1. Introduction
Protic ionic liquids (PILs) are an important subset of ionic liquids (ILs). PILs possess strong dissolvabilities, high thermal stabilities, designable structures and broad electrochemical windows [
1,
2]. They play important roles in fuel cells, electrochemistry, liquid-liquid extraction, gas capture, biological media and so on due to their acidity [
3,
4,
5,
6,
7]. For the existence of active hydrogen in amino acid cations, proton dissociation occurs in different solvents to varying degrees [
8]. Therefore, PILs have been considered as acidic catalysts for the replacement of hazardous acids in many catalytic reactions, including the esterification reaction, biomass conversion, transformation of CO
2 and Diels-Alder reaction. [
9,
10,
11,
12].
Bio-based ILs have been paid more attention in recent years due to their preferable green characters [
13]. Some natural materials, including carboxylate salts, amino acids and sugars or sugar derivatives, have been employed as IL precursors in a green way [
14]. Among these natural materials, amino acids and their derivatives are the most abundant natural sources containing quaternary nitrogens. Amino acid ionic liquids (AAILs) are fascinating for chemists in view of their close associations with chirality and biomolecules [
15,
16]. Some research has found that AAILs may be useful as potential solvents, catalysts, absorbents and selectors, etc. [
17,
18]. At the same time, AAILs can be used as acidic catalysts in the esterification of renewable valeric acid, styrene carbonate synthesis under CO
2, the alkylation of indoles and so on [
19,
20,
21]. Moreover, [AAE]X (AAE means the amino acid ester cations, and X means the corresponding anions) AAILs have higher thermostabilities and lower melting points, as well as lower viscosities than those of their [AA]X (cations are amino acid) analogs and, hence, broader prospects in acid-involving processes [
22].
In general, water and ethanol are considered as green media for acidic catalytic reactions, which is one of the “twelve principles” of green chemistry [
23,
24,
25,
26]. Acidic properties in solvents are very important to industrially relevant reactions [
27,
28]. The common PILs, such as imidazolium salts and quaternary ammonium salts, have been studied in many acidic catalytic reactions in solvents [
29,
30]. However, the precursors of [AAE]X are more abundant and bio-based compared to common PILs, which also possesses Brønsted acidity. Therefore, the proton dissociation of [AAE]X ILs in green solvents are interesting and important for their applications. However, the acidic characteristics of [AAE]X AAILs in solvents are still lacking. The acid dissociation constant (p
Ka) is one of the most significant physiochemical parameters. An accurate p
Ka value is important to select reaction conditions in catalytic chemistry [
27,
31,
32,
33]. Determining the acidity of ILs in water and ethanol has become an intriguing topic, since water and ethanol are promising media for green chemistry [
30,
34,
35,
36,
37]. Herein, four different methods were employed to study the Brønsted acidity of [AAE]X PILs with different amino acid ester cations and anions in water and ethanol carried out.
3. Discussion
To systematically explore the acidity of [AAE]X, eight [AAE]
+ and four anions (including nitrate (NO
3−), chloride (Cl
−), perchlorate (ClO
4−) and trifluoromethanesulfonate (NTf
2−)) were studied from the viewpoint of the structure–property relationship. The cations and anions of [AAE]X used in this work are shown in
Figure 3. All [AAE]X PILs were synthesized and characterized by the referenced method [
15]. Being convenient for studying the structure–property relationship, the glycine methyl ester cation ([GlyC
1]
+) was chosen as the fundamental [AAE]+ framework. Other cations, including the glycine ethyl ester cation ([GlyC
2]
+), serine methyl ester cation ([SerC
1]
+), serine ethyl ester cation ([SerC
2]
+), phenylalanine methyl ester cation ([PheC
1]
+), phenylalanine ethyl ester cation ([PheC
2]
+), valine methyl ester cation ([ValC
1]
+) and D-phenylalanine methyl ester cation ([D-PheC
1]
+), could be viewed as the derivatives of [GlyC
1]
+.
According to the pKa values of [AAE]X and the contrastive compounds in water by the OIM, the acidic strength of [GlyC1]NO3 is the weakest in the studied [AAC1]X (amino acid methyl ILs) and depends on the biggest pKa values. The acidic strengths of [ValC1]NO3, [SerC1]NO3 and [PheC1]NO3 are stronger than that of [GlyC1]NO3, maybe due to the steric effect of the side chains in [ValC1]NO3 and [PheC1]NO3 and the hydrogen bonding in [SerC1]NO3. The acidic strength of [AAE]X has little difference in the same magnitude range, since the side chain of [AAE]X may exist in hydrogen bonding or steric hindrance with the –NH3 group. The pKa values of [GlyC1]NO3, [GlyC2]NO3, [SerC1]NO3, [SerC2]NO3, [PheC1]NO3 and [PheC2]NO3 are 7.67, 7.73, 7.23, 7.10, 7.20 and 7.26, respectively. The methyl ester and ethyl ester group hardly affects the acidity of [AAE]X due to the low maximum difference (0.23) between methyl ester and ethyl ester. The pKa values of [AAE]X range from 7.10 to 7.73, which are smaller than those of glycine (Gly, 9.78), phenylalanine (Phe, 9.31), ethylammonium nitrate (EAN, 10.43), diethylammonium nitrate ([Et2N]NO3, 10.68) and triethylammonium nitrate ([Et3N]NO3, 10.55). The electron-withdrawing inductive effect of the ester group may result in the stronger acidity of [AAE]X than those of their precursors (amino acids), EAN, [Et2N]NO3 and [Et3N]NO3, whose acidity also depend on the protonated amino group.
The pKa values of [AAE]X range from 7.10 to 7.73 in water, which are almost equal to that of imidazolium ([MIM]+, 7.13) salts. The acidic strength of [AAE]X is between pyridinium ([Pyri]+, 5.17) salts and EAN (10.43), [Et2N]NO3 (10.68) and [Et3N]NO3 (10.55).
The p
Ka values of [PheC
1]Cl, [PheC
1]NO
3, [PheC
1]NTf
2 and [PheC
1]ClO
4 are 7.25, 7.20, 7.24 and 7.20 by the OIM, respectively. The p
Ka values of [PheC
1]X with different anions are almost the same, to some extent. It seems like anions have insignificant effects on the acidity of [AAE]X PILs. In other words, the cation and anion of [AAE]X in water may be dissociated, because water is a typical high-polar solvent (ε = 80.100) [
43]. Different from anions, the p
Ka values of [L-PheC
1]NO
3, [D-PheC
1]NO
3, [L-PheC
1]Cl and [D-PheC
1]Cl are similar.
For the same [AAE]X, we found that the △pKa by the PTM obtained by two different determination methods are near to zero. For example, the pKa value of [GlyC1]NO3 determined by the OIM are the same (7.67) to that from the PTM. The data suggest that both the PTM and OIM are reliable to obtain the pKa values of [AAE]X in water.
To ulteriorly study the acidity of [AAE]X, we measured the pKa values in ethanol by the OIM with 2,4-dinitrophenol as the indicator. The pKa values of [AAE]X in ethanol are between 8.54 and 9.05, which are obviously bigger than those in water (7.10 to 7.73). It may be generated by the weaker basicity of ethanol, which means that the weaker intermolecular interactions between the active hydrogens and solvent molecules lead to bigger pKa values. The pKa values of [GlyC1]NO3, [ValC1]NO3, [SerC1]NO3 and [PheC1]NO3 are 9.05, 8.86, 8.88 and 8.54, separately. There is also a tiny difference of the pKa values that may be due to the side chain of [AAE]X. The pKa values of [AAE]X are smaller than [EtNH3]+ (12.0), [Et2NH2]+ (10.7) and [Et3NH]+ (10.22) and bigger than [Pyri]+ (4.30). Therefore, the acidic strength of [AAE]X in ethanol is between [Pyri]+ and [EtNH3]+, [Et2NH2]+ and [Et3NH]+. The acidic strength of [AAE]X is slightly weaker than [MIM]+ (7.50) in ethanol, depending on the pKa values.
To confirm the acidic strength of [AAE]X determined by the PTM and OIM, the Brønsted acidity associated with the Hammett acidity function (H0) of [AAE]X was investigated in water. The maximum absorption peak of sodium 2,4-dinitrophenolate decreased as the acidity of the solution increased. The H0 values of [ValC1]NO3, [PheC1]NO3 and phenylalanine (Phe) are 4.37, 4.33 and 6.44 in water, separately. This means the acidic strength of [AAE]X is almost same and is stronger than that of their precursors (amino acids). The acidity of EAN, [Et2N]NO3 and [Et3N]NO3 are so weak that the decrease of the indicator’s absorbance was not detected. The H0 of [AAE]X is almost the same as [MIM]Cl (4.35) in water. Therefore, the acidic strength of [AAE]X may be almost the same as [MIM]+ salt and stronger than the amino acid, EAN, [Et2N]NO3 and [Et3N]NO3. The results are matched well with the results of the pKa values in water determined by the PTM and OIM.
The relationship between the acidity and concentration is important for many applications, such as catalysis [
9,
37,
51]. The
H0 of [ValC
1]NO
3 and [PheC
1]NO
3 in aqueous solutions at various concentrations were measured. In the UV/Vis spectra, a noticeable decrease of the maximum absorption peak was found, accompanied with adding [ValC
1]NO
3 and [PheC
1]NO
3 (
Figure 4). The
H0 reduced when the concentration increased. The relationship between the concentration of [ValC
1]NO
3 and [PheC
1]NO
3 and the
H0 can be obtained using the fitting equations. Their fitting equations are:
Their nonlinear fittings are shown in
Figure 5. The
H0 of [AAE]X gradually lowered with the concentration of [AAE]X rising. Based on these, the desired acidic strength can be obtained by choosing the appropriate concentrations.
4. Materials and Methods
General methods: All [AAE]Cl were purchased from Energy Chemical (Shanghai, China). Ethylamine (EAN) and ethanol (EtOH) were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). All chemicals were obtained commercially as analytical-grade materials and used as received. Solvents were dried by standard procedures. [AAE]X PILs were synthesized according to a literature procedure by the ion exchange reaction of [AAE]Cl precursors with corresponding salts. The synthesized [AAE]X PILs needed to be dried firstly and kept in vacuum before use.
The standard deviation (SD) was calculated by the equation:
pKa determination by the PTM: A stock solution (0.010 mol·L−1) of [AAE]X PILs was prepared in ultrapure water. Then, the solution was titrated with aqueous KOH solution (0.100 mol·L−1). The electric potential (E) (±1 mV) of the solution was obtained using an Ag–AgCl/glass combination electrode on an OHAUS Starter 2100 pH meter at 25.0 (±0.1) °C. Three standard buffer solutions with the pH values of 4.00, 6.86 and 9.18, respectively, were used to adjust the instrument before titration.
pKa determination by OIM: The method determined the pKa of an “unknown” acid relative to that of an “indicator” acid (whose pKa was known) by monitoring the changes of UV/vis absorption of the indicator during titrations under standard conditions. (The indicators (In) here should show different UV/Vis absorbance between the HIn+ and In species. Besides, in order to produce moderate changes in the titration, the pKa of HIn+ should be close to the measured substance in each solvent.) There were two steps measuring the pKa values by the OIM. Firstly, the linear relation between the concentration of the indicator and absorbance could be achieved by adding the indicator to the alkali solution until the indicator was slightly excessive compared to the alkali. Secondly, an “unknown” acid was quantitatively added to the above solution to achieve the concentrations of HIn, In−, HA+ and A. The UV/vis absorption (A) (±0.0001) of the indicator during the titrations was obtained using a BFRL UV-1601 UV/VIS spectrophotometer 25.0 (±0.1) °C.
Hammett acidity function: The Hammett acidity function of the ILs was investigated on a BFRL UV-1601 UV/VIS spectrophotometer. Samples were measured in sealed 1-cm quartz cuvettes (Helma). The dyes of sodium 2,4-dinitrophenolate were used as the indicator and molecular probe for the determination of the H0 with 5.0 × 10−5 mol·L−1. Absorbance values of the indicator after adding acid in an aqueous solution were recorded between 330 and 500 nm at 25.0 (±0.1) °C. The concentration of [AAE]X PILs was 0.050 mol·L−1.
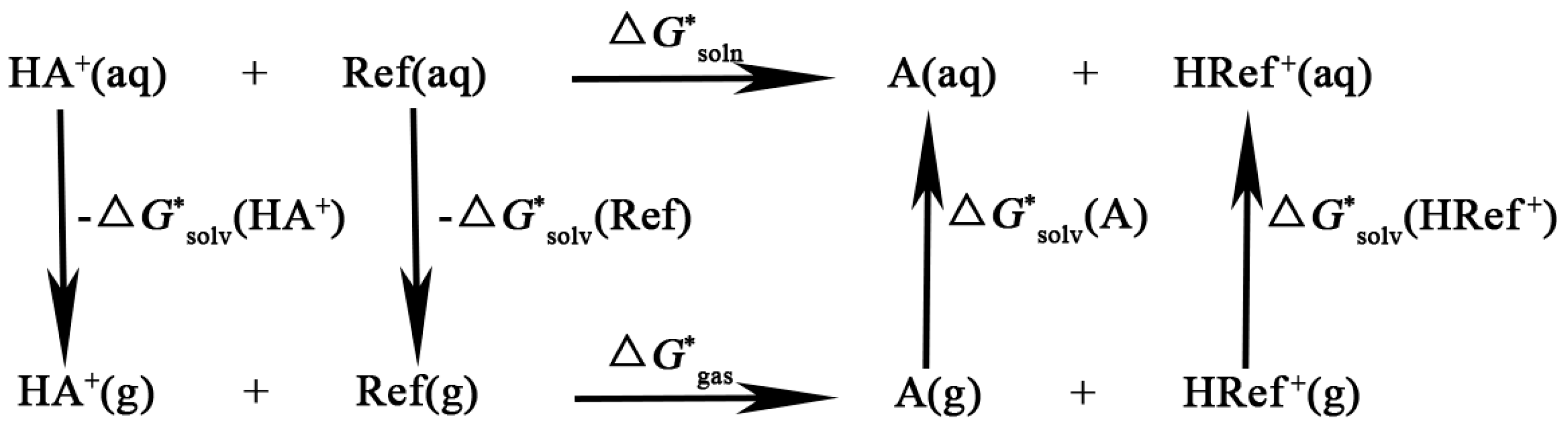
Computational methods: The Brønsted acidity of [AAE]X coming from the –NH
3 group was determined by the above experimental data. In order to obtain a better understanding of the Brønsted acidities of [AAE]X PILs, the p
Ka values of [AAE]X were calculated by the density functional theory (DFT) using the Gaussian 09 suite program [
52]. It was verified that the anions would have hardly any effect on the acidity of [AAE]X in water or ethanol by the experimental data. Therefore, the calculated acidity of [AAE]
+ could be considered as a simple and approximate method for determining the acidity of [AAE]X. The p
Ka values by theoretical calculation could be carried out by using the proton-transfer reaction (
Scheme 3) [
53,
54]:
where the solution-free energy was calculated by:
Then, it led to the following equilibrium in Equation (13):
The calculation of the p
Ka was obtained from Equation (14):
The final expression for the p
Ka can be written as
where K was a correction value dependent on the experimental values. The structures of [AAE]
+, AAE, H
2O, H
3O
+, Et
2OH and Et
2OH
2+ were optimized in the gas phase at the B3LYP/6-311++G(d,p) level [
55,
56,
57]. On the basis of the optimized structures, the solvation-free energies of [AAE]
+ and AAE in water or ethanol were calculated with the polarizable continuum model (PCM) at the B3LYP/6-311++G(d,p) level [
53,
58]. The liquidus Gibbs-free energies of AAEH
+ and AAE were obtained from the sum of the total electronic energies in water or ethanol and the thermal corrections to the gaseous Gibbs-free energies (G
corr).