Recent Progress in Metal–Organic Framework-Derived Nanostructures in the Removal of Volatile Organic Compounds
Abstract
:1. Introduction to the Metal–Organic Framework
- Solvothermal or hydrothermal: in this process, crystals are allowed to grow smoothly over the course of hours to days from a hot solution.
- Microwave-assisted solvothermal synthesis: in this method, microwaves can be used to nucleate MOF crystals rapidly from a solution.
- Chemical vapor deposition method: this is a solvent-free method for the synthesis of MOFs. In this method, initially, metal oxide precursor layers are deposited, followed by exposing to sublimed ligand molecules, which induces a phase transformation to the MOF crystal lattice.
- Post-synthetic modification of MOFs:
2. Volatile Organic Compounds
2.1. Sources and Effect of VOCs
2.2. Classification of VOCs
- A.
- Very Volatile Organic Compounds (VVOCs):
- B.
- Volatile Organic Compounds (VOCs):
- C.
- Semi-Volatile Organic Compounds (SVOCs):
2.3. Removal of VOCs
3. Some Metal–Organic Frameworks:
- A
- ZIF-8 is 2-methylimadizole zinc salt of general chemical formula C8H10N4Zn. The ZIF-8 is Zn(MeIM)2. Here, MeIM is 2-methylimidazolate. The ZIF-8 is composed of zinc atom bonded with 2-methylimidazolate ligands with large cavities (11.4 Å) and small pore (3.4 Å) structures (Figure 1a [62]). The different SEM image of different ZIF structures [63] are shown in the Figure 2.
- B
- Cu-BTC is copper benzene-1,3,5-tricarboxylate with a chemical formula of [Cu3(btc)2] or C16H6Cu3O12. Here, btc is 1,3,5-benzenetricarboxylate. It is commercially available. It consists of three distinct cages: one small octahedral cage with a pore window of 2.0 Å and pore radius of 5.2 Å. Another larger cage is the cuboctahedral cage with a pore radius of 6.1 Å connected by a pore aperture of 2.6 Å radius (Figure 1b).
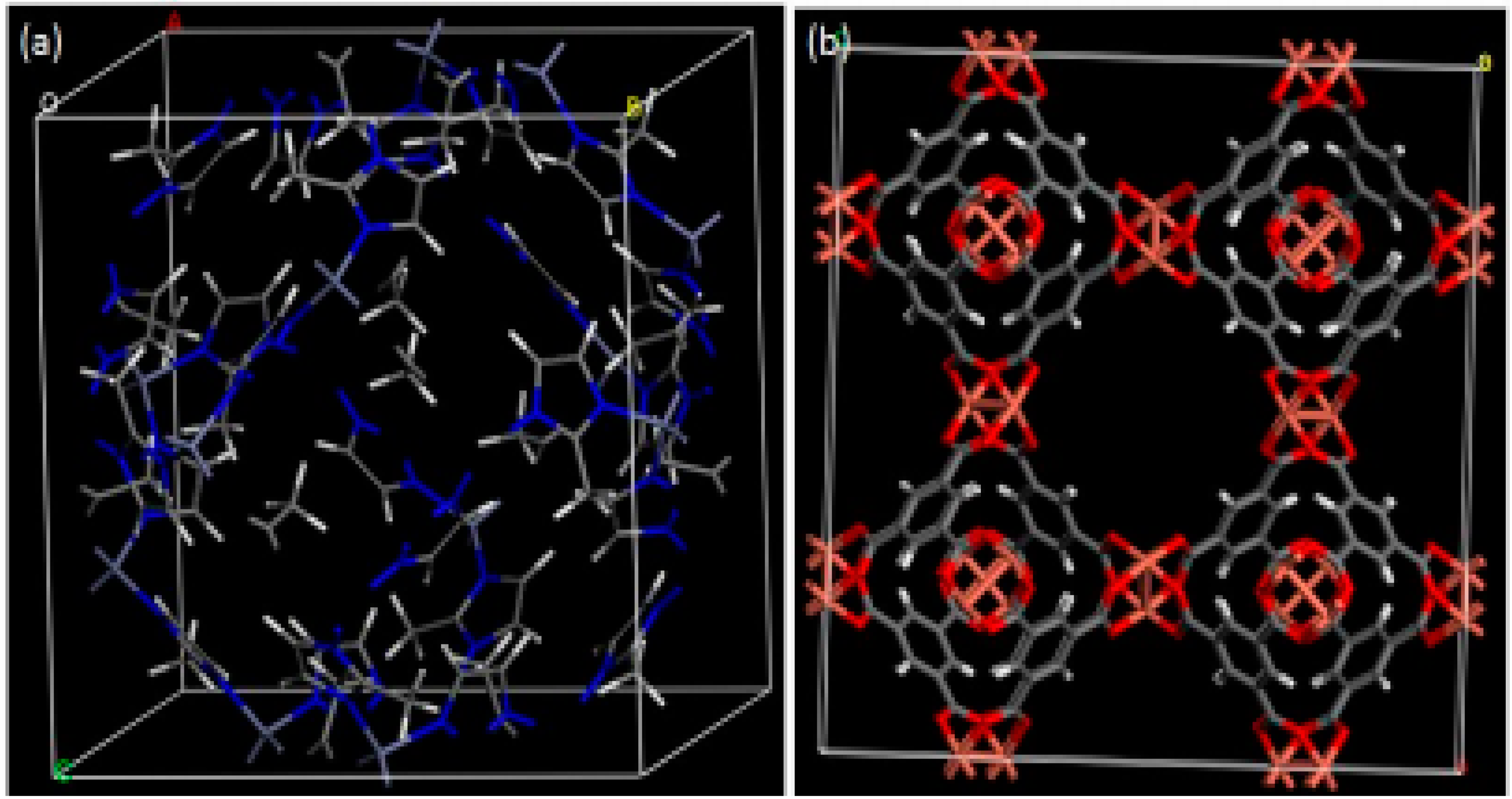

3.1. MOF-Derived Nanomaterials for the Removal of VOCs
3.2. Some Common VOCs and their Removal
- Removal of propane and butane:
- Removal of methyl chloride:
- Removal of ethanol:
- Removal of methanol:
- Removal of formaldehyde:
- Removal of formic acid:
- Removal of acetone:
- Removal of carbon monoxide:
- Removal of toluene:
- Removal of xylene:
- Removal of styrene:
4. Challenges and Future Prospects of MOF-NMs
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BDC | 1:4-Benzenedicarboxylate |
| BTC | 1,3,5-benzenetricarboxylic acid |
| BTEX | Benzene, toluene, ethylbenzene, and xylene |
| COF | Covalent organic framework |
| DDT | Dichlorodiphenyl trichloroethane |
| EPA | Environmental Protection Agency |
| FID | Flame ionization detector |
| LFSE | Ligand field stabilization energy |
| MOF | Metal–organic framework |
| PAH | Polycyclic aromatic hydrocarbons |
| PBB | Polybrominated biphenyl |
| PCB | Polychlorinated biphenyl |
| PCPs | Porous coordination polymer |
| PET | Polyethylene terephthalate |
| PM | Particulate matter |
| PVC | Polyvinyl chloride |
| ROS | Reactive oxygen species |
| SOA | Secondary organic aerosol |
| SVOC | Semi-volatile organic compounds |
| TCD | Thermal conductivity detector |
| UiO | University of Oslo |
| VOC | Volatile organic compound |
| VVOC | Very volatile organic compounds |
| WHO | World Health Organization |
| ZIF | Zeolite imidazolate framework |
| ZIF-8 | Zn(2-methylimidazole)2 |
References
- Lin, Y.; Wan, H.; Wu, D.; Chen, G.; Zhang, N.; Liu, X.; Li, J.; Cao, Y.; Qiu, G.; Ma, R. Metal-organic framework hexagonal nanoplates: Bottom-up synthesis, topotactic transformation, and efficient oxygen evolution reaction. J. Am. Chem. Soc. 2020, 142, 7317–7321. [Google Scholar] [CrossRef]
- Muthurasu, A.; Chae, S.-H.; Kim, T.; Mukhiya, T.; Kim, H.Y. Template-assisted fabrication of ZnO/Co3O4 one-dimensional metal-organic framework array decorated with amorphous iron oxide/hydroxide nanoparticles as an efficient electrocatalyst for the oxygen evolution reaction. Energy Fuels 2020, 34, 7716–7725. [Google Scholar] [CrossRef]
- Rao, K.P.; Higuchi, M.; Duan, J.; Kitagawa, S. pH-dependent interpenetrated, polymorphic, Cd2+- and BTB-based porous coordination polymers with open metal sites. Cryst. Growth Des. 2013, 13, 981–985. [Google Scholar] [CrossRef]
- He, C.-T.; Tian, J.-Y.; Liu, S.-Y.; Ouyang, G.; Zhang, J.-P.; Chen, X.-M. A porous coordination framework for highly sensitive and selective solid-phase microextraction of non-polar volatile organic compounds. Chem. Sci. 2013, 4, 351–356. [Google Scholar] [CrossRef]
- Peng, L.; Wu, S.; Yang, X.; Hu, J.; Fu, X.; Li, M.; Bai, L.; Huo, Q.; Guan, J. Oxidation of benzyl alcohol over metal organic frameworks M-BTC (M = Co, Cu, Fe). New J. Chem. 2017, 41, 2891–2894. [Google Scholar] [CrossRef]
- Ye, Y.; Guo, W.; Wang, L.; Lihua, W.; Song, Z.; Chen, J.; Zhang, Z.; Xiang, S.; Chen, B. Straightforward loading of imidazole molecules into metal-organic framework for high proton conduction. J. Am. Chem. Soc. 2017, 139, 15604–15607. [Google Scholar] [CrossRef] [PubMed]
- Schaate, A.; Dühnen, S.; Platz, G.; Lilienthal, S.; Schneider, A.M.; Behrens, P. A Novel Zr-based porous coordination polymer containing azobenzenedicarboxylate as a linker. Eur. J. Inorg. Chem. 2012, 2012, 790–796. [Google Scholar] [CrossRef]
- Cheng, Y.; Xiao, X.; Guo, X.; Yao, H.; Pang, H. Synthesis of “Quasi-Ce-MOF” electrocatalysts for enhanced urea oxidation reaction performance. ACS Sustain. Chem. Eng. 2020, 8, 8675–8680. [Google Scholar] [CrossRef]
- Li, Y.; Shan, Y.; Pang, H. Design and synthesis of nitrogen-doped hexagonal NiCoO nanoplates derived from Ni-Co-MOF for high-performance electrochemical energy storage. Chin. Chem. Lett. 2020, 31, 2280–2286. [Google Scholar] [CrossRef]
- Liang, Z.; Zhao, R.; Qiu, T.; Zou, R.; Xu, Q. Metal-organic framework-derived materials for electrochemical energy applications. Energy Chem. 2019, 1, 100001. [Google Scholar] [CrossRef]
- Li, D.; Xu, H.-Q.; Jiao, L.; Jiang, H.-L. Metal-organic frameworks for catalysis: State of the art, challenges, and opportunities. Energy Chem. 2019, 1, 100005. [Google Scholar] [CrossRef]
- He, Y.; Wang, Z.; Wang, H.; Wang, Z.; Zeng, G.; Xu, P.; Huang, D.; Chen, M.; Song, B.; Qin, H.; et al. Metal-organic framework-derived nanomaterials in environment related fields: Fundamentals, properties and applications. Coord. Chem. Rev. 2020, 213618. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuan, S.; Feng, X.; Li, H.; Zhou, J.; Wang, B. Preparation of nanofibrous metal-organic framework filters for efficient air pollution control. J. Am. Chem. Soc. 2016, 138, 5785–5788. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The chemistry and applications of metal-organic frameworks. Science 2013, 341, 1230444. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Li, X.; Li, Y.; Zhu, R.; Pang, H. Applications of metal-organic-framework-derived carbon materials. Adv. Mater. 2019, 31, 1804740. [Google Scholar] [CrossRef]
- Min, H.; Wang, J.; Qi, Y.; Zhang, Y.; Han, X.; Xu, Y.; Xu, J.; Li, Y.; Chen, L.; Cheng, K.; et al. Biomimetic metal-organic framework nanoparticles for cooperative combination of antiangiogenesis and photodynamic therapy for enhanced efficacy. Adv. Mater. 2019, 31, 1808200. [Google Scholar] [CrossRef] [PubMed]
- Peera, S.; Koutavarapu, R.; Liu, C.; Rajeshkhanna, G.; Asokan, A.; Reddy, C. Cobalt nanoparticle-embedded nitrogen-doped carbon catalyst derived from a solid-state metal-organic framework complex for OER and HER electrocatalysis. Energies 2021, 14, 1320. [Google Scholar] [CrossRef]
- Salunkhe, R.; Kaneti, Y.; Yamauchi, Y. Metal-organic framework-derived nanoporous metal oxides toward supercapacitor applications: Progress and prospects. ACS Nano 2017, 11, 5293–5308. [Google Scholar] [CrossRef]
- Ojha, G.P.; Muthurasu, A.; Tiwari, A.P.; Pant, B.; Chhetri, K.; Mukhiya, T.; Dahal, B.; Lee, M.; Park, M.; Kim, H.-Y. Vapor solid phase grown hierarchical CuxO NWs integrated MOFs-derived CoS2 electrode for high-performance asymmetric supercapacitors and the oxygen evolution reaction. Chem. Eng. J. 2020, 399, 125532. [Google Scholar] [CrossRef]
- Acharya, J.; Ojha, G.P.; Kim, B.-S.; Pant, B.; Park, M. Modish designation of hollow-tubular rGO-NiMoO4@Ni-Co-S hybrid core-shell electrodes with multichannel superconductive pathways for high-performance asymmetric supercapacitors. ACS Appl. Mater. Interfaces 2021, 13, 17487–17500. [Google Scholar] [CrossRef]
- Acharya, J.; Ko, T.H.; Seo, M.-K.; Khil, M.-S.; Kim, H.-Y.; Kim, B.-S. Engineering the hierarchical heterostructures of Zn-Ni-Co nanoneedles Arrays@Co–Ni-LDH nanosheets core-sheath electrodes for a hybrid asymmetric supercapacitor with high energy density and excellent cyclic stability. ACS Appl. Energy Mater. 2020, 3, 7383–7396. [Google Scholar] [CrossRef]
- Acharya, J.; Pant, B.; Ojha, G.P.; Kong, H.-S.; Park, M. Engineering triangular bimetallic metal-organic-frameworks derived hierarchical zinc-nickel-cobalt oxide nanosheet arrays@reduced graphene oxide-Ni foam as a binder-free electrode for ultra-high rate performance supercapacitors and methanol electro-oxidation. J. Colloid Interface Sci. 2021, 602, 573–589. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Eddaoudi, M.; O’Keeffe, M.; Yaghi, O. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 1999, 402, 276–279. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.; Lee, Y.-S.; Shin, J.-W.; Kim, K.-H.; Kukkar, D.; Tsang, Y.F. Potential applications of graphene-based nanomaterials as adsorbent for removal of volatile organic compounds. Environ. Int. 2020, 135, 105356. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, Q.; Zheng, Y.; Han, R.; Song, C.; Ji, N.; Ma, D. Enhanced catalytic performance for volatile organic compound oxidation over in-situ growth of MnOx on Co3O4 nanowire. Chemosphere 2020, 244, 125532. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.C.; Hopkins, J.R.; Carslaw, D.C.; Hamilton, J.F.; Nelson, B.S.; Stewart, G.; Dernie, J.; Passant, N.; Murrells, T. An increasing role for solvent emissions and implications for future measurements of volatile organic compounds. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2020, 378, 20190328. [Google Scholar] [CrossRef] [PubMed]
- Saini, V.K.; Pires, J. Development of metal organic framework-199 immobilized zeolite foam for adsorption of common indoor VOCs. J. Environ. Sci. 2017, 55, 321–330. [Google Scholar] [CrossRef]
- Jackson, S.; Rananaware, A.; Rix, C.; Bhosale, S.V.; Latham, K. Highly fluorescent metal-organic framework for the sensing of volatile organic compounds. Cryst. Growth Des. 2016, 16, 3067–3071. [Google Scholar] [CrossRef]
- Kandiah, M.; Usseglio, S.; Svelle, S.; Olsbye, U.; Lillerud, K.P.; Tilset, M. Post-synthetic modification of the metal-organic framework compound UiO-66. J. Mater. Chem. 2010, 20, 9848–9851. [Google Scholar] [CrossRef]
- Bari, M.A.; Kindzierski, W.B.; Wheeler, A.J.; Héroux, M.È.; Wallace, L.A. Source apportionment of indoor and outdoor volatile organic compounds at homes in Edmonton, Canada. Build. Environ. 2015, 90, 114–124. [Google Scholar] [CrossRef]
- Contarino, R.; Brighina, S.; Fallico, B.; Cirvilleri, G.; Parafati, L.; Restuccia, C. Volatile organic compounds (VOCs) produced by biocontrol yeasts. Food Microbiol. 2019, 82, 70–74. [Google Scholar] [CrossRef]
- Weisskopf, L.; Schulz, S.; Garbeva, P. Microbial volatile organic compounds in intra-kingdom and inter-kingdom interactions. Nat. Rev. Microbiol. 2021, 19, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Guha, P.; Nandi, S. Essential oil of betel leaf (Piper betle L.): A novel addition to the world food sector. In Essential Oil Research; Springer: Cham, Switzerland, 2019; pp. 149–196. [Google Scholar]
- Zhigzhitzhapova, S.V.; Radnaeva, L.D.; Gao, Q.; Chen, S.; Zhang, F. Chemical composition of volatile organic compounds of Artemisia vulgaris L. (Asteraceae) from the Qinghai–Tibet Plateau. Ind. Crop. Prod. 2016, 83, 462–469. [Google Scholar] [CrossRef]
- Liu, Y.; Song, M.; Liu, X.; Zhang, Y.; Hui, L.; Kong, L.; Zhang, Y.; Zhang, C.; Qu, Y.; An, J.; et al. Characterization and sources of volatile organic compounds (VOCs) and their related changes during ozone pollution days in 2016 in Beijing, China. Environ. Pollut. 2020, 257, 113599. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Kong, S.; Yan, Y.; Chen, N.; Yao, L.; Liu, X.; Wu, F.; Cheng, Y.; Niu, Z.; Zheng, S.; et al. Compositions, sources and health risks of ambient volatile organic compounds (VOCs) at a petrochemical industrial park along the Yangtze River. Sci. Total Environ. 2020, 703, 135505. [Google Scholar] [CrossRef]
- Arı, A.; Arı, P.E.; Yenisoy-Karakaş, S.; Gaga, E.O. Source characterization and risk assessment of occupational exposure to volatile organic compounds (VOCs) in a barbecue restaurant. Build. Environ. 2020, 174, 106791. [Google Scholar] [CrossRef]
- Liu, R.; Chen, J.; Li, G.; Wang, X.; An, T. Cutting down on the ozone and SOA formation as well as health risks of VOCs emitted from e-waste dismantlement by integration technique. J. Environ. Manag. 2019, 249. [Google Scholar] [CrossRef]
- Aklilu, Y.-A.; Cho, S.; Zhang, Q.; Taylor, E. Source apportionment of volatile organic compounds measured near a cold heavy oil production area. Atmos. Res. 2018, 206, 75–86. [Google Scholar] [CrossRef]
- Wei, W.; Cheng, S.; Li, G.; Wang, G.; Wang, H. Characteristics of volatile organic compounds (VOCs) emitted from a petroleum refinery in Beijing, China. Atmos. Environ. 2014, 89, 358–366. [Google Scholar] [CrossRef]
- Sahu, L.K.; Yadav, R.; Pal, D. Source identification of VOCs at an urban site of western India: Effect of marathon events and anthropogenic emissions. J. Geophys. Res. Atmos. 2016, 121, 2416–2433. [Google Scholar] [CrossRef] [Green Version]
- Semenya, K.; Machete, F. Influence of kitchen structures on household exposure to firewood-induced volatile organic compounds in Senwabarwana villages. Air Qual. Atmos. Health 2020, 13, 1193–1201. [Google Scholar] [CrossRef]
- Zhang, K.; Li, L.; Huang, L.; Wang, Y.; Huo, J.; Duan, Y.; Wang, Y.; Fu, Q. The impact of volatile organic compounds on ozone formation in the suburban area of Shanghai. Atmos. Environ. 2020, 232, 117511. [Google Scholar] [CrossRef]
- Gironi, F.; Piemonte, V. VOCs removal from dilute vapour streams by adsorption onto activated carbon. Chem. Eng. J. 2011, 172, 671–677. [Google Scholar] [CrossRef]
- Li, L.; Liu, S.; Liu, J. Surface modification of coconut shell based activated carbon for the improvement of hydrophobic VOC removal. J. Hazard. Mater. 2011, 192, 683–690. [Google Scholar] [CrossRef]
- Cheng, J.; Li, L.; Li, Y.; Wang, Q.; He, C. Fabrication of pillar(5)arene-polymer-functionalized cotton fibers as adsorbents for adsorption of organic pollutants in water and volatile organic compounds in air. Cellulose 2019, 26, 3299–3312. [Google Scholar] [CrossRef]
- Awasthi, G.P.; Bhattarai, D.P.; Maharjan, B.; Kim, K.-S.; Park, C.H.; Kim, C.S. Synthesis and characterizations of activated carbon from Wisteria sinensis seeds biomass for energy storage applications. J. Ind. Eng. Chem. 2019, 72, 265–272. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Pokharel, P.; Xiao, D. Surface functionalization of polymers. In Reactive and Functional Polymers; Springer: New York, NY, USA, 2020; Volume 4, pp. 5–34. [Google Scholar]
- Wang, H.; Sun, Y.; Zhu, T.; Wang, W.; Deng, H. Adsorption of acetaldehyde onto carbide-derived carbon modified by oxidation. Chem. Eng. J. 2015, 273, 580–587. [Google Scholar] [CrossRef]
- Zhuang, Z.; Wang, L.; Tang, J. Efficient removal of volatile organic compound by ball-milled biochars from different preparing conditions. J. Hazard. Mater. 2021, 406, 124676. [Google Scholar] [CrossRef]
- Kansal, A. Sources and reactivity of NMHCs and VOCs in the atmosphere: A review. J. Hazard. Mater. 2009, 166, 17–26. [Google Scholar] [CrossRef]
- Changsuphan, A.; Wahab, M.I.B.; Oanh, N.T.K. Removal of benzene by ZnO nanoparticles coated on porous adsorbents in presence of ozone and UV. Chem. Eng. J. 2012, 181–182, 215–221. [Google Scholar] [CrossRef]
- Lemus, J.; Martinez, M.M.; Palomar, J.F.; Gomez-Sainero, L.; Gilarranz, M.A.; Rodríguez, J.J. Removal of chlorinated organic volatile compounds by gas phase adsorption with activated carbon. Chem. Eng. J. 2012, 211, 246–254. [Google Scholar] [CrossRef] [Green Version]
- Xin, Y.; Ando, Y.; Nakagawa, S.; Nishikawa, H.; Shirai, T. New possibility of hydroxyapatites as noble-metal-free catalysts towards complete decomposition of volatile organic compounds. Catal. Sci. Technol. 2020, 10, 5453–5459. [Google Scholar] [CrossRef]
- Li, J.; Chen, R.; Cui, W.; Dong, X.; Wang, H.; Kim, K.-H.; Chu, Y.; Sheng, J.; Sun, Y.; Dong, F. Synergistic photocatalytic decomposition of a volatile organic compound mixture: High efficiency, reaction mechanism, and long-term stability. ACS Catal. 2020, 10, 7230–7239. [Google Scholar] [CrossRef]
- Debono, O.; Thevenet, F.; Gravejat, P.; Hequet, V.; Raillard, C.; LE Coq, L.; Locoge, N. Toluene photocatalytic oxidation at ppbv levels: Kinetic investigation and carbon balance determination. Appl. Catal. B Environ. 2011, 106, 600–608. [Google Scholar] [CrossRef]
- Yang, X.; Ma, X.; Han, D.; Xiao, M.; Ma, L.; Sun, H.; Yu, X.; Ge, M. Efficient removal of toluene over palladium supported on hierarchical alumina microspheres catalyst. Catal. Today 2020, 375, 352–359. [Google Scholar] [CrossRef]
- Li, P.; Kim, S.; Jin, J.; Do, H.C.; Park, J.H. Efficient photodegradation of volatile organic compounds by iron-based metal-organic frameworks with high adsorption capacity. Appl. Catal. B Environ. 2020, 263, 118284. [Google Scholar] [CrossRef]
- Yu, G.; Mu, M.; Li, J.; Wu, B.; Xu, R.; Liu, N.; Chen, B.; Dai, C. Imidazolium-based ionic liquids introduced into π-electron donors: Highly efficient toluene capture. ACS Sustain. Chem. Eng. 2020, 8, 9058–9069. [Google Scholar] [CrossRef]
- Higashimoto, S.; Katsuura, K.; Yamamoto, M.; Takahashi, M. Photocatalytic activity for decomposition of volatile organic compound on Pt-WO3 enhanced by simple physical mixing with TiO2. Catal. Commun. 2020, 133, 105831. [Google Scholar] [CrossRef]
- Almomani, F.; Bhosale, R.; Shawaqfah, M. Solar oxidation of toluene over Co doped nano-catalyst. Chemosphere 2020, 255, 126878. [Google Scholar] [CrossRef]
- Boudjema, L.; Long, J.; Petitjean, H.; Larionova, J.; Guari, Y.; Trens, P.; Salles, F. Adsorption of volatile organic compounds by ZIF-8, Cu-BTC and a Prussian blue analogue: A comparative study. Inorg. Chim. Acta 2020, 501, 119316. [Google Scholar] [CrossRef]
- Tu, M.; Wannapaiboon, S.; Khaletskaya, K.; Fischer, R.A. Engineering zeolitic-imidazolate framework (ZIF) thin film devices for selective detection of volatile organic compounds. Adv. Funct. Mater. 2015, 25, 4470–4479. [Google Scholar] [CrossRef]
- Maharjan, B.; Kumar, D.; Awasthi, G.P.; Bhattarai, D.P.; Kim, J.Y.; Park, C.H.; Kim, C.S. Synthesis and characterization of gold/silica hybrid nanoparticles incorporated gelatin methacrylate conductive hydrogels for H9C2 cardiac cell compatibility study. Compos. Part B Eng. 2019, 177, 107415. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Kim, B.S. NIR-triggered hyperthermal effect of polythiophene nanoparticles synthesized by surfactant-free oxidative polymerization method on colorectal carcinoma cells. Cells 2020, 9, 2122. [Google Scholar] [CrossRef] [PubMed]
- Rezk, A.I.; Bhattarai, D.P.; Park, J.; Park, C.H.; Kim, C.S. Polyaniline-coated titanium oxide nanoparticles and simvastatin-loaded poly(ε-caprolactone) composite nanofibers scaffold for bone tissue regeneration application. Colloids Surf. B Biointerfaces 2020, 192, 111007. [Google Scholar] [CrossRef]
- Nathanael, A.J.; Kannaiyan, K.; Kunhiraman, A.K.; Kumaravel, V. 8—Nanomaterials for detection and removal of gases. In Nanomaterials for Sustainable Energy and Environmental Remediation; Naushad, M., Saravanan, R., Raju, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 219–260. [Google Scholar]
- Law, T.S.C.; Chao, C.; Chan, G.Y.W.; Law, A.K.Y. Confined catalytic oxidation of volatile organic compounds by transition metal containing zeolites and ionizer. Atmos. Environ. 2003, 37, 5433–5437. [Google Scholar] [CrossRef]
- Lamonier, J.-F.; Boutoundou, A.-B.; Gennequin, C.; Pérez-Zurita, M.J.; Siffert, S.; Aboukais, A. Catalytic removal of toluene in air over Co-Mn-Al nano-oxides synthesized by hydrotalcite route. Catal. Lett. 2007, 118, 165–172. [Google Scholar] [CrossRef]
- Attia, M.F.; Swasy, M.I.; Ateia, M.; Alexis, F.; Whitehead, D.C. Periodic mesoporous organosilica nanomaterials for rapid capture of VOCs. Chem. Commun. 2020, 56, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Xiong, X.; Wan, Z.; Sun, Y.; Tsang, D.C.; Gupta, J.; Gao, B.; Cao, X.; Tang, J.; Ok, Y.S. Ball milling as a mechanochemical technology for fabrication of novel biochar nanomaterials. Bioresour. Technol. 2020, 312, 123613. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, E.; Khapre, A.; Bordoloi, N.; Kumar, S. Sorption of volatile organic compounds on non-activated biochar. Bioresour. Technol. 2020, 297, 122469. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, H.; Mosleh, M.H.; Mandal, P.; Lea-Langton, A.; Sedighi, M. Sorption behaviour of xylene isomers on biochar from a range of feedstock. Chemosphere 2021, 268, 129310. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Awasthi, G.P.; Maharjan, B.; Lee, J.; Kim, B.-S.; Park, C.H.; Kim, C.S. Synthesis of polythiophene nanoparticles by surfactant-free chemical oxidative polymerization method: Characterization, in vitro biomineralization, and cytotoxicity evaluation. J. Ind. Eng. Chem. 2019, 77, 243–252. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Shrestha, S.; Shrestha, B.K.; Park, C.H.; Kim, C.S. A controlled surface geometry of polyaniline doped titania nanotubes biointerface for accelerating MC3T3-E1 cells growth in bone tissue engineering. Chem. Eng. J. 2018, 350, 57–68. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Tiwari, A.P.; Maharjan, B.; Tumurbaatar, B.; Park, C.H.; Kim, C.S. Sacrificial template-based synthetic approach of polypyrrole hollow fibers for photothermal therapy. J. Colloid Interface Sci. 2019, 534, 447–458. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Kim, M.H.; Park, H.; Park, W.H.; Kim, B.S.; Kim, C.S. Coaxially fabricated polylactic acid electrospun nanofibrous scaffold for sequential release of tauroursodeoxycholic acid and bone morphogenic protein2 to stimulate angiogenesis and bone regeneration. Chem. Eng. J. 2020, 389, 123470. [Google Scholar] [CrossRef]
- Bhattarai, D.P.; Hwang, T.I.; Kim, J.I.; Lee, J.H.; Chun, S.; Kim, B.-S.; Park, C.H.; Kim, C.S. Synthesis of polypyrrole nanorods via sacrificial removal of aluminum oxide nanopore template: A study on cell viability, electrical stimulation and neuronal differentiation of PC12 cells. Mater. Sci. Eng. C 2020, 107, 110325. [Google Scholar] [CrossRef] [PubMed]
- Maharjan, B.; Kaliannagounder, V.K.; Jang, S.R.; Awasthi, G.P.; Bhattarai, D.P.; Choukrani, G.; Park, C.H.; Kim, C.S. In-situ polymerized polypyrrole nanoparticles immobilized poly(ε-caprolactone) electrospun conductive scaffolds for bone tissue engineering. Mater. Sci. Eng. C 2020, 114, 111056. [Google Scholar] [CrossRef]
- Cho, W.; Park, S.; Oh, M. Coordination polymer nanorods of Fe-MIL-88B and their utilization for selective preparation of hematite and magnetite nanorods. Chem. Commun. 2011, 47, 4138–4140. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhang, X.; Shioyama, H.; Mukai, T.; Sakai, T.; Xu, Q. Converting cobalt oxide subunits in cobalt metal-organic framework into agglomerated Co3O4 nanoparticles as an electrode material for lithium ion battery. J. Power Sources 2010, 195, 857–861. [Google Scholar] [CrossRef]
- Jiang, H.-L.; Liu, B.; Lan, Y.-Q.; Kuratani, K.; Akita, T.; Shioyama, H.; Zong, F.; Xu, Q. From metal-organic framework to nanoporous carbon: Toward a very high surface area and hydrogen uptake. J. Am. Chem. Soc. 2011, 133, 11854–11857. [Google Scholar] [CrossRef]
- Kaneti, Y.; Tang, J.; Salunkhe, R.; Jiang, X.; Yu, A.; Wu, K.C.-W.; Yamauchi, Y. Nanoarchitectured design of porous materials and nanocomposites from metal-organic frameworks. Adv. Mater. 2017, 29, 1604898. [Google Scholar] [CrossRef]
- Zhang, X.; Li, H.; Hou, F.; Yang, Y.; Dong, H.; Liu, N.; Wang, Y.; Cui, L. Synthesis of highly efficient Mn2O3 catalysts for CO oxidation derived from Mn-MIL-100. Appl. Surf. Sci. 2017, 411, 27–33. [Google Scholar] [CrossRef]
- Sun, H.; Yu, X.; Ma, X.; Yang, X.; Lin, M.; Ge, M. MnOx-CeO2 catalyst derived from metal-organic frameworks for toluene oxidation. Catal. Today 2020, 355, 580–586. [Google Scholar] [CrossRef]
- Lin, D.; Zheng, Y.; Feng, X.; You, Y.; Wu, E.; Luo, Y.; Qian, Q.; Chen, Q. Highly stable Co3O4 nanoparticles-assembled microrods derived from MOF for efficient total propane oxidation. J. Mater. Sci. 2020, 55, 5190–5202. [Google Scholar] [CrossRef]
- Ipadeola, A.K.; Barik, R.; Ray, S.C.; Ozoemena, K.I. Bimetallic Pd/SnO2 nanoparticles on metal organic framework (MOF)-derived carbon as electrocatalysts for ethanol oxidation. Electrocatalysis 2019, 10, 366–380. [Google Scholar] [CrossRef]
- Wu, X.-Q.; Zhao, J.; Wu, Y.-P.; Dong, W.-W.; Li, D.-S.; Li, J.-R.; Zhang, Q. Ultrafine Pt nanoparticles and amorphous nickel supported on 3D mesoporous carbon derived from cu-metal-organic framework for efficient methanol oxidation and nitrophenol reduction. ACS Appl. Mater. Interfaces 2018, 10, 12740–12749. [Google Scholar] [CrossRef]
- Zhang, N.; Lu, Y.; Fan, Y.; Zhou, J.; Li, X.; Adimi, S.; Liu, C.; Ruan, S. Metal-organic framework-derived ZnO/ZnCo2O4 microspheres modified by catalytic PdO nanoparticles for sub-ppm-level formaldehyde detection. Sens. Actuators B Chem. 2020, 315, 128118. [Google Scholar] [CrossRef]
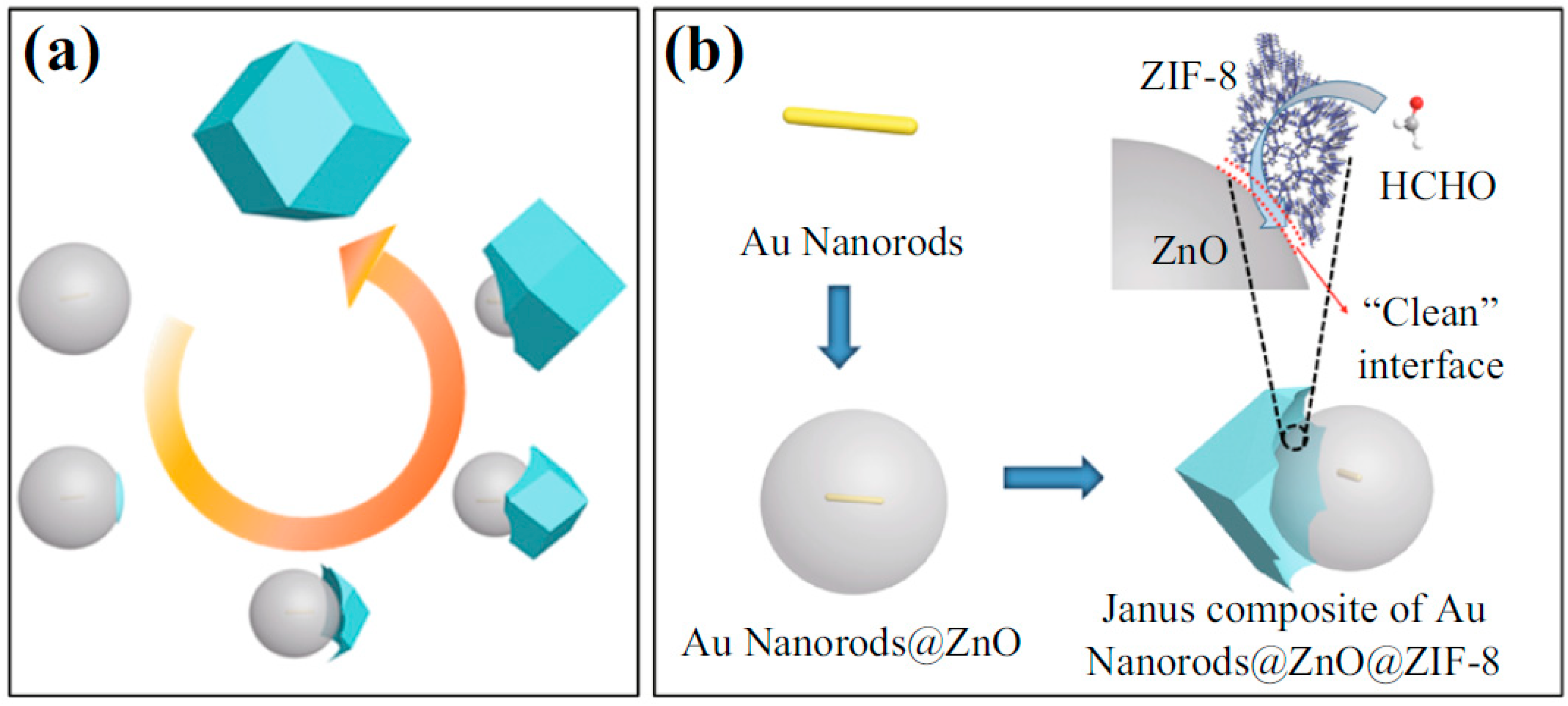
- Wang, D.; Li, Z.; Zhou, J.; Fang, H.; He, X.; Jena, P.; Zeng, J.-B.; Wang, W.-N. Simultaneous detection and removal of formaldehyde at room temperature: Janus Au@ZnO@ZIF-8 nanoparticles. Nano Micro Lett. 2017, 10, 4. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Meng, Q.; Gao, L.; Liu, J.; Ge, J.; Liu, C.; Xing, W. Metal organic framework derived nitrogen-doped carbon anchored palladium nanoparticles for ambient temperature formic acid decomposition. Int. J. Hydrog. Energy 2019, 44, 28402–28408. [Google Scholar] [CrossRef]
- Xia, J.; Diao, K.; Zheng, Z.; Cui, X. Porous Au/ZnO nanoparticles synthesised through a metal organic framework (MOF) route for enhanced acetone gas-sensing. RSC Adv. 2017, 7, 38444–38451. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Li, Y.; Zhang, R.; He, D.; Liu, H.; Liao, S. Metal-organic framework as a host for synthesis of nanoscale Co3O4 as an active catalyst for CO oxidation. Catal. Commun. 2011, 12, 875–879. [Google Scholar] [CrossRef]
- Zhao, J.; Tang, Z.; Dong, F.; Zhang, J. Controlled porous hollow Co3O4 polyhedral nanocages derived from metal-organic frameworks (MOFs) for toluene catalytic oxidation. Mol. Catal. 2019, 463, 77–86. [Google Scholar] [CrossRef]
- Zhang, X.; Lv, X.; Bi, F.; Lu, G.; Wang, Y. Highly efficient Mn2O3 catalysts derived from Mn-MOFs for toluene oxidation: The influence of MOFs precursors. Mol. Catal. 2020, 482, 110701. [Google Scholar] [CrossRef]
- Wang, L.; Yin, G.; Yang, Y.; Zhang, X. Enhanced CO oxidation and toluene oxidation on CuCeZr catalysts derived from UiO-66 metal organic frameworks. React. Kinet. Mech. Catal. 2019, 128, 193–204. [Google Scholar] [CrossRef]
- Zhang, X.; Song, L.; Bi, F.; Zhang, D.; Wang, Y.; Cui, L. Catalytic oxidation of toluene using a facile synthesized Ag nanoparticle supported on UiO-66 derivative. J. Colloid Interface Sci. 2020, 571, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Y.-X.; Liu, L.; Han, Z.-B. Palladium nanoparticles supported on UiO-66-NH2 as heterogeneous catalyst for epoxidation of styrene. Inorg. Chem. Commun. 2019, 100, 51–55. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, D.-D.; Ji, G.-J.; Yuan, S.; Qian, J.-F.; He, M.-Y.; Chen, Q.; Zhang, Z.-H. Efficient adsorption separation of xylene isomers using a facilely fabricated cyclodextrin-based metal-organic framework. J. Chem. Technol. Biotechnol. 2018, 93, 2898–2905. [Google Scholar] [CrossRef]
- Liu, H.; Xu, M.; Li, G.; Zhang, W.; An, T. Solar-light-triggered regenerative adsorption removal of styrene by silver nanoparticles incorporated in metal-organic frameworks. Environ. Sci. Nano 2021, 8, 543–553. [Google Scholar] [CrossRef]
- Cavka, J.H.; Jakobsen, S.; Olsbye, U.; Guillou, N.; Lamberti, C.; Bordiga, S.; Lillerud, K.P. A new zirconium inorganic building brick forming metal organic frameworks with exceptional stability. J. Am. Chem. Soc. 2008, 130, 13850–13851. [Google Scholar] [CrossRef]
- Huang, H.; Huang, W.; Xu, Y.; Ye, X.; Wu, M.; Shao, Q.; Ou, G.; Peng, Z.; Shi, J.; Chen, J.; et al. Catalytic oxidation of gaseous benzene with ozone over zeolite-supported metal oxide nanoparticles at room temperature. Catal. Today 2015, 258, 627–633. [Google Scholar] [CrossRef]
- García, T.; Solsona, B.; Taylor, S.H. Naphthalene total oxidation over metal oxide catalysts. Appl. Catal. B Environ. 2006, 66, 92–99. [Google Scholar] [CrossRef]
- Solsona, B.; García, T.; Sanchis, R.; Soriano, M.D.; Moreno, M.; Rodríguez-Castellón, E.; Agouram, S.; Dejoz, A.; Nieto, J.M.L. Total oxidation of VOCs on mesoporous iron oxide catalysts: Soft chemistry route versus hard template method. Chem. Eng. J. 2016, 290, 273–281. [Google Scholar] [CrossRef]
- Zhang, W.; Cheng, H.; Niu, Q.; Fu, M.; Huang, H.; Ye, D. Microbial targeted degradation pretreatment: A novel approach to preparation of activated carbon with specific hierarchical porous structures, high surface areas, and satisfactory toluene adsorption performance. Environ. Sci. Technol. 2019, 53, 7632–7640. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Zong, B.; Mao, S. Metal-organic framework-based sensors for environmental contaminant sensing. Nano Micro Lett. 2018, 10, 1–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| SN | VOCs | Sources | Removal Method | Ref |
|---|---|---|---|---|
| 1 | Acetaldehyde | Photochemical production | Using carbide-derived carbon | [49] |
| 2 | Acetone and toluene | Oxidation of fuels, biomass burning, geochemical process | Using ball-milled biochar | [50,51] |
| 3 | Benzene | Traffic and various industries | ZnO NPs coated on zeolite and activated carbons | [52] |
| 4 | Chlorinated volatile organic compounds (Cl-VOCs), e.g., monochloromethane, dichloremethane, trichloromethane, etc. | Gas streams | Using activated carbon | [53] |
| 5 | Ethyl acetate, isopropanol, acetone | hydoxyapaptites | [54] | |
| 6 | Formaldehyde and toluene | Photochemical production | Photocatalytic decomposition using SnO2 photocatalyst | [55] |
| 7 | Methyl tert-butyl ether (MTBE) | Motor fuel additive | Using activated carbon | [44] |
| 8 | Toluene | Petrochemical production | Photocatalytic oxidation of toluene to CO2 and H2O | [56] |
| 9 | Toluene | Using Pd supported hierarchical alumina microsphere catalyst | [57] | |
| 10 | Toluene | Using Fe-MOFs as both an adsorbent and photocatalyst | [58] | |
| 11 | Toluene | Toluene capture by using imidazolium-based ionic liquids | [59] | |
| 12 | Toluene | Visible-light-sensitive photocatalytic decomposition of toluene using WO3-deposited Pt | [60] | |
| 13 | Toluene | Solar photocatalytic oxidation of toluene using Co-doped TiO2 | [61] |
| SN | VOCs | Types of Nanomaterials | Efficacy | Preparation Method | Ref. | |
|---|---|---|---|---|---|---|
| 1 | Acetone | Au/ZnO NPs | Gas sensing response of 17.1 ppm–1 | Calcination of ZIF-8 | [92] | |
| 3 | Benzene | ZnO NPs-coated zeolite and AC | Detection limit of 3 ppb | By coating ZnO NPs and AC on zeolite | [52] | |
| 4 | Benzene | MnO2/ZSM-5 zeolite | Benzene can be removed completely. CO2 selectivity reached to 84.7% | Impregnation of metal oxide on ZSM-5 | [102] | |
| 6 | Carbon monoxide | Co3O4 | - | Sacrificial removal of MOFs | [93] | |
| 7 | Carbon monoxide | Mn-MIL-100-derived Mn2O3 nonporous | - | Calcination of MOFs at 700 °C. | [84] | |
| 9 | Ethanol | Pd/SnO2 NPs on MOF-derived carbon | - | Microwave-assisted method | [87] | |
| 10 | Formaldehyde | PdO NPs-decorated ZnO/ZnCo2O4 microsphere | Detection limit of 0.2 ppm | Prussian-blue-based co-precipitation using MOF (Zn3[Co(CN)6]2) | [89] | |
| 11 | Formic acid | N-doped C-anchored Pd NPs | Turn over frequency of the catalyst at 30 °C is 1166 h–1. | Wet chemical method using ZIF-8 | [91] | |
| 12 | Methanol | Pt NPs and amorphous Ni supported 3D mesoporous C | Diverse selectivity on nitrophenol | Carbonization and chemical etching of Cu-MOF | [88] | |
| 13 | Naphthalene | CeO2 | - | Homogeneous precipitation method with urea | [103] | |
| 14 | Propane Toluene | Mesoporous α-Fe2O3 | - | Wet chemical synthesis | [104] | |
| 15 | Propane | Co-BTC | - | Hydrothermal method | [86] | |
| 16 | Styrene | Pd/UiO-66-NH2 | Highest conversion (87%) of styrene and best selectivity (96.5%) in acetonitrile | Solution impregnation method | [98] | |
| 17 | Styrene | Ag/UiO-66 | - | Colloidal deposition method | [100] | |
| 18 | Toluene | Hierarchical porous carbon | Adsorption performance of 2290 m2/g | Microbial lignocellulose decomposition | [105] | |
| 19 | Toluene | Mn2O3 | - | Pyrolysis of MOFs containing Mn salts | [95] | |
| 20 | Toluene | Ag/UiO-66 | - | Liquid phase reduction | [97] | |
| 21 | Toluene | MnOx-CeO2-MOF derived from MOF | - | In situ pyrolysis of MOF-74 | [85] | |
| 22 | Toluene | Hollow Co3O4 polyhedral nanocages | Complete conversion of toluene was observed at 280 °C | Pyrolysis of ZIF-67 MOFs | [94] | |
| 23 | Xylene isomers | Cyclodextrin-alkali metal salt MOFs MIL-101 (Cr) | The equilibrium capacities of o-xylene, m-xylene and p-xylene are 175, 70, and 64 mg/g, respectively | Wet chemical method | [99] | |
| 24 | Toluene and CO | CuCeZr700 | CuCeZr700 exhibited 100% of CO oxidation at 140 °C and 90% toluene oxidation at 310 °C | Direct decomposition of UiO-66 MOFs in air | [96] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhattarai, D.P.; Pant, B.; Acharya, J.; Park, M.; Ojha, G.P. Recent Progress in Metal–Organic Framework-Derived Nanostructures in the Removal of Volatile Organic Compounds. Molecules 2021, 26, 4948. https://doi.org/10.3390/molecules26164948
Bhattarai DP, Pant B, Acharya J, Park M, Ojha GP. Recent Progress in Metal–Organic Framework-Derived Nanostructures in the Removal of Volatile Organic Compounds. Molecules. 2021; 26(16):4948. https://doi.org/10.3390/molecules26164948
Chicago/Turabian StyleBhattarai, Deval Prasad, Bishweshwar Pant, Jiwan Acharya, Mira Park, and Gunendra Prasad Ojha. 2021. "Recent Progress in Metal–Organic Framework-Derived Nanostructures in the Removal of Volatile Organic Compounds" Molecules 26, no. 16: 4948. https://doi.org/10.3390/molecules26164948







