Abstract
Trypanosoma cruzi is the aetiologic agent of Chagas disease, which affects people in the Americas and worldwide. The parasite has a complex life cycle that alternates among mammalian hosts and insect vectors. During its life cycle, T. cruzi passes through different environments and faces nutrient shortages. It has been established that amino acids, such as proline, histidine, alanine, and glutamate, are crucial to T. cruzi survival. Recently, we described that T. cruzi can biosynthesize glutamine from glutamate and/or obtain it from the extracellular environment, and the role of glutamine in energetic metabolism and metacyclogenesis was demonstrated. In this study, we analysed the effect of glutamine analogues on the parasite life cycle. Here, we show that glutamine analogues impair cell proliferation, the developmental cycle during the infection of mammalian host cells and metacyclogenesis. Taken together, these results show that glutamine is an important metabolite for T. cruzi survival and suggest that glutamine analogues can be used as scaffolds for the development of new trypanocidal drugs. These data also reinforce the supposition that glutamine metabolism is an unexplored possible therapeutic target.
1. Introduction
Chagas disease (CD) is caused by the flagellate protist Trypanosoma cruzi and affects approximately 8 million people worldwide [1], with some 25 million further people at risk of infection. It is endemic in the Americas where the vector transmission occurs [2]. However, cases of CD have been reported not only in the Americas but also in Europe, Asia and Oceania, mostly due to migration of infected people from endemic countries. Although the worldwide distribution, the CD treatment available nowadays is based in two drugs discovered many years ago: nifurtimox and beznidazole. The effect of these drugs is controversial, depending on the stage of disease, age and immune response of patient and the susceptibility of the T. cruzi genotype, moreover cause many side effects and the treatment required a prolonged course. [3,4,5,6].
T. cruzi has a complex life cycle, alternating between two hosts, a reduviid insect and many mammalian species, including humans. During its life cycle, this parasite differentiates several times into replicative and non-replicative forms. The insect vectors become infected during the blood meal, when they ingest the non-replicative, infective trypomastigotes present in the host bloodstream. In the insect midgut, the trypomastigotes differentiate into the replicative epimastigotes, which colonize the insect digestive tract. Once in the posterior part of the midgut, epimastigotes differentiate into infective, non-replicative metacyclic trypomastigotes, which are later expelled with faeces during the intake of the next blood meal. The infection of the mammalian host occurs when these parasites enter into contact with mucosae or wounds in the skin. Once inside the host cells, metacyclic trypomastigotes must infect the mammalian host cells to reach the host cell cytoplasm, where they differentiate into the replicative form, the amastigotes. After intracellular proliferation, amastigotes differentiate into trypomastigotes, passing through a transient form called the intracellular epimastigote. Trypomastigotes burst from host cells by lysis and can (i) invade neighbouring cells, colonizing the tissue; (ii) and reach the bloodstream, allowing the infection of other tissues; and (iii) can be ingested by a new triatomine insect during its blood meal [3,4,7,8].
During its life cycle, T. cruzi faces different physicochemical, chemical and nutritional conditions in each environment. In addition, the availability of energy and carbon sources is highly variable, contributing to the selection of parasites with remarkable metabolic flexibility (reviewed elsewhere [9]). For example, it is well documented that T. cruzi is able to consume glucose preferentially at the beginning of the exponential proliferation phase, and after exhaustion of this metabolite, they switch to a metabolism that is mainly based on the consumption of amino acids [10,11,12]. It is well known that a variety of amino acids are robust energy sources [13,14,15,16,17,18]. In addition, amino acids are important to crucial processes such as energy management [19], cell proliferation [20,21], cell differentiation [13,22,23,24], host cell infection [15], intracellular survival [23,25], osmotic control [26] and resistance to different types of stress conditions [14,27,28,29].
Glutamine (Gln) is a non-essential amino acid synthesized by most organisms. Its participation in nitrogen metabolism inside cells has been well documented, mainly in yeast [30] and cyanobacteria [31]. In humans, this amino acid is found at high concentrations in the plasma and in skeletal muscle. In cancer cells, Gln also has a large range of functions, acting as an ATP and carbon source for the biosynthesis of lipids and stimulating cell proliferation, enhancing the redox potential and serving as a substrate for nucleotide biosynthesis [32].
Gln can be internalized from the external medium and/or synthesized through the enzyme glutamine synthetase (GS), which uses ammonium, ATP and glutamate (Glu) as substrates. In turn, Gln is used as an amide- or amine-group donor or as a substrate, being a true metabolic hub for the distribution of -NH2 groups through various biochemical pathways, such as those for amino sugar, guanosine, or pyrimidine biosynthesis. [32,33,34,35]. Recently, we demonstrated that T. cruzi is able to take up Gln from external medium [13] and synthesize it using a canonical biosynthesis pathway [36]. In those studies, we showed that the biosynthesis and uptake of Gln are developmentally regulated according to Gln availability in the extracellular environment and cell demand. We also showed that Gln biosynthesis participates in ammonium detoxification, which is critical for the parasite to escape the parasitophorous vacuole during host-cell infection [13,36].
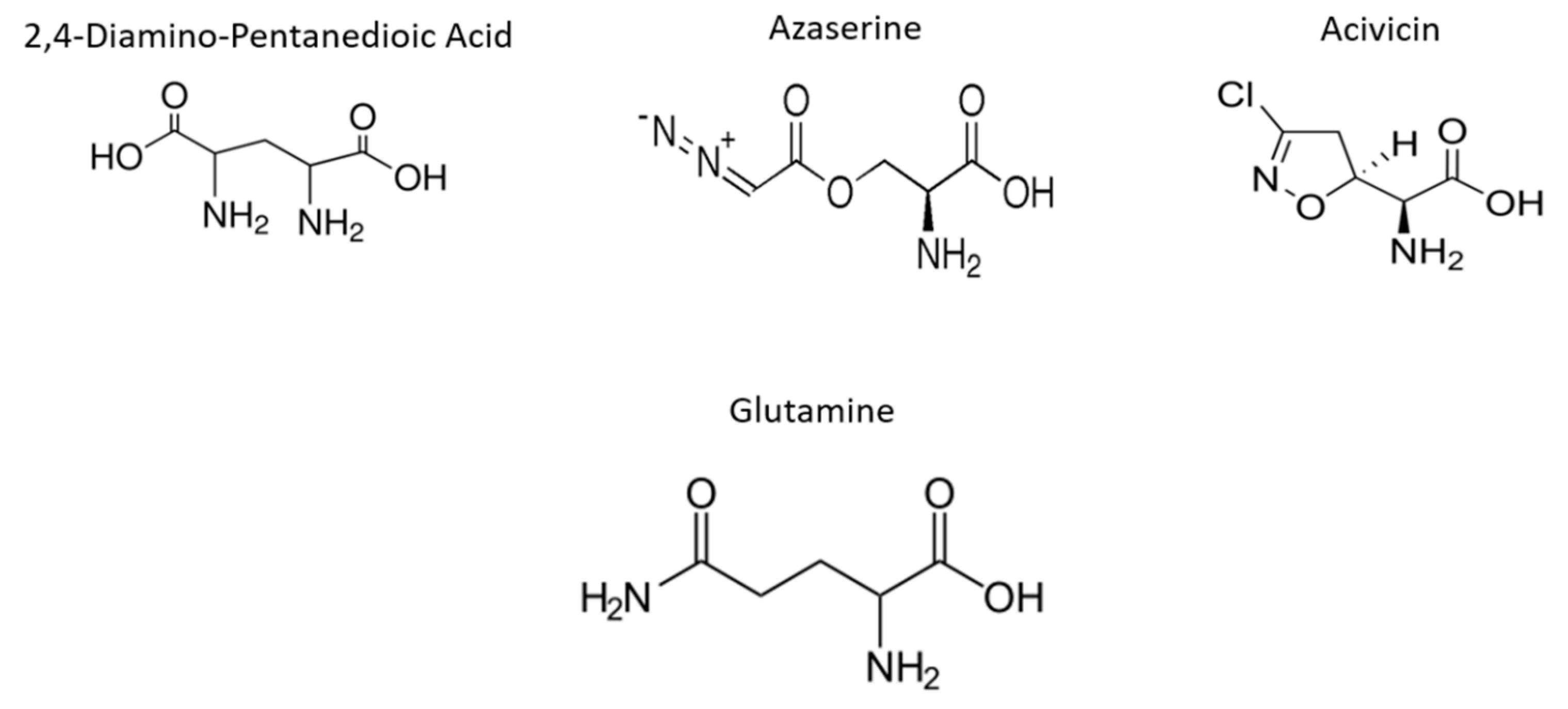
In the present study, we evaluated the impact of the Gln analogues acivicin (ACV), azaserine (AZA) and 2,4-diaminopentanedioic acid (DPA, Figure 1) on the cell biology and life cycle of T. cruzi according to the hypothesis that they will preferentially interfere with enzymes that recognize Gln as a substrate, product or allosteric modulator. Our results showed that these Gln analogues impair parasite proliferation and differentiation and affect the intracellular cycle and metacyclogenesis. Our data demonstrated that Gln metabolism in T. cruzi can be explored as therapeutic target.
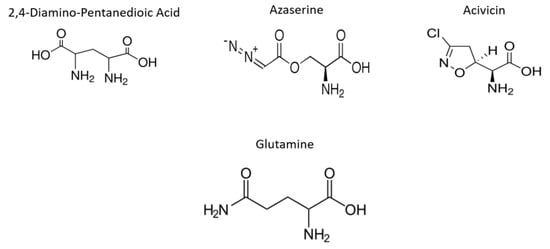
Figure 1.
Structure of Gln and its analogues. Gln analogues used to evaluate its interference in the biological process during the T. cruzi life cycle.
2. Results
2.1. Gln analogues Impair the Proliferation of the Epimastigote Form
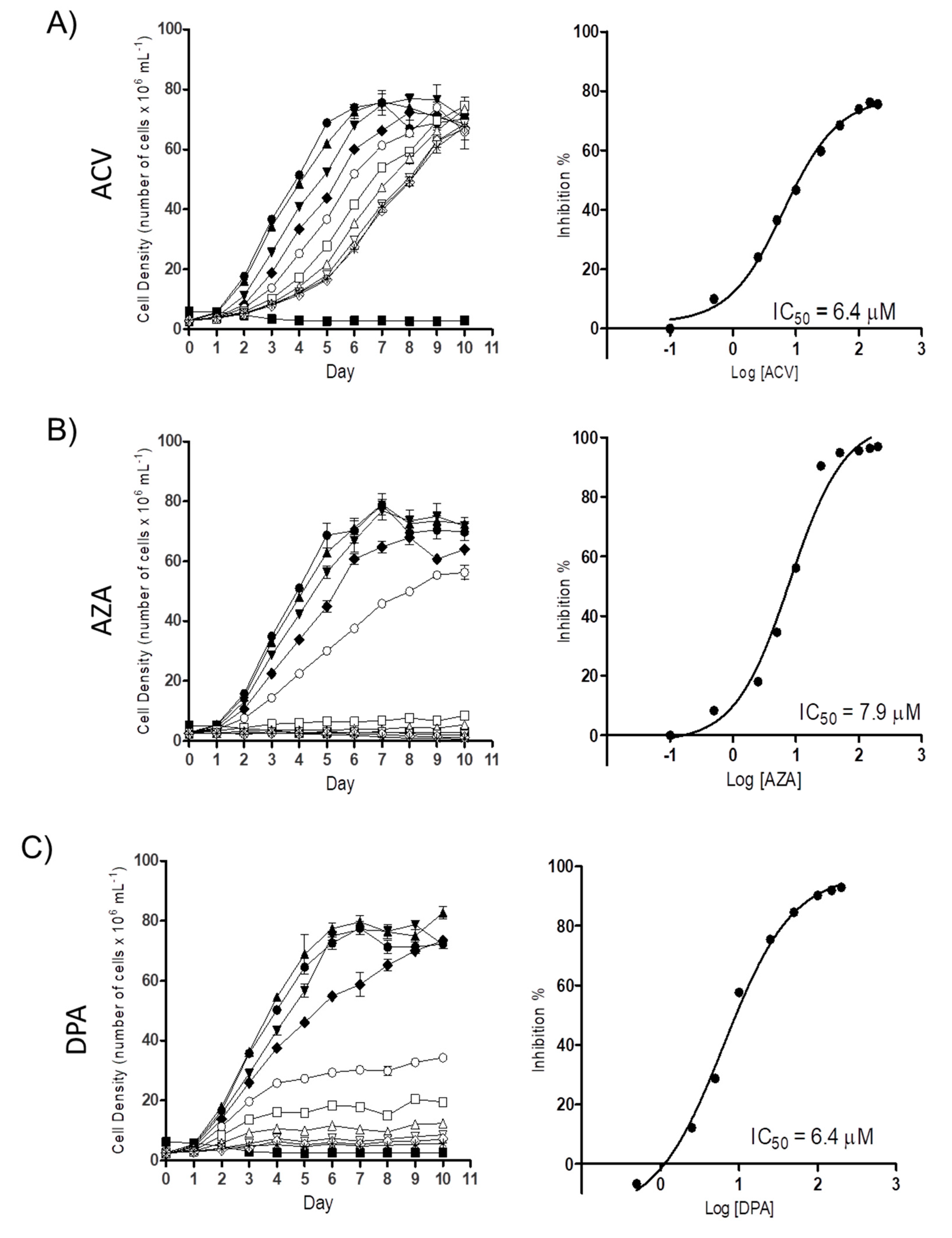
To determine the possible trypanocidal or trypanostatic effect of the Gln analogues on T. cruzi, we initially evaluated their effect on epimastigote proliferation. Proliferating parasites were treated with different concentrations of each Gln analogue (5 µM to 200 µM), and their cell densities were observed for as many as 10 days. All the analogues showed a dose-dependent effect on parasite proliferation. At the time corresponding to the mid-exponential growth phase (5th day of proliferation), the concentration corresponding to 50% inhibition (IC50) was calculated, with values of 6.2 ± 1.01 µM, 6.1 ± 2.1 µM and 6.2 ± 0.72 µM for ACV, AZA and DPA, respectively (Figure 2). These results showed that all three analogues had relevant activity against parasite proliferation at low micromolar range.
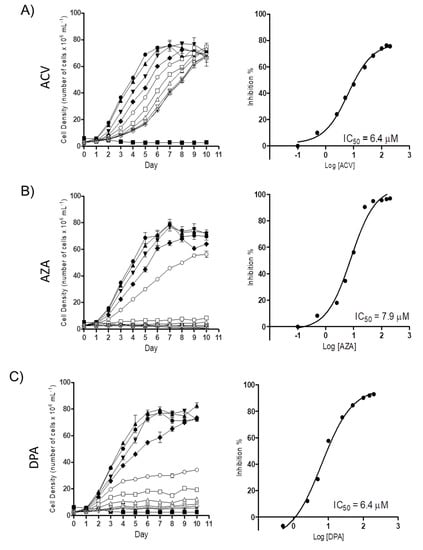
Figure 2.
The effect of the Gln analogues on the proliferation of the epimastigote form. Left panels: Epimastigote proliferation in LIT medium in the presence of ACV (A), AZA (B) or DPA (C) (from 0.5 µM to 200 µM) was observed for as many as ten days. Symbols: black circle: 0 µM; black up-pointing triangle: 0.5 µM; black down-pointing triangle: 2.5 µM; black diamond: 5 µM; white circle: 10 µM; white square: 25 µM; white up-pointing triangle: 50 µM; white down-pointing triangle: 100 µM; white diamond: 150 µM; and star: 200 µM. Inhibition control: 60 µM rotenone and 0.5 µM antimycin. Right panels: dose response curves derived from the proliferation curves (data from the 5th day) when the parasites were in the mid-exponential growth phase. Data were processed and plotted using the GraphPad Prism 6.1 software program. The assay was performed in biological triplicates.
2.2. Gln analogue ACV Induces Extracellular Phosphatidylserine Exposure
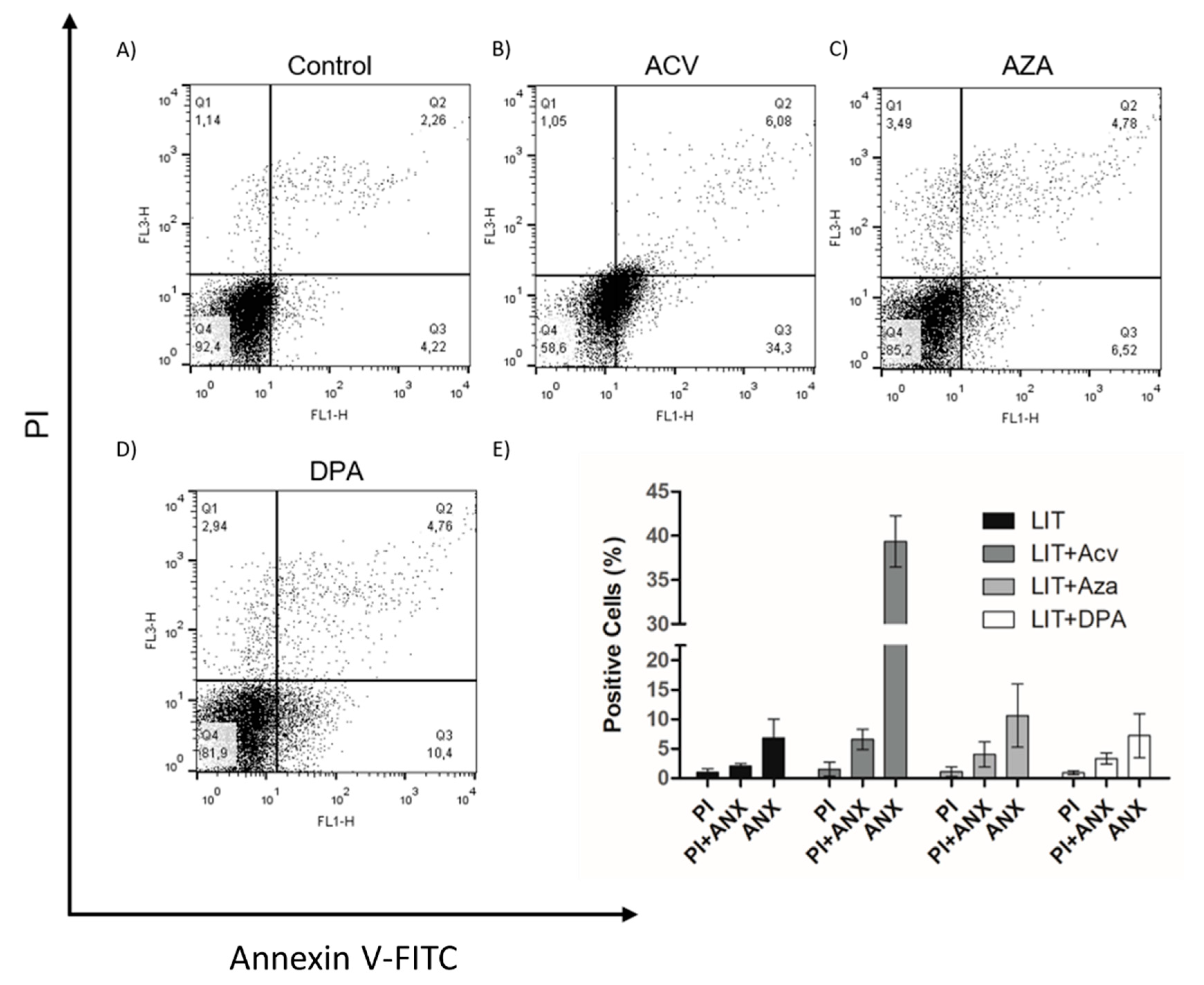
Once Gln analogues impaired cell proliferation, we evaluated whether they induced programmed cell death (PCD) or necrosis in the parasites. PCD is a process characterized by morphological, biochemical, and cellular events, such as extracellular phosphatidylserine exposure, DNA fragmentation, decreased ATP levels, and increased levels of ROS, Ca2+ and cytochrome c release [37,38]. To analyse whether the treatments of cells with the Gln analogues induced PCD, we treated epimastigotes with concentrations of the analogues equivalent to the IC50 value of each for 48 h. After the treatments, the parasites were labelled with propidium iodide (PI) to analyse membrane integrity and annexin V-FITC to evaluate phosphatidylserine exposure (PS) as determined by flow cytometry (Figure 3). Although all Gln analogues impaired cell proliferation, only ACV at a concentration corresponding to the IC50 induced PS exposure in approximately 34% of the cells. AZA and DPA at concentrations corresponding to their respective IC50 had no detectable effect on PS exposure. Under all conditions, the parasites maintained membrane integrity similar to that of the control. These data suggest that treatment with ACV affects parasite metabolism, triggering PCD, whereas AZA and DPA likely had only a trypanostatic effect.
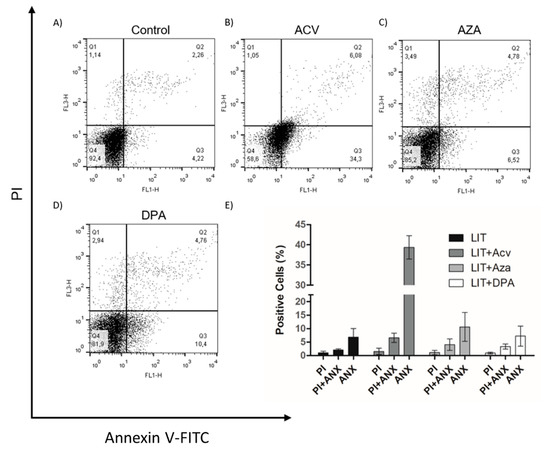
Figure 3.
The effect of Gln analogues on the extracellular phosphatidylserine exposure and membrane integrity. Parasites in the exponential growth phase were treated with Gln analogues (IC50 value) for 48 h. After incubation, the parasites were labelled with propidium iodide (PI) and annexin V-FITC (ANX) and analysed by flow cytometry; (A) Control, (B) ACV, (C) AZA, (D) DPA. (E) Bars correspond to three independent mean values from three independent experiments. Error bars represent the standard deviation among biological replicates.
2.3. Recovery of Parasites after Treatment with Gln analogues
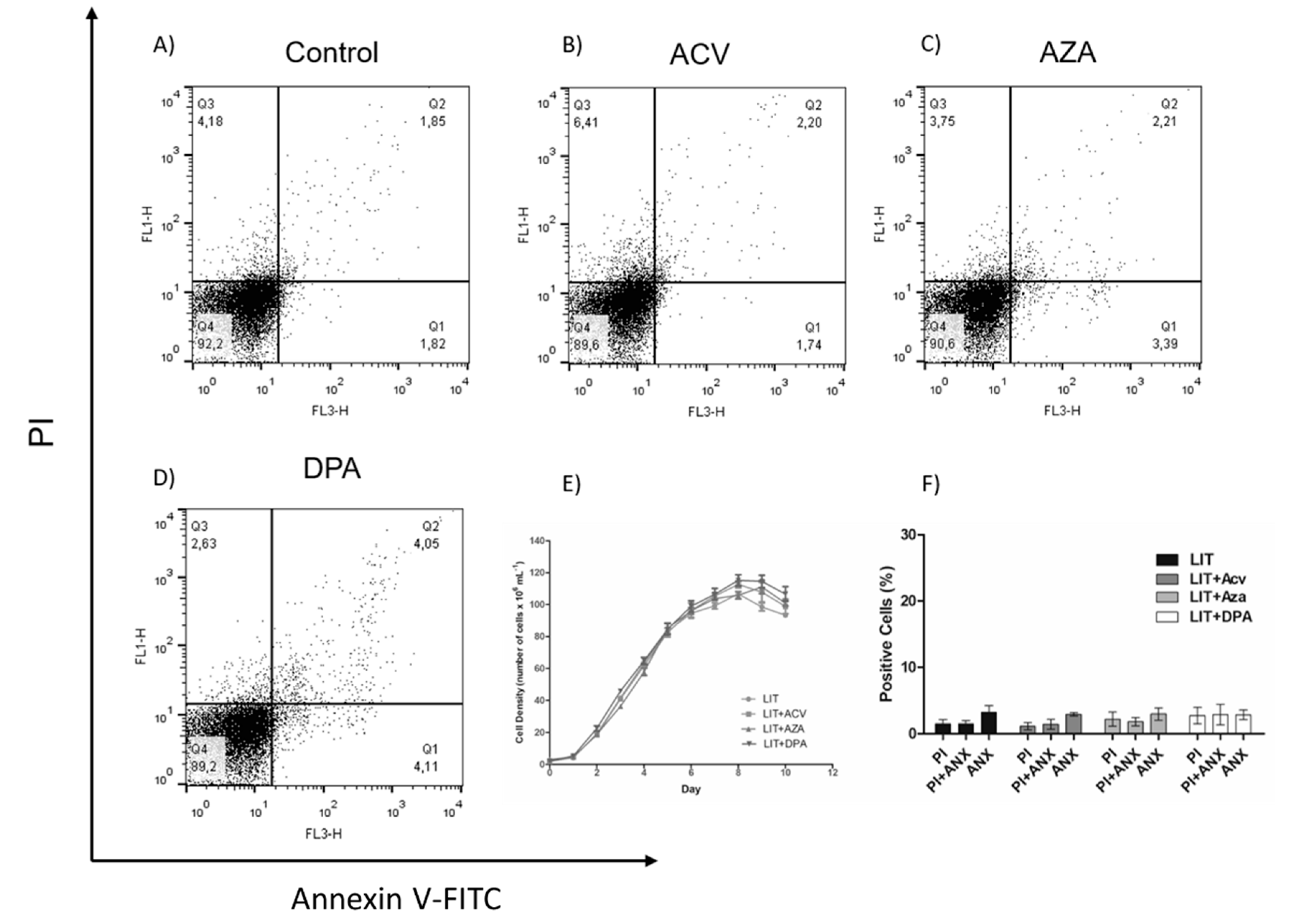
To evaluate whether the effect of the Gln analogues is reversible, epimastigotes were treated for 48 h with Gln analogues (concentrations corresponding to their IC50 values). Then, the compounds were washed away, and the parasites were maintained in LIT medium without Gln analogues. Parasite proliferation was observed for as many as ten days to analyse the recovery of their proliferative profile (Figure 4E). We also evaluated parasite cell death on the fifth day of recovery. To make these observations, the parasites were labelled with PI and annexin V-FITC (Figure 4A–D). Surprisingly, the obtained data showed that, in the case of ACV-induced membrane PS exposure, the PS exposure was reversed, and in all cases, the proliferation was recovered to the expected level when the treatments were removed. These data show that the Gln analogue effect (at the used concentrations) is trypanostatic rather than trypanocidal.
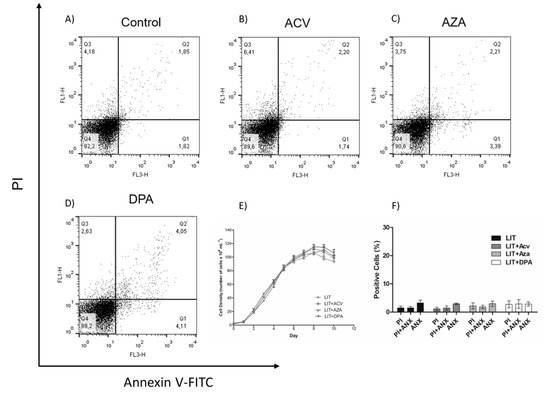
Figure 4.
Recovery of the epimastigotes after Gln analogues treatment. The parasites were treated for 48 h with Gln analogues (IC50 value concentration). Then, the treatment was removed, and the parasites were cultivated in LIT medium. Proliferation was observed for as many as 10 days (E). On the fifth day, the parasites were labelled with propidium iodide (PI) and annexin V-FITC to evaluate cell death by flow cytometry: (A) control, (B) ACV, (C) AZA and (D) DPA. (F) Bars correspond to three independent mean values from three independent experiments. Error bars represent the standard deviation among biological replicates.
2.4. Effect of Gln analogues on Metacyclogenesis
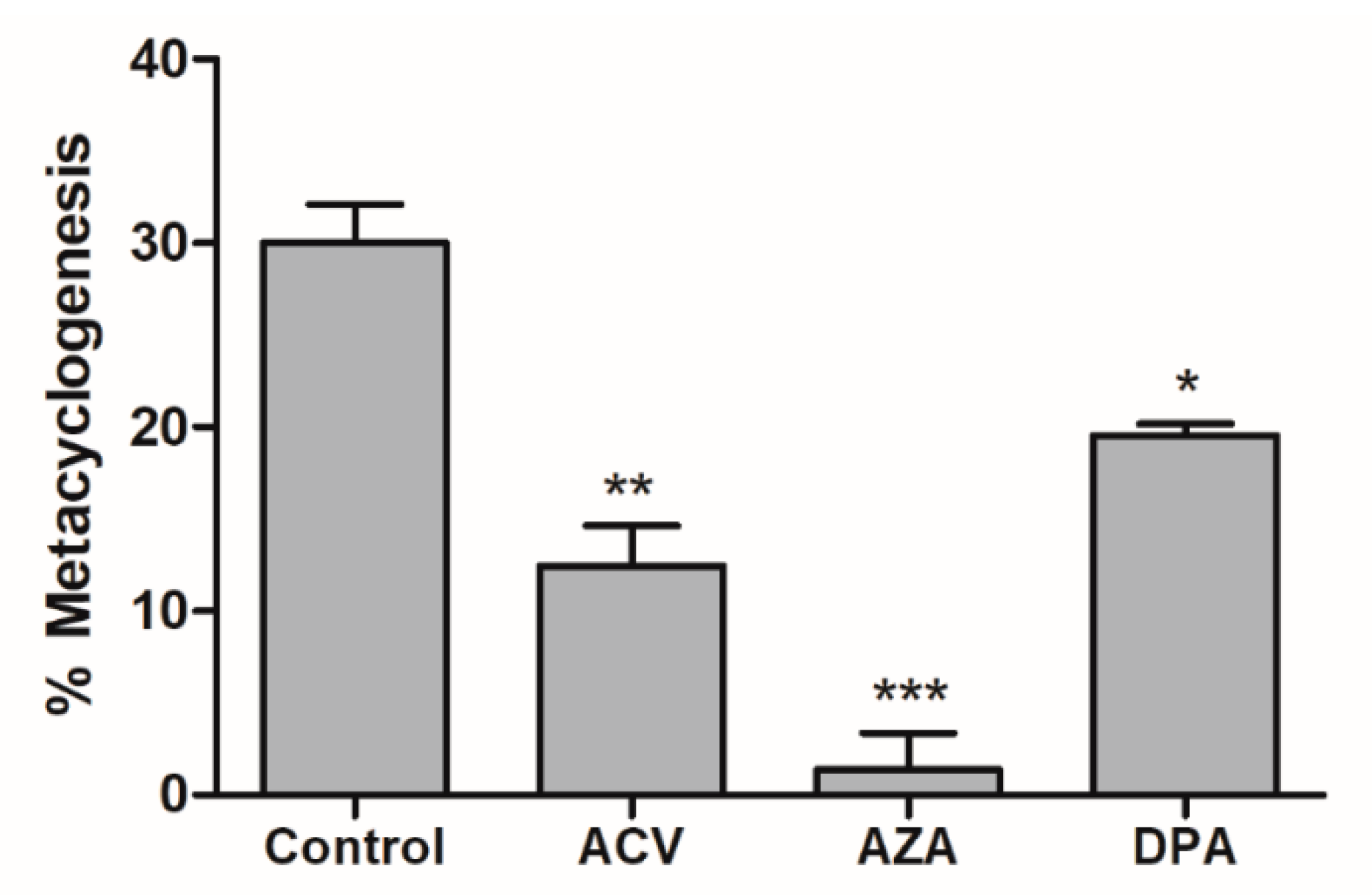
Metacyclogenesis is a crucial differentiation process involving intensive cell remodelling. This process can be performed in vitro by submitting epimastigotes to controlled (2 h) metabolic stress in TAU medium and then supplying carbon and energy sources. Contreras et al. showed that a combination of glucose, aspartate, glutamate and proline trigger and support this differentiation [22]; more recently, it was shown that Gln by itself is able to support metacyclogenesis to levels comparable to those of parasites in standard medium (TAU 3AAG) [13]. To evaluate the effect of the Gln analogues on the metacyclogenesis promoted by Gln, parasites in the epimastigote form in the stationary growth phase were induced to differentiation in TAU-Gln in the presence of each Gln analogue (IC50 value). The obtained data showed that the Gln analogues acted as metacyclogenesis inhibitors, decreasing the differentiation from the epimastigote to the metacyclic trypomastigote form. The inhibition was approximately 59%, 90% and 35% for ACV, AZA and DPA, respectively (Figure 5).
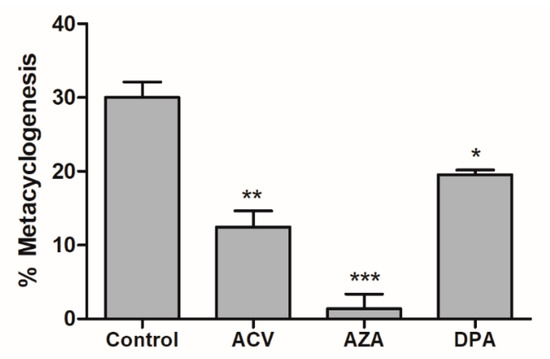
Figure 5.
Effect of Gln analogues on the metacyclogenesis process. The epimastigote parasites (stationary growth phase) were maintained in TAU Gln medium in the presence of each Gln analogue (IC50 value concentrations) or not (control). The differentiation was evaluated by counting in a Neubauer chamber on the 6th day. This experiment was performed in triplicate. Statistical analysis: One-way ANOVA followed by Tukey post-test; p < 0.05, using the GraphPad Prism 6.1 software program. The assays were performed in biological triplicates and error bars represent the standard deviation. We represent in this figure the level of statistical significance as follow: *** p value < 0.001; ** p value < 0.01; * p value < 0.05.
2.5. Effect of the Gln analogues on the Intracellular Infection of T. cruzi
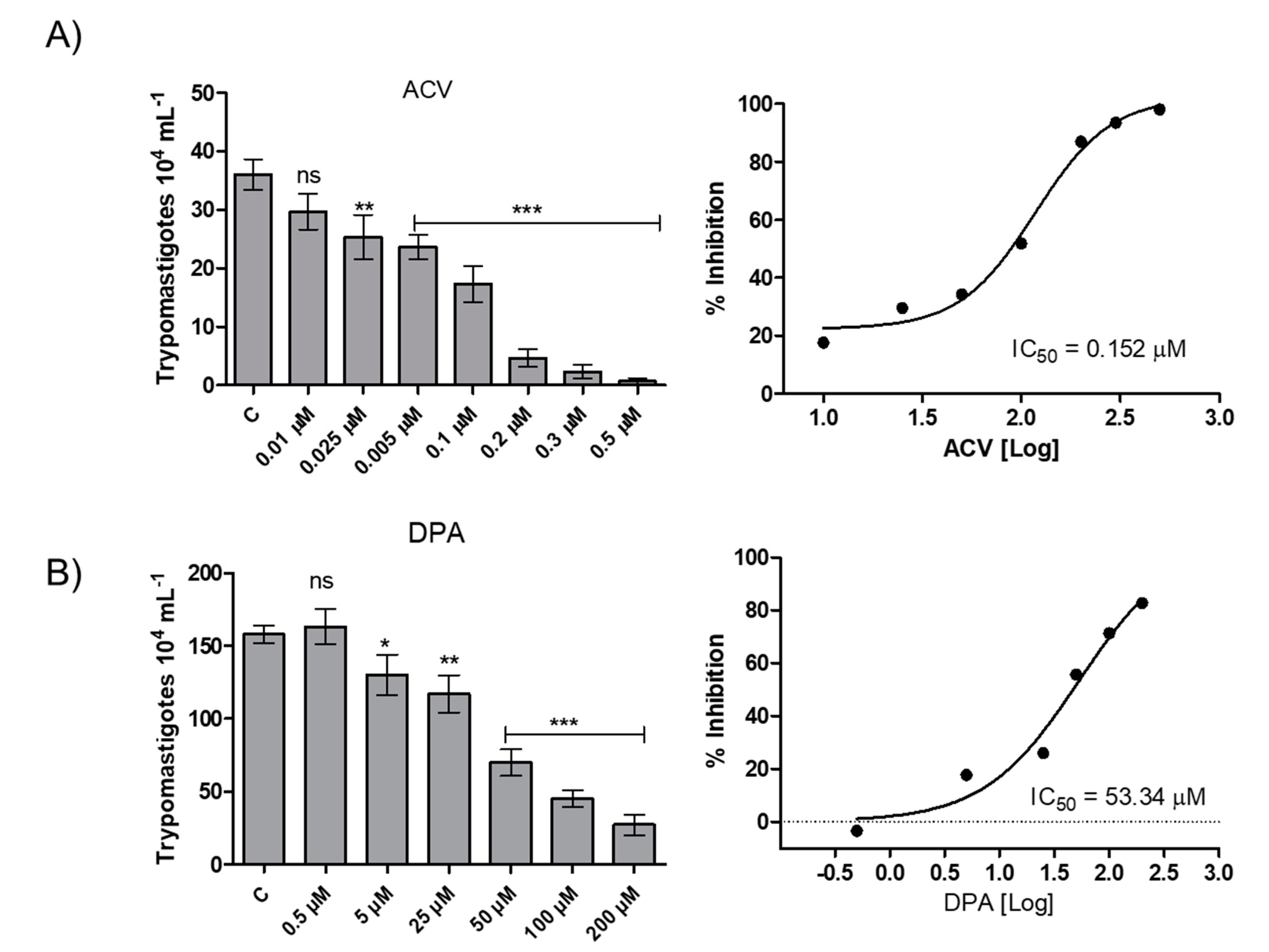
To evaluate the effect of the Gln analogues on the infection in mammalian host cells, we initially evaluated the analogues toxicity in CHO-K1 cells, the cell system chosen as the parasitic host cell. The AZA and ACV analogues were toxic at concentrations above 0.5 µM, whereas the DPA analogue had no toxic effect at concentrations up to 200 µM (Figure S1). Then, CHO-K1 cells were infected with trypomastigotes and treated with Gln analogues at concentrations up to their toxicity limit, and the number of trypomastigotes released into the culture medium from the infected cells was quantified. AZA at a concentration of 0.5 µM inhibited 20% of trypomastigote burst (Figure S2). ACV and DPA showed a dose-dependent inhibition of trypomastigote burst in the infected cells, with IC50 values of 0.152 µM and 53.3 µM, respectively (Figure 6). Given that DPA has a relevant effect on intracellular parasites at a high concentration, our data re-establish interest in the use of ACV as a potential starting compound for developing a trypanocidal agent against T. cruzi.
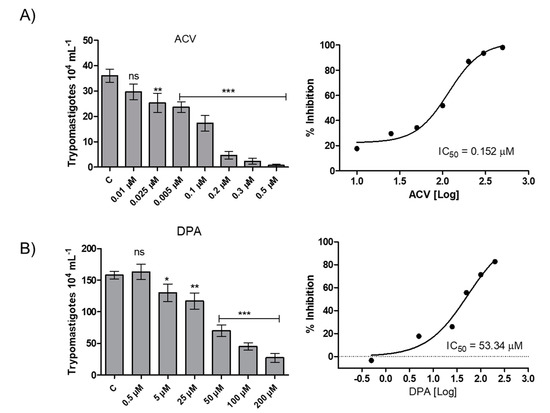
Figure 6.
Effect of the Gln analogues on the intracellular cycle of T. cruzi. CHO-K1-infected cells were treated with different concentrations of each Gln analogue. On the fifth day post-infection, the burst trypomastigotes were counted in a Neubauer chamber. (A) ACV, (B) DPA. The IC50 value was calculated using a classical sigmoidal dose-response curve, and the statistical analysis was performed with one-way ANOVA followed by Tukey post-test p < 0.05, using the GraphPad Prism 6.1 software program. We represent in this figure the level of statistical significance as follow: *** p value < 0.001; ** p value < 0.01; * p value < 0.05. For p value > 0.05 we consider the differences not significant (ns).
3. Discussion
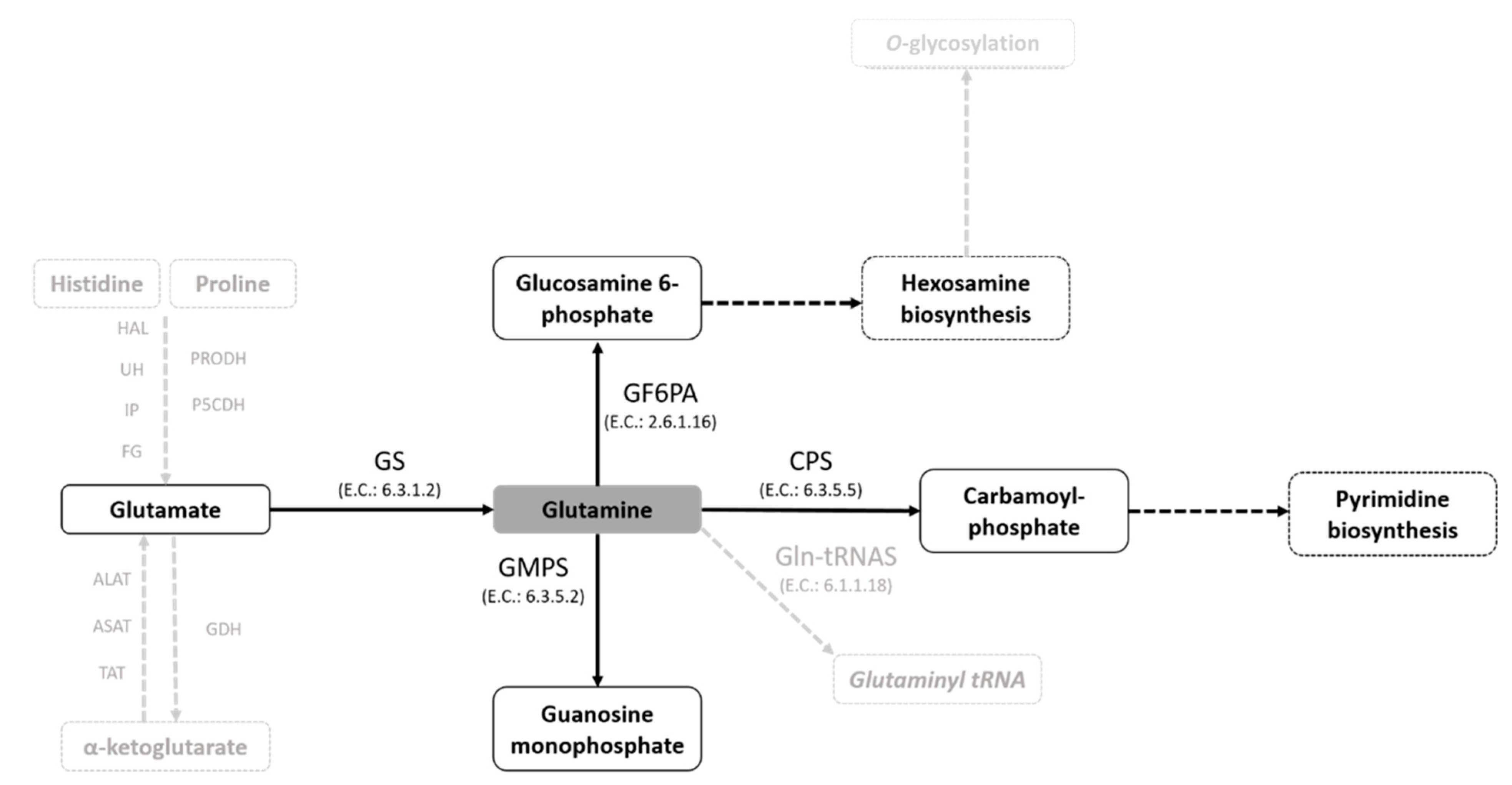
In this study, we demonstrate the effect of the Gln analogues ACV, AZA and DPA on the life cycle of T. cruzi. We had previously shown that T. cruzi can take up Gln and/or engage in biosynthesis in all stages of the T. cruzi life cycle, except for the bloodstream trypomastigotes. We also demonstrated that Gln supports the differentiation from the epimastigote to the metacyclic trypomastigote form, maintaining cell viability during metacyclogenesis and participating in energy metabolism [13,36]. In addition, in T. cruzi, Gln can be the substrate for extremely important metabolic pathways of cell survival (Figure 7).
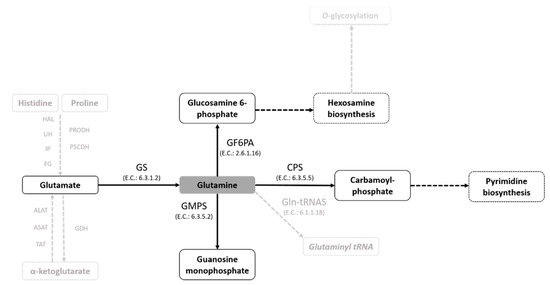
Figure 7.
Glutamine-dependent metabolic pathways in T. cruzi. A schematic view of Gln metabolism in T. cruzi based on the gene sequences annotated in genome databases (TriTrypDB and KEGG pathway databases).
The Gln analogues used in this study did not impair Gln uptake [13] or the T. cruzi GS activity (Figure S3). However, all of them inhibited the T. cruzi enzyme glutamine fructose-6-phosphate aminotransferase (GF6PA) activity (Figure S4). GF6PA is an enzyme that utilizes Gln as an amino donor for glucosamine biosynthesis and is the first step in the hexosamine biosynthetic pathway (HBP). Particularly, AZA has been described as an inhibitor of HBP, and its effects are related to glucose homeostasis and glycosylation of proteins associated with insulin resistance in type II diabetes [39,40]. In trypanosomatids, AZA was studied many years ago mainly to assess its action as an inhibitor of nucleic acid metabolism, with the findings demonstrating that AZA does not affect purine or pyrimidine biosynthesis [41]. In early infections in mice with Trypanosoma equiperdum, treatment with AZA diminished the number of parasites [42]. In this parasite, the analogue inhibits the incorporation of sugar nucleotides, impairing nucleic acid metabolism. The same study showed that Gln and other amino acids diminish the effects of AZA [43]. Interestingly, the findings reported herein on the inhibition of HBP by AZA are in agreement with the absence of effects previously reported for T. cruzi nucleic acid metabolism.
The analogue DPA also had an anti-proliferative effect on the epimastigote forms, inhibiting GF6PA activity and impairing the intracellular cycle of the parasite. Moreover, DPA was well tolerated by the host cell compared with other analogues studied in this study (four hundred-fold less toxicity). Interestingly, we did not find any description in the literature about the mechanism of action for this compound, and to our knowledge, this is the first study to report the possible cell targets for this Gln analogue.
ACV is a Gln analogue produced by fermentation of Streptomyces sviceus and was previously studied as a potential anticancer drug [44]. The best-known mechanism of action depends on the inhibition of CTP synthetase, an enzyme critical for the biosynthesis of sugar nucleotides. Many side effects were reported from the clinical trials of ACV as an anticancer drug and are explained by several instances of off-targeting inside the cell. ACV is an inhibitor of not only Gln-dependent enzymes but also enzymes with aldehyde dehydrogenase activity [45]. When studied in trypanosomatids, this Gln analogue was found to act as an inhibitor of CTPs in T. brucei, and was thus proposed as a compound useful for guiding the synthesis of new anti-T. brucei drugs [46]. Additionally, for T. brucei, ACV was described as an inhibitor of GMP synthase, another enzyme that utilizes Gln as an amide donor [47]. Our results showed that ACV is a T. cruzi proliferation inhibitor, specifically of those in the epimastigote form, in a dose-dependent manner. In addition, this analogue induced extracellular phosphatidylserine exposure, indicating that the PCD process was triggered. PCD in trypanosomatids is a well-characterized process, and parasites can trigger PCD after drug treatment or cultivation [48,49,50]. Interestingly, in this case, the effect of phosphatidylserine exposure was reversed after the ACV treatment was removed. ACV has also been shown to be effective in the intracellular cycle, diminishing the number of burst trypomastigotes in a dose-dependent response at concentrations in the nanomolar range. Importantly, ACV inhibits TcGF6PA activity, making TcGF6PA a putative new target for ACV. These data suggest that, as described for T. brucei, ACV can be used as a scaffold compound to guide the synthesis of new molecules against T. cruzi with few or no side effects in the host.
Finally, we demonstrated that Gln analogues impair the differentiation from the epimastigote to the metacyclic trypomastigote form that had been promoted by Gln, reinforcing the importance of this amino acid in the metacyclogenesis process. Taken together, these results showed the importance of Gln for T. cruzi survival. Gln analogues impaired important biological processes, such as cell proliferation, differentiation and the intracellular cycle.
4. Materials and Methods
4.1. Parasites and Mammalian Cells
The epimastigote form of the T. cruzi CL strain and 14 clones were maintained in the exponential phase by sub-culturing every 48 h in liver infusion tryptose (LIT) medium supplemented with heat-inactivated FCS (foetal calf serum) 10% at 28 °C [51]. Chinese hamster ovary cells (CHO-K1 cells) were maintained in RPMI medium supplemented with FCS 10%, 0.15% (w/v) NaCO3, 100 units·mL−1 penicillin and 100 g·mL−¹ streptomycin in a humidified atmosphere with 5% CO2 [23].
4.2. Proliferation Inhibition Assays
T. cruzi epimastigote forms in the exponential growth phase (4.0–6.0 × 107 mL−1) were treated with different concentrations of each analogue: ACV, AZA and DPA (purchased from Sigma-Aldrich, San Luis, MO, USA) or not treated (negative control) in LIT medium. As a positive control, we used a combination of rotenone (60 µM) and antimycin (0.5 µM) [28]. The parasites (2.5 × 106 mL−1) were transferred to 96-well plates and incubated at 28 °C. Quantification of cell proliferation was performed by reading the optical density (OD) at λ 620 nm for eight days. The OD values were converted to cell numbers using a linear regression equation previously obtained under the same conditions. The IC50 was calculated by using the cell numbers obtained in the exponential growth phase (5th day of proliferation). To determine the IC50 value, the obtained values were fitted to a nonlinear regression function: a sigmoidal dose-response curve [28,48]. Each compound was evaluated in biological triplicates with technical quadruplicates in each experiment.
4.3. Extracellular Phosphatidylserine Exposure Analysis
Epimastigotes in the exponential growth phase were maintained in LIT and treated with Gln analogues (concentrations corresponding to the IC50) for 48 h. Then, the parasites were washed in annexin buffer (10 mM HEPES, 140 mM NaCl, and 5 mM CaCl2, pH 7.4) and resuspended in 50 µL of annexin buffer. The cells were incubated with a mixture of propidium iodide (1 µg·µL−1) and annexin (1 µM) for 15 min at room temperature. After incubation, the reaction was stopped by the addition of annexin buffer (450 µL), and the cells were analysed by flow cytometry (FACSCalibur, BD Biosciences, Franklin Lakes, NJ, USA) as previously described [48].
4.4. Effect of the Gln analogues on Metacyclogenesis
Epimastigotes were maintained in LIT medium until reaching the stationary phase (~1.0 × 108 cells·mL−1), washed twice with PBS, resuspended in triatomine artificial urine (TAU) medium (190 mM NaCl, 17 mM KCl, 2 mM MgCl2, 2 mM CaCl2, and 8 mM potassium phosphate buffer, pH 6.0) [22], and incubated for 2 h at 28 °C. Then, the cells were centrifuged (5 min, 1600× g at room temperature) and resuspended in TAU Gln (190 mM NaCl, 17 mM KCl, 2 mM MgCl2, 2 mM CaCl2, 8 mM potassium phosphate buffer, and 10 mM Gln, pH 6.0) [13] in the presence or absence (control) of each Gln analogue: ACV, AZA or DPA (concentration corresponding to the IC50). On the 6th day, after triggering metacyclogenesis, the parasites were counted in a Neubauer chamber, and the number and percentage of the MTs were calculated.
4.5. Effect of the Gln analogues on Mammalian Cell Viability
Chinese hamster ovary cell line (CHO-K1) cells (5.0 × 105 per well) were maintained in 24-well plates in RPMI medium supplemented with FCS (10% v/v) at 37 °C and 5% CO2 in the presence or absence (control) of different concentrations of each Gln analogue for 48 h. After the incubation time, cell viability was evaluated by MTT assay [52]. The cells were washed twice in PBS, and PBS (200 µl) and MTT (0.25 mg/mL) were added to each well. The cells were incubated for 3 h (37 °C in 5% CO2), and the reaction was stopped by the addition of SDS (200 µL of 10% SDS per well). The cells were lysed by up and down pipetting. The viability (colorimetric reaction) was measured by spectrophotometry at 595–690 nm in a SpectraMax i3 spectrophotometer (Molecular Devices, San Jose, CA, USA).
4.6. Effect of the Gln analogues on Trypomastigote Bursting
CHO-K1 cells (5.0 × 104 per well) were maintained in 24-well plates as described previously. Briefly, the cells were infected with the trypomastigote form (2.5 × 106 trypomastigotes per well) for 3 h. Then, the parasites remaining in the supernatant were removed, the cells were washed twice with PBS, and RPMI medium was added to the culture. The infected cells were treated with different concentrations of each Gln analogue and kept in an incubator (37 °C with a 5% CO2 humid atmosphere). On the fifth day after infection, the trypomastigotes present in the supernatant were counted in a Neubauer chamber.
Supplementary Materials
The following are available online: Figure S1: Viability of CHO-K1 cells treated with different concentrations of ACV and DPA. Figure S2: Effect of AZA on cell viability and trypomastigote burst. Figure S3: Effects of Gln analogues on GS activity in the extracts from the epimastigotes. Figure S4: Effects of Gln analogues on GF6PA activity in the extracts from the epimastigotes.
Author Contributions
Conceptualization, data curation and formal analysis: R.O.O.S., A.M.S. and F.S.D.; methodology and investigation: R.O.O.S., M.C. and F.S.D.; writing-original draft preparation: R.O.O.S.; supervision, writing-review and editing: A.M.S. and F.S.D.; project administration and funding acquisition: A.M.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo, grant numbers 2016/06034-2; Conselho Nacional de Pesquisas Científicas e Tecnológicas (CNPq), grant number 308351/2013-4 and 404769/2018-7; the UK Research and Innovation c, grant number MR/P027989/1.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- WHO. WHO|Chagas Disease (American Trypanosomiasis); WHO: Geneva, Switzerland, 2018; Available online: http://www.who.int/chagas/en (accessed on 29 January 2019).
- Browne, A.J.; Guerra, C.A.; Alves, R.V.; da Costa, V.M.; Wilson, A.L.; Pigott, D.M.; Hay, S.I.; Lindsay, S.W.; Golding, N.; Moyes, C.L. The contemporary distribution of Trypanosoma cruzi infection in humans, alternative hosts and vectors. Sci. Data 2017, 4, 170050. [Google Scholar] [CrossRef]
- Lisvane Silva, P.; Mantilla, B.S.; Barison, M.J.; Wrenger, C.; Silber, A.M. The uniqueness of the Trypanosoma cruzi mitochondrion: Opportunities to identify new drug target for the treatment of Chagas disease. Curr. Pharm. Des. 2011, 17, 2074–2099. [Google Scholar] [CrossRef] [PubMed]
- Perez-Molina, J.A.; Molina, I. Chagas disease. Lancet 2018, 391, 82–94. [Google Scholar] [CrossRef]
- Lidani, K.C.F.; Andrade, F.A.; Bavia, L.; Damasceno, F.S.; Beltrame, M.H.; Messias-Reason, I.J.; Sandri, T.L. Chagas disease: From discovery to a worldwide health problem. Front. Public Health 2019, 7, 166. [Google Scholar] [CrossRef]
- Urbina, J.A. Specific chemotherapy of Chagas disease: Relevance, current limitations and new approaches. Acta Trop. 2010, 115, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Rassi, A., Jr.; Rassi, A.; Marcondes de Rezende, J. American trypanosomiasis (Chagas disease). Infect. Dis. Clin. N. Am. 2012, 26, 275–291. [Google Scholar] [CrossRef] [PubMed]
- Bern, C. Chagas’ Disease. N. Engl. J. Med. 2015, 373, 1882. [Google Scholar] [CrossRef] [PubMed]
- Marchese, L.; Nascimento, J.; Damasceno, F.; Bringaud, F.; Michels, P.; Silber, A. The uptake and metabolism of amino acids, and their unique role in the biology of pathogenic trypanosomatids. Pathogens 2018, 7, 36. [Google Scholar] [CrossRef]
- Cazzulo, J.J. Energy metabolism in Trypanosoma cruzi. Subcell Biochem. 1992, 18, 235–257. [Google Scholar]
- Silber, A.M.; Colli, W.; Ulrich, H.; Alves, M.J.; Pereira, C.A. Amino acid metabolic routes in Trypanosoma cruzi: Possible therapeutic targets against Chagas’ disease. Curr. Drug Targets Infect. Disord. 2005, 5, 53–64. [Google Scholar] [CrossRef][Green Version]
- Barison, M.J.; Rapado, L.N.; Merino, E.F.; Furusho Pral, E.M.; Mantilla, B.S.; Marchese, L.; Nowicki, C.; Silber, A.M.; Cassera, M.B. Metabolomic profiling reveals a finely tuned, starvation-induced metabolic switch in Trypanosoma cruzi epimastigotes. J. Biol. Chem. 2017, 292, 8964–8977. [Google Scholar] [CrossRef] [PubMed]
- Damasceno, F.S.; Barison, M.J.; Crispim, M.; Souza, R.O.O.; Marchese, L.; Silber, A.M. L-Glutamine uptake is developmentally regulated and is involved in metacyclogenesis in Trypanosoma cruzi. Mol. Biochem. Parasitol. 2018, 224, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Paes, L.S.; Suarez Mantilla, B.; Zimbres, F.M.; Pral, E.M.; Diogo de Melo, P.; Tahara, E.B.; Kowaltowski, A.J.; Elias, M.C.; Silber, A.M. Proline dehydrogenase regulates redox state and respiratory metabolism in Trypanosoma cruzi. PLoS ONE 2013, 8, e69419. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.M.; Covarrubias, C.; Rojas, R.G.; Silber, A.M.; Yoshida, N. Use of L-proline and ATP production by Trypanosoma cruzi metacyclic forms as requirements for host cell invasion. Infect. Immun. 2009, 77, 3023–3032. [Google Scholar] [CrossRef] [PubMed]
- Sylvester, D.; Krassner, S.M. Proline metabolism in Trypanosoma cruzi epimastigotes. Comp. Biochem. Physiol. B 1976, 55, 443–447. [Google Scholar] [CrossRef]
- Barisón, M.J.; Damasceno, F.S.; Mantilla, B.S.; Silber, A.M. The active transport of histidine and its role in ATP production in Trypanosoma cruzi. J. Bioenerg. Biomembr. 2016, 48, 437–449. [Google Scholar]
- Mantilla, B.S.; Paes, L.S.; Pral, E.M.F.; Martil, D.E.; Thiemann, O.H.; Fernández-Silva, P.; Bastos, E.L.; Silber, A.M. Role of Δ1-pyrroline-5-carboxylate dehydrogenase supports mitochondrial metabolism and host-cell invasion of Trypanosoma cruzi. J. Biol. Chem. 2015, 290, 7767–7790. [Google Scholar] [CrossRef]
- Pereira, C.A.; Alonso, G.D.; Paveto, M.C.; Flawia, M.M.; Torres, H.N. L-arginine uptake and L-phosphoarginine synthesis in Trypanosoma cruzi. J. Eukaryot. Microbiol. 1999, 46, 566–570. [Google Scholar] [CrossRef]
- Pereira, C.A.; Alonso, G.D.; Ivaldi, S.; Silber, A.; Alves, M.J.M.; Bouvier, L.A.; Flawiá, M.M.; Torres, H.N. Arginine metabolism in Trypanosoma cruzi is coupled to parasite stage and replication. FEBS Lett. 2002, 526, 111–114. [Google Scholar] [CrossRef]
- Pereira, C.A.; Alonso, G.D.; Ivaldi, S.; Silber, A.M.; Alves, M.J.M.; Torres, H.N.; Flawiá, M.M. Arginine kinase overexpression improves Trypanosoma cruzi survival capability. FEBS Lett. 2003, 554, 201–205. [Google Scholar] [CrossRef]
- Contreras, V.T.; Salles, J.M.; Thomas, N.; Morel, C.M.; Goldenberg, S. In vitro differentiation of Trypanosoma cruzi under chemically defined conditions. Mol. Biochem Parasitol. 1985, 16, 315–327. [Google Scholar] [CrossRef]
- Tonelli, R.R.; Silber, A.M.; Almeida-de-Faria, M.; Hirata, I.Y.; Colli, W.; Alves, M.J. L-proline is essential for the intracellular differentiation of Trypanosoma cruzi. Cell Microbiol. 2004, 6, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Homsy, J.J.; Granger, B.; Krassner, S.M. Some factors inducing formation of metacyclic stages of Trypanosoma cruzi. J. Protozool. 1989, 36, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Silber, A.M.; Tonelli, R.R.; Lopes, C.G.; Cunha-e-Silva, N.; Torrecilhas, A.C.; Schumacher, R.I.; Colli, W.; Alves, M.J. Glucose uptake in the mammalian stages of Trypanosoma cruzi. Mol. Biochem Parasitol. 2009, 168, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Rohloff, P.; Rodrigues, C.O.; Docampo, R. Regulatory volume decrease in Trypanosoma cruzi involves amino acid efflux and changes in intracellular calcium. Mol. Biochem Parasitol. 2003, 126, 219–230. [Google Scholar] [CrossRef]
- Magdaleno, A.; Suarez Mantilla, B.; Rocha, S.C.; Pral, E.M.; Silber, A.M. The Involvement of Glutamate Metabolism in the Resistance to Thermal, Nutritional, and Oxidative Stress in Trypanosoma cruzi. Enzym. Res. 2011, 2011, 486928. [Google Scholar] [CrossRef]
- Magdaleno, A.; Ahn, I.Y.; Paes, L.S.; Silber, A.M. Actions of a proline analogue, L-thiazolidine-4-carboxylic acid (T4C), on Trypanosoma cruzi. PLoS ONE 2009, 4, e4534. [Google Scholar] [CrossRef]
- Saye, M.; Miranda, M.R.; di Girolamo, F.; de los Milagros Camara, M.; Pereira, C.A. Proline modulates the Trypanosoma cruzi resistance to reactive oxygen species and drugs through a novel D, L-proline transporter. PLoS ONE 2014, 9, e92028. [Google Scholar] [CrossRef]
- Soberon, M.; Gonzalez, A. Physiological role of glutaminase activity in Saccharomyces cerevisiae. J. Gen. Microbiol. 1987, 133, 1–8. [Google Scholar] [CrossRef]
- Ames, T.D.; Breaker, R.R. Bacterial aptamers that selectively bind glutamine. RNA Biol. 2011, 8, 82–89. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Cheng, T. Q’s next: The diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 2010, 29, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, J.; Lou, L. Biochemical characterization of human GMP synthetase. J. Biol. Chem. 1995, 270, 7347–7353. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, J.; Straub, K.; Wu, J.; Lou, L. The glutamine hydrolysis function of human GMP synthetase. Identification of an essential active site cysteine. J. Biol. Chem. 1995, 270, 23450–23455. [Google Scholar] [CrossRef] [PubMed]
- Raushel, F.M.; Thoden, J.B.; Holden, H.M. The amidotransferase family of enzymes: Molecular machines for the production and delivery of ammonia. Biochemistry 1999, 38, 7891–7899. [Google Scholar] [CrossRef]
- Crispim, M.; Damasceno, F.S.; Hernandez, A.; Barison, M.J.; Pretto Sauter, I.; Souza Pavani, R.; Santos Moura, A.; Pral, E.M.F.; Cortez, M.; Elias, M.C.; et al. The glutamine synthetase of Trypanosoma cruzi is required for its resistance to ammonium accumulation and evasion of the parasitophorous vacuole during host-cell infection. PLoS Negl. Trop. Dis. 2018, 12, e0006170. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Aaronson, S.A.; Abrams, J.; Alnemri, E.S.; Andrews, D.W.; Baehrecke, E.H.; Bazan, N.G.; Blagosklonny, M.V.; Blomgren, K.; Borner, C.; et al. Guidelines for the use and interpretation of assays for monitoring cell death in higher eukaryotes. Cell Death Differ. 2009, 16, 1093–1107. [Google Scholar] [CrossRef]
- van Zandbergen, G.; Luder, C.G.; Heussler, V.; Duszenko, M. Programmed cell death in unicellular parasites: A prerequisite for sustained infection? Trends Parasitol. 2010, 26, 477–483. [Google Scholar] [CrossRef]
- O’Rourke, R.W.; Meyer, K.A.; Gaston, G.; White, A.E.; Lumeng, C.N.; Marks, D.L. Hexosamine biosynthesis is a possible mechanism underlying hypoxia’s effects on lipid metabolism in human adipocytes. PLoS ONE 2013, 8, e71165. [Google Scholar]
- Rajapakse, A.G.; Ming, X.F.; Carvas, J.M.; Yang, Z. The hexosamine biosynthesis inhibitor azaserine prevents endothelial inflammation and dysfunction under hyperglycemic condition through antioxidant effects. Am. J. Physiol. Hear. Circ. Physiol. 2009, 296, H815–H822. [Google Scholar] [CrossRef]
- Yoneda, S. Some aspects of the purine metabolism of Trypanosoma cruzi in tissue culture. Rev. Bras. Pesqui. Med. Biol. 1971, 4, 205–218. [Google Scholar]
- Jaffe, J.J. Sensitivity of Trypanosoma equiperdum to the action of tumor-inhibitory antibiotics in vitro. Biochem. Pharmacol. 1965, 14, 1867–1881. [Google Scholar] [CrossRef]
- Momparler, R.L.; Jaffe, J.J. Effect of Azaserine on the Incorporation of 14-C-Labeled Purines and pyrimidines into acid-soluble and nucleic acid fractions of Trypanosoma equiperdum. Biochem. Pharmacol. 1965, 14, 255–262. [Google Scholar] [CrossRef]
- Poster, D.S.; Bruno, S.; Penta, J.; Neil, G.L.; McGovren, J.P. Acivicin. An antitumor antibiotic. Cancer Clin. Trials 1981, 4, 327–330. [Google Scholar] [PubMed]
- Kreuzer, J.; Bach, N.C.; Forler, D.; Sieber, S.A. Target discovery of acivicin in cancer cells elucidates its mechanism of growth inhibition. Chem. Sci. 2014, 6, 237–245. [Google Scholar] [CrossRef]
- Oliveira de Souza, J.; Dawson, A.; Hunter, W.N. An Improved Model of the Trypanosoma brucei CTP Synthetase Glutaminase Domain-Acivicin Complex. ChemMedChem 2017, 12, 577–579. [Google Scholar] [CrossRef]
- Li, Q.; Leija, C.; Rijo-Ferreira, F.; Chen, J.; Cestari, I.; Stuart, K.; Tu, B.P.; Phillips, M.A. GMP synthase is essential for viability and infectivity of Trypanosoma brucei despite a redundant purine salvage pathway. Mol. Microbiol. 2015, 97, 1006–1020. [Google Scholar] [CrossRef]
- Damasceno, F.S.; Barison, M.J.; Pral, E.M.; Paes, L.S.; Silber, A.M. Memantine, an antagonist of the NMDA glutamate receptor, affects cell proliferation, differentiation and the intracellular cycle and induces apoptosis in Trypanosoma cruzi. PLoS Negl. Trop Dis. 2014, 8, e2717. [Google Scholar] [CrossRef]
- Duszenko, M.; Figarella, K.; Macleod, E.T.; Welburn, S.C. Death of a trypanosome: A selfish altruism. Trends Parasitol. 2006, 22, 536–542. [Google Scholar] [CrossRef]
- Figarella, K.; Rawer, M.; Uzcategui, N.L.; Kubata, B.K.; Lauber, K.; Madeo, F.; Wesselborg, S.; Duszenko, M. Prostaglandin D2 induces programmed cell death in Trypanosoma brucei bloodstream form. Cell Death Differ. 2005, 12, 335–346. [Google Scholar] [CrossRef]
- Camargo, E.P. Growth and differentiation in Trypanosoma cruzi. I. Origin of metacyclic trypanosomes in liquid media. Rev. Inst. Med. Trop. Sao Paulo 1964, 6, 93–100. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds can be purchased by SIGMA. Limited quantities are available from the authors. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).