Immunocytochemical Localization of XRCC1 and γH2AX Foci Induced by Tightly Focused Femtosecond Laser Radiation in Cultured Human Cells
Abstract
:1. Introduction
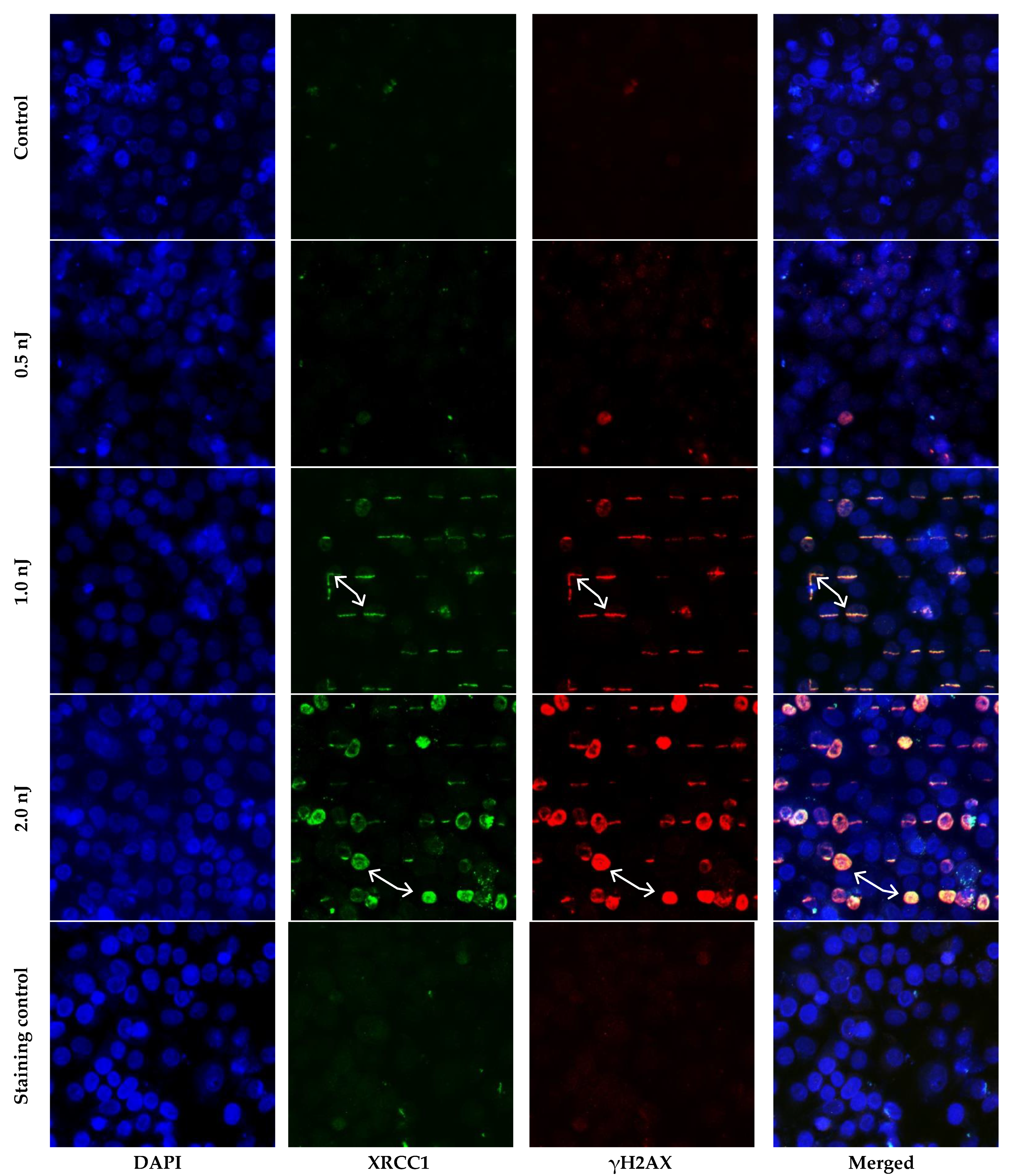
2. Results
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Culture Conditions
4.2. Irradiation of Cells
4.3. Immunocytochemical Analysis of DNA Double-Strand Break Localization
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Kuetemeyer, K.; Lucas-Hahn, A.; Petersen, B.; Lemme, E.; Hassel, P.; Niemann, H.; Heisterkamp, A. Combined multiphoton imaging and automated functional enucleation of porcine oocytes using femtosecond laser pulses. J. Biomed. Opt. 2010, 15, 046006. [Google Scholar] [CrossRef] [PubMed]
- Osychenko, A.; Zalessky, A.; Astafiev, A.; Shakhov, A.; Kostrov, A.; Krivokharchenko, A.; Nadtochenko, V. Femtosecond laser-induced blastomere fusion results in embryo tetraploidy by common metaphase plate formation. Exp. Cell Res. 2020, 389, 111887. [Google Scholar] [CrossRef] [PubMed]
- Vogel, A.; Noack, J.; Hüttman, G.; Paltauf, G. Mechanisms of femtosecond laser nanosurgery of cells and tissues. Appl. Phys. B 2005, 81, 1015–1047. [Google Scholar] [CrossRef]
- Babayan, N.; Vorobyeva, N.; Grigoryan, B.; Grekhova, A.; Pustovalova, M.; Rodneva, S.; Fedotov, Y.; Tsakanova, G.; Aroutiounian, R.; Osipov, A. Low Repair Capacity of DNA Double-Strand Breaks Induced by Laser-Driven Ultrashort Electron Beams in Cancer Cells. Int. J. Mol. Sci. 2020, 21, 9488. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.E.; Sunder, S. Double-strand breaks in motion: Implications for chromosomal rearrangement. Curr. Genet. 2020, 66, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Babayan, N.; Grigoryan, B.; Khondkaryan, L.; Tadevosyan, G.; Sarkisyan, N.; Grigoryan, R.; Apresyan, L.; Aroutiounian, R.; Vorobyeva, N.; Pustovalova, M.; et al. Laser-Driven Ultrashort Pulsed Electron Beam Radiation at Doses of 0.5 and 1.0 Gy Induces Apoptosis in Human Fibroblasts. Int. J. Mol. Sci. 2019, 20, 5140. [Google Scholar] [CrossRef]
- Ulyanenko, S.; Pustovalova, M.; Koryakin, S.; Beketov, E.; Lychagin, A.; Ulyanenko, L.; Kaprin, A.; Grekhova, A.; Ozerova, A.M.; Ozerov, I.V.; et al. Formation of gammaH2AX and pATM Foci in Human Mesenchymal Stem Cells Exposed to Low Dose-Rate Gamma-Radiation. Int. J. Mol. Sci. 2019, 20, 2645. [Google Scholar] [CrossRef]
- Osipov, A.N.; Pustovalova, M.; Grekhova, A.; Eremin, P.; Vorobyova, N.; Pulin, A.; Zhavoronkov, A.; Roumiantsev, S.; Klokov, D.Y.; Eremin, I. Low doses of X-rays induce prolonged and ATM-independent persistence of γH2AX foci in human gingival mesenchymal stem cells. Oncotarget 2015, 6, 27275–27287. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.; Haber, J.E. Sources of DNA double-strand breaks and models of recombinational DNA repair. Cold Spring Harb. Perspect. Biol. 2014, 6, a016428. [Google Scholar] [CrossRef]
- Abbotts, R.; Wilson, D.M., 3rd. Coordination of DNA single strand break repair. Free Radic. Biol. Med. 2017, 107, 228–244. [Google Scholar] [CrossRef]
- Nuzzo, V.; Maxwell, I.; Chung, S.; Mazur, E.; Heisterkamp, A. Subcellular Surgery and Nanoneurosurgery Using Femtosecond Laser Pulses. In Biophotonics: Spectroscopy, Imaging, Sensing, and Manipulation; Springer: Dordrecht, The Netherlands, 2011; pp. 203–218. [Google Scholar] [CrossRef]
- Osychenko, A.A.; Zalesskii, A.D.; Krivokharchenko, A.S.; Zhakhbazyan, A.K.; Ryabova, A.V.; Nadtochenko, V.A. Fusion of blastomeres in mouse embryos under the action of femtosecond laser radiation. Efficiency of blastocyst formation and embryo development. Quantum Electron. 2015, 45, 498–502. [Google Scholar] [CrossRef]
- Osychenko, A.A.; Tochilo, U.A.; Astafiev, A.A.; Zalessky, A.D.; Shakhov, A.M.; Krivokharchenko, A.S.; Nadtochenko, V.A. Determining the Range of Noninvasive Near-Infrared Femtosecond Laser Pulses for Mammalian Oocyte Nanosurgery. Sovrem. Tehnol. Med. 2017, 9. [Google Scholar] [CrossRef]
- Botchway, S.W.; Reynolds, P.; Parker, A.W.; O’Neill, P. Laser-Induced Radiation Microbeam Technology and Simultaneous Real-Time Fluorescence Imaging in Live Cells. In Imaging and Spectroscopic Analysis of Living Cells-Optical and Spectroscopic Techniques; Academic Press: Cambridge, MA, USA, 2012; pp. 3–28. [Google Scholar] [CrossRef]
- Harper, J.V.; Reynolds, P.; Leatherbarrow, E.L.; Botchway, S.W.; Parker, A.W.; O’Neill, P. Induction of Persistent Double Strand Breaks Following Multiphoton Irradiation of Cycling and G1-arrested Mammalian Cells-Replication-induced Double Strand Breaks. Photochem. Photobiol. 2008, 84, 1506–1514. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Zhang, Y.; Wang, J.; Zhang, X.; Gao, Y.; Yin, L.; Li, Q.; Li, J.; Chen, H. Induction and inhibition of the pan-nuclear gamma-H2AX response in resting human peripheral blood lymphocytes after X-ray irradiation. Cell Death Discov. 2016, 2, 16011. [Google Scholar] [CrossRef]
- Moeglin, E.; Desplancq, D.; Conic, S.; Oulad-Abdelghani, M.; Stoessel, A.; Chiper, M.; Vigneron, M.; Didier, P.; Tora, L.; Weiss, E. Uniform Widespread Nuclear Phosphorylation of Histone H2AX Is an Indicator of Lethal DNA Replication Stress. Cancers 2019, 11, 355. [Google Scholar] [CrossRef]
- Venugopalan, V.; Guerra, A.; Nahen, K.; Vogel, A. Role of Laser-Induced Plasma Formation in Pulsed Cellular Microsurgery and Micromanipulation. Phys. Rev. Lett. 2002, 88, 078103. [Google Scholar] [CrossRef]
- Vogel, A.; Venugopalan, V. Mechanisms of Pulsed Laser Ablation of Biological Tissues. Chem. Rev. 2003, 103, 577–644. [Google Scholar] [CrossRef] [PubMed]
- Hammer, D.X.; Thomas, R.J.; Noojin, G.D.; Rockwell, B.A.; Kennedy, P.K.; Roach, W.P. Experimental investigation of ultrashort pulse laser-induced breakdown thresholds in aqueous media. IEEE J. Quantum Electron. 1996, 32, 670–678. [Google Scholar] [CrossRef]
- Kudryashov, S.I.; Zvorykin, V.D. Microscale nanosecond laser-induced optical breakdown in water. Phys. Rev. E 2008, 78, 036404. [Google Scholar] [CrossRef]
- Oraevsky, A.A.; Da Silva, L.B.; Rubenchik, A.M.; Feit, M.D.; Glinsky, M.E.; Perry, M.D.; Mammini, B.M.; Small, W.; Stuart, B.C. Plasma mediated ablation of biological tissues with nanosecond-to-femtosecond laser pulses: Relative role of linear and nonlinear absorption. IEEE J. Quantum Electron. 1996, 2, 801–809. [Google Scholar] [CrossRef]
- Sarpe, C.; Köhler, J.; Winkler, T.; Wollenhaupt, M.; Baumert, T. Real-time observation of transient electron density in water irradiated with tailored femtosecond laser pulses. New J. Phys. 2012, 14, 075021. [Google Scholar] [CrossRef]
- Stuart, B.C.; Feit, M.D.; Herman, S.; Rubenchik, A.M.; Shore, B.W.; Perry, M.D. Nanosecond-to-femtosecond laser-induced breakdown in dielectrics. Phys. Rev. B 1996, 53, 1749–1761. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.F.; Zhang, P.; Nunes, F.B.; Jordan, I.; von Conta, A.; Wörner, H.J. Ionization Energy of Liquid Water Revisited. J. Phys. Chem. Lett. 2020, 11, 1789–1794. [Google Scholar] [CrossRef] [PubMed]
- Fernando, H.; Papadantonakis, G.A.; Kim, N.S.; LeBreton, P.R. Conduction-band-edge ionization thresholds of DNA components in aqueous solution. Proc. Natl. Acad. Sci. USA 1998, 95, 5550–5555. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.K. A first-order model for computation of laser-induced breakdown thresholds in ocular and aqueous media. I. Theory. IEEE J. Quantum Electron. 1995, 31, 2241–2249. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, Q.; Li, D.; Xu, S.; Cao, P.; Zhou, Y.; Cao, W.; Lu, P. Identification of tunneling and multiphoton ionization in intermediate Keldysh parameter regime. Optics Express 2019, 27, 6471. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.H.; Sun, J.; Longtin, J.P. Breakdown threshold and localized electron density in water induced by ultrashort laser pulses. J. Appl. Phys. 2002, 91, 2530–2536. [Google Scholar] [CrossRef]
- Lenzner, M.; Krüger, J.; Sartania, S.; Cheng, Z.; Spielmann, C.; Mourou, G.; Kautek, W.; Krausz, F. Femtosecond Optical Breakdown in Dielectrics. Phys. Rev. Lett. 1998, 80, 4076–4079. [Google Scholar] [CrossRef]
- Boudaiffa, B.; Cloutier, P.; Hunting, D.; Huels, M.A.; Sanche, L. Resonant formation of DNA strand breaks by low-energy (3 to 20 eV) electrons. Science 2000, 287, 1658–1660. [Google Scholar] [CrossRef]
- Gohlke, S.; Illenberger, E. Probing biomolecules: Gas phase experiments and biological relevance. Europhys. News 2002, 33, 207–209. [Google Scholar] [CrossRef]
- Huels, M.A.; Boudaïffa, B.; Cloutier, P.; Hunting, D.; Sanche, L. Single, Double, and Multiple Double Strand Breaks Induced in DNA by 3−100 eV Electrons. J. Am. Chem. Soc. 2003, 125, 4467–4477. [Google Scholar] [CrossRef] [PubMed]
- Chin, S.L.; Hosseini, S.A.; Liu, W.; Luo, Q.; Théberge, F.; Aközbek, N.; Becker, A.; Kandidov, V.P.; Kosareva, O.G.; Schroeder, H. The propagation of powerful femtosecond laser pulses in opticalmedia: Physics, applications, and new challenges. Can. J. Phys. 2005, 83, 863–905. [Google Scholar] [CrossRef]
- König, K. Review: Clinical in vivo multiphoton FLIM tomography. Methods Appl. Fluoresc. 2020, 8, 034002. [Google Scholar] [CrossRef] [PubMed]
- Müller, D.; Hagenah, D.; Biswanath, S.; Coffee, M.; Kampmann, A.; Zweigerdt, R.; Heisterkamp, A.; Kalies, S.M.K. Femtosecond laser-based nanosurgery reveals the endogenous regeneration of single Z-discs including physiological consequences for cardiomyocytes. Sci. Rep. 2019, 9, 3625. [Google Scholar] [CrossRef]
- Chung, S.; Nuzzo, V.; Mazur, E. Nanosurgery with Femtosecond Lasers. In Proceedings of the Frontiers in Optics 2009/Laser Science XXV/Fall 2009 OSA Optics & Photonics Technical Digest, San Jose, CA, USA, 11–15 October 2009. [Google Scholar]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zalessky, A.; Fedotov, Y.; Yashkina, E.; Nadtochenko, V.; Osipov, A.N. Immunocytochemical Localization of XRCC1 and γH2AX Foci Induced by Tightly Focused Femtosecond Laser Radiation in Cultured Human Cells. Molecules 2021, 26, 4027. https://doi.org/10.3390/molecules26134027
Zalessky A, Fedotov Y, Yashkina E, Nadtochenko V, Osipov AN. Immunocytochemical Localization of XRCC1 and γH2AX Foci Induced by Tightly Focused Femtosecond Laser Radiation in Cultured Human Cells. Molecules. 2021; 26(13):4027. https://doi.org/10.3390/molecules26134027
Chicago/Turabian StyleZalessky, Alexandr, Yuriy Fedotov, Elizaveta Yashkina, Viktor Nadtochenko, and Andreyan N. Osipov. 2021. "Immunocytochemical Localization of XRCC1 and γH2AX Foci Induced by Tightly Focused Femtosecond Laser Radiation in Cultured Human Cells" Molecules 26, no. 13: 4027. https://doi.org/10.3390/molecules26134027






