Melatonin-Loaded Nanocarriers: New Horizons for Therapeutic Applications
Abstract
:1. Introduction
1.1. Tuning the Connection between Nanocarriers and Melatonin
1.2. Melatonin: Chemical Modulation and Solubility
2. Melatonin Incorporated into Lipid-Based NCs/Nanosystems
2.1. Liposomes NPs as Carriers of Melatonin
2.2. Solid Lipid NCs/Nanosystems for Melatonin Delivery
2.3. Hybrid NCs/Nanosystems Composed of Lipids and Polymers as Carriers of Melatonin
3. Non-Ionic Surfactant-Based Vesicles (Niosomes) for Melatonin Delivery
4. Melatonin-Loaded Silica-Based NPs
5. Graphene and Melatonin Delivery
6. Nanofibers and Nanocapsules as Biomaterial for Melatonin Controlled Release
7. Chitosan-Based NPs for Melatonin Delivery
8. Synthetic Polymeric NPs as Carriers of Melatonin
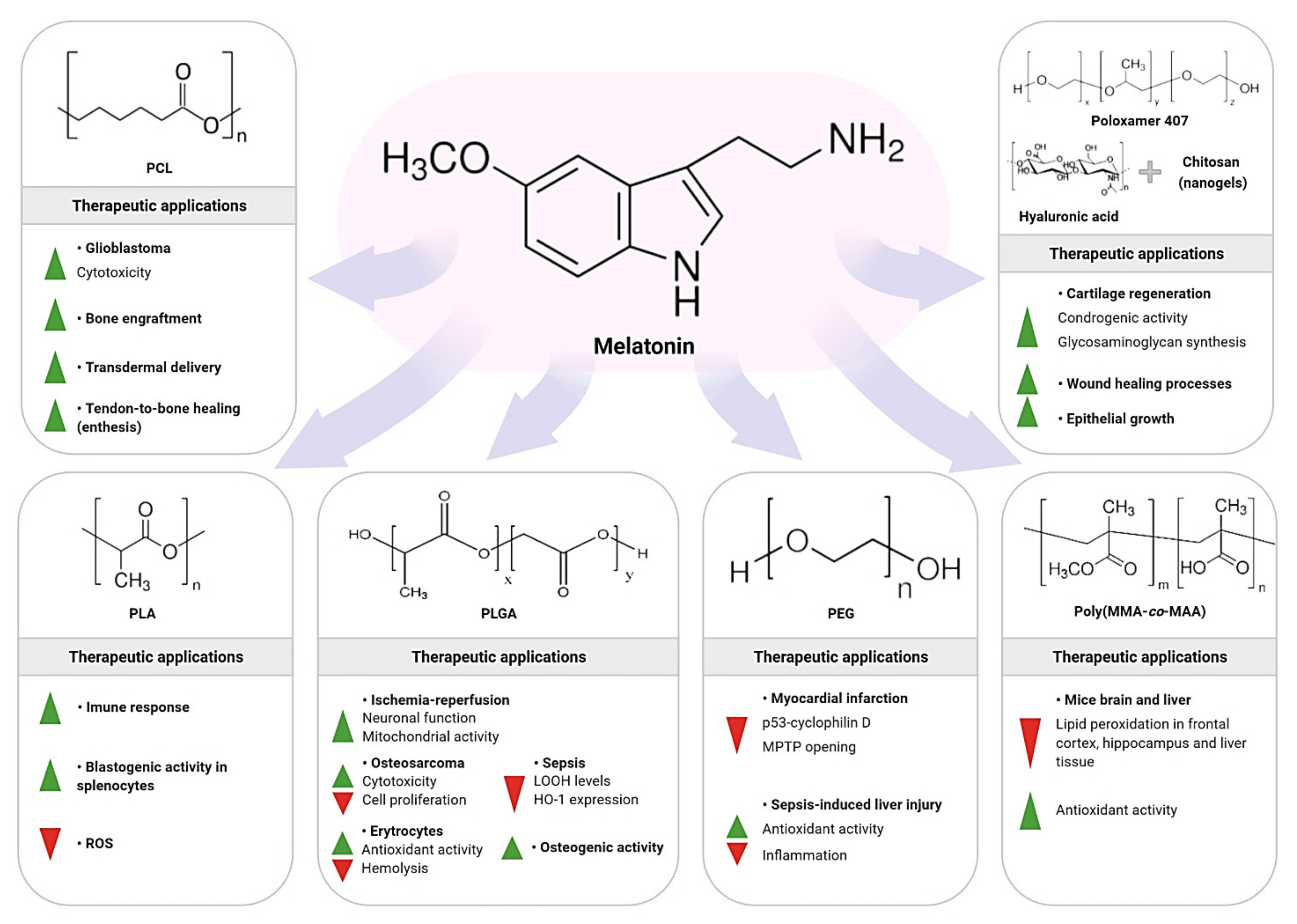
8.1. Polycaprolactone/Melatonin
8.2. Poly-lactic Acid/Melatonin
8.3. Poly (lactic-co-glycolic Acid)/Melatonin
8.4. Polyethylene Glycol/Melatonin
8.5. Poly (methacrylic acid-co-methyl methacrylate)/Melatonin
8.6. Polymeric Nanogel (Hydrogel) for Melatonin Delivery
9. Metallic NPs and Melatonin Delivery
10. Melatonin-Associated Metallic and Non-Metallic Nanocomposites (NCP)
11. Concluding Remarks and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Feynman, R.P. There’s Plenty of Room at the Bottom. Eng. Sci. 1960, 23, 22–36. [Google Scholar]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic Iron Oxide Nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical Characterizations, and Biological Applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef] [PubMed]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef] [Green Version]
- Tiwari, J.N.; Tiwari, R.N.; Kim, K.S. Zero-dimensional, one-dimensional, two-dimensional and three-dimensional nanostructured materials for advanced electrochemical energy devices. Prog. Mater. Sci. 2012, 57, 724–803. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Shin, W.-K.; Cho, J.; Kannan, A.G.; Lee, Y.-S.; Kim, D.-W. Cross-linked Composite Gel Polymer Electrolyte using Mesoporous Methacrylate-Functionalized SiO2 Nanoparticles for Lithium-Ion Polymer Batteries. Sci. Rep. 2016, 6, 26332. [Google Scholar] [CrossRef] [PubMed]
- Barrak, H.; Saied, T.; Chevallier, P.; Laroche, G.; M’nif, A.; Hamzaoui, A.H. Synthesis, characterization, and functionalization of ZnO nanoparticles by N-(trimethoxysilylpropyl) ethylenediamine triacetic acid (TMSEDTA): Investigation of the interactions between Phloroglucinol and ZnO@TMSEDTA. Arab. J. Chem. 2019, 12, 4340–4347. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.E.; Lee, N.; Kim, T.; Kim, J.; Hyeon, T. Multifunctional Mesoporous Silica Nanocomposite Nanoparticles for Theranostic Applications. Acc. Chem. Res. 2011, 44, 893–902. [Google Scholar] [CrossRef]
- Ullah, H.; Khan, I.; Yamani, Z.H.; Qurashi, A. Sonochemical-driven ultrafast facile synthesis of SnO2 nanoparticles: Growth mechanism structural electrical and hydrogen gas sensing properties. Ultrason. Sonochem. 2017, 34, 484–490. [Google Scholar] [CrossRef]
- Ganesh, M.; Hemalatha, P.; Peng, M.M.; Jang, H.T. One pot synthesized Li, Zr doped porous silica nanoparticle for low temperature CO2 adsorption. Arab. J. Chem. 2017, 10, S1501–S1505. [Google Scholar] [CrossRef] [Green Version]
- Shaalan, M.; Saleh, M.; El-Mahdy, M.; El-Matbouli, M. Recent progress in applications of nanoparticles in fish medicine: A review. Nanomedicine 2016, 12, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, A.; Azoia, N.G.; Gomes, A.C.; Cavaco-Paulo, A. Albumin-Based Nanodevices as Drug Carriers. Curr. Pharm. Des. 2016, 22, 1371–1390. [Google Scholar] [CrossRef]
- Alexis, F.; Pridgen, E.; Molnar, L.K.; Farokhzad, O.C. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol. Pharm. 2008, 5, 505–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Longmire, M.R.; Ogawa, M.; Choyke, P.L.; Kobayashi, H. Biologically optimized nanosized molecules and particles: More than just size. Bioconjug. Chem. 2011, 22, 993–1000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiter, R.J.; Sharma, R.; Ma, Q.; Rosales-Corral, S.; Manucha, W. Circadian and non-circadian melatonin: Influences on glucose metabolism in cancer cells. J. Curr. Sci. Technol. 2020, 10, 85–98. [Google Scholar] [CrossRef]
- De Almeida Chuffa, L.G.; Lupi, L.A.; Cucielo, M.S.; Silveira, H.S.; Reiter, R.J.; Seiva, F.R.F. Melatonin Promotes Uterine and Placental Health: Potential Molecular Mechanisms. Int. J. Mol. Sci. 2019, 21, 300. [Google Scholar] [CrossRef] [Green Version]
- Manchester, L.C.; Coto-Montes, A.; Boga, J.A.; Andersen, L.P.H.; Zhou, Z.; Galano, A.; Vriend, J.; Tan, D.-X.; Reiter, R.J. Melatonin: An ancient molecule that makes oxygen metabolically tolerable. J. Pineal Res. 2015, 59, 403–419. [Google Scholar] [CrossRef]
- Salehi, B.; Sharopov, F.; Fokou, P.V.T.; Kobylinska, A.; Jonge, L.D.; Tadio, K.; Sharifi-Rad, J.; Posmyk, M.M.; Martorell, M.; Martins, N.; et al. Melatonin in Medicinal and Food Plants: Occurrence, Bioavailability, and Health Potential for Humans. Cells 2019, 8, 681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Zhao, X.; Zu, Y.; Wang, L.; Wu, W.; Deng, Y.; Zu, C.; Liu, Y. Melatonin-loaded silica coated with hydroxypropyl methylcellulose phthalate for enhanced oral bioavailability: Preparation, and in vitro-in vivo evaluation. Eur. J. Pharm. Biopharm. 2017, 112, 58–66. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.; Tan, D.X.; Jou, M.J.; Galano, A.; Xu, B. Melatonin as a mitochondria-targeted antioxidant: One of evolution’s best ideas. Cell. Mol. Life Sci. 2017, 74, 3863–3881. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.A.; Tan, D.-X.; Acuna-Castroviejo, D.; Qin, L.; Yang, S.-F.; Xu, K. Melatonin, a Full Service Anti-Cancer Agent: Inhibition of Initiation, Progression and Metastasis. Int. J. Mol. Sci. 2017, 18, 843. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Chuffa, L.G.; Seiva, F.R.F.; Cucielo, M.S.; Silveira, H.S.; Reiter, R.J.; Lupi, L.A. Mitochondrial functions and melatonin: A tour of the reproductive cancers. Cell. Mol. Life Sci. 2019, 76, 837–863. [Google Scholar] [CrossRef] [PubMed]
- Pandiperumal, S.; Trakht, I.; Srinivasan, V.; Spence, D.; Maestroni, G.; Zisapel, N.; Cardinali, D. Physiological effects of melatonin: Role of melatonin receptors and signal transduction pathways. Prog. Neurobiol. 2008, 85, 335–353. [Google Scholar] [CrossRef] [PubMed]
- Zetner, D.; Andersen, L.P.H.; Rosenberg, J. Pharmacokinetics of Alternative Administration Routes of Melatonin: A Systematic Review. Drug Res. 2016, 66, 169–173. [Google Scholar] [CrossRef] [Green Version]
- Wade, A.G.; Ford, I.; Crawford, G.; McConnachie, A.; Nir, T.; Laudon, M.; Zisapel, N. Nightly treatment of primary insomnia with prolonged release melatonin for 6 months: A randomized placebo controlled trial on age and endogenous melatonin as predictors of efficacy and safety. BMC Med. 2010, 8, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Razali, S.; Bose, A.; Chong, P.W.; Benetti, C.; Colombo, P.; Wong, T.W. Design of multi-particulate “Dome matrix” with sustained-release melatonin and delayed-release caffeine for jet lag treatment. Int. J. Pharm. 2020, 587, 119618. [Google Scholar] [CrossRef]
- Milan, A.S.; Calpena Campmany, A.C.; Naveros, B.C. Antioxidant Nanoplatforms for Dermal Delivery: Melatonin. Curr. Drug. Metab. 2017, 18, 437–453. [Google Scholar] [CrossRef]
- Martín Giménez, V.M.; Prado, N.; Diez, E.; Manucha, W.; Reiter, R.J. New proposal involving nanoformulated melatonin targeted to the mitochondria as a potential COVID-19 treatment. Nanomedicine 2020, 15, 2819–2821. [Google Scholar] [CrossRef]
- Akanmu, M.A.; Songkram, C.; Kagechika, H.; Honda, K. A novel melatonin derivative modulates sleep–wake cycle in rats. Neurosci. Lett. 2004, 364, 199–202. [Google Scholar] [CrossRef]
- Calastretti, A.; Gatti, G.; Lucini, V.; Dugnani, S.; Canti, G.; Scaglione, F.; Bevilacqua, A. Melatonin Analogue Antiproliferative and Cytotoxic Effects on Human Prostate Cancer Cells. Int. J. Mol. Sci. 2018, 19, 1505. [Google Scholar] [CrossRef] [Green Version]
- He, P.; Ouyang, X.; Zhou, S.; Yin, W.; Tang, C.; Laudon, M.; Tian, S. A novel melatonin agonist Neu-P11 facilitates memory performance and improves cognitive impairment in a rat model of Alzheimer’ disease. Horm. Behav. 2013, 64, 1–7. [Google Scholar] [CrossRef]
- Zhang, J.; Yan, X.; Tian, Y.; Li, W.; Wang, H.; Li, Q.; Li, Y.; Li, Z.; Wu, T. Synthesis of a New Water-Soluble Melatonin Derivative with Low Toxicity and a Strong Effect on Sleep Aid. ACS Omega 2020, 5, 6494–6499. [Google Scholar] [CrossRef]
- Hamed, M.Y.; Mostafa, E.M.; Tous, S.S. Antifertility effect of orally formulated melatonin tablets in mice. Int. J. Pharm. 1991, 69, 93–102. [Google Scholar] [CrossRef]
- Cavallo, A.; Hassan, M. Stability of melatonin in aqueous solution. J. Pineal Res. 1995, 18, 90–92. [Google Scholar] [CrossRef]
- Daya, S.; Walker, R.B.; Glass, B.D.; Anoopkumar-Dukie, S. The effect of variations in pH and temperature on stability of melatonin in aqueous solution. J. Pineal Res. 2001, 31, 155–158. [Google Scholar] [CrossRef]
- Zetner, D.; Rosenberg, J. Solubility and stability of melatonin in propylene glycol, glycofurol, and dimethyl sulfoxide. F1000Research 2020, 9, 85. [Google Scholar] [CrossRef] [Green Version]
- Proietti, S.; Carlomagno, G.; Dinicola, S.; Bizzarri, M. Soft gel capsules improve melatonin’s bioavailability in humans. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1193–1198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teixeira, M.C.; Carbone, C.; Souto, E.B. Beyond liposomes: Recent advances on lipid based nanostructures for poorly soluble/poorly permeable drug delivery. Prog. Lipid Res. 2017, 68, 1–11. [Google Scholar] [CrossRef]
- Carbone, C.; Leonardi, A.; Cupri, S.; Puglisi, G.; Pignatello, R. Pharmaceutical and biomedical applications of lipid-based nanocarriers. Pharm. Pat. Anal. 2014, 3, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Laouini, A.; Jaafar-Maalej, C.; Limayem-Blouza, I.; Sfar, S.; Charcosset, C.; Fessi, H. Preparation, Characterization and Applications of Liposomes: State of the Art. J. Colloid Sci. Biotechnol. 2012, 1, 147–168. [Google Scholar] [CrossRef]
- Molska, A.; Nyman, A.K.G.; Sofias, A.M.; Kristiansen, K.A.; Hak, S.; Widerøe, M. In vitro and in vivo evaluation of organic solvent-free injectable melatonin nanoformulations. Eur. J. Pharm. Biopharm. 2020, 152, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, E.; Gomes, A.C.; Preto, A.; Cavaco-Paulo, A. Design of liposomal formulations for cell targeting. Colloids Surf. B Biointerfaces 2015, 136, 514–526. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Ou, C.; Ye, S.; Song, X.; Luo, S. Construction of nanoscale liposomes loaded with melatonin via supercritical fluid technology. J. Microencapsul. 2017, 34, 687–698. [Google Scholar] [CrossRef]
- Lin, W.; Kampf, N.; Goldberg, R.; Driver, M.J.; Klein, J. Poly-phosphocholinated Liposomes Form Stable Superlubrication Vectors. Langmuir 2019, 35, 6048–6054. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Ren, B.; Zhang, D.; Xie, S.; Chang, Y.; Yang, J.; Wu, J.; Xu, L.; Zheng, J. Fundamentals and applications of zwitterionic antifouling polymers. J. Phys. D Appl. Phys. 2019, 52, 403001. [Google Scholar] [CrossRef]
- Cao, Z.; Zhang, L.; Jiang, S. Superhydrophilic zwitterionic polymers stabilize liposomes. Langmuir 2012, 28, 11625–11632. [Google Scholar] [CrossRef]
- Gonçalves, M.C.F.; Mertins, O.; Pohlmann, A.R.; Silveira, N.P.; Guterres, S.S. Chitosan Coated Liposomes as an Innovative Nanocarrier for Drugs. J. Biomed. Nanotechnol. 2012, 8, 240–250. [Google Scholar] [CrossRef]
- Dubey, V.; Mishra, D.; Jain, N.K. Melatonin loaded ethanolic liposomes: Physicochemical characterization and enhanced transdermal delivery. Eur. J. Pharm. Biopharm. 2007, 67, 398–405. [Google Scholar] [CrossRef]
- Marepally, S.; Boakye, C.H.; Shah, P.P.; Etukala, J.R.; Vemuri, A.; Singh, M. Design, synthesis of novel lipids as chemical permeation enhancers and development of nanoparticle system for transdermal drug delivery. PLoS ONE 2013, 8, e82581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sana, S.; Ghosh, S.; Das, N.; Sarkar, S.; Mandal, A.K. Vesicular melatonin efficiently downregulates sodium fluoride-induced rat hepato- and broncho-TNF-α, TGF-β expressions, and associated oxidative injury: A comparative study of liposomal and nanoencapsulated forms. Int. J. Nanomed. 2017, 12, 4059–4071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Souto, E.B.; Doktorovová, S. Solid Lipid Nanoparticle Formulations: Pharmacokinetic and Biopharmaceutical Aspects in Drug Delivery. In Methods Enzymol; Elsevier: Amsterdam, The Netherlands, 2009; pp. 105–129. [Google Scholar]
- Subramaniam, B.; Siddik, Z.H.; Nagoor, N.H. Optimization of nanostructured lipid carriers: Understanding the types, designs, and parameters in the process of formulations. J. Nanopart. Res. 2020, 22. [Google Scholar] [CrossRef]
- Carbone, C.; Cupri, S.; Leonardi, A.; Puglisi, G.; Pignatello, R. Lipid-based nanocarriers for drug delivery and targeting: A patent survey of methods of production and characterization. Pharm. Pat. Anal. 2013, 2, 665–677. [Google Scholar] [CrossRef]
- Albertini, B.; Di Sabatino, M.; Melegari, C.; Passerini, N. Formulating SLMs as oral pulsatile system for potential delivery of melatonin to pediatric population. Int. J. Pharm. 2014, 469, 67–79. [Google Scholar] [CrossRef]
- Musazzi, U.M.; Dolci, L.S.; Albertini, B.; Passerini, N.; Cilurzo, F. A new melatonin oral delivery platform based on orodispersible films containing solid lipid microparticles. Int. J. Pharm. 2019, 559, 280–288. [Google Scholar] [CrossRef] [Green Version]
- Tursilli, R.; Casolari, A.; Iannuccelli, V.; Scalia, S. Enhancement of melatonin photostability by encapsulation in lipospheres. J. Pharm. Biomed. Anal. 2006, 40, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Kanikkannan, N.; Singh, M. Skin permeation enhancement effect and skin irritation of saturated fatty alcohols. Int. J. Pharm. 2002, 248, 219–228. [Google Scholar] [CrossRef]
- Priano, L.; Esposti, D.; Esposti, R.; Castagna, G.; De Medici, C.; Fraschini, F.; Gasco, M.R.; Mauro, A. Solid Lipid Nanoparticles Incorporating Melatonin as New Model for Sustained Oral and Transdermal Delivery Systems. J. Nanosci. Nanotechnol. 2007, 7, 3596–3601. [Google Scholar] [CrossRef] [Green Version]
- Hatem, S.; Nasr, M.; Moftah, N.H.; Ragai, M.H.; Geneidi, A.S.; Elkheshen, S.A. Clinical cosmeceutical repurposing of melatonin in androgenic alopecia using nanostructured lipid carriers prepared with antioxidant oils. Expert Opin. Drug. Deliv. 2018, 15, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Bucolo, C.; Drago, F.; Salomone, S.; Pignatello, R. Cationic solid lipid nanoparticles enhance ocular hypotensive effect of melatonin in rabbit. Int. J. Pharm. 2015, 478, 180–186. [Google Scholar] [CrossRef]
- Rezzani, R.; Rodella, L.F.; Fraschini, F.; Gasco, M.R.; Demartini, G.; Musicanti, C.; Reiter, R.J. Melatonin delivery in solid lipid nanoparticles: Prevention of cyclosporine A induced cardiac damage. J. Pineal Res. 2009, 46, 255–261. [Google Scholar] [CrossRef]
- Mirhoseini, M.; Rezanejad Gatabi, Z.; Saeedi, M.; Morteza-Semnani, K.; Talebpour Amiri, F.; Kelidari, H.R.; Karimpour Malekshah, A.A. Protective effects of melatonin solid lipid nanoparticles on testis histology after testicular trauma in rats. Res. Pharm. Sci. 2019, 14, 201–208. [Google Scholar] [CrossRef]
- Sabzichi, M.; Samadi, N.; Mohammadian, J.; Hamishehkar, H.; Akbarzadeh, M.; Molavi, O. Sustained release of melatonin: A novel approach in elevating efficacy of tamoxifen in breast cancer treatment. Colloids Surf. B Biointerfaces 2016, 145, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Hallan, S.S.; Kaur, P.; Kaur, V.; Mishra, N.; Vaidya, B. Lipid polymer hybrid as emerging tool in nanocarriers for oral drug delivery. Artif. Cells Nanomed. Biotechnol. 2014, 44, 334–349. [Google Scholar] [CrossRef]
- Pohlmann, A.R.; Schaffazick, S.R.; Creczynski-Pasa, T.B.; Guterres, S.S. Preparation of Drug-Loaded Polymeric Nanoparticles and Evaluation of the Antioxidant Activity Against Lipid Peroxidation. Methods Mol. Biol. 2010, 610, 109–121. [Google Scholar]
- Schaffazick, S.R.; Siqueira, I.R.; Badejo, A.S.; Jornada, D.S.; Pohlmann, A.R.; Netto, C.A.; Guterres, S.S. Incorporation in polymeric nanocapsules improves the antioxidant effect of melatonin against lipid peroxidation in mice brain and liver. Eur. J. Pharm. Biopharm. 2008, 69, 64–71. [Google Scholar] [CrossRef]
- Külkamp, I.C.; Rabelo, B.D.; Berlitz, S.J.; Isoppo, M.; Bianchin, M.D.; Schaffazick, S.R.; Pohlmann, A.R.; Guterres, S.S. Nanoencapsulation Improves the In Vitro Antioxidant Activity of Lipoic Acid. J. Biomed. Nanotechnol. 2011, 7, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Charão, M.F.; Baierle, M.; Gauer, B.; Goethel, G.; Fracasso, R.; Paese, K.; Brucker, N.; Moro, A.M.; Bubols, G.B.; Dias, B.B.; et al. Protective effects of melatonin-loaded lipid-core nanocapsules on paraquat-induced cytotoxicity and genotoxicity in a pulmonary cell line. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2015, 784–785, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Charão, M.F.; Goethel, G.; Brucker, N.; Paese, K.; Eifler-Lima, V.L.; Pohlmann, A.R.; Guterres, S.S.; Garcia, S.C. Melatonin-loaded lipid-core nanocapsules protect against lipid peroxidation caused by paraquat through increased SOD expression in Caenorhabditis elegans. BMC Pharmacol. Toxicol. 2019, 20, 80. [Google Scholar] [CrossRef]
- Charão, M.F.; Souto, C.; Brucker, N.; Barth, A.; Jornada, D.S.; Fagundez, D.; Ávila, D.S.; Eifler-Lima, V.L.; Guterres, S.S.; Pohlmann, A.R.; et al. Caenorhabditis elegans as an alternative in vivo model to determine oral uptake, nanotoxicity, and efficacy of melatonin-loaded lipid-core nanocapsules on paraquat damage. Int. J. Nanomed. 2015, 10, 5093–5106. [Google Scholar] [CrossRef] [Green Version]
- Komninou, E.R.; Remião, M.H.; Lucas, C.G.; Domingues, W.B.; Basso, A.C.; Jornada, D.S.; Deschamps, J.C.; Beck, R.C.R.; Pohlmann, A.R.; Bordignon, V.; et al. Effects of Two Types of Melatonin-Loaded Nanocapsules with Distinct Supramolecular Structures: Polymeric (NC) and Lipid-Core Nanocapsules (LNC) on Bovine Embryo Culture Model. PLoS ONE 2016, 11, e0157561. [Google Scholar] [CrossRef]
- Remião, M.H.; Lucas, C.G.; Domingues, W.B.; Silveira, T.; Barther, N.N.; Komninou, E.R.; Basso, A.C.; Jornada, D.S.; Beck, R.C.R.; Pohlmann, A.R.; et al. Melatonin delivery by nanocapsules during in vitro bovine oocyte maturation decreased the reactive oxygen species of oocytes and embryos. Reprod. Toxicol. 2016, 63, 70–81. [Google Scholar] [CrossRef]
- Schaffazick, S.R.; Pohlmann, A.R.; de Cordova, C.A.S.; Creczynski-Pasa, T.B.; Guterres, S.S. Protective properties of melatonin-loaded nanoparticles against lipid peroxidation. Int. J. Pharm. 2005, 289, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Carbone, C.; Manno, D.; Serra, A.; Musumeci, T.; Pepe, V.; Tisserand, C.; Puglisi, G. Innovative hybrid vs polymeric nanocapsules: The influence of the cationic lipid coating on the “4S”. Colloids Surf. B Biointerfaces 2016, 141, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Bartelds, R.; Nematollahi, M.H.; Pols, T.; Stuart, M.C.A.; Pardakhty, A.; Asadikaram, G.; Poolman, B. Niosomes, an alternative for liposomal delivery. PLoS ONE 2018, 13, e0194179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.; Hanning, S.; Falconer, J.; Locke, M.; Wen, J. Recent advances in non-ionic surfactant vesicles (niosomes): Fabrication, characterization, pharmaceutical and cosmetic applications. Eur. J. Pharm. Biopharm. 2019, 144, 18–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Priprem, A.; Nukulkit, C.; Johns, N.P.; Laohasiriwong, S.; Yimtae, K.; Soontornpas, C. Transmucosal delivery of melatonin-encapsulated niosomes in a mucoadhesive gel. Ther. Deliv. 2018, 9, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Damrongrungruang, T.; Panpitakkul, P.; Somudorn, J.; Sangchart, P.; Mahakunakorn, P.; Uthaiwat, P.; Daduang, J.; Panyatip, P.; Puthongking, P.; Priprem, A. Glutaryl Melatonin Niosome Gel for Topical Oral Mucositis: Anti- Inflammatory and Anticandidiasis. Curr. Drug Deliv. 2020, 17, 195–206. [Google Scholar] [CrossRef]
- Uthaiwat, P.; Daduang, J.; Priprem, A.; Settasatian, C.; Chio-Srichan, S.; Lee, Y.-C.; Mahakunakorn, P.; Boonsiri, P. Topical Melatonin Niosome Gel for the Treatment of 5-FU-Induced Oral Mucositis in Mice. Curr. Drug Deliv. 2021, 18, 199–211. [Google Scholar] [CrossRef]
- Priprem, A.; Johns, J.R.; Limsitthichaikoon, S.; Limphirat, W.; Mahakunakorn, P.; Johns, N.P. Intranasal melatonin nanoniosomes: Pharmacokinetic, pharmacodynamics and toxicity studies. Ther. Deliv. 2017, 8, 373–390. [Google Scholar] [CrossRef]
- Arslan Azizoglu, G.; Tuncay Tanriverdi, S.; Aydin Kose, F.; Ballar Kirmizibayrak, P.; Ozer, O. Dual-Prevention for UV-Induced Skin Damage: Incorporation of Melatonin-Loaded Elastic Niosomes into Octyl Methoxycinnamate Pickering Emulsions. AAPS PharmSciTech. 2017, 18, 2987–2998. [Google Scholar] [CrossRef]
- Achilleos, D.S.; Vamvakaki, M. End-Grafted Polymer Chains onto Inorganic Nano-Objects. Materials 2010, 3, 1981–2026. [Google Scholar] [CrossRef] [Green Version]
- Gann, J.P.; Yan, M. A versatile method for grafting polymers on nanoparticles. Langmuir 2008, 24, 5319–5323. [Google Scholar] [CrossRef] [Green Version]
- Bitar, A.; Ahmad, N.M.; Fessi, H.; Elaissari, A. Silica-based nanoparticles for biomedical applications. Drug Discov. Today 2012, 17, 1147–1154. [Google Scholar] [CrossRef]
- Khattabi, A.M.; Talib, W.H.; Alqdeimat, D.A. The effect of polymer length on the in vitro characteristics of a drug loaded and targeted silica nanoparticles. Saudi Pharm. J. 2018, 26, 1022–1026. [Google Scholar] [CrossRef]
- Niu, G.; Yousefi, B.; Qujeq, D.; Marjani, A.; Asadi, J.; Wang, Z.; Mir, S.M. Melatonin and doxorubicin co-delivered via a functionalized graphene-dendrimeric system enhances apoptosis of osteosarcoma cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111554. [Google Scholar] [CrossRef]
- Mirmajidi, T.; Chogan, F.; Rezayan, A.H.; Sharifi, A.M. In vitro and in vivo evaluation of a nanofiber wound dressing loaded with melatonin. Int. J. Pharm. 2021, 596, 120213. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, X.; Liu, Y.; Yang, J.; Zhang, Q.; Wang, L.; Wu, W.; Yang, Q.; Liu, B. Melatonin loaded with bacterial cellulose nanofiber by Pickering-emulsion solvent evaporation for enhanced dissolution and bioavailability. Int. J. Pharm. 2019, 559, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Bessone, C.D.V.; Martinez, S.M.; Luna, J.D.; Marquez, M.A.; Ramírez, M.L.; Allemandi, D.A.; Carpentieri, Á.R.; Quinteros, D.A. Neuroprotective effect of melatonin loaded in ethylcellulose nanoparticles applied topically in a retinal degeneration model in rabbits. Exp. Eye Res. 2020, 200, 108222. [Google Scholar] [CrossRef]
- Vlachou, M.; Kikionis, S.; Siamidi, A.; Tragou, K.; Kapoti, S.; Ioannou, E.; Roussis, V.; Tsotinis, A. Fabrication and Characterization of Electrospun Nanofibers for the Modified Release of the Chronobiotic Hormone Melatonin. Curr. Drug Deliv. 2019, 16, 79–85. [Google Scholar] [CrossRef]
- Vlachou, M.; Kikionis, S.; Siamidi, A.; Tragou, K.; Ioannou, E.; Roussis, V.; Tsotinis, A. Modified In Vitro Release of Melatonin Loaded in Nanofibrous Electrospun Mats Incorporated Into Monolayered and Three-Layered Tablets. J. Pharm. Sci. 2019, 108, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Salehi, M.; Naseri-Nosar, M.; Ebrahimi-Barough, S.; Nourani, M.; Khojasteh, A.; Farzamfar, S.; Mansouri, K.; Ai, J. Polyurethane/Gelatin Nanofibrils Neural Guidance Conduit Containing Platelet-Rich Plasma and Melatonin for Transplantation of Schwann Cells. Cell. Mol. Neurobiol. 2017, 38, 703–713. [Google Scholar] [CrossRef]
- Kumar Yadav, S.; Kumar Srivastava, A.; Dev, A.; Kaundal, B.; Roy Choudhury, S.; Karmakar, S. Nanomelatonin triggers superior anticancer functionality in a human malignant glioblastoma cell line. Nanotechnology 2017, 28, 365102. [Google Scholar] [CrossRef] [PubMed]
- Blažević, F.; Milekić, T.; Romić, M.D.; Juretić, M.; Pepić, I.; Filipović-Grčić, J.; Lovrić, J.; Hafner, A. Nanoparticle-mediated interplay of chitosan and melatonin for improved wound epithelialisation. Carbohydr. Polym. 2016, 146, 445–454. [Google Scholar] [CrossRef]
- Lopes Rocha Correa, V.; Assis Martins, J.; Ribeiro de Souza, T.; de Castro Nunes Rincon, G.; Pacheco Miguel, M.; Borges de Menezes, L.; Correa Amaral, A. Melatonin loaded lecithin-chitosan nanoparticles improved the wound healing in diabetic rats. Int. J. Biol. Macromol. 2020, 162, 1465–1475. [Google Scholar] [CrossRef] [PubMed]
- Shokrzadeh, M.; Ghassemi-Barghi, N. Melatonin Loading Chitosan-Tripolyphosphate Nanoparticles: Application in Attenuating Etoposide-Induced Genotoxicity in HepG2 Cells. Pharmacology 2018, 102, 74–80. [Google Scholar] [CrossRef]
- Hafner, A.; Lovrić, J.; Pepić, I.; Filipović-Grčić, J. Lecithin/chitosan nanoparticles for transdermal delivery of melatonin. J. Microencapsul. 2011, 28, 807–815. [Google Scholar] [CrossRef]
- Hafner, A.; Lovrić, J.; Voinovich, D.; Filipović-Grčić, J. Melatonin-loaded lecithin/chitosan nanoparticles: Physicochemical characterisation and permeability through Caco-2 cell monolayers. Int. J. Pharm. 2009, 381, 205–213. [Google Scholar] [CrossRef]
- El-Gibaly, I. Novel B melatonin-loaded chitosan microcapsules: In vitro characterization and antiapoptosis efficacy for aflatoxin B1-induced apoptosis in rat liver. Int. J. Pharm. 2003, 260, 5–22. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Fal′ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef]
- Novoselov, K.S. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [Green Version]
- Usman, M.S.; Hussein, M.Z.; Kura, A.U.; Fakurazi, S.; Masarudin, M.J.; Ahmad Saad, F.F. Graphene Oxide as a Nanocarrier for a Theranostics Delivery System of Protocatechuic Acid and Gadolinium/Gold Nanoparticles. Molecules 2018, 23, 500. [Google Scholar] [CrossRef] [Green Version]
- Singh, M.R.; Saraf, S.; Vyas, A.; Jain, V.; Singh, D. Innovative approaches in wound healing: Trajectory and advances. Artif. Cells Nanomed. Biotechnol. 2013, 1–11. [Google Scholar] [CrossRef]
- Rahmani Del Bakhshayesh, A.; Annabi, N.; Khalilov, R.; Akbarzadeh, A.; Samiei, M.; Alizadeh, E.; Alizadeh-Ghodsi, M.; Davaran, S.; Montaseri, A. Recent advances on biomedical applications of scaffolds in wound healing and dermal tissue engineering. Artif. Cells Nanomed. Biotechnol. 2017, 46, 691–705. [Google Scholar] [CrossRef] [PubMed]
- Almaaytah, A.; Qaoud, M.T.; Khalil Mohammed, G.; Abualhaijaa, A.; Knappe, D.; Hoffmann, R.; Al-Balas, Q. Antimicrobial and Antibiofilm Activity of UP-5, an Ultrashort Antimicrobial Peptide Designed Using Only Arginine and Biphenylalanine. Pharmaceuticals 2018, 11, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rassu, G.; Porcu, E.P.; Fancello, S.; Obinu, A.; Senes, N.; Galleri, G.; Migheli, R.; Gavini, E.; Giunchedi, P. Intranasal Delivery of Genistein-Loaded Nanoparticles as a Potential Preventive System against Neurodegenerative Disorders. Pharmaceutics 2018, 11, 8. [Google Scholar] [CrossRef] [Green Version]
- Saeed, R.M.; Dmour, I.; Taha, M.O. Stable Chitosan-Based Nanoparticles Using Polyphosphoric Acid or Hexametaphosphate for Tandem Ionotropic/Covalent Crosslinking and Subsequent Investigation as Novel Vehicles for Drug Delivery. Front. Bioeng. Biotechnol. 2020, 8, 4. [Google Scholar] [CrossRef] [Green Version]
- Du, H.; Yang, X.; Pang, X.; Zhai, G. The synthesis, self-assembling, and biocompatibility of a novel O-carboxymethyl chitosan cholate decorated with glycyrrhetinic acid. Carbohydr. Polym. 2014, 111, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Hafner, A.; Lovrić, J.; Romić, M.D.; Juretić, M.; Pepić, I.; Cetina-Čižmek, B.; Filipović-Grčić, J. Evaluation of cationic nanosystems with melatonin using an eye-related bioavailability prediction model. Eur. J. Pharm. Sci. 2015, 75, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Romić, M.D.; Sušac, A.; Lovrić, J.; Cetina-Čižmek, B.; Filipović-Grčić, J.; Hafner, A. Evaluation of stability and in vitro wound healing potential of melatonin loaded (lipid enriched) chitosan based microspheres. Acta Pharmaceutica 2019, 69, 635–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hafner, A.; Dürrigl, M.; Pepić, I.; Filipović-Grčić, J. Short- and Long-Term Stability of Lyophilised Melatonin-Loaded Lecithin/Chitosan Nanoparticles. Pharm. Bull. 2011, 59, 1117–1123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Wang, L.; Belwal, T.; Jiang, Y.; Li, D.; Xu, Y.; Luo, Z.; Li, L. Chitosan-based melatonin bilayer coating for maintaining quality of fresh-cut products. Carbohydr. Polym. 2020, 235, 115973. [Google Scholar] [CrossRef]
- Ahlawat, J.; Henriquez, G.; Narayan, M. Enhancing the Delivery of Chemotherapeutics: Role of Biodegradable Polymeric Nanoparticles. Molecules 2018, 23, 2157. [Google Scholar] [CrossRef] [Green Version]
- Vauthier, C.; Bouchemal, K. Methods for the Preparation and Manufacture of Polymeric Nanoparticles. Pharm. Res. 2008, 26, 1025–1058. [Google Scholar] [CrossRef]
- Amgoth, C.; Phan, C.; Banavoth, M.; Rompivalasa, S.; Tang, G. Polymer Properties: Functionalization and Surface Modified Nanoparticles. In Role of Novel Drug Delivery Vehicles in Nanobiomedicine; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Schaffazick, S.R.; Pohlmann, A.R.; Guterres, S.S. Nanocapsules, nanoemulsion and nanodispersion containing melatonin: Preparation, characterization and stability evaluation. Pharmazie 2007, 62, 354–360. [Google Scholar]
- De Oliveira Junior, E.R.; Nascimento, T.L.; Salomão, M.A.; da Silva, A.C.G.; Valadares, M.C.; Lima, E.M. Increased Nose-to-Brain Delivery of Melatonin Mediated by Polycaprolactone Nanoparticles for the Treatment of Glioblastoma. Pharm. Res. 2019, 36. [Google Scholar] [CrossRef] [PubMed]
- Martin Gimenez, V.M.; Russo, M.G.; Narda, G.E.; Fuentes, L.B.; Mazzei, L.; Gamarra-Luques, C.; Kassuha, D.E.; Manucha, W. Synthesis, physicochemical characterisation and biological activity of anandamide/varepsilon-polycaprolactone nanoparticles obtained by electrospraying. IET Nanobiotechnol. 2020, 14, 86–93. [Google Scholar] [CrossRef]
- Martin Gimenez, V.M.; Diaz-Rodriguez, P.; Sanz, R.L.; Vivero-Lopez, M.; Concheiro, A.; Diez, E.; Prado, N.; Enrique Kassuha, D.; Alvarez-Lorenzo, C.; Manucha, W. Anandamide-nanoformulation obtained by electrospraying for cardiovascular therapy. Int. J. Pharm. 2019, 566, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Martin Gimenez, V.M.; Mocayar Maron, F.J.; Garcia, S.; Mazzei, L.; Guevara, M.; Yunes, R.; Manucha, W. Central nervous system, peripheral and hemodynamic effects of nanoformulated anandamide in hypertension. Adv. Med. Sci. 2021, 66, 72–80. [Google Scholar] [CrossRef]
- Gimenez, V.M.; Sperandeo, N.; Faudone, S.; Noriega, S.; Manucha, W.; Kassuha, D. Preparation and characterization of bosentan monohydrate/epsilon-polycaprolactone nanoparticles obtained by electrospraying. Biotechnol. Prog. 2019, 35, e2748. [Google Scholar] [CrossRef]
- Gurler, E.B.; Ergul, N.M.; Ozbek, B.; Ekren, N.; Oktar, F.N.; Haskoylu, M.E.; Oner, E.T.; Eroglu, M.S.; Ozbeyli, D.; Korkut, V.; et al. Encapsulated melatonin in polycaprolactone (PCL) microparticles as a promising graft material. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 100, 798–808. [Google Scholar] [CrossRef] [PubMed]
- Massella, D.; Leone, F.; Peila, R.; Barresi, A.A.; Ferri, A. Functionalization of Cotton Fabrics with Polycaprolactone Nanoparticles for Transdermal Release of Melatonin. J. Funct. Biomater. 2017, 9, 1. [Google Scholar] [CrossRef] [Green Version]
- Penott-Chang, E.; Walther, A.; Millard, P.; Jäger, A.; Jäger, E.; Müller, A.H.E.; Guterres, S.S.; Pohlmann, A.R. Amphiphilic Diblock Copolymer and Polycaprolactone Blends to Produce New Vesicular Nanocarriers. J. Biomed. Nanotechnol. 2012, 8, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Ma, Z.; Wang, C.; Li, H.; He, Y. Pro-chondrogenic and immunomodulatory melatonin-loaded electrospun membranes for tendon-to-bone healing. J. Mater. Chem. B 2019, 7, 6564–6575. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.G.; Khalil, N.M.; Mainardes, R.M. Application of a validated HPLC-PDA method for the determination of melatonin content and its release from poly(lactic acid) nanoparticles. J. Pharm. Anal. 2017, 7, 388–393. [Google Scholar] [CrossRef]
- Pandey, S.K.; Haldar, C.; Vishwas, D.K.; Maiti, P. Synthesis andin vitroevaluation of melatonin entrapped PLA nanoparticles: An oxidative stress and T-cell response using golden hamster. J. Biomed. Mater. Res. A 2015, 103, 3034–3044. [Google Scholar] [CrossRef] [PubMed]
- Rezvantalab, S.; Drude, N.I.; Moraveji, M.K.; Guvener, N.; Koons, E.K.; Shi, Y.; Lammers, T.; Kiessling, F. PLGA-Based Nanoparticles in Cancer Treatment. Front. Pharmacol. 2018, 9, 1260. [Google Scholar] [CrossRef] [Green Version]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Ghitman, J.; Biru, E.I.; Stan, R.; Iovu, H. Review of hybrid PLGA nanoparticles: Future of smart drug delivery and theranostics medicine. Mater. Des. 2020, 193, 108805. [Google Scholar] [CrossRef]
- Martins, L.G.; Khalil, N.M.; Mainardes, R.M. PLGA Nanoparticles and Polysorbate-80-Coated PLGA Nanoparticles Increase the In vitro Antioxidant Activity of Melatonin. Curr. Drug Deliv. 2018, 15, 554–563. [Google Scholar] [CrossRef]
- Altındal, D.Ç.; Gümüşderelioğlu, M. Melatonin releasing PLGA micro/nanoparticles and their effect on osteosarcoma cells. J. Microencapsul. 2015, 33, 53–63. [Google Scholar] [CrossRef]
- Jarrar, H.; Çetin Altındal, D.; Gümüşderelioğlu, M. Scaffold-based osteogenic dual delivery system with melatonin and BMP-2 releasing PLGA microparticles. Int. J. Pharm. 2021, 600, 120489. [Google Scholar] [CrossRef]
- Li Volti, G.; Musumeci, T.; Pignatello, R.; Murabito, P.; Barbagallo, I.; Carbone, C.; Gullo, A.; Puglisi, G. Antioxidant potential of different melatonin-loaded nanomedicines in an experimental model of sepsis. Exp. Biol. Med. 2012, 237, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Mukherjee, A.; Das, N.; Swarnakar, S. Protective roles of nanomelatonin in cerebral ischemia-reperfusion of aged brain: Matrixmetalloproteinases as regulators. Exp. Gerontol. 2017, 92, 13–22. [Google Scholar] [CrossRef]
- Ma, Q.; Yang, J.; Huang, X.; Guo, W.; Li, S.; Zhou, H.; Li, J.; Cao, F.; Chen, Y. Poly(Lactide-Co-Glycolide)-Monomethoxy-Poly-(Polyethylene Glycol) Nanoparticles Loaded with Melatonin Protect Adipose-Derived Stem Cells Transplanted in Infarcted Heart Tissue. Stem Cells 2018, 36, 540–550. [Google Scholar] [CrossRef] [Green Version]
- Chen, G.; Deng, H.; Song, X.; Lu, M.; Zhao, L.; Xia, S.; You, G.; Zhao, J.; Zhang, Y.; Dong, A.; et al. Reactive oxygen species-responsive polymeric nanoparticles for alleviating sepsis-induced acute liver injury in mice. Biomaterials 2017, 144, 30–41. [Google Scholar] [CrossRef]
- López-Muñoz, R.; Treviño, M.E.; Morales, G.; Valdez-Garza, J.A.; Díaz de León, R.; Saade, H.; Enríquez-Medrano, F.J.; López, R.G. Ultrafine Nanoparticles of Poly(Methyl Methacrylate-co-Methacrylic Acid) Loaded with Aspirin. J. Nanomater. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Atoufi, Z.; Kamrava, S.K.; Davachi, S.M.; Hassanabadi, M.; Saeedi Garakani, S.; Alizadeh, R.; Farhadi, M.; Tavakol, S.; Bagher, Z.; Hashemi Motlagh, G. Injectable PNIPAM/Hyaluronic acid hydrogels containing multipurpose modified particles for cartilage tissue engineering: Synthesis, characterization, drug release and cell culture study. Int. J. Biol. Macromol. 2019, 139, 1168–1181. [Google Scholar] [CrossRef]
- Soriano, J.L.; Calpena, A.C.; Rincón, M.; Pérez, N.; Halbaut, L.; Rodríguez-Lagunas, M.J.; Clares, B. Melatonin nanogel promotes skin healing response in burn wounds of rats. Nanomedicine 2020, 15, 2133–2147. [Google Scholar] [CrossRef] [PubMed]
- Alphandéry, E. Biodistribution and targeting properties of iron oxide nanoparticles for treatments of cancer and iron anemia disease. Nanotoxicology 2019, 13, 573–596. [Google Scholar] [CrossRef]
- Moody, A.S.; Sharma, B. Multi-metal, Multi-wavelength Surface-Enhanced Raman Spectroscopy Detection of Neurotransmitters. ACS Chem Neurosci. 2018, 9, 1380–1387. [Google Scholar] [CrossRef] [PubMed]
- Krpeticć, Z.; Porta, F.; Caneva, E.; Dal Santo, V.; Scariì, G. Phagocytosis of Biocompatible Gold Nanoparticles. Langmuir 2010, 26, 14799–14805. [Google Scholar] [CrossRef] [PubMed]
- El-Megharbel, S.M.; Almalki, A.S.A.; Hamza, R.Z.; Gobouri, A.A.; Alhadhrami, A.A.; Al-Humaidi, J.Y.; Refat, M.S. Synthesis and suggestion of a new nanometric gold(III) melatonin drug complex: An interesting model for testicular protection. Future Med. Chem. 2018, 10, 1693–1704. [Google Scholar] [CrossRef]
- Gurunathan, S.; Jeyaraj, M.; Kang, M.-H.; Kim, J.-H. Melatonin Enhances Palladium-Nanoparticle-Induced Cytotoxicity and Apoptosis in Human Lung Epithelial Adenocarcinoma Cells A549 and H1229. Antioxidants 2020, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Soltani, N.; Tavakkoli, N.; Shahdost-fard, F.; Salavati, H.; Abdoli, F. A carbon paste electrode modified with Al2O3-supported palladium nanoparticles for simultaneous voltammetric determination of melatonin, dopamine, and acetaminophen. Microchimica Acta 2019, 186. [Google Scholar] [CrossRef]
- Vázquez-Gil, M.J.; Mesonero, M.J.; Flores, O.; Criado, M.; Hidalgo, F.; Arévalo, M.A.; Sánchez-Rodrıíguez, A.; Tuñón, M.J.; López-Novoa, J.M.; Esteller, A. Sequential changes in redox status and nitric oxide synthases expression in the liver after bile duct ligation. Life Sci. 2004, 75, 717–732. [Google Scholar] [CrossRef]
- Wang, H.; Wei, W.; Zhang, S.-Y.; Shen, Y.-X.; Yue, L.; Wang, N.-P.; Xu, S.-Y. Melatonin-selenium nanoparticles inhibit oxidative stress and protect against hepatic injury induced by Bacillus Calmette-Guérin/lipopolysaccharide in mice. J. Pineal Res. 2005, 39, 156–163. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Roy Choudhury, S.; Karmakar, S. Near-Infrared Responsive Dopamine/Melatonin-Derived Nanocomposites Abrogating in Situ Amyloid β Nucleation, Propagation, and Ameliorate Neuronal Functions. ACS Appl. Mater. Interfaces. 2020, 12, 5658–5670. [Google Scholar] [CrossRef]
- Xie, W.; Gao, Q.; Wang, D.; Wang, W.; Yuan, J.; Guo, Z.; Yan, H.; Wang, X.; Sun, X.; Zhao, L. Melatonin potentiates “inside-out” nano-thermotherapy in human breast cancer cells: A potential cancer target multimodality treatment based on melatonin-loaded nanocomposite particles. Int. J. Nanomed. 2017, 12, 7351–7363. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Wang, X.; Wang, J.; Yuan, J.; Zhang, J.; Zhu, X.; Lei, C.; Yang, Q.; Wang, B.; Cao, F.; et al. A Peptide-Functionalized Magnetic Nanoplatform-Loaded Melatonin for Targeted Amelioration of Fibrosis in Pressure Overload-Induced Cardiac Hypertrophy. Int. J. Nanomed. 2020, 15, 1321–1333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sruthi, S.; Millot, N.; Mohanan, P.V. Zinc oxide nanoparticles mediated cytotoxicity, mitochondrial membrane potential and level of antioxidants in presence of melatonin. Int. J. Biol. Macromol. 2017, 103, 808–818. [Google Scholar] [CrossRef]
- Torabi, F.; Malekzadeh Shafaroudi, M.; Rezaei, N. Combined protective effect of zinc oxide nanoparticles and melatonin on cyclophosphamide-induced toxicity in testicular histology and sperm parameters in adult Wistar rats. Int. J. Reprod. BioMed. 2017, 15, 403–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.; Liu, J.; Bai, R.; Shi, J.; Zhu, X.; Liu, J.; Guo, J.; Zhang, W.; Liu, H.; Liu, Z. Mitochondria-Inspired Nanoparticles with Microenvironment-Adapting Capacities for On-Demand Drug Delivery after Ischemic Injury. ACS Nano. 2020, 14, 11846–11859. [Google Scholar] [CrossRef] [PubMed]
- Ruszkiewicz, J.A.; Pinkas, A.; Ferrer, B.; Peres, T.V.; Tsatsakis, A.; Aschner, M. Neurotoxic effect of active ingredients in sunscreen products, a contemporary review. Toxicol. Rep. 2017, 4, 245–259. [Google Scholar] [CrossRef] [PubMed]


| NC/Nanoplatform | Cell Type/Tissue/Nanomethod | Main Actions | Ref |
|---|---|---|---|
| Silica | HeLa cells | Longer polymers improved melatonin release and cell toxicity | [85] |
| Gaphene-dendrimeric system | Saos-2 and MG-63 cells | Antitumor action against osteosarcoma (higher cellular uptake and apoptosis) | [86] |
| Chitosan-polycaprolactone (PCL)/polyvinylalcohol (PVA)-melatonin | Rat skin | Improved skin regeneration, wound remodeling, and reduced inflammation | [87] |
| Bacterial cellulose nanofiber | Rat blood | Increased oral dissolution and bioavailability of melatonin | [88] |
| Ethylcellulose nanocapsules | Rabbit retinal ganglion cells | Slow in vitro release of melatonin and high corneal penetration | [89] |
| Cellulose acetate (CA), polyvinylpyrrolidinone (PV), and hydroxypropylmethylcellulose (HP) | Gastric-like fluids showing pH variations | Increased bioavailability of melatonin to treat sleep dysfunctions; control of the sleep-onset | [90,91] |
| Polyurethane (PU) and gelatin nanofibrils (GNFs) | Defected sciatic nerve of Wistar rats | Enhanced regenerative capacity of nerve and muscle function by melatonin | [92] |
| Lecithin/chitosan | U87MG and HepG2 cells | Improved melatonin release and permeability; reduced tumor growth and the genotoxic effect of drugs | [93,94,95,96] |
| Lecithin/chitosan | Lyophilization for storage | Prevented melatonin degradation and increased photostability | [56,97] |
| Lecithin/chitosan | Encapsulation efficiency in the nanoformulation | Promoted a slow release of melatonin | [95,96,98] |
| Chitosan buoyant microcapsules | Rats exposed to aflatoxin B1 | Promoted superior antiapoptotic activity of melatonin | [99] |
| NC/Nanoplatform | Cell Type/Tissue/ Nanomethod | Main Actions | Ref |
|---|---|---|---|
| Gold NP | Murine macrophage cells | Improved cellular uptake | [143] |
| Gold NP | Rat testis tissue | Protected against testicular damage by reducing lipid peroxidation, TNF-α, and IL-1β level, and enhancing antioxidant capacity | [144] |
| Palladium NP | A549 cells | Increased lung cell toxicity via apoptosis and DNA oxidation, and reduced ATP content, and mitochondrial membrane potential | [145] |
| Selenium NP | Mouse model of liver injury | Improved hepatocellular protection by reducing the activity of aminotransferase, the extent of hepatic cell damage, and migration rate of inflammatory cells | [148] |
| Dopamine-melatonin nanocomposite | SH-SY5 cells; Balb/c mice | Suppressed ROS and intracellular Aβ production and aggregation in cultured midbrain cells of adult mice with Alzheimer’s disease | [149] |
| Magnetic nanocomposite | MCF-7 cells | Increased the antiproliferative effect of melatonin | [150] |
| Superparamagnetic iron oxide NP with PLGA-COOH | Animal model of myocardial hypertrophy | Low doses of melatonin ameliorated fibrosis and myocardial hypertrophy | [151] |
| Zinc oxide NP | Mice brain tissue | Increased the activities of antioxidant enzymes | [152] |
| Zinc oxide NP | Rat testis | Protected against cyclophosphamide-induced reproductive damage | [153] |
| Mitochondria-resembling NP | Multistimuli-responsive NP | Favored melatonin release after ischemia improving ROS scavenging and preventing apoptosis | [154] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuffa, L.G.d.A.; Seiva, F.R.F.; Novais, A.A.; Simão, V.A.; Martín Giménez, V.M.; Manucha, W.; Zuccari, D.A.P.d.C.; Reiter, R.J. Melatonin-Loaded Nanocarriers: New Horizons for Therapeutic Applications. Molecules 2021, 26, 3562. https://doi.org/10.3390/molecules26123562
Chuffa LGdA, Seiva FRF, Novais AA, Simão VA, Martín Giménez VM, Manucha W, Zuccari DAPdC, Reiter RJ. Melatonin-Loaded Nanocarriers: New Horizons for Therapeutic Applications. Molecules. 2021; 26(12):3562. https://doi.org/10.3390/molecules26123562
Chicago/Turabian StyleChuffa, Luiz Gustavo de Almeida, Fábio Rodrigues Ferreira Seiva, Adriana Alonso Novais, Vinícius Augusto Simão, Virna Margarita Martín Giménez, Walter Manucha, Debora Aparecida Pires de Campos Zuccari, and Russel J. Reiter. 2021. "Melatonin-Loaded Nanocarriers: New Horizons for Therapeutic Applications" Molecules 26, no. 12: 3562. https://doi.org/10.3390/molecules26123562










