β-Cyclodextrin Inclusion Complexes with Catechol-Containing Antioxidants Protocatechuic Aldehyde and Protocatechuic Acid—An Atomistic Perspective on Structural and Thermodynamic Stabilities
Abstract
:1. Introduction
2. Results and Discussion
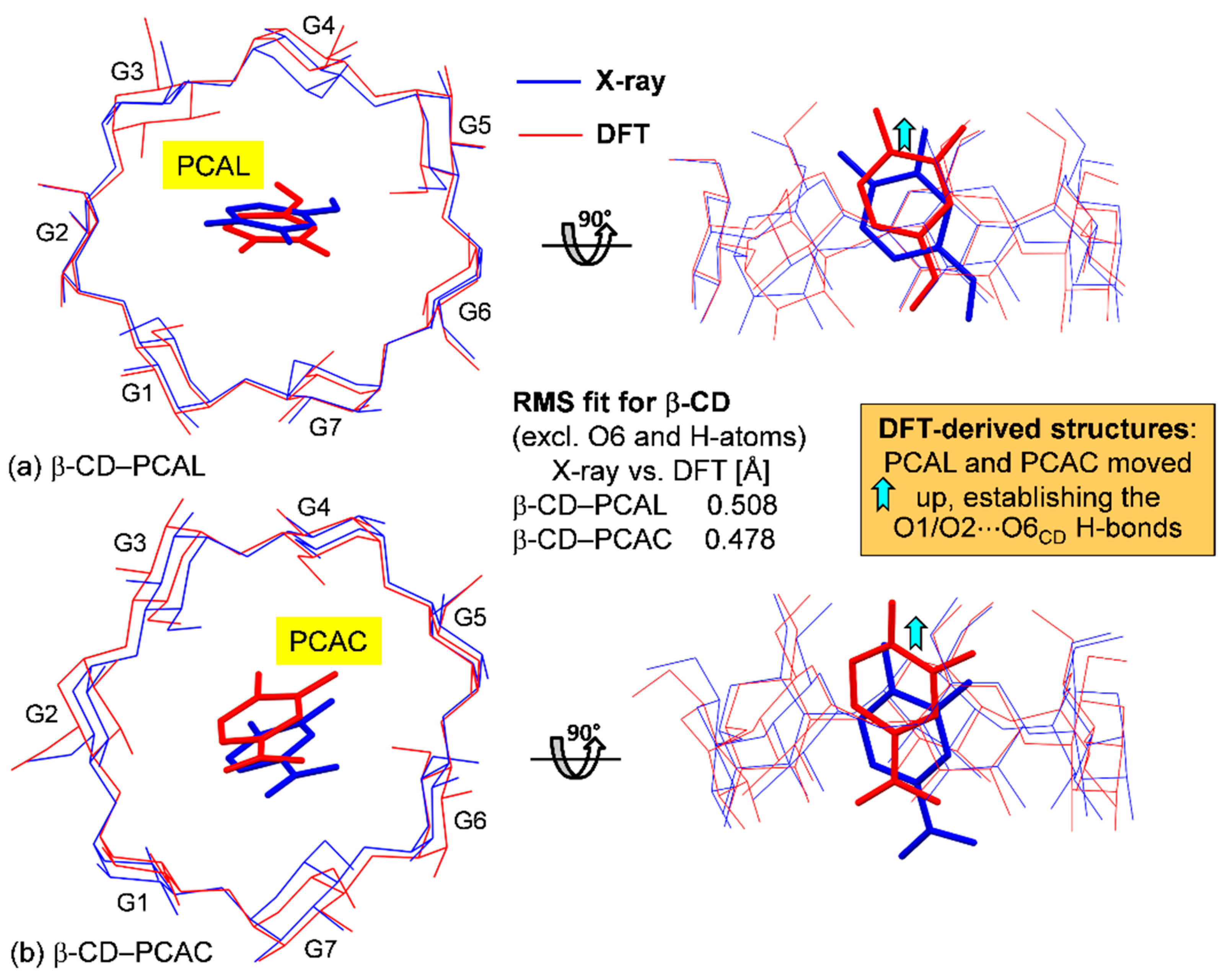
2.1. β-CD Macrocycles Are Affected to Some Extent by Inclusion of PCAL and PCAC
2.2. Common Inclusion Geometry of Catechol-Containing Antioxidants
2.3. COOH Group of PCAC Makes the Distinction in Hydrogen Bonding Network
2.4. Thermodynamic Stabilities of β-CD–PCAL and β-CD–PCAC
2.5. Bioactive PCAL and PCAC Are Rather Rigid in Distinct Lattice Circumstances
3. Materials and Methods
3.1. Materials
3.2. Single Crystal X-ray Diffraction Analysis
3.2.1. Crystallization
3.2.2. Diffraction Data Collection
3.2.3. Structure Solution and Refinement
3.3. DFT Full-Geometry Optimization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability: Samples of the compounds are available from the authors. |
References
- Rababah, T.M.; Hettiarachchy, N.S.; Horax, R. Total phenolics and antioxidant activities of fenugreek, green tea, black tea, grape seed, ginger, rosemary, gotu kola, and ginkgo extracts, vitamin E, and tert-butylhydroquinone. J. Agric. Food Chem. 2004, 52, 5183–5186. [Google Scholar] [CrossRef]
- Pacheco-Palencia, L.A.; Mertens-Talcott, S.; Talcott, S.T. Chemical composition, antioxidant properties, and thermal stability of a phytochemical enriched oil from Acai (Euterpe oleracea Mart.). J. Agric. Food Chem. 2008, 56, 4631–4636. [Google Scholar] [CrossRef]
- Chao, C.Y.; Yin, M.C. Antibacterial effects of roselle calyx extracts and protocatechuic acid in ground beef and apple juice. Foodborne Pathog. Dis. 2009, 6, 201–206. [Google Scholar] [CrossRef] [Green Version]
- Palacios, I.; Lozano, M.; Moro, C.; D’arrigo, M.; Rostagno, M.A.; Martínez, J.A.; García-Lafuente, A.; Guillamón, E.; Villares, A. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem. 2011, 128, 674–678. [Google Scholar] [CrossRef]
- Vitaglione, P.; Donnarumma, G.; Napolitano, A.; Galvano, F.; Gallo, A.; Scalfi, L.; Fogliano, V. Protocatechuic acid is the major human metabolite of cyanidin-glucosides. J. Nutr. 2007, 137, 2043–2048. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kang, Y.H.; Jung, J.Y.; Lee, S.; Ohuchi, K.; Shin, K.H.; Kang, I.J.; Park, J.H.; Shin, H.K.; Lim, S.S. Protein glycation inhibitors from the fruiting body of Phellinus linteus. Biol. Pharm. Bull. 2008, 31, 1968–1972. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; Liu, Y.; Miao, A.D.; Wang, S.Q. Protocatechuic aldehyde suppresses TNF-α-induced ICAM-1 and VCAM-1 expression in human umbilical vein endothelial cells. Eur. J. Pharmacol. 2005, 513, 1–8. [Google Scholar] [CrossRef]
- Etoh, H.; Murakami, K.; Yogoh, T.; Ishikawa, H.; Fukuyama, Y.; Tanaka, H. Anti-oxidative compounds in barley tea. Biosci. Biotechnol. Biochem. 2004, 68, 2616–2618. [Google Scholar] [CrossRef] [Green Version]
- Xu, M.; Zhang, Z.; Fu, G.; Sun, S.; Sun, J.; Yang, M.; Liu, A.; Han, J.; Guo, D. Liquid chromatography–tandem mass spectrometry analysis of protocatechuic aldehyde and its phase I and II metabolites in rat. J. Chromatogr. B 2007, 856, 100–107. [Google Scholar] [CrossRef]
- Conde, E.; Cadahía, E.; García-Vallejo, M.C.; Fernández de Simón, B. Polyphenolic composition of Quercus suber cork from different Spanish provenances. J. Agric. Food Chem. 1998, 46, 3166–3171. [Google Scholar] [CrossRef]
- Masella, R.; Santangelo, C.; D’archivio, M.; LiVolti, G.; Giovannini, C.; Galvano, F. Protocatechuic acid and human disease prevention: Biological activities and molecular mechanisms. Curr. Med. Chem. 2012, 19, 2901–2917. [Google Scholar] [CrossRef]
- Kakkar, S.; Bais, S. A review on protocatechuic acid and its pharmacological potential. ISRN Pharmacol. 2014, 952943. [Google Scholar] [CrossRef] [Green Version]
- Yan, X. (Ed.) Dan Shen (Salvia miltiorrhiza) in Medicine; Biology and Chemistry; Springer: Berlin/Heidelberg, Germany, 2014; Volume 1. [Google Scholar]
- Semaming, Y.; Pannengpetch, P.; Chattipakorn, S.C.; Chattipakorn, N. Pharmacological properties of protocatechuic acid and its potential roles as complementary medicine. Evid Based Compl. Alt. Med. 2015, 593902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, A.K.; Rashid, R.; Fatima, N.; Mahmood, S.; Mir, S.; Khan, S.; Jabeen, N.; Murtaza, G. Pharmacological activities of protocatechuic acid. Acta Pol. Pharm. 2015, 72, 643–650. Available online: https://ptfarm.pl/pub/File/Acta_Poloniae/2015/4/643.pdf (accessed on 12 May 2021). [PubMed]
- Song, J.; He, Y.; Luo, C.; Feng, B.; Ran, F.; Xu, H.; Cia, Z.; Xua, R.; Han, L.; Zhang, D. New progress in the pharmacology of protocatechuic acid: A compound ingested in daily foods and herbs frequently and heavily. Pharmacol. Res. 2020, 105109. [Google Scholar] [CrossRef] [PubMed]
- Chang, Z.Q.; Gebru, E.; Lee, S.P.; Rhee, M.H.; Kim, J.C.; Cheng, H.; Park, S.C. In vitro antioxidant and anti-inflammatory activities of protocatechualdehyde isolated from Phellinus gilvus. J. Nutr. Sci. Vitaminol. 2011, 57, 118–122. [Google Scholar] [CrossRef] [Green Version]
- Jeong, J.B.; Lee, S.H. Protocatechualdehyde possesses anti-cancer activity through downregulating cyclin D1 and HDAC2 in human colorectal cancer cells. Biochem. Biophys. Res. Commun. 2013, 430, 381–386. [Google Scholar] [CrossRef]
- Wang, B.Q. Salvia miltiorrhiza: Chemical and pharmacological review of a medicinal plant. J. Med. Plant. Res. 2010, 4, 2813–2820. [Google Scholar] [CrossRef]
- Wang, L.; Ma, R.; Liu, C.; Liu, H.; Zhu, R.; Guo, S.; Tang, M.; Li, Y.; Niu, J.; Fu, M.; et al. Salvia miltiorrhiza: A potential red light to the development of cardiovascular diseases. Curr. Pharm. Des. 2017, 23, 1077–1097. [Google Scholar] [CrossRef] [Green Version]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Chen, D.; Chen, S. Antioxidant activity and mechanism of protocatechuic acid in vitro. Funct. Food Health Dis. 2011, 1, 232–244. [Google Scholar] [CrossRef]
- Saito, S.; Kawabata, J. DPPH (= 2, 2-diphenyl-1-picrylhydrazyl) radical-scavenging reaction of protocatechuic acid (= 3, 4-dihydroxybenzoic acid): Difference in reactivity between acids and their esters. Helv. Chim. Acta 2006, 89, 1395–1407. [Google Scholar] [CrossRef]
- Saenger, W. Cyclodextrin inclusion compounds in research and industry. Angew. Chem. Int. Ed. Engl. 1980, 19, 344–362. [Google Scholar] [CrossRef]
- Dodziuk, H. Cyclodextrins and Their Complexes: Chemistry, Analytical Methods, Applications; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Pinho, E.; Grootveld, M.; Soares, G.; Henriques, M. Cyclodextrins as encapsulation agents for plant bioactive compounds. Carbohydr. Polym. 2014, 101, 121–135. [Google Scholar] [CrossRef] [Green Version]
- Rescifina, A.; Chiacchio, U.; Iannazzo, D.; Piperno, A.; Romeo, G. β-cyclodextrin and caffeine complexes with natural polyphenols from olive and olive oils: NMR, thermodynamic, and molecular modeling studies. J. Agric. Food Chem. 2010, 58, 11876–11882. [Google Scholar] [CrossRef]
- Irwin, P.L.; Pfeffer, P.E.; Doner, L.W.; Sapers, G.M.; Brewster, J.D.; Nagahashi, G.; Hicks, K.B. Binding geometry, stoichiometry, and thermodynamics of cyclomalto-oligosaccharide (cyclodextrin) inclusion complex formation with chlorogenic acid, the major substrate of apple polyphenol oxidase. Carbohydr. Res. 1994, 256, 13–27. [Google Scholar] [CrossRef]
- Divakar, S.; Maheswaran, M.M. Structural studies on inclusion compounds of β-cyclodextrin with some substituted phenols. J. Incl. Phenom. Macrocycl. Chem. 1997, 27, 113–126. [Google Scholar] [CrossRef]
- Cao, J.; Wei, J.; Tian, K.; Su, H.; Wan, J.; Li, P. Simultaneous determination of seven phenolic acids in three Salvia species by capillary zone electrophoresis with β-cyclodextrin as modifier. J. Sep. Sci. 2014, 37, 3738–3744. [Google Scholar] [CrossRef]
- Mourtzinos, I.; Makris, D.P.; Yannakopoulou, K.; Kalogeropoulos, N.; Michali, I.; Karathanos, V.T. Thermal stability of anthocyanin extract of Hibiscus sabdariffa L. in the presence of β-cyclodextrin. J. Agric. Food Chem. 2008, 56, 10303–10310. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Konteles, S.; Mourtzinos, I.; Troullidou, E.; Chiou, A.; Karathanos, V.T. Encapsulation of complex extracts in β-cyclodextrin: An application to propolis ethanolic extract. J. Microencapsul. 2009, 26, 603–613. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Yannakopoulou, K.; Gioxari, A.; Chiou, A.; Makris, D.P. Polyphenol characterization and encapsulation in β-cyclodextrin of a flavonoid-rich Hypericum perforatum (St John’s wort) extract. LWT Food Sci. Technol. 2010, 43, 882–889. [Google Scholar] [CrossRef]
- Ibrahim, S.; Bowra, S. Improving Oxidative Stability of Polyphenolic Fraction of Apple Pomace by Encapsulation Using Naturally Occurring Polymers. J. Encapsul. Adsorp. Sci. 2019, 9, 83–108. [Google Scholar] [CrossRef] [Green Version]
- Aree, T.; Jongrungruangchok, S. Structure–antioxidant activity relationship of β-cyclodextrin inclusion complexes with olive tyrosol, hydroxytyrosol and oleuropein: Deep insights from X-ray analysis, DFT calculation and DPPH assay. Carbohydr. Polym. 2018, 199, 661–669. [Google Scholar] [CrossRef]
- Aree, T. Understanding structures and thermodynamics of β-cyclodextrin encapsulation of chlorogenic, caffeic and quinic acids: Implications for enriching antioxidant capacity and masking bitterness in coffee. Food Chem. 2019, 293, 550–560. [Google Scholar] [CrossRef]
- Aree, T. β-Cyclodextrin encapsulation of nortriptyline HCl and amitriptyline HCl: Molecular insights from single-crystal X-ray diffraction and DFT calculation. Int. J. Pharm. 2020, 575, 118899. [Google Scholar] [CrossRef]
- Aree, T. β-Cyclodextrin inclusion complexation with tricyclic antidepressants desipramine and imipramine: A structural chemistry perspective. J. Pharm. Sci. 2020, 109, 3086–3094. [Google Scholar] [CrossRef]
- Aree, T. Supramolecular Complexes of β-Cyclodextrin with Clomipramine and Doxepin: Effect of the Ring Substituent and Component of Drugs on Their Inclusion Topologies and Structural Flexibilities. Pharmaceuticals 2020, 13, 278. [Google Scholar] [CrossRef]
- Cremer, D.T.; Pople, J.A. General definition of ring puckering coordinates. J. Am. Chem. Soc. 1975, 97, 1354–1358. [Google Scholar] [CrossRef]
- French, A.D.; Johnson, G.P. Linkage and pyranosyl ring twisting in cyclodextrins. Carbohydr. Res. 2007, 342, 1223–1237. [Google Scholar] [CrossRef]
- Lindner, K.; Saenger, W. Crystal and molecular structure of cyclohepta-amylose dodecahydrate. Carbohydr. Res. 1982, 99, 103–115. [Google Scholar] [CrossRef]
- Aree, T.; Jongrungruangchok, S. Crystallographic evidence for β-cyclodextrin inclusion complexation facilitating the improvement of antioxidant activity of tea (+)-catechin and (−)-epicatechin. Carbohydr. Polym. 2016, 140, 362–373. [Google Scholar] [CrossRef]
- Saenger, W.; Betzel, C.; Hingerty, B.; Brown, G.M. Flip-flop hydrogen bonding in a partially disordered system. Nature 1982, 296, 581–583. [Google Scholar] [CrossRef]
- Terao, J. Dietary flavonoids as antioxidants. Food Fact. Health Promot. 2009, 61, 87–94. [Google Scholar] [CrossRef]
- Lucarini, M.; Pedulli, G.F.; Guerra, M. A Critical Evaluation of the Factors Determining the Effect of Intramolecular Hydrogen Bonding on the O–H Bond Dissociation Enthalpy of Catechol and of Flavonoid Antioxidants. Chem. Eur. J. 2004, 10, 933–939. [Google Scholar] [CrossRef]
- Koshland, D.E. Protein shape and biological control. Sci. Am. 1973, 229, 52–67. Available online: https://www.jstor.org/stable/24923220 (accessed on 12 May 2021). [CrossRef]
- Jeffrey, G.A.; Saenger, W. Hydrogen Bonding in Biological Structures; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge structural database. Acta Cryst. 2016, 72, 171–179. [Google Scholar] [CrossRef]
- Rose, P.W.; Prlić, A.; Bi, C.; Bluhm, W.F.; Christie, C.H.; Dutta, S.; Green, R.K.; Goodsell, D.S.; Westbrook, J.D.; Woo, J.; et al. The RCSB Protein Data Bank: Views of structural biology for basic and applied research and education. Nucleic Acids Res. 2015, 43, D345–D356. [Google Scholar] [CrossRef]
- Sarma, J.A.; Nagaraju, A.; Majumdar, K.K.; Samuel, P.M.; Das, I.; Roy, S.; McGhie, A.J. Solid state nuclear bromination with N-bromosuccinimide. Part 2. Experimental and theoretical studies of reactions with some substituted benzaldehydes. J. Chem. Soc. Perkin Trans. 2000, 2, 1119–1123. [Google Scholar] [CrossRef]
- Horneffer, V.; Dreisewerd, K.; Lüdemann, H.C.; Hillenkamp, F.; Läge, M.; Strupat, K. Is the incorporation of analytes into matrix crystals a prerequisite for matrix-assisted laser desorption/ionization mass spectrometry? A study of five positional isomers of dihydroxybenzoic acid. Int. J. Mass Spectrom. 1999, 185, 859–870. [Google Scholar] [CrossRef]
- Ng, S.W. A triclinic modification of 3,4-dihydroxybenzoic acid monohydrate. Acta Cryst. 2011, 67, o2476. [Google Scholar] [CrossRef]
- Sarma, B.; Sanphui, P.; Nangia, A. Polymorphism in isomeric dihydroxybenzoic acids. Cryst. Growth Des. 2010, 10, 2388–2399. [Google Scholar] [CrossRef]
- Borbulevych, O.Y.; Jankun, J.; Selman, S.H.; Skrzypczak-Jankun, E. Lipoxygenase interactions with natural flavonoid, quercetin, reveal a complex with protocatechuic acid in its X-ray structure at 2.1 Å resolution. Proteins: Struct. Funct. Bioinf. 2004, 54, 13–19. [Google Scholar] [CrossRef]
- Zheng, Y.; Haworth, I.S.; Zuo, Z.; Chow, M.S.; Chow, A.H. Physicochemical and structural characterization of quercetin-β-cyclodextrin complexes. J. Pharm. Sci. 2005, 94, 1079–1089. [Google Scholar] [CrossRef]
- Bruker. APEX2, SADABS and SHELXTL; Bruker AXS Inc.: Madison, WI, USA, 2014. [Google Scholar]
- Bruker. SAINT and XPREP; Bruker AXS Inc.: Madison, WI, USA, 2008. [Google Scholar]
- Aree, T.; Jongrungruangchok, S. Enhancement of antioxidant activity of green tea epicatechins in β-cyclodextrin cavity: Single-crystal X-ray analysis, DFT calculation and DPPH assay. Carbohydr. Polym. 2016, 151, 1139–1151. [Google Scholar] [CrossRef]
- Schnupf, U.; Momany, F.A. DFT energy optimization of a large carbohydrate: Cyclomaltohexaicosaose (CA-26). J. Phys. Chem. B 2011, 116, 6618–6627. [Google Scholar] [CrossRef]
- Allen, F.H.; Bruno, I.J. Bond lengths in organic and metal-organic compounds revisited: X—H bond lengths from neutron diffraction data. Acta Cryst. 2010, B66, 380–386. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, G.; Scalmani, V.; Barone, G.A.; Petersson, H.; Nakatsuji; et al. GAUSSIAN09, Revision A.01; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Salvatierra, D.; Jaime, C.; Virgili, A.; Sánchez-Ferrando, F. Determination of the inclusion geometry for the β-cyclodextrin/benzoic acid complex by NMR and molecular modeling. J. Org. Chem. 1996, 61, 9578–9581. [Google Scholar] [CrossRef]
- Aree, T.; Chaichit, N. Crystal structure of β-cyclodextrin–benzoic acid inclusion complex. Carbohydr. Res. 2003, 338, 439–446. [Google Scholar] [CrossRef]
- Aree, T.; Chaichit, N.; Engkakul, C. Polymorphism in β-cyclodextrin–benzoic acid inclusion complex: A kinetically controlled crystal growth according to the Ostwald’s rule. Carbohydr. Res. 2008, 343, 2451–2458. [Google Scholar] [CrossRef] [PubMed]








| PCAL (1) | PCAC (2) | HTY b | OLE b | CFA c | CGA c | |
|---|---|---|---|---|---|---|
| AR vs. β-CD-O4 interplanar angle [°] d | 74.9(1) | 71.5(1) | 85.0(1) | 59.2 | 87.9(7) | 89.3(1) |
| Moiety nearby/protruded from β-CD O6-side | OH | OH | OH | OH | COOH | QNA |
| Moiety enclosed in β-CD cavity | AR | AR | AR | AR | C=C−COOH | C=C−COO |
| No. of free OH e | ||||||
| In each polyphenol | 2 | 3 | 3 | 6 | 3 | 6 |
| Shielded in β-CD [dimeric] cavity | 2 | 3 | 0 | 0 | [3] | [6] |
| Distance from guest to β-CD [Å] | ||||||
| AR centroid to O4 centroid (diagonal) f | 0.837 | −0.779 | 0.990 | 1.016 | −3.987 | −4.450 |
| AR centroid to O4 plane (vertical) | 0.787 | −0.739 | 0.986 | 0.871 | −3.453 | −4.182 |
| Interaction | O−H | O⋅⋅⋅O | ∠(OHO) | Interaction | O−H | O⋅⋅⋅O | ∠(OHO) |
|---|---|---|---|---|---|---|---|
| X-ray | DFT c | ||||||
| β-CD⋅PCAL (1) | β-CD⋅PCAL d | ||||||
| O2L−H⋅⋅⋅O1L | 0.82 | 2.700(6) | 114.1 | O2L−H⋅⋅⋅O1L | 0.98 | 2.69 | 114.4 |
| O1L−H⋅⋅⋅O1W | 0.82 | 2.658(6) | 177.7 | O61−H⋅⋅⋅O2L | 0.97 | 2.95 | 164.7 |
| O2L−H⋅⋅⋅O34 i b | 0.82 | 2.874(6) | 166.0 | O33−H⋅⋅⋅O3L | 0.98 | 2.93 | 164.7 |
| O31−H⋅⋅⋅O3L ii | 0.82 | 2.842(6) | 134.4 | O67−H⋅⋅⋅O1L | 0.97 | 3.02 | 169.3 |
| O1L−H⋅⋅⋅O65 | 0.99 | 2.74 | 177.8 | ||||
| β-CD⋅PCAC (2) | β-CD⋅PCAC d | ||||||
| O1D−H⋅⋅⋅O2D | 0.82 | 2.703(5) | 108.7 | O1D−H⋅⋅⋅O2D | 0.99 | 2.69 | 108.7 |
| O1D−H⋅⋅⋅O1W | 0.82 | 2.705(5) | 168.9 | O65−H⋅⋅⋅O1D | 0.97 | 3.02 | 177.5 |
| O2D−H⋅⋅⋅O2W | 0.82 | 2.670(6) | 167.3 | O1D−H⋅⋅⋅O67 | 0.99 | 3.01 | 145.1 |
| O64−H⋅⋅⋅O3D iii | 0.82 | 2.937(6) | 167.4 | O2D−H⋅⋅⋅O61 | 0.99 | 2.78 | 161.9 |
| O67−H⋅⋅⋅O3D iv | 0.82 | 2.767(5) | 154.3 | O32−H⋅⋅⋅O3D | 0.98 | 2.90 | 169.4 |
| O4D−H⋅⋅⋅O3W v | 0.82 | 2.631(5) | 171.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aree, T. β-Cyclodextrin Inclusion Complexes with Catechol-Containing Antioxidants Protocatechuic Aldehyde and Protocatechuic Acid—An Atomistic Perspective on Structural and Thermodynamic Stabilities. Molecules 2021, 26, 3574. https://doi.org/10.3390/molecules26123574
Aree T. β-Cyclodextrin Inclusion Complexes with Catechol-Containing Antioxidants Protocatechuic Aldehyde and Protocatechuic Acid—An Atomistic Perspective on Structural and Thermodynamic Stabilities. Molecules. 2021; 26(12):3574. https://doi.org/10.3390/molecules26123574
Chicago/Turabian StyleAree, Thammarat. 2021. "β-Cyclodextrin Inclusion Complexes with Catechol-Containing Antioxidants Protocatechuic Aldehyde and Protocatechuic Acid—An Atomistic Perspective on Structural and Thermodynamic Stabilities" Molecules 26, no. 12: 3574. https://doi.org/10.3390/molecules26123574







