bioaccessibility. Molecules 2018, 23, 1227. [Google Scholar] [CrossRef] [Green Version]
- Mahdavi, S.A.; Jafari, S.M.; Assadpoor, E.; Dehnad, D. Microencapsulation optimization of natural anthocyanins with maltodextrin, gum Arabic and gelatin. Int. J. Biol. Macromol. 2016, 85, 379–385. [Google Scholar] [CrossRef]
- Tan, C.; Selig, M.J.; Abbaspourrad, A. Anthocyanin stabilization by chitosan-chondroitin sulfate polyelectrolyte complexation integrating catechin co-pigmentation. Carbohyd polym 2018, 181, 124–131. [Google Scholar] [CrossRef]
- Da Rosa, J.R.; Nunes, G.L.; Motta, M.H.; Fortes, J.P.; Weis, G.C.C.; Hecktheuer, L.H.R.; Muller, E.; de Menezes, C.; da Rosa, C.S. Microencapsulation of anthocyanin compounds extracted from blueberry (Vaccinium spp.) by spray drying: Characterization, stability and simulated gastrointestinal conditions. Food Hydrocolloid. 2019, 89, 742–748. [Google Scholar] [CrossRef]
- Ahmed, M.; Akter, M.S.; Lee, J.C.; Eun, J.B. Encapsulation by spray drying of bioactive components, physicochemical and morphological properties from purple sweet potato. LWT Food Sci. Technol. 2010, 43, 1307–1312. [Google Scholar] [CrossRef]
- Cai, X.; Du, X.; Cui, D.; Wang, X.; Yang, Z.; Zhu, G. Improvement of stability of blueberry anthocyanins by carboxymethyl starch/xanthan gum combinations microencapsulation. Food Hydrocolloid. 2019, 91, 238–245. [Google Scholar] [CrossRef]
- Fredes, C.; Osorio, M.J.; Parada, J.; Robert, P. Stability and bioaccessibility of anthocyanins from maqui (Aristotelia chilensis [Mol.] Stuntz) juice microparticles. LWT Food Sci. Technol. 2018, 91, 549–556. [Google Scholar] [CrossRef]
- Das, A.B.; Goud, V.V.; Das, C. Microencapsulation of anthocyanin extract from purple rice bran using modified rice starch and its effect on rice dough rheology. Int. J. Biol. Macromol. 2019, 124, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Anthocyanin stability and antioxidant activity of spray-dried açai (Euterpe oleracea Mart.) juice produced with different carrier agents. Food Res Int. 2010, 43, 907–914. [Google Scholar] [CrossRef]
- Tan, C.; Arshadi, M.; Lee, M.C.; Godec, M.; Azizi, M.; Yan, B.; Eskandarloo, H.; Deisenroth, T.W.; Darji, R.; Van Pho, T.; et al. A Robust Aqueous Core–Shell–Shell Coconut-like Nanostructure for Stimuli-Responsive Delivery of Hydrophilic Cargo. ACS Nano. 2019, 13, 9016–9027. [Google Scholar] [CrossRef]
- De Vos, P.; Faas, M.M.; Spasojevic, M.; Sikkema, J. Encapsulation for preservation of functionality and targeted delivery of bioactive food components. Int Dairy J. 2010, 20, 292–302. [Google Scholar] [CrossRef]
- Mohd Nawi, N.; Muhamad, I.I.; Mohd Marsin, A. The physicochemical properties of microwave-assisted encapsulated anthocyanins from Ipomoea batatas as affected by different wall materials. Food Sci. Nutr. 2015, 3, 91–99. [Google Scholar] [CrossRef]
- Laila, U.; Rochmadi, R.; Pudjiraharti, S. Microencapsulation of Purple-Fleshed Sweet Potato Anthocyanins with Chitosan-Sodium Tripolyphosphate by Using Emulsification-Crosslinking Technique. J. Math Fund Sci. 2019, 51, 29–46. [Google Scholar] [CrossRef] [Green Version]
- Ersus, S.; Yurdagel, U. Microencapsulation of anthocyanin pigments of black carrot (Daucus carota L.) by spray drier. J. Food Eng. 2007, 80, 805–812. [Google Scholar] [CrossRef]
- Robert, P.; Fredes, C. The encapsulation of anthocyanins from berry-type fruits. Trends in foods. Molecules 2015, 20, 5875–5888. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, B.; Kermasha, S.; Alli, I.; Mulligan, C. Encapsulation in the food industry: A review. Int. J. Food Sci. Nutr. 1999, 50, 213–224. [Google Scholar] [PubMed]
- Lachman, J.; Hamouz, K. Red and purple coloured potatoes as a significant antioxidant source in human nutrition-a review. Plant Soil. Environ. 2005, 51, 477. [Google Scholar] [CrossRef] [Green Version]
- Hillebrand, S.; Naumann, H.; Kitzinski, N.; Köhler, N.; Winterhalter, P. Isolation and characterization of anthocyanins from blue-fleshed potatoes (Solanum tuberosum L.). Food 2009, 3, 96–101. [Google Scholar]
- Gharsallaoui, A.; Roudaut, G.; Chambin, O.; Voilley, A.; Saurel, R. Applications of spray-drying in microencapsulation of food ingredients: An overview. Food Res. Int. 2007, 40, 1107–1121. [Google Scholar] [CrossRef]
- Robert, P.; Gorena, T.; Romero, N.; Sepulveda, E.; Chavez, J.; Saenz, C. Encapsulation of polyphenols and anthocyanins from pomegranate (Punica granatum) by spray drying. Int. J. Food Sci. Tech. 2010, 45, 1386–1394. [Google Scholar] [CrossRef]
- Fang, Z.; Bhandari, B. Effect of spray drying and storage on the stability of bayberry polyphenols. Food Chem. 2011, 129, 1139–1147. [Google Scholar] [CrossRef]
- Yinbin, L.; Wu, L.; Weng, M.; Tang, B.; Lai, P.; Chen, J. Effect of different encapsulating agent combinations on physicochemical properties and stability of microcapsules loaded with phenolics of plum (Prunus salicina lindl.). Powder Technol. 2018, 340, 459–464. [Google Scholar] [CrossRef]
- Alamilla-Beltrán, L.; Chanona-Pérez, J.J.; Jiménez-Aparicio, A.R.; Gutiérrez-López, G.F. Description of morphological changes of particles along spray drying. J. Food Eng. 2005, 6, 179–184. [Google Scholar] [CrossRef]
- Alvim, I.D.; Stein, M.A.; Koury, I.P.; Dantas, F.B.H.; Cruz, C.L.D.C.V. Comparison between the spray drying and spray chilling microparticles contain ascorbic acid in a baked product application. LWT Food Sci. Technol. 2016, 65, 689–694. [Google Scholar] [CrossRef]
- Rhim, J.W. Kinetics of thermal degradation of anthocyanin pigment solutions driven from red flower cabbage. Food Sci. Biotech. 2002, 11, 361–364. [Google Scholar]
- Idham, Z.; Muhamad, I.I.; Sarmidi, M.R. Degradation kinetics and color stability of spray-dried encapsulates anthocyanins from Hibiscus Sabdariffa L. J. Food Process Eng. 2012, 35, 522–542. [Google Scholar] [CrossRef]
- Wang, W.D.; Xu, S.Y. Degradation kinetics of anthocyanins in blackberry juice and concentrate. J. Food Eng. 2007, 82, 271–275. [Google Scholar] [CrossRef]
- Lila, M.A.; Ribnicky, D.M.; Rojo, L.E.; Rojas-Silva, P.; Oren, A.; Havenaar, R.; Janle, E.M.; Raskin, I.; Yousef, G.G.; Grace, M.H. Complementary approaches to gauge the bioavailability and distribution of ingested berry polyphenolics. J Agric. Food Chem. 2011, 60, 5763–5771. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar]
- Cai, Y.; Corke, H. Production and properties of spray-dried Amaranthus betacyanin pigments. J. Food Sci. 2000, 65, 1248–1252. [Google Scholar] [CrossRef]
- Vergara, C.; Saavedra, J.; Sáenz, C.; García, P.; Robert, P. Microencapsulation of pulp and ultrafiltered cactus pear (Opuntia ficus-indica) extracts and betanin stability during storage. Food Chem. 2014, 157, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Aravena, G.; García, O.; Muñoz, O.; Pérez-Correa, J.R.; Parada, J. The impact of cooking and delivery modes of thymol and carvacrol on retention and bioaccessibility in starchy foods. Food Chem. 2016, 196, 848–852. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2007, 76, 965–977. [Google Scholar] [CrossRef]
- Yolmeh, M.; Jafari, S.M. Applications of response surface methodology in the food industry processes. Food Bioproc. Tech. 2017, 10, 413–433. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |



| Sample | PP | PPE |
|---|---|---|
| Moisture content (%) | 75.6 ± 3.7 a | 35.0 ± 0.4 b |
| Soluble solids (°Brix at 20 °C) | 5.2 ± 0.1 b | 65.0 ± 0.5 a |
| Total Anthocyanins (mg cy-3-glu/g) | 0.24 ± 0.02 b | 2.0 ± 0.1 a |
| Antioxidant capacity (FRAP) (mg TE/g) | 4.7 ± 0.3 b | 21.1 ± 0.5 a |
| Factors | Response Variables | |||
|---|---|---|---|---|
| Runs | Inlet Air Temperature (°C) [X1] | PPE:MD ratio [X2] | EE (%) | Yield (%) |
| 1 | 100 (−1) | 1:1 (–1) | 72.8 ± 4.1 | 60.0 ± 4.1 |
| 2 | 180 (+1) | 1:1 (–1) | 62.3 ± 0.4 | 39.6 ± 0.3 |
| 3 | 100 (−1) | 1:4 (+1) | 93.2 ± 1.2 | 47.1 ± 1.2 |
| 4 | 180 (+1) | 1:4 (+1) | 84.7 ± 1.5 | 40.0 ± 0.5 |
| 5 | 92 (−1.21) | 1:2.5 (0) | 85.6 ± 0.3 | 20.0 ± 0.7 |
| 6 | 188 (+1.21) | 1:2.5 (0) | 85.0 ± 1.6 | 22.0 ± 0.2 |
| 7 | 140 (0) | 1:0.7 (−1.21) | 59.4 ± 0.4 | 64.7 ± 0.4 |
| 8 | 140 (0) | 1:4.3 (+1.21) | 78.8 ± 0.2 | 81.0 ±1.6 |
| 9 | 140 (0) | 1:2.5 (0) | 88.5 ± 0.7 | 57.1 ± 1.6 |
| 10 | 140 (0) | 1:2.5 (0) | 87.0 ± 1.6 | 56.0 ± 1.6 |
| 11 | 140 (0) | 1:2.5 (0) | 89.0 ± 1.6 | 50.0 ± 1.6 |
| 12 | 140 (0) | 1:2.5 (0) | 85.1 ± 1.6 | 56.6 ± 1.6 |
| System | PPE-MD |
|---|---|
| Inlet air temperature (°C) | 130 |
| PPE:MD ratio | 1:4 |
| EE (%) | 86.0 ± 0.6 |
| Yield (%) | 58.9 ± 1.0 |
| Moisture content (%) | 5.6 ± 0.4 |
| Water activity (aw) | 0.225 ± 0.001 |
| Hygroscopicity (g/100 g) | 33.6 ± 2.7 |
| Particle size (D4,3) | 6.51 ± 0.1 |
| Total Anthocyanin (mg cy-3-glu/g) | 1.34 ± 0.02 |
| Antioxidant capacity (FRAP) (mg TE/g) | 10.1 ± 0.6 |
| System | k(obs) ± DS (days-1) | r2 | ΔΕ |
|---|---|---|---|
| PPE | 11.18 × 102 ± 0.10 × 102 a | 0.971 | 58.3 ± 0.3 a |
| PPE-MD | 0.53 × 102 ± 0.02 × 102 b | 0.974 | 18.4 ± 0.2 b |
| System | Anthocyanins (mg cy-3-glu/g) | Gastric BA | Final BA (%) | ||
|---|---|---|---|---|---|
| Before Digestion | After Gastric Digestion | After Intestinal Digestion | (%) | ||
| PPE | 2.010 ± 0.050 | 0.957 ± 0.048 | 0.913 ± 0.040 | 47.6 ± 3.5 b | 45.4 ± 2.3 b |
| PPE-MD | 1.340 ± 0.020 | 1.028 ± 0.064 | 0.887 ± 0.099 | 76.7 ± 4.2 a | 66.2 ± 9.1 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vergara, C.; Pino, M.T.; Zamora, O.; Parada, J.; Pérez, R.; Uribe, M.; Kalazich, J. Microencapsulation of Anthocyanin Extracted from Purple Flesh Cultivated Potatoes by Spray Drying and Its Effects on In Vitro Gastrointestinal Digestion. Molecules 2020, 25, 722. https://doi.org/10.3390/molecules25030722
Vergara C, Pino MT, Zamora O, Parada J, Pérez R, Uribe M, Kalazich J. Microencapsulation of Anthocyanin Extracted from Purple Flesh Cultivated Potatoes by Spray Drying and Its Effects on In Vitro Gastrointestinal Digestion. Molecules. 2020; 25(3):722. https://doi.org/10.3390/molecules25030722
Chicago/Turabian StyleVergara, Cristina, María Teresa Pino, Olga Zamora, Javier Parada, Ricardo Pérez, Marco Uribe, and Julio Kalazich. 2020. "Microencapsulation of Anthocyanin Extracted from Purple Flesh Cultivated Potatoes by Spray Drying and Its Effects on In Vitro Gastrointestinal Digestion" Molecules 25, no. 3: 722. https://doi.org/10.3390/molecules25030722