1. Introduction
Microalgae of
Arthrospira species (i.e.
Arthrospira platensis), known as
Spirulina have been employed for centuries by several African populations as food, and today are studied for various applications in nutraceutical and food industry [
1], thanks to the content of bioactive compounds such as proteins, peptides, carotenoids, chlorophylls, polyphenols, and polyunsaturated fatty acids [
2]. Phycocyanin, the main protein of
Spirulina, has several bioactivities, namely, hepatoprotective, anti-inflammatory, immunomodulating, antioxidant, and anticancer effects [
3,
4].
Anyway,
Spirulina biomass and extracts are usually not appreciated by consumers for their organoleptic characteristics and critical sensory attributes (penetrating smell and flavour, dark colour, and undesirable taste). Although sensory attributes do not have a direct relationship with the potential adverse human health effects, they could result in unpalatable products limiting the incorporation in the target food or food supplements. There are many cases reported in the literature that show the difficulty to obtain
Spirulina bakery products, palatable for the consumers. Shahbazizadeh et al. produced cookies with a supplement of
S. platensis in a 0.5–1.5 (
w/
w) concentration resulting in unpleasant colour, bitter taste and low acceptability [
5]; Niccolai et al. developed a microalgae-based bakery product with different concentrations of
Spirulina biomass (2–6–10%
w/
w); whereas the bioactivity was kept with the incorporation of at least 6–10%
w/
w, acceptable sensory scores were achieved only up to 2%
w/
w of the biomass [
6]. Moreover, the bioactivity of its heat-sensitive and low-stability molecules could be compromised during both the storage and heat-treatment manufacturing.
Another limitation in oral administration of protein-rich extracts is related both to the protein nature itself and their high molecular weight. The proteins in the gastrointestinal environment undergo enzymatic degradations, leading to the production of peptides with a molecular weight distribution of about 1000 Da, which is more easily absorbed [
7] and generally responsible for the nutritional value and bioactivity. For this reason, it is very common to subject protein extracts to an in vitro gastrointestinal digestion to obtain a standardized product, less targeted by enzymatic degradations once orally administered [
7]. A peptide blend, ideal candidate for the oral intake, should, therefore, have a low molecular weight, high potency, enzymatic/chemical stability as well as high intestinal permeability [
8]. Hydrolyzed
Spirulina extract (HSE) is the digestion product of a
Spirulina water extract; it is rich in low molecular weight peptides, exerting a significant vasorelaxation activity either on normotensive mice or spontaneously hypertensive rats [
9]. HSE contains hundreds of peptides. In detail, 97 peptides, initially encrypted in Phycocyanin and Allophycocyanin (α and β chain), were identified in a mass range from 487.2642 to 2084.0095. As previously reported, to gain insight into the bioactive compounds responsible for this effect, a complex peptidome multistep approach was used to fractionate the crude digest giving five peptide fractions (A–E), of which only fraction E evoked vasorelaxation. The high-resolution mass spectrometry-based screening revealed the presence of four main peptides (spirulina peptides SP3, SP4, SP5, and SP6) in the fraction E, of which only SP6 (GIVAGDVTPI) exerted direct endothelium-dependent vasodilation of ex vivo vessels associated with enhanced serum nitrite levels [
9]. Despite the interesting bioactivity, HSE has high water solubility implicating low intestinal absorption due to insufficient epithelial membrane permeability [
8,
10]. In this case, a formulative strategy to improve permeability, stability, and organoleptic characteristics can be undertaken.
In the present research, HSE was, for the first time, microencapsulated following a particle engineering approach involving the development of a delivery system based on chitosan/mannitol (CM) matrix able to both enhance the permeation of hydrophilic peptides and convert HSE in a more handling and stable product. CM-HSE was produced by spray-drying with adequate additives providing a physical barrier that protects the core extract from the outside environment reducing its degradation and reactivity [
11,
12,
13]. Additives, such as sugars, polysaccharides, gums, proteins, and lipids may guarantee an easy handling powder with high process yield, low moisture content, and good flowability [
14,
15]. Mannitol, a stable powdered sugar, was chosen as a loading carrier, as it resists moisture resorption at relatively high humidity and, therefore, it is ideal to load moisture-sensitive extracts [
16]. Chitosan was chosen as coating biopolymer, able to promote the formation of microparticles and the efficiency of the spray-drying process [
17]; also able to positively affect the membrane permeation as it can open epithelial tight junctions due to its interaction with the negative charges [
18], leading to an improvement of the para-cellular transport and increase in the bioavailability of hydrophilic compounds [
19]. Due to these characteristics, it has been widely employed for the encapsulation of active ingredients such as peptides and extracts [
20,
21]. The choice to use a combination of chitosan and mannitol was corroborated by the study of Artursson et al. which demonstrated the mannitol absorption in a Caco-2 monolayers system [
22]. Hydrophilic and inert mannitol itself does not distribute in the intestinal mucosa, but in combination with chitosan, an enhancement in permeability was observed [
22]. Selecting the matrix and appropriate process conditions, it was possible to obtain engineered particles CM-HSE by spray-drying with interesting characteristics, mainly solid-state, dimensional distribution, and morphology. The final CM-HSE delivery system showed improved technological characteristics, stability, and enhancement of permeation suggesting potential improvement of absorption after oral administration with respect to the unprocessed HSE.
3. Materials and Methods
3.1. Chemicals
Spirulina platensis powder was obtained from FarmaLabor Srl (Canosa di Puglia, Barletta-Trani, Italy).
Ultra-pure water (H
2O) was obtained by a Direct-8 Milli-Q system (Millipore, Milan, Italy); LC-MS grade acetonitrile (ACN) and water (H
2O), formic acid (HCOOH), trifluoroacetic acid (TFA), hydrochloric acid, ammonium formate (HCOONH
4) pepsin, chymotrypsin (bovine pancreas), pancreatin (porcine pancreas), bile salts, Chitosan (C) low molecular weight (LMW ≈150 kDa), degree of deacetylation ≥75% were all purchased from Sigma-Aldrich (St. Louis, MO, USA). Mannitol (M) was purchased from Carlo Erba Reagenti (Rodano, Milano, Italy). Permeapad
® barriers were kindly gifted by InnoME GmbH (Espelkamp, Germany). Peptide SP6 (GIVAGDVTPI) was synthesized as reported by Carrizzo et al., 2019 [
9], and kindly provided by the authors.
3.2. HSE Preparation
The pre-treatment of 15 g of
Spirulina powder and aqueous extraction of the protein fraction was carried out following the method reported in our previous work [
9]; a recovery over than 50% (7.712 g of lyophilized protein fraction) was obtained. Also the subsequent in vitro gastrointestinal digestion was carried out with the same procedure and apparatus described in reference [
8]; the crude GID (gastro-intestinal digestion) hydrolysate was purified by solid-phase extraction (SPE), partially evaporated and then lyophilized giving the final product HSE.
3.3. Spray-Drying Process
3.3.1. Liquid Feed Preparation
The solution of HSE and mannitol was prepared by dissolving 5 g of mannitol in 50 mL of deionized H2O adding 0.5 g of HSE and diluting to a volume of 100 mL in a graduated cylinder under magnetic stirring.
The solution of chitosan was prepared by dissolving 0.5 g of chitosan in 50 mL of acidified water (HCl 0.1 M, pH = 1.27) and diluting to a volume of 100 mL in a graduated cylinder, under magnetic stirring
Both solutions were pooled with a final volume of 200 mL. Each liquid feed was prepared in triplicate.
3.3.2. Spray-Drying Conditions
The feed solution was spray-dried in a mini spray dryer (Büchi B-191-Büchi Laboratoriums-Tecnik, Flawil, Switzerland). The experimental conditions were: inlet temperature of 120 °C, the outlet temperature of 70 °C, air pressure of 6.5 × 105 Pa, drying airflow of 500/600 L/h, aspirator at 90%, spray flow feed rate of 5 mL/min and the nozzle diameter of 0.5 mm.
Each spray-dried powder was produced in triplicate. All the obtained powders were collected and stored under vacuum for 48 h at room temperature before characterization.
3.3.3. Yield of Process
The process yield was gravimetrically determined (balance Crystal 100 CAL–Gibertini (max 110 g; d= 0.1 mg; +15 °C/30 °C)) and expressed as the weight percentage of the final product compared to the total amount of the materials sprayed:
3.3.4. Drug Content
The content of SP6, used as the marker for the characterization of HSE and the engineered particles was estimated by both UV and UHPLC-MS/MS analysis and resulted in 1% (±0.2).
Briefly, for UHPLC-MS/MS-method analysis were carried out with a Shimadzu Nexera (Shimadzu, Milan, Italy) UHPLC consisting of two LC 30 AD pumps, a SIL 30AC autosampler, a CTO 20AC column oven, a CBM 20A controller, and the system was coupled online to a triple quadrupole LCMS 8050 (Shimadzu, Kyoto, Japan) by an ESI source. The conditions of the separations were: BIOshell TM A160 Peptide C18 column with geometry (L × I.D) 100 × 2.1 mm, 2.7 μm (Supelco, Bellefonte, PA, USA) employing as mobile phases: A) 0.1% HCOOH in H2O v/v, B) ACN plus 0.1% HCOOH, with the following gradient starting 0–7 min, 5–40% B; 7–7.01 min, 40–99% B, flow rate set to 0.5 mL/min and the column oven set to 35 °C, with 5 µL of sample injected. Interface temperature, Desolvation line temperature and heat block temperature were set, respectively, to 250 °C, 200 °C and 400 °C. Nebulizing gas, drying (N2) and heating gas (air) was set, respectively, to 3, 10 and 10 L/min.
Specificity
To analyze SP6 in raw HSE and CM-HSE, ESI was operated in positive ionization. MS/MS analysis were conducted in multiple reaction monitoring (MRM) (
Supplementary Materials, Figure S2), employing as transitions: 941.40 > 731.40 (quantifier ion), Q1 pre-bias −28.0 V, collision energy: −30.0 V, Q3 pre-bias −34.0 V; 941.40 > 612.30 (qualifier ion), Q1 pre-bias −36.0 V, collision energy: −30.0 V, Q3 pre-bias −22.0 V; 941.40 > 343.20, Q1 pre-bias −38.0 V, collision energy: −47.0 V, Q3 pre-bias −23.0 V; Dwell time 50 msec.
For UV-method, a UV double beam spectrophotometer Specord 200 Plus (Analytik Jena, Germany) was equipped with an automatic sampling system using quartz cells (Hellma) with 1 mm pathway. Calibration curves of SP6 (y = 0.001x − 2,1683 r
2 = 0.9992) and HSE (y = 4090.2x – 2.843 r
2 = 0.9995) (
Supplementary Materials Figures S3 and S4) were previously worked out at 260 nm (λ
max). The SP6 stock solution was prepared by dissolving 1 mg of the peptide with 1 mL of distilled water. Then, several concentrations in the range of 0.5–500 ng/mL were prepared. The HSE stock solution was prepared by dissolving 1 mg of HSE with 1 mL of distilled water; then several concentrations in the range 5–250 μg/mL. Samples of the particles were prepared by dissolving 5 mg of the spray-dried powders in 5 mL of deionized H
2O and the absorbance measured spectrophotometrically at λ
max 260 nm.
3.3.5. Encapsulation Efficiency
The extract content, encapsulation efficiency, and loading capacity were calculated by the following equations.
The theoretical extract content (TEC) was calculated as the percentage of the amount of the extract initially loaded in the formulation compared to the amount of all feed components:
The actual extract content (AEC, amount of HSE in microparticles) was obtained by data interpolation of the powder formulation with calibration curves previously described using SP6 as the marker of HSE (UV method confirmed by UHPLC-MS).
The loading capacity was calculated considering the process yield, considering the loss of carrier mass during the process. So the weight (expressed in g) of the carrier in microparticles was calculated as:
3.4. Powder Characterization—Solid-State Evaluation
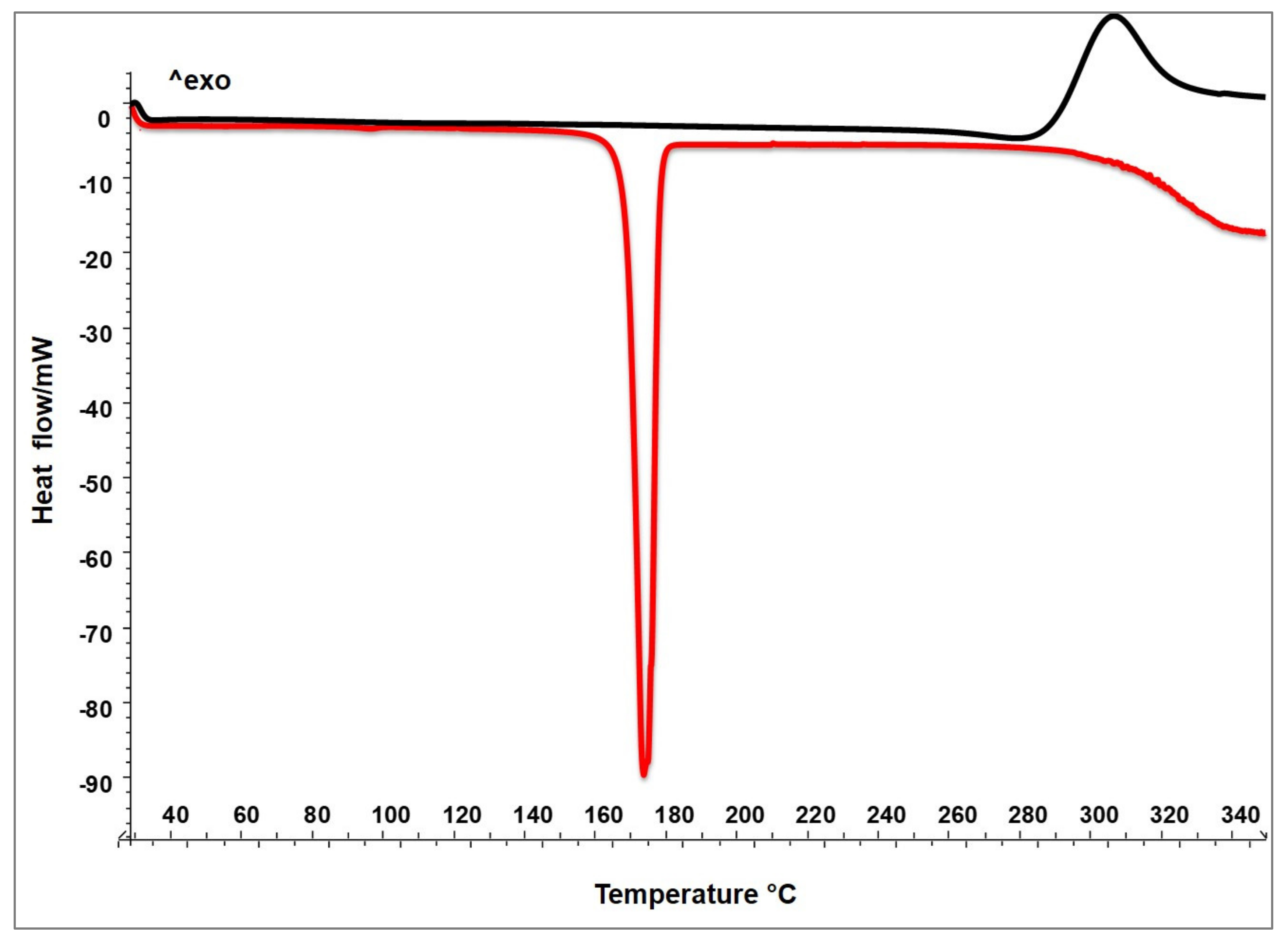
3.4.1. Thermal Analysis—Differential Scanning Calorimetry (DSC)
CM-HSE, blank and raw materials (C, M, and HSE) were analyzed by Differential Scanning Calorimetry on an indium calibrated Mettler Toledo DSC 822e (Mettler Toledo, OH, USA). Thermograms were recorded by placing accurately weighed quantities (8–10 mg weighed with a microbalance MTS, Mettler Toledo, OH, USA) of each sample in a 40 µL aluminium pan, which was sealed and pierced. The blank curve was automatically determined by the instrument. The samples underwent one dynamic thermal cycle; they were heated from 25 °C to 350 °C at a heating rate of 10 °C/min.
3.4.2. Dimensional Distribution (LLS)
Sizes and dimensional distributions of CM-HSE and raw materials (C, M, HSE) were carried out with a Laser Light Scattering (LLS) granulometer (Beckman Counter LS 230, Particle Volume Module Plus, Brea, CA, USA).
CM-HSE, blank, and mannitol (15, 20, and 50 mg, respectively) were suspended in 2 mL of ethanol. Approximately 300, 150, and 50 μL, respectively, were added in a small volume cell to obtain an obscuration between 8 and 12%.
A total of 20 mg of Chitosan was suspended in 2 mL of distilled H2O and only a few drops were poured to the same cell obtaining a correct obscuration value.
Particle size distributions were calculated using the Fraunhofer model. The results are expressed as d
10, d
50, and d
90, indicating the volume diameters at the 10th, 50th and 90th percentiles, respectively, of the particle size distribution. The analyses were made in triplicate. The span is defined as:
3.4.3. Morphology (SEM-FM)
The morphologies of raw materials and produced particles were analyzed via scanning electron microscopy (SEM) using a Carl Zeiss EVO MA 10 microscope operating at 17 kV; the powders were coated with Au/Pd and eventually observed at different extensions.
The fluorescent microscopy assays (FM) were performed observing the samples with a Zeiss Axiophot fluorescence microscope, with 40, 63 and 100 × 1.4 NA plan Apochromat oil immersion objectives (Carl Zeiss Vision, München-Hallbergmoos, Germany) using standard DAPI (40, 6-diamidino-2-phenylindole) optics that adsorb violet radiation (max 372 nm) and emit a blue fluorescence (max 456 nm).
3.4.4. Flowability
The bulk and tap densities of CM-HSE was measured as reported in the U.S. Pharmacopeia by a slightly modified method as reported by Sansone et al. [
29]. Briefly, CM-HSE was loaded into a bottom-sealed 1 mL plastic syringe (Terumo Europe, Leuven, Belgium) capped with laboratory film (Parafilm1“M”, Pechiney Plastic Packaging, Chicago, IL, USA) and tapped until no change in the volume of the powder was observed. The bulk and the tap densities were calculated, respectively, from the ratio between the net weight of the plastic syringe content and the volume in the syringe before and after tapping. Experiments were performed in triplicate.
Compressibility index (CI) and Hausner ratio (HR) were calculated using measured values for the bulk and tap densities, as follows:
The powder flow character was evaluated according to the following classification [
29](
Table 6).
3.4.5. Mixture Homogeneity
Mixture homogeneity test was performed on CM-HSE, Lactose/Mg Stearate 97:3 (w/w), CM-HSE plus Lactose/Mg Stearate 97:3 (w/w) and HSE plus Lactose/Mg Stearate 97:3 (w/w) (D) after 120 s of mixing.
4 g of Lactose/Mg Stearate (97:3 w/w) were gently ground for one minute in a mortar with and 2 g of CM-HSE or HSE, then were loaded in a 15 mL falcon, and, finally, mixed with a super mixer for two minutes, to evaluate the resultant mixture homogeneity by macroscopic evaluation.
Uniformity of content was calculated taking three different aliquots from the upper, middle, and lower part of the falcon. Each sample was solubilized in the water a final concentration of 0.500 mg/mL and the HSE content determined spectrophotometrically following the wavelength of 260 nm.
3.5. Stability Studies
Glass vials containing 1 g of CM-HSE was stored for six months at 40 °C ± 2 °C with 75% RH ± 5% (Accelerated stability test, ICH guidelines) [
32], in tapped and untapped vials. A climatic chamber was used (Climatic and Thermostatic Chamber, Mod. CCP37, AMT Srl, Milan, Italy).
SEM, LLS, and flowability analyses before storage (t0), and for six months (t180) were performed.
Hygroscopicity was gravimetrically calculated according to the procedure described by [
29,
40] and the following formula:
where m
0 and m
180 express, respectively, the moisture of the samples before (t
0) and after (t
180 days) the storage period.
3.6. In Vitro Permeability Tests
In vitro permeability assay of HSE and CM-HSE was performed using a conventional Franz type vertical diffusion cell (Hanson Research Corporation, Chatsworth, CA, USA), employing Permeapad
® (innoME GmbH, Espelkamp, Germany, UE) barrier constituted of Cellulose membrane + Lecithin (S100), able to simulate the passive mass transport through the intestinal membrane [
41].
At first, the receptor compartment was filled with 7 mL of PBS (pH = 7.31) and incubated at a temperature of 37 °C ± 0.5 in a water bath. In this compartment, a magnetic stirrer was placed to ensure a constant stirring (250 rpm). The acceptor compartment was, then, covered with Permeapad® barrier, leaving the first hour under stirring to equilibrate the membrane.
Subsequently, the powder (CM-HSE or HSE) was placed on the barrier in the donor compartment until the affected area of mass transfer was totally filled (1.767 cm2). The quantity of CM-HSE loaded on the membrane was established in order to fill the surface available for the absorption. As a result, the CM-HSE powder used for the test was of 45 mg, consequently, HSE was loaded with an equivalent amount of 3.8 mg.
The permeation started when 100 μL of PBS was added to the powder and was followed for 6 hours. At time points 5, 10, 15, 20, 30, 40, 50, 60, 75, 90, 105, 120, 135, 150, 165, 180, 195, 210, 225, 240, 255, 270, 285, 300, 315, 330, 345, 360 min, an aliquot of 500 μL was collected from the outlet of acceptor compartment and, simultaneously, replaced from the inlet with fresh PBS at 37 °C.
Samples collected during the experiment were analyzed spectrophotometrically at a wavelength of 260 nm. Each experiment was performed in triplicate.
An aliquot collected at the end of the equilibration period was used as blank.
To confirm results by UV analysis, two samples for each hour were also analyzed by UHPLC-MS/MS
The amount of the extract permeated per area (Q) for each time interval was calculated, according to [
42], using the following equation:
where V
R is the receiver volume, C
N is HSE concentration in the receiver at the time n, V
p is the volume of the removed sample, C
i is HSE concentration in the receiver at the time n-1.
Permeation data were reported as the quantity of permeated HSE per permeation area related to time. All the permeation tests were made in triplicate; only the mean values are reported (standard evaluation <1%).
3.7. Statistical Analysis
All results are shown as mean ± standard deviation of three experiments performed in triplicate. Statistical comparison between groups was made using ANOVA followed by the Bonferroni parametric test. Differences were considered significant if p < 0.05. The SP6 and HSE content data were subjected to one-way analysis of variance (ANOVA) followed by and Tukey HSD test (p < 0.05), using GraphPad Prism version 7.00 for Windows.
4. Conclusions
A chitosan/mannitol (CM) matrix was studied for its ability to encapsulate via spray drying and to deliver HSE, sensitive peptide-rich Spirulina extract. Using a 1:10 C/M ratio, highly efficient encapsulation (100%) of HSE extract was obtained with a good process yield (70%) and production of stable powder made up of well-formed and micronized particles. Bio-active extract HSE was loaded within the microparticles core and enveloped by CM system. Even under harsh storage conditions, neither the dimension, content, morphology, nor derived properties of the particles appeared modified. The spray dried system is potentially able to mask the unpleasant smell and unfavourable taste of HSE and shows a decrease in the initial dark green colour of HSE. Therefore, the engineered particles appear suitable for HSE delivery with good storage, organoleptic, and handling performance. Moreover, the system shows an enhanced permeation of the peptide-rich extract through an in vitro simulating intestinal barrier with respect to the unprocessed HSE. The final product may be proposed as an ingredient for functional food and food supplements for maintaining cardiovascular homeostasis, improving endothelial function, and controlling blood pressure with good technological and manufacturing characteristics.
The developed method may be generally proposed and applied to carry and deliver extracts with solubility and permeability characteristics like HSE, furnishing a stable product with extended shelf-life and optimized organoleptic, technological, and permeation properties.