Metal Complex Formation and Anticancer Activity of Cu(I) and Cu(II) Complexes with Metformin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Potentiometry-UV-Vis
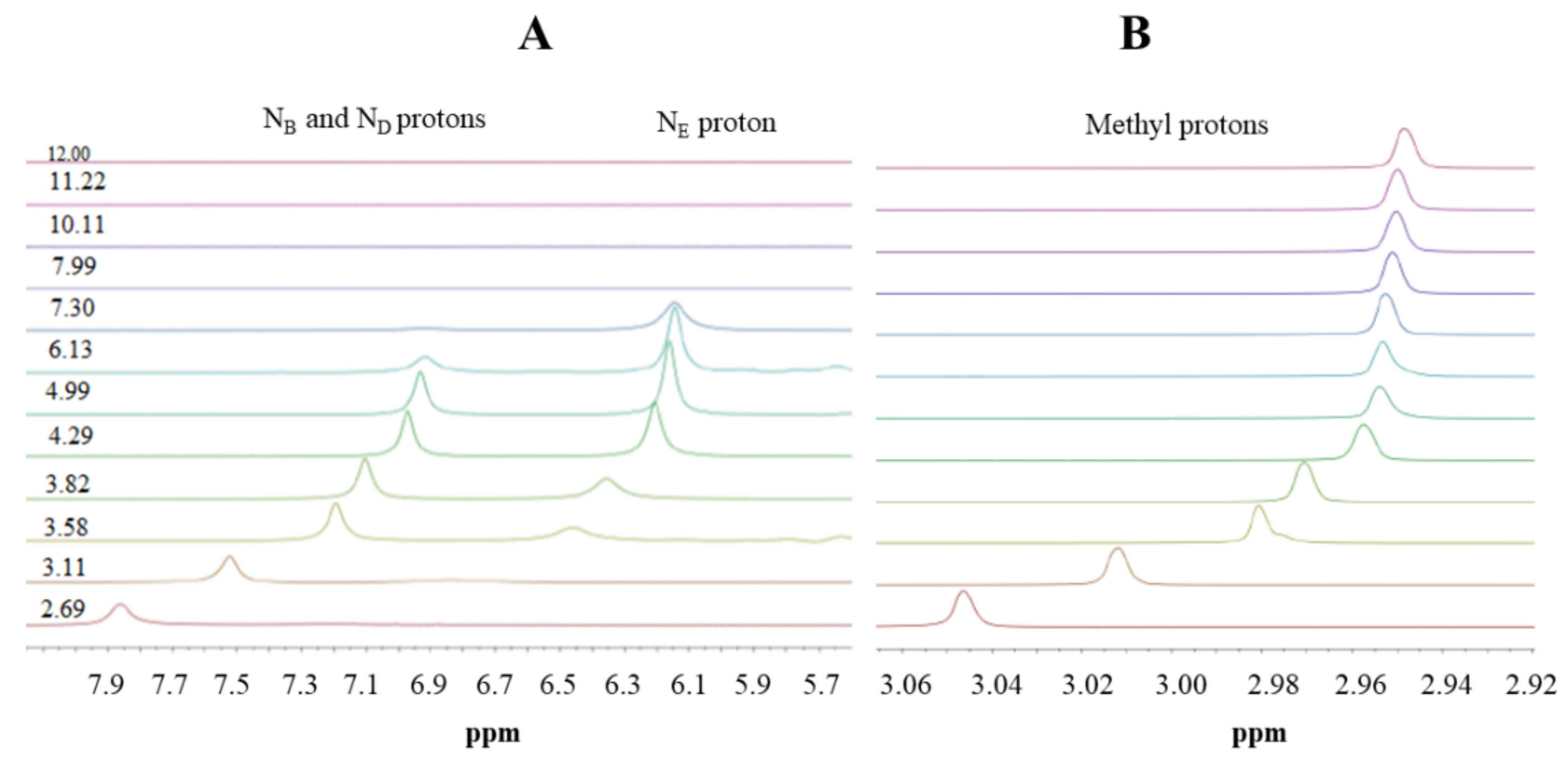
2.3. Nuclear Magnetic Resonance (NMR) Spectroscopy
2.4. Cellular Studies
2.4.1. Cell Culture of SW1222, K562, and K562-200
2.4.2. Assessment of the Effect of Metformin on the Proliferation of Cancer Cell Lines in the Presence and Absence of Copper Sulfate
2.4.3. Immunostaining of Actin Stress Fibers
3. Results and Discussion
3.1. Metformin Protonation Constants



| Technique/Species | Experimental Data | Literature Data | ||||
|---|---|---|---|---|---|---|
| LH3 | LH2 | LH | LH3 | LH2 | LH | |
| Potentiometry | 2.67(2) | 8.00(1) | 10.57(1) | - | 2.79 [37] | 11.02 [37] |
| - | 2.90 [38] | 11.10 [38] | ||||
| UV | - | - | - | |||
| NMR | 2.8(1) | NA | NA | - | 3.1 [39] | 13.8 [39] |
3.2. Copper Complexes
| Technique/Species | Logβ (Experimental Data) | LogK (Literature Data) | |||
|---|---|---|---|---|---|
| CuLH | CuL2H | CuL2 | CuL | CuL2 | |
| Potentiometry | 17.87(5) | 28.3(1) | 22.20(7) | 7.17 [42] | 5.13 [42] |
| 8.59 [38] | 6.35 [38] | ||||
| Vis | - | - | |||


3.3. Metformin and Copper Anticancer Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Viollet, B.; Guigas, B.; Garcia, N.S.; Leclerc, J.; Foretz, M.; Andreelli, F. Cellular and molecular mechanisms of metformin: An overview. Clin. Sci. 2012, 122, 253–270. [Google Scholar] [CrossRef] [Green Version]
- Repiščák, P.; Erhardt, S.; Rena, G.; Paterson, M.J. Biomolecular mode of action of metformin in relation to its copper binding properties. Biochemistry 2014, 53, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Myers, R.; Li, Y.; Chen, Y.; Shen, X.; Fenyk-Melody, J.; Wu, M.; Ventre, J.; Doebber, T.; Fujii, N.; et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Investig. 2001, 108, 1167–1174. [Google Scholar] [CrossRef]
- Logie, L.; Harthill, J.; Patel, K.; Bacon, S.; Hamilton, D.L.; Macrae, K.; McDougall, G.; Wang, H.-H.; Xue, L.; Jiang, H.; et al. Cellular responses to the metal-binding properties of metformin. Diabetes 2012, 61, 1423–1433. [Google Scholar] [CrossRef] [Green Version]
- Congiu, T.; Alghrably, M.; Emwas, A.-H.; Jaremko, L.; Lachowicz, J.I.; Piludu, M.; Monica, P.; Faa, G.; Pichiri, G.; Jaremko, M.; et al. Undercover toxic ménage à trois of Amylin, Copper(II) and Metformin in human embryonic kidney cells. Pharmaceutics 2021, 13, 830. [Google Scholar] [CrossRef]
- Chowdhury, T. Diabetes and cancer. QJM Int. J. Med. 2010, 103, 905–915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stattin, P.; Björ, O.; Ferrari, P.; Lukanova, A.; Lenner, P.; Lindahl, B.; Hallmans, G.; Kaaks, R. Prospective study of hyperglycemia and cancer risk. Diabetes Care 2007, 30, 561–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hemminki, K.; Li, X.; Sundquist, J.; Sundquist, K. Risk of cancer following hospitalization for type 2 diabetes. Oncologist 2010, 15, 548. [Google Scholar] [CrossRef] [Green Version]
- Wideroff, L.; Gridley, G.; Chow, W.-H.; Linet, M.; Mellemkjaer, L.; Olsen, J.H.; Keehn, S.; Borch-Johnsen, K. Cancer incidence in a population-based cohort of patients hospitalized with diabetes mellitus in Denmark. J. Natl. Cancer Inst. 1997, 89, 1360–1365. [Google Scholar] [CrossRef]
- Gandini, S.; Puntoni, M.; Heckman-Stoddard, B.M.; Dunn, B.K.; Ford, L.; DeCensi, A.; Szabo, E. Metformin and cancer risk and mortality: A systematic review and meta-analysis taking into account biases and confounders. Cancer Prev. Res. 2014, 7, 867–885. [Google Scholar] [CrossRef] [Green Version]
- DeCensi, A.; Puntoni, M.; Goodwin, P.; Cazzaniga, M.; Gennari, A.; Bonanni, B.; Gandini, S. Metformin and cancer risk in diabetic patients: A systematic review and meta-analysis. Cancer Prev. Res. 2010, 3, 1451–1461. [Google Scholar] [CrossRef] [Green Version]
- Chen, K.; Li, Y.; Guo, Z.; Zeng, Y.; Zhang, W.; Wang, H. Metformin: Current clinical applications in nondiabetic patients with cancer. Aging 2020, 12, 3993. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.M.; Donnelly, L.A.; Emslie-Smith, A.M.; Alessi, D.R.; Morris, A.D. Metformin and reduced risk of cancer in diabetic patients. Bmj 2005, 330, 1304–1305. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.; Yin, L.; Jiang, X.; Sun, X.; Wu, J.; Tian, H.; Gao, X.; He, X. Effect of metformin on cancer risk and treatment outcome of prostate cancer: A meta-analysis of epidemiological observational studies. PLoS ONE 2014, 9, e116327. [Google Scholar] [CrossRef] [Green Version]
- Heckman-Stoddard, B.M.; DeCensi, A.; Sahasrabuddhe, V.V.; Ford, L.G. Repurposing metformin for the prevention of cancer and cancer recurrence. Diabetologia 2017, 60, 1639–1647. [Google Scholar] [CrossRef] [Green Version]
- Murff, H.J.; Roumie, C.L.; Greevy, R.A.; Hackstadt, A.J.; McGowan, L.E.; Hung, A.M.; Grijalva, C.G.; Griffin, M.R. Metformin use and incidence cancer risk: Evidence for a selective protective effect against liver cancer. Cancer Causes Control 2018, 29, 823–832. [Google Scholar] [CrossRef]
- Wang, Z.; Lai, S.; Xie, L.; Zhao, J.; Ma, N.; Zhu, J.; Ren, Z.; Jiang, G. Metformin is associated with reduced risk of pancreatic cancer in patients with type 2 diabetes mellitus: A systematic review and meta-analysis. Diabetes Res. Clin. Pract. 2014, 106, 19–26. [Google Scholar] [CrossRef]
- Bradley, M.C.; Ferrara, A.; Achacoso, N.; Ehrlich, S.F.; Quesenberry, C.P.; Habel, L.A. A cohort study of metformin and colorectal cancer risk among patients with diabetes mellitus. Cancer Epidemiol. Prev. Biomark. 2018, 27, 525–530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bodmer, M.; Meier, C.; Krähenbühl, S.; Jick, S.S.; Meier, C.R. Long-term metformin use is associated with decreased risk of breast cancer. Diabetes Care 2010, 33, 1304–1308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tseng, C.-H. Use of metformin and risk of kidney cancer in patients with type 2 diabetes. Eur. J. Cancer 2016, 52, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.; Zhang, Y.; Gong, Y.; He, J.; Chen, X. Metformin and lung cancer risk of patients with type 2 diabetes mellitus: A meta-analysis. Biomed. Rep. 2015, 3, 235–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coyle, C.; Cafferty, F.; Vale, C.; Langley, R. Metformin as an adjuvant treatment for cancer: A systematic review and meta-analysis. Ann. Oncol. 2016, 27, 2184–2195. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Chen, K.; Jiang, Z.; Chen, X.; Sun, L.; Li, J.; Lei, J.; Xu, Q.; Ma, J.; Li, X.; et al. Desmoplasia suppression by metformin-mediated AMPK activation inhibits pancreatic cancer progression. Cancer Lett. 2017, 385, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Kheder, S.; Sisley, K.; Hadad, S.; Balasubramanian, S.P. Effects of prolonged exposure to low dose metformin in thyroid cancer cell lines. J. Cancer 2017, 8, 1053. [Google Scholar] [CrossRef] [Green Version]
- Shahabadi, N.; Heidari, L. Binding studies of the antidiabetic drug, metformin to calf thymus DNA using multispectroscopic methods. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 97, 406–410. [Google Scholar] [CrossRef]
- Shoair, A.G.F.; Ibrahim, M.M.; Soliman, M.; Abu-Melha, K.A. Spectroscopic, electrochemical, DNA binding and antioxidant biomimetic catalytic activities of metformin-based copper(II) complexes. J. Mol. Liq. 2015, 212, 865–871. [Google Scholar] [CrossRef]
- Anthony, E.J.; Bolitho, E.M.; Bridgewater, H.E.; Carter, O.W.; Donnelly, J.M.; Imberti, C.; Lant, E.C.; Lermyte, F.; Needham, R.J.; Palau, M.; et al. Metallodrugs are unique: Opportunities and challenges of discovery and development. Chem. Sci. 2020, 11, 12888–12917. [Google Scholar] [CrossRef]
- Pitucha, M.; Korga-Plewko, A.; Czylkowska, A.; Rogalewicz, B.; Drozd, M.; Iwan, M.; Kubik, J.; Humeniuk, E.; Adamczuk, G.; Karczmarzyk, Z.; et al. Influence of complexation of thiosemicarbazone derivatives with Cu(II) ions on their antitumor activity against melanoma cells. Int. J. Mol. Sci. 2021, 22, 3104. [Google Scholar] [CrossRef]
- Zeng, Z.; Huang, Q.; Cai, J.; Zheng, G.; Huang, Q.; Liu, Z.; Chen, Z.; Wei, Y. Synthesis, characterization, DNA/HSA interactions, and anticancer activity of two novel Copper(II) complexes with 4-Chloro-3-Nitrobenzoic acid ligand. Molecules 2021, 26, 4028. [Google Scholar] [CrossRef]
- Toso, L.; Crisponi, G.; Nurchi, V.M.; Crespo-Alonso, M.; Lachowicz, J.I.; Santos, M.A.; Marques, S.M.; Niclós-Gutiérrez, J.; González-Pérez, J.M.; Domínguez-Martín, A.; et al. A family of hydroxypyrone ligands designed and synthesized as iron chelators. J. Inorg. Biochem. 2013, 127, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Lachowicz, J.I.; Emwas, A.-H.; Delpiano, G.R.; Salis, A.; Piludu, M.; Jaremko, L.; Jaremko, M. Improving metal adsorption on Triethylenetetramine (TETA) functionalized SBA-15 Mesoporous silica using potentiometry, EPR and ssNMR. Adv. Mater. Interfaces 2020, 7, 2000544. [Google Scholar] [CrossRef]
- HypSpec2014. Available online: http://www.hyperquad.co.uk/HypSpec2014.htm (accessed on 9 July 2021).
- Ibrahim, S.M.; Bakhashab, S.; Ilyas, A.M.; Pushparaj, P.N.; Karim, S.; Khan, J.A.; Abuzenadah, A.M.; Chaudhary, A.G.; Al-Qahtani, M.H.; Ahmed, F. WYE-354 restores Adriamycin sensitivity in multidrug-resistant acute myeloid leukemia cell lines. Oncol. Rep. 2019, 41, 3179–3188. [Google Scholar] [CrossRef] [Green Version]
- Estimation Program Interface (EPI) Suite. Ver. 4.1; November 2012. Available online: https://www2.epa.gov/tsca-screening-tools (accessed on 8 November 2016).
- Hernández, B.; Pflüger, F.; Kruglik, S.G.; Cohen, R.; Ghomi, M. Protonation–deprotonation and structural dynamics of antidiabetic drug metformin. J. Pharm. Biomed. Anal. 2015, 114, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Frassineti, C.; Ghelli, S.; Gans, P.; Sabatini, A.; Moruzzi, M.S.; Vacca, A. Nuclear magnetic resonance as a tool for determining protonation constants of natural polyprotic bases in solution. Anal. Biochem. 1995, 231, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Karaderi, S.; Mazi, C.; Yilmazbilek, S. Determination of the stability constants comlexes of Metformin Hydrochloride with Cu(II), Cd(II) and Zn(II). Int. J. Pharm. Res. Allied Sci. 2020, 9, 121–129. [Google Scholar]
- Thakur, S.V.; Farooqui, M.; Sakhare, M.; Naikwade, S. Thermodynamic studies of oxytetracycline with some transition and rare earth metal ions in mixed solvent media. Am. Int. J. Res. Form. Appl. Nat. Sci. 2013, 3, 123–127. [Google Scholar]
- Orgován, G.; Noszál, B. Electrodeless, accurate pH determination in highly basic media using a new set of 1H-NMR pH indicators. J. Pharm. Biomed. Anal. 2011, 54, 958–964. [Google Scholar] [CrossRef]
- Bharatam, P.V.; Patel, D.S.; Iqbal, P. Pharmacophoric features of biguanide derivatives: An electronic and structural analysis. J. Med. Chem. 2005, 48, 7615–7622. [Google Scholar] [CrossRef]
- Ray, P. Complex compounds of biguanides and guanylureas with metallic elements. Chem. Rev. 1961, 61, 313–359. [Google Scholar] [CrossRef]
- Abu-El-Wafa, S.; El-Ries, M.; Ahmed, F. Formation of metformin complexes with some transition metal ions: Their biological activity. Inorg. Chim. Acta 1987, 136, 127–131. [Google Scholar] [CrossRef]
- Alderighi, L.; Gans, P.; Ienco, A.; Peters, D.; Sabatini, A.; Vacca, A. Hyperquad simulation and speciation (HySS): A utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 1999, 184, 311–318. [Google Scholar] [CrossRef]
- Zhu, M.; Lu, L.; Yang, P.; Jin, X. Bis(1,1-dimethylbiguanido) copper(II) octahydrate. Acta Crystallogr. Sect. E Struct. Rep. Online 2002, 58, m217–m219. [Google Scholar] [CrossRef]
- Lu, L.-P.; Yang, P.; Qin, S.-D.; Zhu, M.-L. Bis [1,1-dimethylbiguanide (1–)-κ2N2, N5] copper (II) monohydrate. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2004, 60, m219–m220. [Google Scholar] [CrossRef] [PubMed]
- Olar, R.; Badea, M.; Marinescu, D.; Chifiriuc, C.-M.; Bleotu, C.; Grecu, M.N.; Iorgulescu, E.E.; Bucur, M.; Lazar, V.; Finaru, A. Prospects for new antimicrobials based on N,N-dimethylbiguanide complexes as effective agents on both planktonic and adhered microbial strains. Eur. J. Med. Chem. 2010, 45, 2868–2875. [Google Scholar] [CrossRef]
- Olar, R.; Badea, M.; Marinescu, D.; Chifiriuc, M.-C.; Bleotu, C.; Grecu, M.N.; Iorgulescu, E.-E.; Lazar, V. N,N-dimethylbiguanide complexes displaying low cytotoxicity as potential large spectrum antimicrobial agents. Eur. J. Med. Chem. 2010, 45, 3027–3034. [Google Scholar] [CrossRef]
- Solomon, E.I.; Szilagyi, R.K.; DeBeer, G.S.; Basumallick, L. Electronic structures of metal sites in proteins and models: Contributions to function in blue copper proteins. Chem. Rev. 2004, 104, 419–458. [Google Scholar] [CrossRef]
- Balendiran, G.K.; Dabur, R.; Fraser, D. The role of glutathione in cancer. Cell Biochem. Funct. Cell. Biochem. Modul. Act. Agents Dis. 2004, 22, 343–352. [Google Scholar] [CrossRef]
- Midander, J.; Deschavanne, P.-J.; Malaise, E.-P.; Revesz, L. Survival curves of irradiated glutathione-deficient human fibroblasts: Indication of a reduced enhancement of radiosensitivity by oxygen and misonidazole. Int. J. Radiat. Oncol. Biol. Phys. 1982, 8, 443–446. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Munshi, A.; Hobbs, M.L.; Story, M.D.; Meyn, R.D., Jr. Paclitaxel restores radiation-induced apoptosis in a bcl-2-expressing, radiation-resistant lymphoma cell line. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 1127–1132. [Google Scholar] [CrossRef]
- Vukovic, V.; Nicklee, T.; Hedley, D.W. Microregional heterogeneity of non-protein thiols in cervical carcinomas assessed by combined use of HPLC and fluorescence image analysis. Clin. Cancer Res. 2000, 6, 1826–1832. [Google Scholar]
- Vukovic, V.; Nicklee, T.; Hedley, D.W. Differential effects of buthionine sulphoximine in hypoxic and non-hypoxic regions of human cervical carcinoma xenografts. Radiother. Oncol. 2001, 60, 69–73. [Google Scholar] [CrossRef]
- Kato, N.; Nakamura, M.; Uchiyama, T. 1H-NMR studies of the reactions of copper (I) and copper (II) with D-penicillamine and glutathione. J. Inorg. Biochem. 1999, 75, 117–121. [Google Scholar] [CrossRef]
- Miyoshi, K.; Sugiura, Y.; Ishizu, K.; Iitaka, Y.; Nakamura, H. Crystal structure and spectroscopic properties of violet glutathione-copper(II) complex with axial sulfur coordination and two copper sites via a disulfide bridge. J. Am. Chem. Soc. 1980, 102, 6130–6136. [Google Scholar] [CrossRef]
- Lu, C.-C.; Chiang, J.-H.; Tsai, F.-J.; Hsu, Y.-M.; Juan, Y.-N.; Yang, J.-S.; Chiu, H.-Y. Metformin triggers the intrinsic apoptotic response in human AGS gastric adenocarcinoma cells by activating AMPK and suppressing mTOR/AKT signaling. Int. J. Oncol. 2019, 54, 1271–1281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hess, C.; Unger, M.; Madea, B.; Stratmann, B.; Tschoepe, D. Range of therapeutic metformin concentrations in clinical blood samples and comparison to a forensic case with death due to lactic acidosis. Forensic Sci. Int. 2018, 286, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Zheng, Y.; Xi, T.; Zhang, C.; Song, W.; Burugapalli, K.; Yang, H.; Ma, Y. Concentration-dependent cytotoxicity of copper ions on mouse fibroblasts in vitro: Effects of copper ion release from TCu380A vs TCu220C intra-uterine devices. Biomed. Microdevices 2012, 14, 709–720. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.; Jang, S.Y.; Kim, C.; Choi, Y.; Kim, A. Anticancer effect of metformin on estrogen receptor-positive and tamoxifen-resistant breast cancer cell lines. Oncol. Rep. 2016, 35, 2553–2560. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Lee, J.; Jang, S.Y.; Kim, C.; Choi, Y.; Kim, A. Metformin sensitizes anticancer effect of dasatinib in head and neck squamous cell carcinoma cells through AMPK-dependent ER stress. Oncotarget 2014, 5, 298. [Google Scholar]
- Lee, J.-Y.; Shin, D.H.; Kim, J.-S. Anticancer effect of metformin in herceptin-conjugated liposome for breast cancer. Pharmaceutics 2020, 12, 11. [Google Scholar] [CrossRef] [Green Version]
- Safe, S.; Nair, V.; Karki, K. Metformin-induced anticancer activities: Recent insights. Biol. Chem. 2018, 399, 321–335. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, Z.; Wang, H.; Chen, X.; Jin, C. Activation of AMPK by metformin promotes renal cancer cell proliferation under glucose deprivation through its interaction with PKM2. Int. J. Biol. Sci. 2019, 15, 617. [Google Scholar] [CrossRef]
- Zakikhani, M.; Dowling, R.; Fantus, I.G.; Sonenberg, N.; Pollak, M. Metformin is an AMP kinase–dependent growth inhibitor for breast cancer cells. Cancer Res. 2006, 66, 10269–10273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, L.; Wei, J.; Wan, J.; Wang, W.; Wang, L.; Yuan, Y.; Yang, Z.; Liu, X.; Ming, L. Low glucose and metformin-induced apoptosis of human ovarian cancer cells is connected to ASK1 via mitochondrial and endoplasmic reticulum stress-associated pathways. J. Exp. Clin. Cancer Res. 2019, 38, 77. [Google Scholar] [CrossRef] [PubMed]
- Kacar, S.; Unver, H.; Sahinturk, V. A mononuclear copper(II) complex containing benzimidazole and pyridyl ligands: Synthesis, characterization, and antiproliferative activity against human cancer cells. Arab. J. Chem. 2020, 13, 4310–4323. [Google Scholar] [CrossRef]
- Hussain, A.; AlAjmi, M.F.; Rehman, M.T.; Amir, S.; Husain, F.M.; Alsalme, A.; Siddiqui, M.A.; AlKhedhairy, A.A.; Khan, R.A. Copper(II) complexes as potential anticancer and Nonsteroidal anti-inflammatory agents: In Vitro and In Vivo studies. Sci. Rep. 2019, 9, 5237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, N.K.; Kumbhar, A.A.; Pokharel, Y.R.; Yadav, P.N. Anticancer potency of copper (II) complexes of thiosemicarbazones. J. Inorg. Biochem. 2020, 210, 111134. [Google Scholar] [CrossRef]
- Molinaro, C.; Martoriati, A.; Pelinski, L.; Cailliau, K. Copper complexes as anticancer agents targeting topoisomerases I and II. Cancers 2020, 12, 2863. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelrahman, S.; Alghrably, M.; Campagna, M.; Hauser, C.A.E.; Jaremko, M.; Lachowicz, J.I. Metal Complex Formation and Anticancer Activity of Cu(I) and Cu(II) Complexes with Metformin. Molecules 2021, 26, 4730. https://doi.org/10.3390/molecules26164730
Abdelrahman S, Alghrably M, Campagna M, Hauser CAE, Jaremko M, Lachowicz JI. Metal Complex Formation and Anticancer Activity of Cu(I) and Cu(II) Complexes with Metformin. Molecules. 2021; 26(16):4730. https://doi.org/10.3390/molecules26164730
Chicago/Turabian StyleAbdelrahman, Sherin, Mawadda Alghrably, Marcello Campagna, Charlotte Armgard Emma Hauser, Mariusz Jaremko, and Joanna Izabela Lachowicz. 2021. "Metal Complex Formation and Anticancer Activity of Cu(I) and Cu(II) Complexes with Metformin" Molecules 26, no. 16: 4730. https://doi.org/10.3390/molecules26164730
APA StyleAbdelrahman, S., Alghrably, M., Campagna, M., Hauser, C. A. E., Jaremko, M., & Lachowicz, J. I. (2021). Metal Complex Formation and Anticancer Activity of Cu(I) and Cu(II) Complexes with Metformin. Molecules, 26(16), 4730. https://doi.org/10.3390/molecules26164730






