Inhibitory Activity and Mechanism Investigation of Hypericin as a Novel α-Glucosidase Inhibitor
Abstract
:1. Introduction
2. Results and Discussion
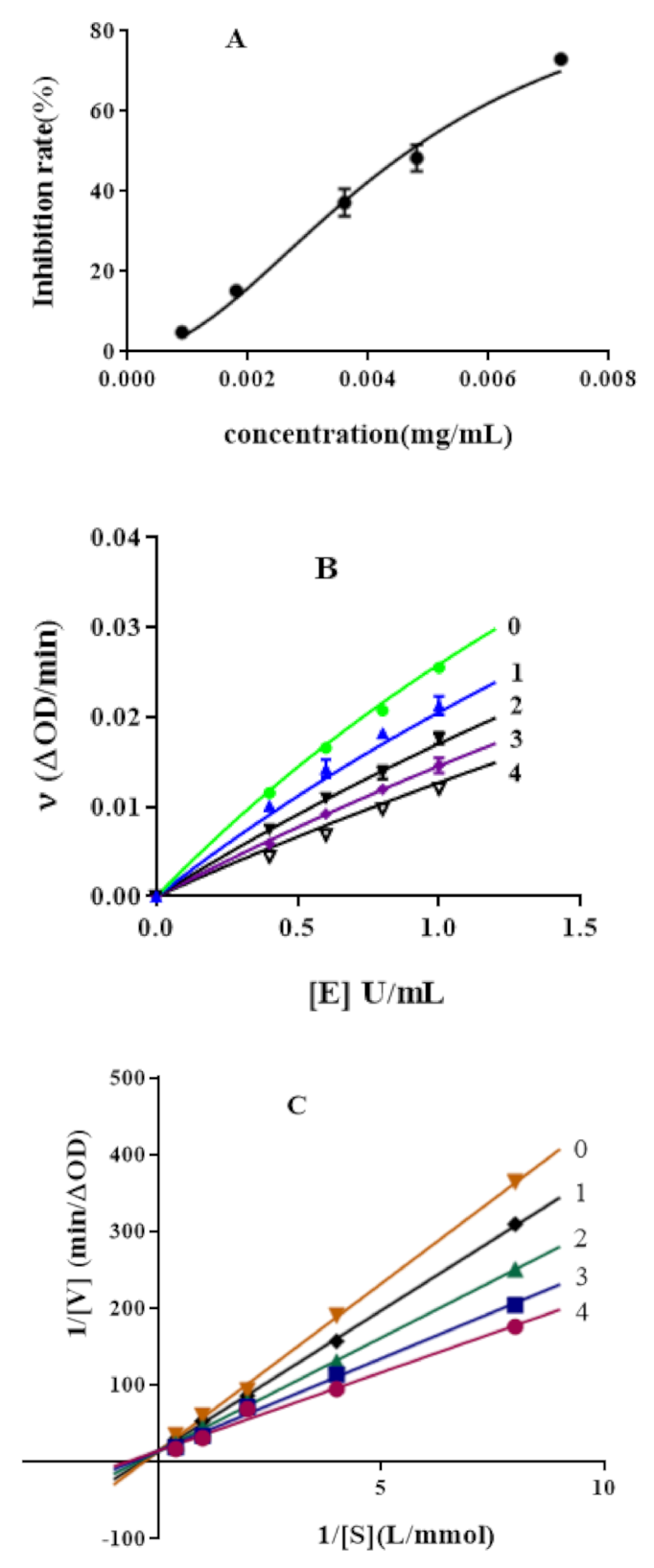
2.1. Inhibition of α-Glucosidase by Hypericin and Acarbose
2.2. Inhibitory Mechanism of Hypericin on α-Glucosidase Activity
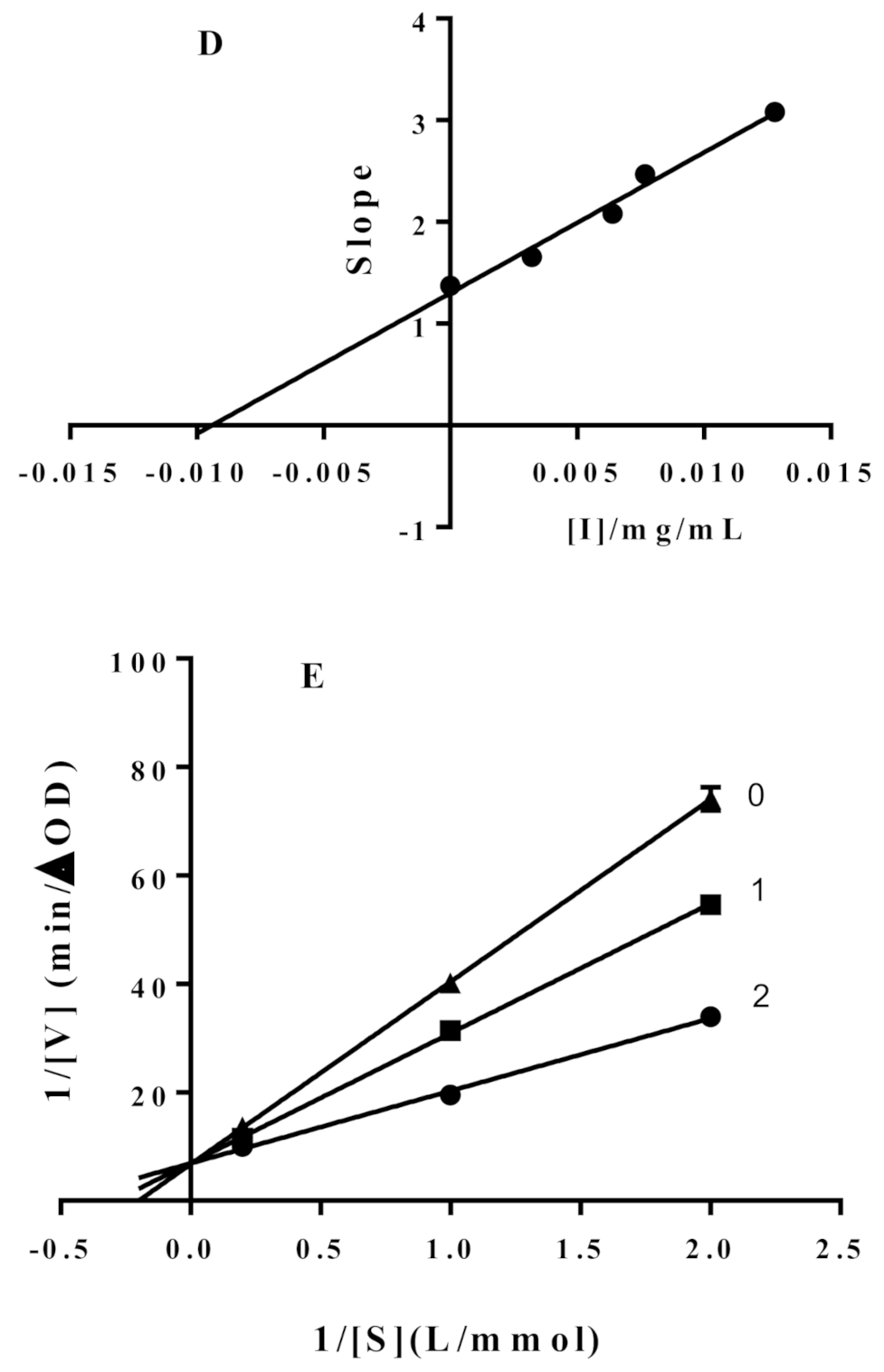
2.3. Enzymatic Kinetics of α-Glucosidase Inhibition
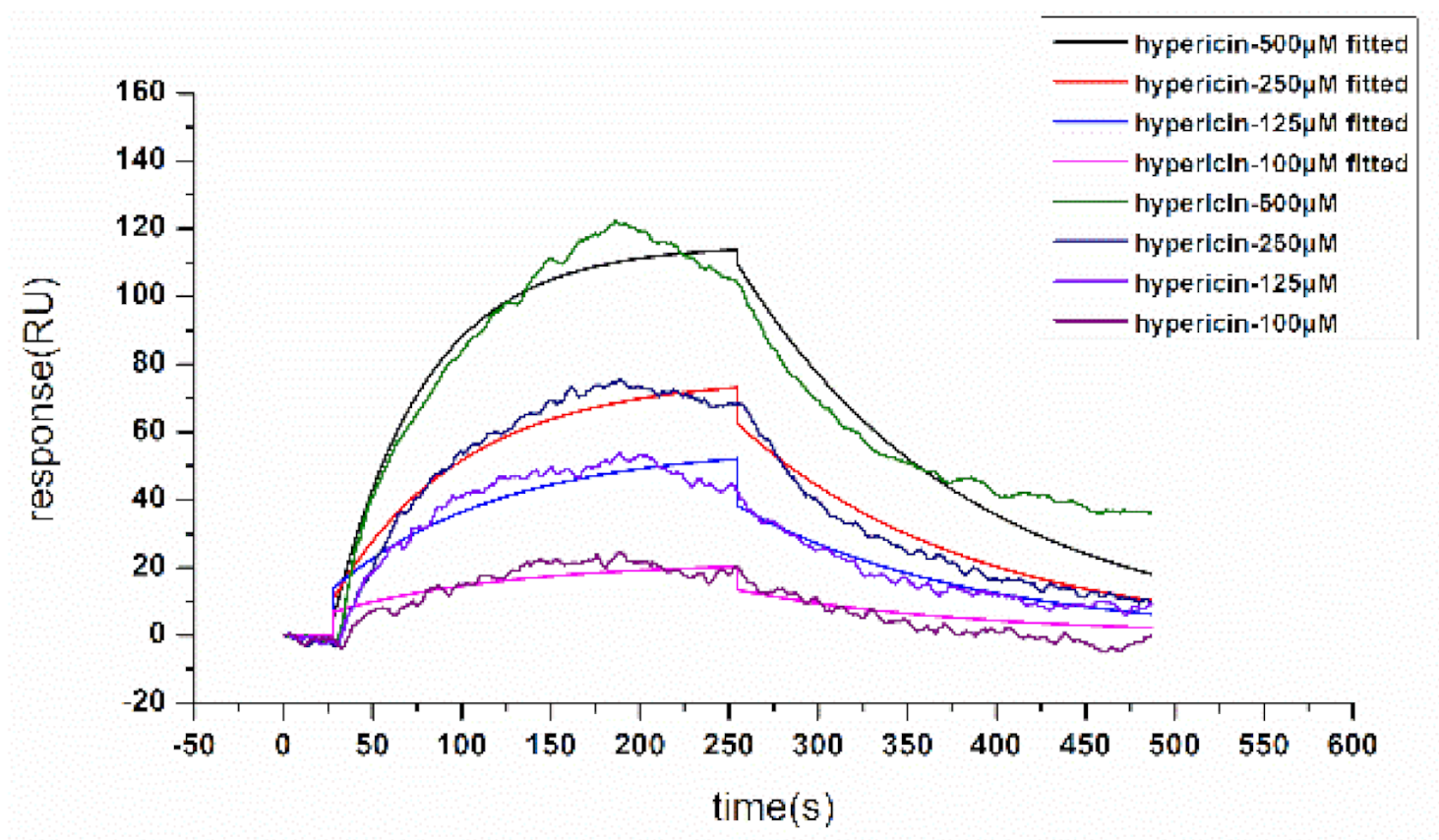
2.4. SPR Analysis of the Interaction of Hypericin and α-Glucosidase
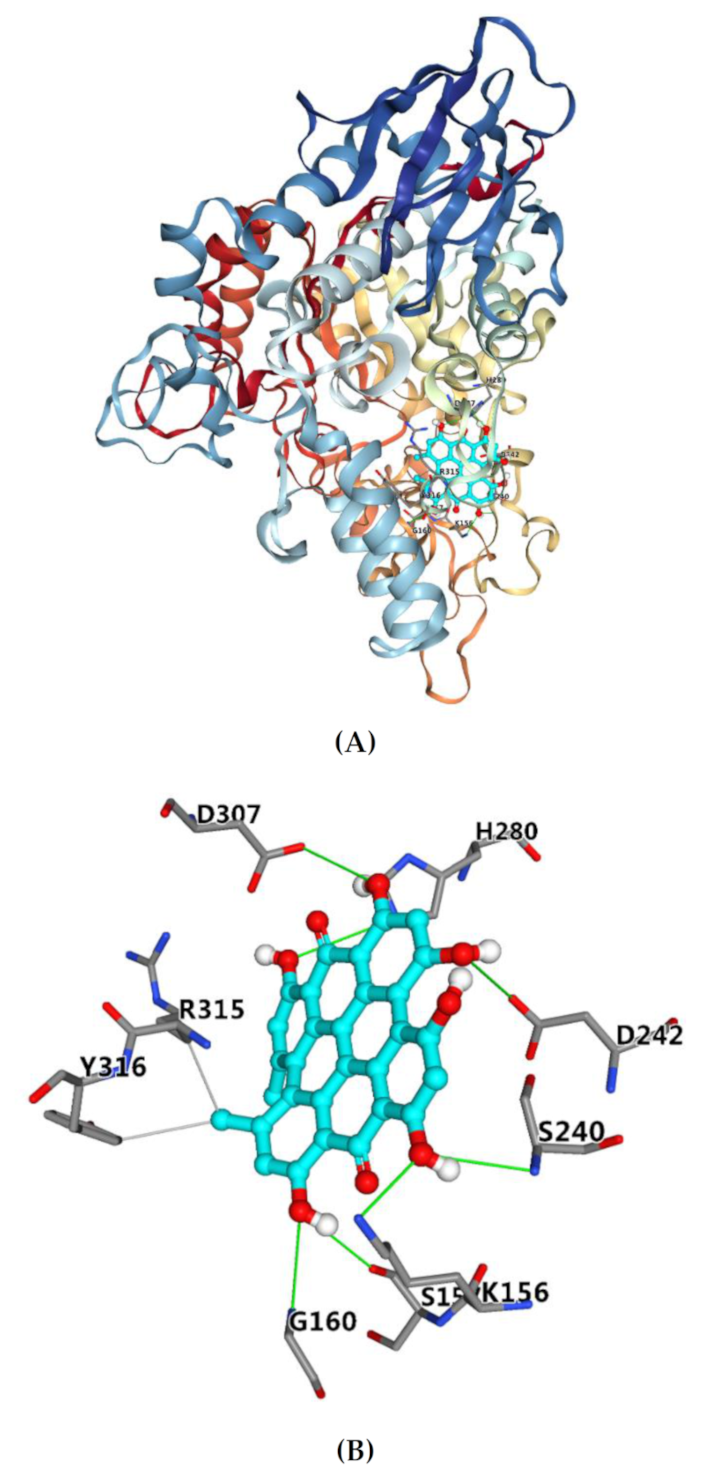
2.5. Molecular Docking
3. Materials and Methods
3.1. Materials
3.2. Determination of α-Glucosidase Activity Using Hypericin
3.3. Inhibitory Kinetic Analysis of α-Glucosidase Inhibition
3.4. Hypericin and α-Glucosidase Interactions by SPR Measurements
3.5. Molecular Docking Analysis
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Zhang, J.; Li, L.; Deng, Y.; Liang, F.; Shi, Z.; Zhang, H. The analysis of the epidemic status and risk factors of diabetes mellitus. World Latest Med. Inf. 2018, 18, 94–96. [Google Scholar] [CrossRef]
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF diabetes atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.C.; Millns, H.; Neil, H.A.W.; Stratton, I.M.; Manley, S.E.; Matthews, D.R.; Holman, R.R. Risk factors for coronary artery disease in non-insulin dependent diabetes mellitus: United Kingdom prospective diabetes study (UKPDS: 23). BMJ 1998, 316, 823–828. [Google Scholar] [CrossRef] [Green Version]
- Ceriello, A.; Genovese, S. Atherogenicity of postprandial hyperglycemia and lipotoxicity Atherogenicity of postprandial hyperglycemia and lipotoxicity. Rev. Endocr. Metab. Dis. 2016, 17, 111–116. [Google Scholar] [CrossRef]
- Mahler, R.J.; Adler, M.L. Type 2 diabetes mellitus: Update on diagnosis, pathophysiology, and treatment. J. Clin. Endocrinol. Metab. 1999, 84, 1165–1171. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Giugliano, D.; Nappo, F. Regression of carotid atherosclerosis by control of postprandial hyperglycemia in type 2 diabetes mellitus. Circulation 2004, 110, 214–219. [Google Scholar] [CrossRef]
- Bello, N.A.; Pfeffer, M.A.; Skali, H.; McGill, J.B.; Rossert, J.; Olson, K.A.; Weinrauch, L.; Cooper, M.E.C.; Zeeuw, D.D. Retinopathy and clinical outcomes in patients with type 2 diabetes mellitus, chronic kidney disease, and anemia. BMJ Open Diab. Res. Care 2014, 2, e000011. [Google Scholar] [CrossRef] [Green Version]
- Jiao, Y.; Hua, D.; Huang, D.; Zhang, Q.; Yan, C. Characterization of a new heteropolysaccharide from green guava and its application as an α-glucosidase inhibitor for the treatment of type II diabetes. Food Funct. 2018, 9, 3997–4007. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.-H.; Rose, D.R.; Lin, A.H.-M.; Quezada-Calvillo, R.; Nichols, B.L.; Hamaker, B.R. Contribution of the individual small intestinal α-glucosidases to digestion of unusual α-linked glycemic disaccharides. J. Agric. Food Chem. 2016, 64, 6487–6494. [Google Scholar] [CrossRef]
- Mohiuddin, M.; Arbain, D.; Islam, A.K.M.S.; Ahmad, M.S.; Ahmad, M.N. Alpha-glucosidase enzyme biosensor for the electrochemical measurement of antidiabetic potential of medicinal plants. Nanoscale Res. Lett. 2016, 11, 95. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Cheng, X.; Ren, G. α-Glucosidase inhibitory activity of protein-rich extracts from extruded adzuki bean in diabetic KK-Ay mice. Food Funct. 2014, 5, 966. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wang, S.; Wu, J.; Zhang, L.; Wang, Z.; Gan, L.; He, J.; Shia, H.M.; Hou, J.L. The kinetics and mechanism of α-glucosidase inhibition by F5-SP, a novel compound derived from sericin peptides. Food Funct. 2017, 8, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Chiba, S. Molecular mechanism in α-glucosidase and glucoamylase. Biosci. Biotech. Bioch. 1997, 61, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Mauldin, R.; Byers, L. Multiple sugar binding sites in α-glucosidase. BBA Proteins Proteom. 2003, 1645, 22–29. [Google Scholar] [CrossRef]
- Borges de Melo, E.; da Silveira Gomes, A.; Carvalho, I. α- and β-Glucosidase inhibitors: Chemical structure and biological activity. Tetrahedron 2006, 62, 10277–10302. [Google Scholar] [CrossRef]
- Casirola, D.M.; Ferraris, R.P. α-Glucosidase inhibitors prevent diet-induced increases in intestinal sugar transport in diabetic mice. Metabolism 2006, 55, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Narwal, S.; Kumar, V.O.; Prakash, O. α-Glucosidase inhibitors from plants: A natural approach to treat diabetes. Pharmacogn. Rev. 2011, 5, 19–29. [Google Scholar] [CrossRef] [Green Version]
- Barnes, J.; Anderson, L.A.; Phillipson, J.D. St John’s wort (Hypericum perforatum L.): A review of its chemistry, pharmacology and clinical properties. J. Pharm. Pharmacol. 2001, 53, 583–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahimi, R.; Kiani, S. Chemical compound and therapeutic effects of Hypericum perforatum. Der Pharm. Lett. 2016, 8, 237–241. [Google Scholar]
- Hasanein, P.; Shahidi, S. Effects of Hypericum perforatum extract on diabetes-induced learning and memory impairment in rats. Phytother. Res. 2010, 25, 544–549. [Google Scholar] [CrossRef]
- Arokiyaraj, S.; Balamurugan, R.; Augustian, P. Antihyperglycemic effect of Hypericum perforatum ethyl acetate extract on streptozotocin–induced diabetic rats. Asian Pac. J. Trop. Biomed. 2011, 1, 386–390. [Google Scholar] [CrossRef] [Green Version]
- Gu, J.; Chen, L.; Yuan, G.; Xu, X. A drug-target network-based approach to evaluate the efficacy of medicinal plants for type II diabetes mellitus. Evid. Based Complement. Altern. Med. 2013, 2013, 203614. [Google Scholar] [CrossRef] [Green Version]
- Hudson, J.B.; Lopez-Bazzocchi, I.; Towers, G.H.N. Antiviral activities of hypericin. Antivir. Res. 1991, 15, 101–112. [Google Scholar] [CrossRef]
- Lenard, J.; Rabson, A.; Vanderoef, R. Photodynamic inactivation of infectivity of human immunodeficiency virus and other enveloped viruses using hypericin and rose bengal: Inhibition of fusion and syncytia formation. Proc. Natl. Acad. Sci. USA 1993, 90, 158–162. [Google Scholar] [CrossRef] [Green Version]
- Yip, L.; Hudson, J.B.; Gruszecka-Kowalik, E.; Zalkow, L.H.; Neil Towers, G.H. Antiviral activity of a derivative of the photosensitive compound Hypericin. Phytomedicine 1996, 3, 185–190. [Google Scholar] [CrossRef]
- Laffers, W.; Busse, A.-C.; Mahrt, J.; Nguyen, P.; Gerstner, A.O.H.; Bootz, F.; Wessels, J.T. Photosensitizing effects of hypericin on head neck squamous cell carcinoma in vitro. Eur. Arch. Oto Rhino Laryngol. 2014, 272, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.; Singer, A.; Wonnemann, M. Hyperforin—Antidepressant activity by a novel mechanism of action. Pharmacopsychiatry 2001, 34, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.S.; Bhattacharya, S.K.; Wonnemann, M.; Singer, A.; Müller, W.E. Hyperforin as a possible antidepressant component of hypericum extracts. Life Sci. 1998, 63, 499–510. [Google Scholar] [CrossRef]
- Gibon, J.; Deloulme, J.C.; Chevallier, T.; Ladevèze, E.; Abrous, D.N.; Bouron, A. The antidepressant hyperforin increases the phosphorylation of CREB and the expression of TrkB in a tissue-specific manner. Int. J. Neuropsychopharmacol. 2013, 16, 189–198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kashef, N.; Borghei, Y.S.; Djavid, G.E. Photodynamic effect of hypericin on the microorganisms and primary human fibroblasts. Photodiagn. Photodyn. Ther. 2013, 10, 150–155. [Google Scholar] [CrossRef]
- Yin, R.; Hamblin, M.R. Antimicrobial photosensitizers: Drug discovery under the spotlight. Curr. Med. Chem. 2015, 22, 2159–2185. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Lu, Y.-H. Comparison of inhibitory activities and mechanisms of five mulberry plant bioactive components against α-glucosidase. J. Agric. Food Chem. 2013, 61, 8110–8119. [Google Scholar] [CrossRef]
- Choi, S.B.; Ko, B.S.; Park, S.K.; Jang, J.S.; Park, S. Insulin sensitizing and α-glucoamylase inhibitory action of sennosides, rheins and rhaponticin in Rhei Rhizoma. Life Sci. 2006, 78, 934–942. [Google Scholar] [CrossRef]
- Proença, C.; Freitas, M.; Ribeiro, D.; Oliveira, E.F.T.; Sousa, J.L.C.; Tomé, S.M.; Ramos, M.J.; Silva, A.M.S.; Fernandes, P.A.; Fernandes, E. α-glucosidase inhibition by flavonoids: An in vitro and in silico structure-activity relationship study. J. Enzym. Inhib. Med. Chem. 2017, 32, 1216–1228. [Google Scholar] [CrossRef] [Green Version]
- Chai, W.M.; Huang, Q.; Lin, M.Z.; Ou-Yang, C.; Huang, W.Y.; Wang, Y.X.; Xu, K.L.; Feng, H.L. Condensed tannins from longan bark as inhibitor of tyrosinase: Structure, activity, and mechanism. J. Agric. Food Chem. 2018, 66, 908–917. [Google Scholar] [CrossRef]
- Chi, G.; Qi, Y.; Li, J.; Wang, L.; Hu, J. Polyoxomolybdates as α-glucosidase inhibitors: Kinetic and molecular modeling studies. J. Inorg. Biochem. 2019, 193, 173–179. [Google Scholar] [CrossRef]
- Ding, H.; Hu, X.; Xu, X.; Zhang, G.; Gong, D. Inhibitory mechanism of two allosteric inhibitors, oleanolic acid and ursolic acid on α-glucosidase. Int. J. Biol. Macromol. 2018, 107, 1844–1855. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.B.; Annamalai, T.; Cheng, B.; Narula, G.; Wang, X.; Tse-Dinh, Y.C.; He, J.; Darici, Y. A surface plasmon resonance study of the intermolecular interaction between Escherichia coli topoisomerase I and pBAD/Thio supercoiled plasmid DNA. Biochem. Biophys. Res. Commun. 2014, 445, 445–450. [Google Scholar] [CrossRef] [Green Version]
- Nahálková, J.; Švitel, J.; Gemeiner, P.; Danielsson, B.; Pribulová, B.; Petruš, L. Affinity analysis of lectin interaction with immobilized C- and O- gylcosides studied by surface plasmon resonance assay. J. Biochem. Bioph. Methods 2002, 52, 11–18. [Google Scholar] [CrossRef]
- Liu, X.; Luo, F.; Li, P.; She, Y.; Gao, W. Investigation of the interaction for three Citrus flavonoids and α-amylase by surface plasmon resonance. Food Res. Int. 2017, 97, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.C.; Matsuoka, S.; Ano, H.; Ishida, H.; Hirose, M.; Sato, F.; Sugiyama, S.; Murata, M. Interaction kinetics of liposome-incorporated unsaturated fatty acids with fatty acid-binding protein 3 by surface plasmon resonance. Bioorg. Med. Chem. 2014, 22, 1804–1808. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zhang, G.; Pan, J.; Wang, Y. α-Glucosidase inhibition by luteolin: Kinetics, interaction and molecular docking. Int. J. Biol. Macromol. 2014, 64, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Jiang, J.; Tian, J.; Chen, S.; Ye, X.; Hu, Y.; Chen, J. Inhibitory mechanism of novel allosteric inhibitor, Chinese bayberry (Myrica rubra Sieb. et Zucc.) leaves proanthocyanidins against α-glucosidase. J. Funct. Foods 2019, 56, 286–294. [Google Scholar] [CrossRef]
- Zeng, L.; Zhang, G.; Liao, Y.; Gong, D. Inhibitory mechanism of morin on α-glucosidase and its anti-glycation properties. Food Funct. 2016, 7, 3953–3963. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Fang, C.; Zhu, R.; Peng, Q.; Li, D.; Wang, M. Inhibitory effect of phloretin on α-glucosidase: Kinetics, interaction mechanism and molecular docking. Int. J. Biol. Macromol. 2017, 95, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Ding, H.; Hu, X.; Zhang, G.; Gong, D. Galangin inhibits α-glucosidase activity and formation of non-enzymatic glycation products. Food Chem. 2019, 271, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Miyake, H.; Kusunoki, M.; Osaki, S. Crystal structures of isomaltase from Saccharomyces cerevisiae and in complex with its competitive inhibitor maltose. FEBS J. 2010, 277, 4205–4214. [Google Scholar] [CrossRef] [PubMed]
- Vachali, P.; Li, B.; Nelson, K.; Bernstein, P.S. Surface plasmon resonance (SPR) studies on the interactions of carotenoids and their binding proteins. Arch. Biochem. Biophys. 2012, 519, 32–37. [Google Scholar] [CrossRef] [Green Version]
- Cai, C.Y.; Rao, L.; Rao, Y.; Guo, J.X.; Xiao, Z.Z.; Cao, J.Y.; Huang, Z.S.; Wang, B. Analogues of xanthones—Chalcones and bis-chalcones as α-glucosidase inhibitors and anti-diabetes candidates. Eur. J. Med. Chem. 2017, 130, 51–59. [Google Scholar] [CrossRef]
- Lin, M.Z.; Chai, W.M.; Zheng, Y.L.; Huang, Q.; Ou-Yang, C. Inhibitory kinetics and mechanism of rifampicin on α-Glucosidase: Insights from spectroscopic and molecular docking analyses. Int. J. Biol. Macromol. 2019, 122, 1244–1252. [Google Scholar] [CrossRef]
- Ghani, U.; Nur-e-Alam, M.; Yousaf, M.; Ul-Haq, Z.; Noman, O.M.; Al-Rehaily, A.J. Natural flavonoid α-glucosidase inhibitors from Retama raetam: Enzyme inhibition and molecular docking reveal important interactions with the enzyme active site. Bioorg. Chem. 2019, 87, 736–742. [Google Scholar] [CrossRef]
- Song, S.S.; Sun, C.P.; Zhou, J.J.; Chu, L. Flavonoids as human carboxylesterase 2 inhibitors: Inhibition potentials and molecular docking simulations. Int. J. Biol. Macromol. 2019, 131, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, M.; Nusrat, S.; Alam, P.; Malik, S.; Chaturvedi, S.K.; Ajmal, M.R.; Abdelhameed, A.S.; Khan, R.H. Investigating the site selective binding of busulfan to human serum albumin: Biophysical and molecular docking approaches. Int. J. Biol. Macromol. 2018, 107, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, Q.; Hu, N.; Yue, H.; Wang, H. Inhibitory Activity and Mechanism Investigation of Hypericin as a Novel α-Glucosidase Inhibitor. Molecules 2021, 26, 4566. https://doi.org/10.3390/molecules26154566
Dong Q, Hu N, Yue H, Wang H. Inhibitory Activity and Mechanism Investigation of Hypericin as a Novel α-Glucosidase Inhibitor. Molecules. 2021; 26(15):4566. https://doi.org/10.3390/molecules26154566
Chicago/Turabian StyleDong, Qi, Na Hu, Huilan Yue, and Honglun Wang. 2021. "Inhibitory Activity and Mechanism Investigation of Hypericin as a Novel α-Glucosidase Inhibitor" Molecules 26, no. 15: 4566. https://doi.org/10.3390/molecules26154566





