The Valence-Bond (VB) Model and Its Intimate Relationship to the Symmetric or Permutation Group
Abstract
1. Introduction
2. The VB Model and the Symmetric Group
total wave function spatial part spin part
- (a)
- The total wave function must be a product of a spatial and a spin part;
- (b)
- Both the spatial and spin parts must independently exhibit permutation symmetry, i.e., must transform like the irreducible representations of the symmetric (SN) group;
- (c)
- The total wave function must be anti-symmetric.
χ1 = (αβ − βα) (αβ − βα)
χ2 = 2ααββ + 2ββαα − (αβ + αβ) (αβ + βα).
3. Results and Discussion
3.1. Molecular Structure and Chemical Structure
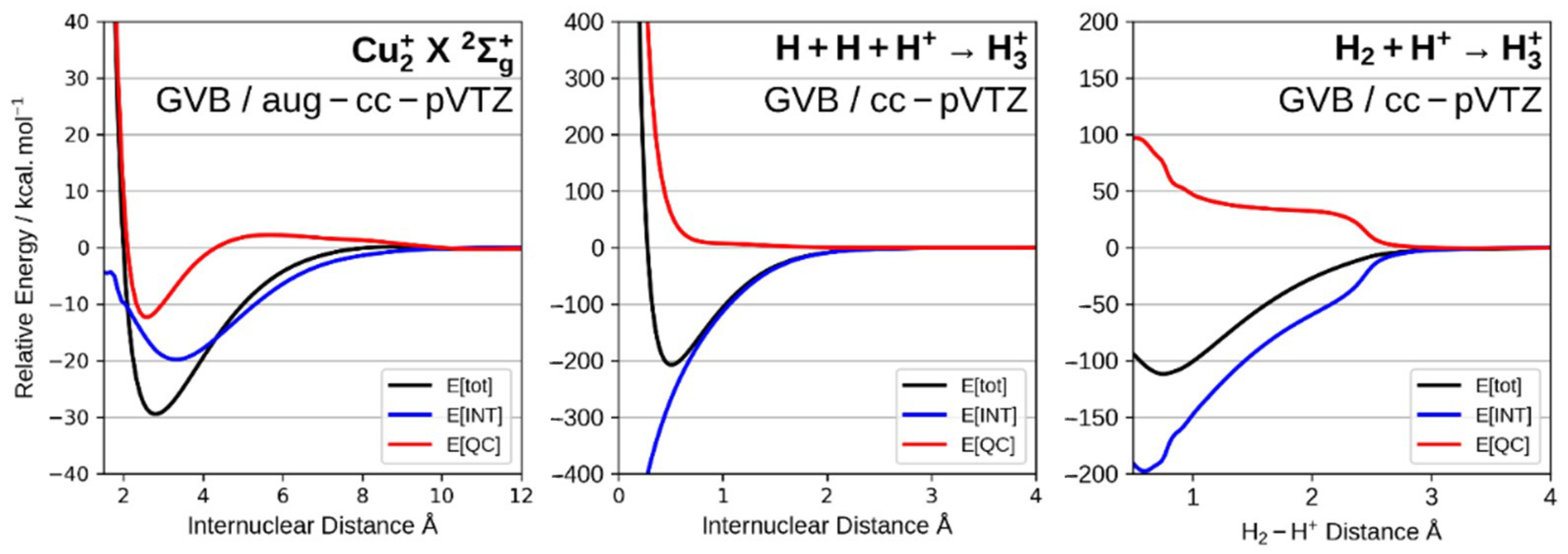
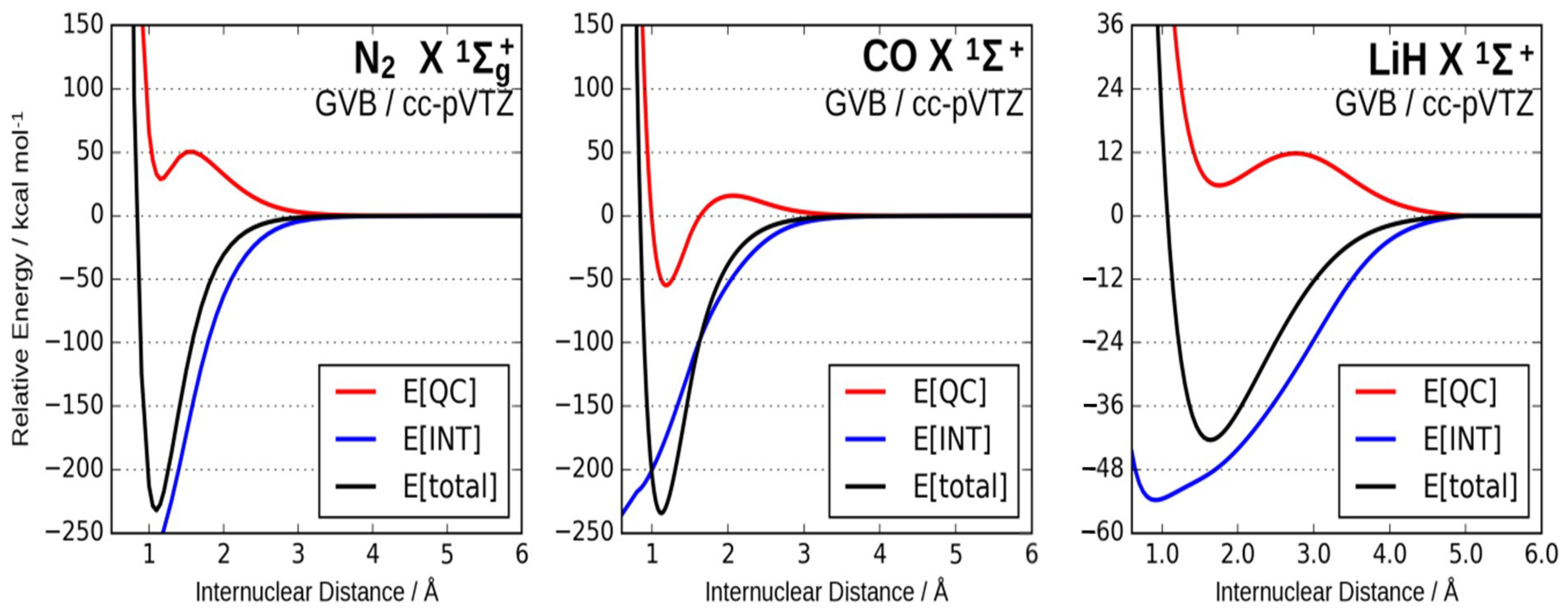
3.2. The Nature of the Chemical Bond. Quantum Interference
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Pauling, L. Fifty Years of Progress in Structural Chemistry and Molecular Biology. Daedalus 1970, 99, 998–1014. [Google Scholar]
- Pauling, L. The Theoretical Prediction of the Physical Properties of Many-Electron Atoms and Ions: Mole Refraction, Diamagnetic Susceptibility, and Extension in Space. Proc. Roy. Soc. Lond. A 1927, 114, 181–211. [Google Scholar]
- Heitler, W.; London, F. Wechselwirkung neutraler Atome und homopolare Bindung nach der Quantenmechanik. Z. Physik. 1927, 44, 455–472. [Google Scholar] [CrossRef]
- Pauling, L. The shared-electron chemical bond. Proc. Natl. Acad. Sci. USA 1928, 14, 359–362. [Google Scholar] [CrossRef]
- Pauling, L. The application of the quantum mechanics to the structure of the hydrogen molecule and hydrogen molecule-ion and to related problems. Chem. Rev. 1928, 5, 173–213. [Google Scholar] [CrossRef]
- Pauling, L. Quantum mechanics and the chemical bond. Phys. Rev. 1931, 37, 1185–1186. [Google Scholar] [CrossRef]
- Pauling, L. The nature of the chemical bond. Application of results obtained from the quantum mechanics and from a theory of paramagnetic susceptibility to the structure of molecules. J. Am. Chem. Soc. 1931, 53, 1367–1400. [Google Scholar] [CrossRef]
- Pauling, L. The nature of the chemical bond II. The one-electron bond and the three-electron bond. J. Am. Chem. Soc. 1931, 53, 3225–3237. [Google Scholar] [CrossRef]
- Pauling, L. The nature of the chemical bond III. The transition from one extreme bond type to another. J. Am. Chem. Soc. 1932, 54, 988–1003. [Google Scholar] [CrossRef]
- Pauling, L. The nature of the chemical bond. IV. The energy of single bonds and the relative electronegativity of atoms. J. Am. Chem. Soc. 1932, 54, 3570–3582. [Google Scholar] [CrossRef]
- Pauling, L.; Wheland, W. The nature of the chemical bond. V. The quantum-mechanical calculation of the resonance energy of benzene and naphthalene and the hydrocarbon free radicals. J. Chem. Phys. 1933, 1, 362–374. [Google Scholar] [CrossRef]
- Pauling, L.; Sherman, J. The nature of the chemical bond. VI. The calculation from thermochemical data of the energy of resonance of molecules among several electronic structures. J. Chem. Phys. 1933, 1, 606–617. [Google Scholar] [CrossRef]
- Pauling, L.; Sherman, J. The nature of the chemical bond. VII. The calculation of resonance energy in conjugated systems. J. Chem. Phys. 1933, 1, 679–686. [Google Scholar] [CrossRef]
- Heisenberg, W. Mehrkörpenproblem und Resonanz in der Quantumechanik. Z. Phys. 1926, 38, 411–426. [Google Scholar] [CrossRef]
- Heisenberg, W. Über die Spektra von Atomsystemen mit zwei Elektronen. Z. Phys. 1926, 39, 499–518. [Google Scholar] [CrossRef]
- Heisenberg, W. Mehrkörpenproblem und Resonanz in der Quantumechanik II. Z. Phys. 1926, 41, 239–267. [Google Scholar] [CrossRef]
- Wigner, E. Über nicht kombinierende Terme in der neueren Quantumtheory. I Teil. Z. Phys. 1927, 40, 492–500. [Google Scholar] [CrossRef]
- Wigner, E. Über nicht kombinierende Terme in der neueren Quantumtheory. II Teil. Z. Phys. 1927, 40, 833–892. [Google Scholar]
- Slater, J.C. The Theory of Complex Spectra. Phys. Rev. 1929, 34, 1293–1322. [Google Scholar] [CrossRef]
- Nascimento, M.A.C. The consequences of neglecting permutation symmetry in the description of many-electrons systems. Int. J. Quantum Chem. 2018, e25765. [Google Scholar] [CrossRef]
- Goddard, W.A., III. Improved Quantum Theory of Many-Electron Systems. I. Construction of Eigenfunctions of which Satisfy Pauli’s Principle. Phys. Rev. 1967, 157, 73–80. [Google Scholar] [CrossRef]
- Goddard, W.A., III. Improved Quantum Theory of many-electron systems. II. The basic method. Phys. Rev. 1967, 157, 81–93. [Google Scholar] [CrossRef]
- Gerratt, J.; Lipscomb, W.N. Spin-Coupled Wave Functions for Atoms and Molecules. Proc. Natl. Acad. Sci. USA 1968, 59, 332–335. [Google Scholar] [CrossRef][Green Version]
- Born, M.; Oppenheimer, J.R. Zur Quantentheorie der Molekeln. Ann. Phys. 1927, 84, 457–484. [Google Scholar] [CrossRef]
- Coulson, C.A. The electronic structure of some polyenes and aromatic molecules. VII, Bonds of fractional order by the molecular orbital method. Proc. R. Soc. Lond. A 1939, 169, 413–428. [Google Scholar]
- Mulliken, R.S. Electronic population analysis on LCAO-MO molecular wave functions I. J. Chem. Phys. 1955, 23, 1833–1840. [Google Scholar] [CrossRef]
- Hurley, A.C. The molecular orbital theory of valency. Proc. R. Soc. Lond. A 1953, 216, 424–433. [Google Scholar]
- Lennard-Jones, J. The molecular orbital theory of chemical valency I. The determination of molecular orbitals. Proc. R. Soc. Lond. A 1949, 198, 1–13. [Google Scholar]
- Hall, G.G.; Lennard-Jones, J. The molecular orbital theory of chemical valency III. Properties of molecular orbitals. Proc. R. Soc. Lond. A 1950, 202, 166–180. [Google Scholar]
- Edmiston, C.; Ruedenberg, K. Localized atomic and molecular orbitals. Rev. Mod. Phys. 1963, 35, 457. [Google Scholar] [CrossRef]
- Weinhold, F.; Landis, C.R. Valency and Bonding: A Natural Bond Orbital Donor−Acceptor Perspective; Cambridge University Press: New York, NY, USA, 2005. [Google Scholar]
- Ruedenberg, K. The physical nature of the chemical bond. Rev. Mod. Phys. 1962, 34, 326–376. [Google Scholar] [CrossRef]
- Goddard, W.A.; Wilson, C.W. The role of kinetic energy in chemical binding. Theor. Chim. Acta 1972, 26, 211–230. [Google Scholar] [CrossRef]
- Wilson, C.A.; Goddard, W.A. The role of kinetic energy in chemical binding II. Theor. Chim. Acta 1972, 26, 195–210. [Google Scholar] [CrossRef]
- Kutzelnigg, W. Theoretical Models of Chemical Bonding: Part. 2; Maksic, Z.B., Ed.; Springer: Berlin, Germany, 1990; pp. 1–43. [Google Scholar]
- Kutzelnigg, W. The Physical Mechanism of the Chemical Bond. Angew. Chem. Int. Ed. Engl. 1973, 12, 546–562. [Google Scholar] [CrossRef]
- Feinberg, M.J.; Ruedenberg, K. Paradoxical Role of the Kinetic-Energy Operator in the Formation of the Covalent Bond. J. Chem. Phys. 1971, 54, 1495–1511. [Google Scholar] [CrossRef]
- Ruedenberg, K.; Schmidt, M.W.; Gilbert, M.M.; Elbert, S.T. Are Atoms Intrinsic to Molecular Electronic Wavefunctions? I. The FORS Model. Chem. Phys. 1982, 71, 41–49. [Google Scholar] [CrossRef]
- Lu, W.C.; Wang, C.Z.; Schmidt, M.W.; Bytautas, L.; Ho, K.M.; Ruedenberg, K. Molecule Intrinsic Minimal Basis Sets. I. Exact Resolution of Ab Initio Optimized Molecular Orbitals in Terms of Deformed Atomic Minimal-Basis Orbitals. J. Chem. Phys. 2004, 120, 2629–2637. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.C.; Wang, C.Z.; Schmidt, M.W.; Bytautas, L.; Ho, K.M.; Ruedenberg, K. Molecule Intrinsic Minimal Basis Sets. II. Bonding Analyses for Si4H6 and Si2 to Si10. J. Chem. Phys. 2004, 120, 2638–2651. [Google Scholar] [CrossRef]
- Ruedenberg, K.; Schmidt, M.W. Why Does Electron Sharing Lead to Covalent Bonding? A Variational Analysis. J. Comput. Chem. 2007, 28, 391–410. [Google Scholar] [CrossRef]
- Ivanic, J.; Atchity, G.J.; Ruedenberg, K.; Ivanic, J.; Atchity, G.J.; Ruedenberg, K. Intrinsic Local Constituents of Molecular Electronic Wave Functions. I. Exact Representation of the Density Matrix in Terms of Chemically Deformed and Oriented Atomic Minimal Basis Set Orbitals. Theor. Chem Acc. 2008, 120, 281–294. [Google Scholar] [CrossRef]
- West, A.C.; Schmidt, M.W.; Gordon, M.S.; Ruedenberg, K. A Comprehensive Analysis of Molecule-Intrinsic Quasi-Atomic, Bonding, and Correlating Orbitals. I. Hartree-Fock Wave Functions. J. Chem. Phys. 2013, 139, 234107. [Google Scholar] [CrossRef]
- Schmidt, M.W.; Ivanic, J.; Ruedenberg, K. Covalent Bonds Are Created by the Drive of Electron Waves to Lower Their Kinetic Energy through Expansion. J. Chem. Phys. 2014, 140, 204104. [Google Scholar] [CrossRef]
- West, A.C.; Schmidt, M.W.; Gordon, M.S.; Ruedenberg, K. A Comprehensive Analysis in Terms of Molecule-Intrinsic, Quasi-Atomic Orbitals. III. the Covalent Bonding Structure of Urea. J. Phys. Chem. A 2015, 119, 10368–10375. [Google Scholar] [CrossRef] [PubMed]
- West, A.C.; Duchimaza-Heredia, J.J.; Gordon, M.S.; Ruedenberg, K. Identification and Characterization of Molecular Bonding Structures by Ab Initio Quasi-Atomic Orbital Analyses. J. Phys. Chem. A 2017, 121, 8884–8898. [Google Scholar] [CrossRef] [PubMed]
- Duchimaza Heredia, J.J.; Sadow, A.D.; Gordon, M.S. A Quasi-Atomic Analysis of Three-Center Two-Electron Zr-H-Si Interactions. J. Phys. Chem. A 2018, 122, 9653–9669. [Google Scholar] [CrossRef]
- Schoendorff, G.; Schmidt, M.W.; Ruedenberg, K.; Gordon, M.S. Quasi-Atomic Bond Analyses in the Sixth Period: II. Bond Analyses of Cerium Oxides. J. Phys. Chem. A 2019. [Google Scholar] [CrossRef] [PubMed]
- Guidez, E.B.; Gordon, M.S.; Ruedenberg, K. Why Is Si2H2 Not Linear? An Intrinsic Quasi-Atomic Bonding Analysis. J. Am. Chem. Soc. 2020, 142, 13729–13742. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, T.M.; Nascimento, M.A.C. Energy Partitioning for Generalized Product Functions: The Interference Contribution to the Energy of Generalized Valence Bond and Spin Coupled Wave Functions. J. Chem. Phys. 2009, 130, 104102. [Google Scholar] [CrossRef]
- Cardozo, T.M.; Nascimento, M.A.C. Chemical Bonding in the N2 Molecule and the Role of the Quantum Mechanical Interference Effect. J. Phys. Chem. A 2009, 113, 12541–12548. [Google Scholar] [CrossRef]
- De Sousa, D.W.O.; Nascimento, M.A.C. Are One-Electron Bonds Any Different from Standard Two-Electron Covalent Bonds? Acc. Chem. Res. 2017, 50, 2264–2272. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, D.W.O.; Nascimento, M.A.C. One-Electron Bonds Are Not “Half-Bonds”. Phys. Chem. Chem. Phys. 2019, 21, 13319–13336. [Google Scholar] [CrossRef] [PubMed]
- Fantuzzi, F.; de Sousa, D.W.O.; Nascimento, M.A.C. The Nature of the Chemical Bond from a Quantum Mechanical Interference Perspective. Chem. Sel. 2017, 2, 604–619. [Google Scholar] [CrossRef]
- Cardozo, T.M.; Fantuzzi, F.; Nascimento, M.A.C. The Non-Covalent Nature of the Molecular Structure of the Benzene Molecule. Phys. Chem. Chem. Phys. 2014, 16, 11024. [Google Scholar] [CrossRef]
- Fantuzzi, F.; Cardozo, T.M.; Nascimento, M.A.C. Nature of the Chemical Bond and Origin of the Inverted Dipole Moment in Boron Fluoride: A Generalized Valence Bond Approach. J. Phys. Chem. A 2015, 119, 5335–5343. [Google Scholar] [CrossRef]
- De Sousa, D.W.O.; Nascimento, M.A.C. Is There a Quadruple Bond in C2? J. Chem. Theory Comput. 2016, 12, 2234–2241. [Google Scholar] [CrossRef]
- Fantuzzi, F.; Cardozo, T.M.; Nascimento, M.A.C. The Nature of the Singlet and Triplet States of Cyclobutadiene as Revealed by Quantum Interference. Chem. Phys. Chem. 2016, 17, 288–295. [Google Scholar] [CrossRef]
- Fantuzzi, F.; de Sousa, D.W.O.; Nascimento, M.A.C. Chemical Bonding in the Pentagonal-Pyramidal Benzene Dication and Analogous Isoelectronic Hexa-Coordinate Species. Comput. Theor. Chem. 2017, 1116, 225–233. [Google Scholar] [CrossRef]
- Fantuzzi, F.; Cardozo, T.M.; Nascimento, M.A.C. On the Metastability of Doubly Charged Homonuclear Diatomics. Phys. Chem. Chem. Phys. 2017, 19, 19352–19359. [Google Scholar] [CrossRef]
- De Sousa, D.W.O.; Nascimento, M.A.C. Quantum Interference Contribution to the Dipole Moment of Diatomic Molecules. J. Phys. Chem. A 2018, 122, 1406–1412. [Google Scholar] [CrossRef]
- Fantuzzi, F.; Wolff, W.; Quitián-Lara, H.M.; Boechat-Roberty, H.M.; Hilgers, G.; Rudek, B.; Nascimento, M.A.C. Unexpected Reversal of Stability in Strained Systems Containing One-Electron Bonds. Phys. Chem. Chem. Phys. 2019, 21, 24984–24992. [Google Scholar] [CrossRef]
- Fantuzzi, F.; Nascimento, M.A.C. Description of Polar Chemical Bonds from the Quantum Mechanical Interference Perspective. J. Chem. Theory Comput. 2014, 10, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Lipscomb, W. The boranes and their relatives. Science 1977, 196, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, B.; Nethercot, A.; Townes, C. Isotopic Mass Ratios, Magnetic Moments and the Sign of the Electric Dipole Moment in Carbon Monoxide. Phys. Rev. 1958, 109, 400. [Google Scholar] [CrossRef]
- Muenter, J.S. Electric Dipole Moment of Carbon Monoxide. J. Mol. Spectrosc. 1975, 55, 490. [Google Scholar] [CrossRef]
- Wharton, L.; Gold, L.P.; Klemperer, W. Dipole Moment of Lithium Hydride. J. Chem. Phys. 1960, 33, 1255. [Google Scholar] [CrossRef]
- Johnson, R.D., III. NIST Computational Chemistry Comparison and Benchmark Database. NIST Standard Reference, Database Number 101, Release 16ª. 2013. Available online: http://cccbdb.nist.gov/ (accessed on 8 February 2014).
- Thomson, R.; Dalby, F.W. An Experimental Determination of the Dipole Moments of the X(1Σ) and A(1Π) States of the BH Molecule. Can. J. Phys. 1969, 47, 1155. [Google Scholar] [CrossRef]
- Phelps, D.; Dalby, F. Experimental Determination of the Electric Dipole Moment of the Ground Electronic State of CH. Phys. Rev. Lett. 1966, 16, 3. [Google Scholar] [CrossRef]
- Powell, F.X.; Lide, D.R. Improved Measurement of the Electric-Dipole Moment of the Hydroxyl Radical. J. Chem. Phys. 1965, 42, 4201. [Google Scholar] [CrossRef]
- Weiss, R. Stark Effect and Hyperfine Structure of Hydrogen Fluoride. Phys. Rev. 1963, 131, 659. [Google Scholar] [CrossRef]

| |ΔXAB| | (μexp/D) a | |
|---|---|---|
| LiH (1Σ+) | 1.1 | −5.882 |
| BeH(2Σ+) | 0.6 | −0.228 |
| BH (1Σ+) | 0.2 | 1.270 |
| CH (2Π) | 0.4 | 1.460 |
| NH (3Σ−) | 0.9 | 1.539 |
| OH (2Π) | 1.4 | 1.660 |
| HF (1Σ+) | 1.9 | −1.820 |
| CO | 1.0 | 0.120 |
| LiF | 3.0 | −6.325 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento, M.A.C. The Valence-Bond (VB) Model and Its Intimate Relationship to the Symmetric or Permutation Group. Molecules 2021, 26, 4524. https://doi.org/10.3390/molecules26154524
Nascimento MAC. The Valence-Bond (VB) Model and Its Intimate Relationship to the Symmetric or Permutation Group. Molecules. 2021; 26(15):4524. https://doi.org/10.3390/molecules26154524
Chicago/Turabian StyleNascimento, Marco Antonio Chaer. 2021. "The Valence-Bond (VB) Model and Its Intimate Relationship to the Symmetric or Permutation Group" Molecules 26, no. 15: 4524. https://doi.org/10.3390/molecules26154524
APA StyleNascimento, M. A. C. (2021). The Valence-Bond (VB) Model and Its Intimate Relationship to the Symmetric or Permutation Group. Molecules, 26(15), 4524. https://doi.org/10.3390/molecules26154524





