A–X⋯σ Interactions—Halogen Bonds with σ-Electrons as the Lewis Base Centre
Abstract
:1. Introduction
2. Results and Discussions
2.1. Examples of Crystal Structures
2.2. Energies and Geometries of Complexes Linked by A–X⋯σ Interactions
2.3. C–X⋯σ and X–X⋯σ Interactions in Complexes of Dihydrogen and Cyclopropane
2.4. Energy Decomposition Analysis
3. Computational Approaches
4. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Metrangolo, P.; Resnati, G. Halogen bonding: A paradigm in supramolecular chemistry. Chem. Eur. J. 2001, 7, 2511–2519. [Google Scholar] [CrossRef]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. The Halogen Bond. Chem. Rev. 2016, 116, 2478–2601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clark, T.; Hennemann, M.; Murray, J.S.; Politzer, P. Halogen bonding: The σ-hole. J. Mol. Model. 2007, 13, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Lane, P.; Concha, M.C.; Ma, Y.; Murray, J.S. An overview of halogen bonding. J. Mol. Model. 2007, 13, 305–311. [Google Scholar] [CrossRef]
- Politzer, P.; Riley, K.E.; Bulat, F.A.; Murray, J.S. Perspectives on halogen bonding and other σ-hole interactions: Lex parsimoniae (Occam’s Razor). Comput. Theor. Chem. 2012, 998, 2–8. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S. Halogen Bonding: An Interim Discussion. ChemPhysChem 2013, 14, 278–294. [Google Scholar] [CrossRef]
- Murray, J.S.; Lane, P.; Politzer, P. Expansion of the σ-hole concept. J. Mol. Model. 2009, 15, 723–729. [Google Scholar] [CrossRef]
- Eskandari, K.; Zariny, H. Halogen bonding: A lump-hole interaction. Chem. Phys. Lett. 2010, 492, 9–13. [Google Scholar] [CrossRef]
- Dong, W.; Li, Q.; Scheiner, S. Comparative Strengths of Tetrel, Pnicogen, Chalcogen, and Halogen Bonds and Contributing Factors. Molecules 2018, 23, 1681. [Google Scholar] [CrossRef] [Green Version]
- Scheiner, S. Detailed Comparison of the Pnicogen Bond with Chalcogen, Halogen, and Hydrogen Bonds. Int. J. Quantum Chem. 2013, 113, 1609–1620. [Google Scholar] [CrossRef] [Green Version]
- Scheiner, S. The Pnicogen Bond: Its Relation to Hydrogen, Halogen, and Other Noncovalent Bonds. Acc. Chem. Res. 2013, 46, 280–288. [Google Scholar] [PubMed]
- Grabowski, S.J. Hydrogen and halogen bonds are ruled by the same mechanisms. Phys. Chem. Chem. Phys. 2013, 15, 7249–7259. [Google Scholar] [CrossRef]
- Turunen, L.; Erdélyi, M. Halogen Bonds and Halonium Ions. Chem. Soc. Rev. 2020, 49, 2688–2700. [Google Scholar] [CrossRef]
- Zordan, F.; Brammer, L.; Sherwood, P. Supramolecular Chemistry of Halogens: Complementary Features of Inorganic (M-X) and Organic (C-X´) Halogens Applied to M-X···X´-C halogen Bond Formation. J. Am. Chem. Soc. 2005, 127, 5979–5989. [Google Scholar]
- Nyburg, S.C.; Wong-Ng, W. Anisotropic atom-atom forces in the space group of solid chlorine. Proc. R Soc. London A 1979, 367, 29–45. [Google Scholar]
- Bilewicz, E.; Rybarczyk-Pirek, A.J.; Dubis, A.T.; Grabowski, S.J. Halogen bonding in crystal structure of 1-methylpyrrol-2-yl trichloromethyl ketone. J. Mol. Struct. 2007, 829, 208–211. [Google Scholar] [CrossRef]
- Grabowski, S.J. Halogen Bond and Its Counterparts: Bent’s Rule Explains the Formation of Nonbonding Interactions. J. Phys. Chem. A 2011, 115, 12340–12347. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, M.L. Strength of the [Z–I⋯Hal]− and [Z–Hal⋯I]− Halogen Bonds: Electron Density Properties and Halogen Bond Length as Estimators of Interaction Energy. Molecules 2021, 26, 2083. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Murray, J.S.; Clark, T. Halogen bonding: An electrostatically-driven highly directional noncovalent interaction. Phys. Chem. Chem. Phys. 2010, 12, 7748–7758. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Clark, T. Halogen bonding and other σ-hole interactions: A perspective. Phys. Chem. Chem. Phys. 2013, 15, 11178–11189. [Google Scholar] [CrossRef]
- Bauzá, A.; Frontera, A. Aerogen Bonding Interaction: A New Supramolecular Force. Angew. Chem. Int. Ed. 2015, 54, 7340–7343. [Google Scholar] [CrossRef]
- Grabowski, S.J. Hydrogen bonds, and σ-hole and π-hole bonds—Mechanisms protecting doublet and octet electron structures. Phys. Chem. Chem. Phys. 2017, 19, 29742–29759. [Google Scholar] [CrossRef]
- Grabowski, S.J. Boron and other triel Lewis acid centers: From hypovalency to hypervalency. ChemPhysChem 2014, 15, 2985–2993. [Google Scholar] [CrossRef] [PubMed]
- Grabowski, S.J. Triel bond and coordination of triel centres—Comparison with hydrogen bond interaction. Coord. Chem. Rev. 2020, 407, 213171. [Google Scholar] [CrossRef]
- Brinck, T.; Murray, J.S.; Politzer, P. A computational analysis of the bonding in boron trifluoride and boron trichloride and their complexes with ammonia. Inorg. Chem. 1993, 32, 2622–2625. [Google Scholar] [CrossRef]
- Bessac, F.; Frenking, G. Why Is BCl3 a Stronger Lewis Acid with Respect to String Bases than BF3? Inorg. Chem. 2003, 42, 7990–7994. [Google Scholar] [CrossRef]
- Jonas, V.; Frenking, G.; Reetz, M.T. Comparative Theoretical Study of Lewis Acid-Base Complexes of BH3, BF3, BCl3, AlCl3, and SO2. J. Am. Chem. Soc. 1994, 116, 8741–8753. [Google Scholar] [CrossRef]
- Van der Veken, B.J.; Sluyts, E.J. Reversed Lewis Acidity of Mixed Boron Halides: An Infrared Study of the Van der Waals Complexes of BFxCly with CH3F in Cryosolution. J. Am. Chem. Soc. 1997, 119, 11516–11522. [Google Scholar] [CrossRef]
- Fau, S.; Frenking, G. Theoretical investigation of the weakly bonded donor—Acceptor complexes X3B-H2, X3B-C2H4, and X3B-C2H2 (X = H, F, Cl). Mol. Phys. 1999, 96, 519–527. [Google Scholar]
- Grabowski, S.J. Hydrogen Bond and Other Lewis Acid—Lewis Base Interactions as Preliminary Stages of Chemical Reactions. Molecules 2020, 25, 4668. [Google Scholar] [CrossRef] [PubMed]
- Richardson, T.B.; de Gala, S.; Crabtree, R.H. Unconventional Hydrogen Bonds: Intermolecular B-H…H-N Interactions. J. Am. Chem. Soc. 1995, 117, 12875–12876. [Google Scholar] [CrossRef]
- Klooster, W.T.; Koetzle, T.F.; Siegbahn, P.E.M.; Richardson, T.B.; Crabtree, R.H. Study of the N−H···H−B Dihydrogen Bond Including the Crystal Structure of BH3NH3 by Neutron Diffraction. J. Am. Chem. Soc. 1999, 121, 6337–6343. [Google Scholar] [CrossRef]
- Grabowski, S.J. Dihydrogen bond and X-H⋯σ interaction as sub-classes of hydrogen bond. J. Phys. Org. Chem. 2013, 26, 452–459. [Google Scholar] [CrossRef]
- Szymczak, J.J.; Grabowski, S.J.; Roszak, S.; Leszczynski, J. H…σ interactions—An ab initio and ’atoms in molecules’ study. Chem. Phys. Lett. 2004, 393, 81–86. [Google Scholar] [CrossRef]
- Grabowski, S.J. A-H⋯σ Hydrogen Bonds: Dihydrogen and Cycloalkanes as Proton Acceptors. Chem. Phys. Chem. 2019, 20, 565–574. [Google Scholar] [CrossRef]
- Grabowski, S.J. Molecular Hydrogen as a Lewis Base in Hydrogen Bonds and Other Interactions. Molecules 2020, 25, 3294. [Google Scholar] [CrossRef]
- Hamilton, C.W.; Baker, R.T.; Staubitz, A.; Manners, I. B-N compounds for chemical hydrogen storage. Chem. Soc. Rev. 2009, 38, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Keaton, R.J.; Blacquiere, J.M.; Baker, R.T. Base Metal Catalyzed Dehydrogenation of Ammonia−Borane for Chemical Hydrogen Storage. J. Am. Chem. Soc. 2007, 129, 1844–1845. [Google Scholar] [CrossRef]
- Staubitz, A.; Besora, M.; Harvey, J.N.; Manners, I. Computational Analysis of Amine− Borane Adducts as Potential Hydrogen Storage Materials with Reversible Hydrogen Uptake. Inorg. Chem. 2008, 47, 5910–5918. [Google Scholar] [CrossRef]
- Raghavendra, B.; Arunan, E. Unpaired and σ Bond Electrons as H, Cl, and Li Bond Acceptors: An Anomalous One-Electron Blue-Shifting Chlorine Bond. J. Phys. Chem. A 2007, 111, 9699–9706. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge structural database. Acta Cryst. 2016, 72, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.; Allen, F.H.; Willett, P. The scientific impact of the Cambridge Structural Database: A citation-based study. J. Appl. Cryst. 2010, 43, 811–824. [Google Scholar] [CrossRef] [Green Version]
- Bondi, J. van der Waals Volumes and Radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]
- Kubas, G.J. Metal Dihydrogen and σ-Bond Complexes—Structure, Theory, and Reactivity; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2001. [Google Scholar]
- Crabtree, R.H. The Organometallic Chemistry of the Transition Metals; John Wiley & Sons. Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Kubas, G.J. Dihydrogen complexes as prototypes for the coordination chemistry of saturated molecules. Proc. Natl. Acad. Sci. USA 2007, 104, 6901–6907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, C.C. Single Crystal Neutron Diffraction from Molecular Materials; World Scientific Publishing, Co. Pre. Ltd.: Singapore, 2000. [Google Scholar]
- Luger, P. Modern X-ray Analysis on Single Crystals, 2nd ed.; Walter de Gruyter: Berlin, Germany, 2014. [Google Scholar]
- Grabowski, S.J. Hydrogen bonds with BF4− anion as a proton acceptor. Crystals 2020, 10, 460. [Google Scholar] [CrossRef]
- Galano, A.; Alvarez-Idaboy, J.R.; Vivier-Bunge, A. Non-alkane behavior of cyclopropane and its derivatives: Characterization of unconventional hydrogen bond interactions. Theor. Chem. Acc. 2007, 118, 597–606. [Google Scholar] [CrossRef]
- Piela, L. Ideas of Quantum Chemistry; Elsevier Science Publishers: Amsterdam, The Netherlands, 2007; pp. 684–691. [Google Scholar]
- Boys, S.F.; Bernardi, F. The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol. Phys. 1970, 19, 553–561. [Google Scholar] [CrossRef]
- Rozas, I.; Alkorta, I.; Elguero, J. Unusual Hydrogen Bonds: H⋯π Interactions. J. Phys. Chem. A 1997, 101, 9457–9463. [Google Scholar] [CrossRef]
- Jeffrey, G.A.; Saenger, W. Hydrogen Bonding in Biological Structures; Springer: Berlin, Germany, 1991. [Google Scholar]
- Jeffrey, G.A. An Introduction to Hydrogen Bonding; Oxford University Press: New York, NY, USA, 1997. [Google Scholar]
- Pinchas, S. Infrared Absorption of Aldehydic C-H Group. Anal. Chem. 1955, 27, 2–6. [Google Scholar] [CrossRef]
- Pinchas, S. Infrared Absorption of Aldehydic C-H Group. Ortho-Substituted Benzaldehydes. Anal. Chem. 1957, 29, 334–339. [Google Scholar] [CrossRef]
- Hobza, P.; Havlas, Z. Blue-shifting hydrogen bonds. Chem. Rev. 2000, 100, 4253–4264. [Google Scholar] [CrossRef]
- Gu, Y.; Kar, T.; Scheiner, S. Fundamental Properties of the CH···O Interaction: Is It a True Hydrogen Bond? J. Am. Chem. Soc. 1999, 121, 9411–9422. [Google Scholar] [CrossRef]
- Kryachko, E.S. Neutral Blue-Shifting and Blue-Shifted Hydrogen Bonds. In Hydrogen Bonding—New Insights; Grabowski, S.J., Ed.; Springer: Berlin, Germany, 2006. [Google Scholar]
- Zhou, J.-W.; Jiang, Y.-J.; Guo, M.; Hu, G.-X.; Zhang, B.; Liu, H.-C.; Yu, Q.-S. Ab Initio Study of the Complexes of Halogen-Containing Molecules RX (X = Cl, Br, and I) and NH3: Towards Understanding the Nature of Halogen Bonding and the Electron-Accepting Propensities of Covalently Bonded Halogen Atoms. Chem. Eur. J. 2005, 11, 740–751. [Google Scholar] [CrossRef]
- Zierkiewicz, W.; Bieńko, D.C.; Michalska, D.; Zeegers-Huyskens, T. Theoretical Investigation of the Halogen Bonded Complexes Between Carbonyl Bases and Molecular Chlorine. J. Comput. Chem. 2015, 36, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Reed, E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
- Weinhold, F.; Landis, C. Valency and Bonding, a Natural Bond Orbital Donor—Acceptor Perspective; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Bader, R.F.W. Atoms in Molecules, A Quantum Theory; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Matta, C.; Boyd, R.J. (Eds.) Quantum Theory of Atoms in Molecules: Recent Progress in Theory and Application; Wiley-VCH Weinheim: Berlin, Germany, 2007. [Google Scholar]
- Poater, J.; Solá, M.; Bickelhaupt, F.M. Hydrogen–Hydrogen Bonding in Planar Biphenyl, Predicted by Atoms-In-Molecules Theory, Does Not Exist. Chem. Eur. J. 2006, 12, 2889–2896. [Google Scholar] [CrossRef] [PubMed]
- Poater, J.; Solá, M.; Bickelhaupt, F.M. A Model of the Chemical Bond Must Be Rooted in Quantum Mechanics, Provide Insight, and Possess Predictive Power. Chem. Eur. J. 2006, 12, 2902–2905. [Google Scholar] [CrossRef]
- Bader, R.F.W. Pauli repulsions exist only in the eye of the beholder. Chem. Eur. J. 2006, 12, 2896–2901. [Google Scholar] [CrossRef] [PubMed]
- Bader, R.F.W. A bond path: A universal indicator of bonded interactions. J. Phys. Chem. A 1998, 102, 7314–7323. [Google Scholar] [CrossRef]
- Bader, R.F.W. Bond paths are not chemical bonds. J. Phys. Chem. A 2009, 113, 10391–10396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espinosa, E.; Molins, E.; Lecomte, C. Hydrogen bond strengths revealed by topological analyses of experimentally observed electron densities. Chem. Phys. Lett. 1998, 285, 170–173. [Google Scholar] [CrossRef]
- Espinosa, E.; Molins, E. Retrieving interaction potentials from the topology of the electron density distribution: The case of hydrogen bonds. J. Chem. Phys. 2000, 113, 5686–5694. [Google Scholar] [CrossRef]
- Parthasarathi, R.; Subramanian, V.; Sathyamurthy, N. Hydrogen bonding without borders: An atoms-in-molecules perspective. J. Phys. Chem. A 2006, 110, 3349–3351. [Google Scholar] [CrossRef] [PubMed]
- Grabowski, S.J. Ab Initio Calculations on Conventional and Unconventional Hydrogen Bonds—Study of the Hydrogen Bond Strength. J. Phys. Chem. A 2001, 105, 10739–10746. [Google Scholar] [CrossRef]
- Ziegler, T.; Rauk, A. CO, CS, N2, PF3, and CNCH3 as σ Donors and π Acceptors. A Theoretical Study by the Hartree-Fock-Slater Transition-State Method. Inorg. Chem. 1979, 18, 1755–1759. [Google Scholar] [CrossRef]
- Velde, G.T.E.; Bickelhaupt, F.M.; Baerends, E.J.; Guerra, C.F.; van Gisbergen, S.J.A.; Snijders, J.G.; Ziegler, T. Chemistry with ADF. J. Comput. Chem. 2001, 22, 931–967. [Google Scholar] [CrossRef]
- Grabowski, S.J.; Sokalski, W.A.; Dyguda, E.; Leszczynski, J. Quantitative classification of covalent and noncovalent H-bonds. J. Phys. Chem. B 2006, 110, 6444–6446. [Google Scholar] [CrossRef]
- Grabowski, S.J. What is the Covalency of Hydrogen Bonding? Chem. Rev. 2011, 11, 2597–2625. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 16, Revision A.03; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Minenkov, Y.; Singstad, A.; Occhipinti, G.; Jensen, V.R. The accuracy of DFT-optimized geometries of functional transition metal compounds: A validation study of catalysts for olefin metathesis and other reactions in the homogeneous phase. Dalton Trans. 2012, 41, 5526–5541. [Google Scholar] [CrossRef]
- Burns, L.A.; Vázquez-Mayagoitia, A.; Sumpter, B.G.; Sherrill, C.D. Density-functional approaches to noncovalent interactions: A comparison of dispersion corrections (DFT-D), exchange-hole dipole moment (XDM) theory, and specialized functionals. J. Chem. Phys. 2011, 134, 084107. [Google Scholar] [CrossRef]
- Kendall, R.A.; Dunning, T.H., Jr.; Harrison, R.J. Electron Affinities of the First-Row Atoms Revisited. Systematic Basis Sets and Wave Functions. J. Chem. Phys. 1992, 96, 6796–6806. [Google Scholar] [CrossRef] [Green Version]
- Peterson, K.A.; Figgen, D.; Goll, E.; Stoll, H.; Dolg, M. Systematically convergent basis sets with relativistic pseudopotentials. II. Small-core pseudopotentials and correlation consistent basis sets for the post-d group 16–18 elements. J. Chem. Phys. 2003, 119, 11113. [Google Scholar] [CrossRef] [Green Version]
- Peterson, K.A. Systematically convergent basis sets with relativistic pseudopotentials. I. Correlation consistent basis sets for the post-d group 13–15 elements. J. Chem. Phys. 2003, 119, 11099. [Google Scholar] [CrossRef] [Green Version]
- Jensen, F. Introduction to Computational Chemistry, 2nd ed.; John Wiley & Sons Ltd.: Chichester, UK, 2007. [Google Scholar]
- Keith, T.A. AIMAll; Version 11.08.23; TK Gristmill Software: Overland Park, KS, USA, 2011. [Google Scholar]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Perdew, J.P. Density-functional approximation for the correlation energy of the inhomogeneous electron gas. Phys. Rev. B 1986, 33, 8822–8824. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [Green Version]
- Van Lenthe, E.; Baerends, E.J. Optimized Slater-type basis sets for the elements 1-118. J. Comput. Chem. 2003, 24, 1142–1156. [Google Scholar] [CrossRef]
- Baerends, E.J.; Ziegler, T.; Atkins, A.J.; Autschbach, J.; Baseggio, O.; Bashford, D.; Bérces, A.; Bickelhaupt, F.M.; Bo, C.; Boerrigter, P.M.; et al. ADF2019, SCM, Theoretical Chemistry; Vrije Universiteit: Amsterdam, The Netherlands, 2019; Available online: http://www.scm.com (accessed on 7 July 2021).
- Glendening, E.D.; Landis, C.-R.; Weinhold, F. NBO 6.0: Natural bond orbital analysis program. J. Comput. Chem. 2013, 34, 1429–1437. [Google Scholar] [CrossRef] [PubMed]



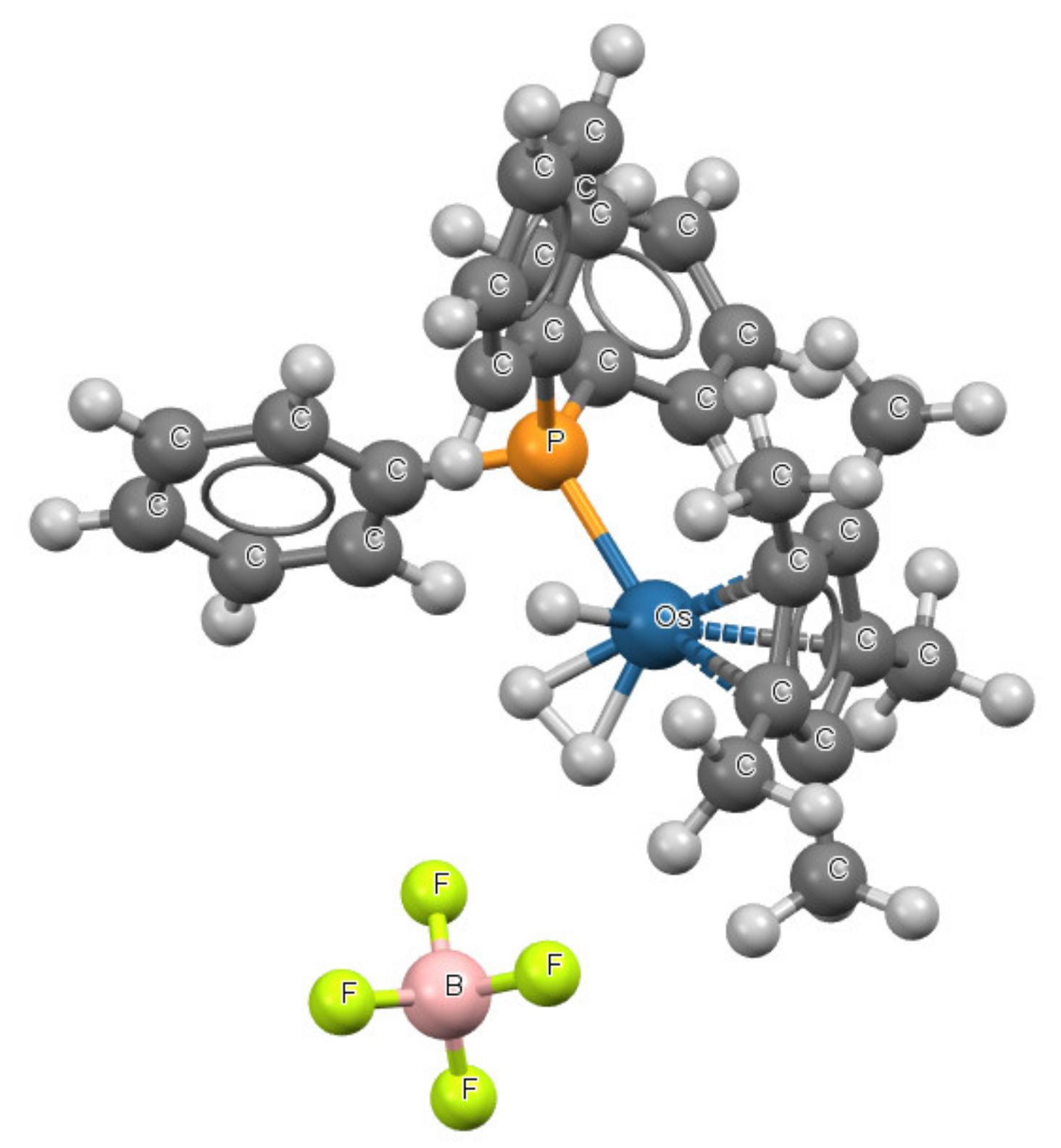

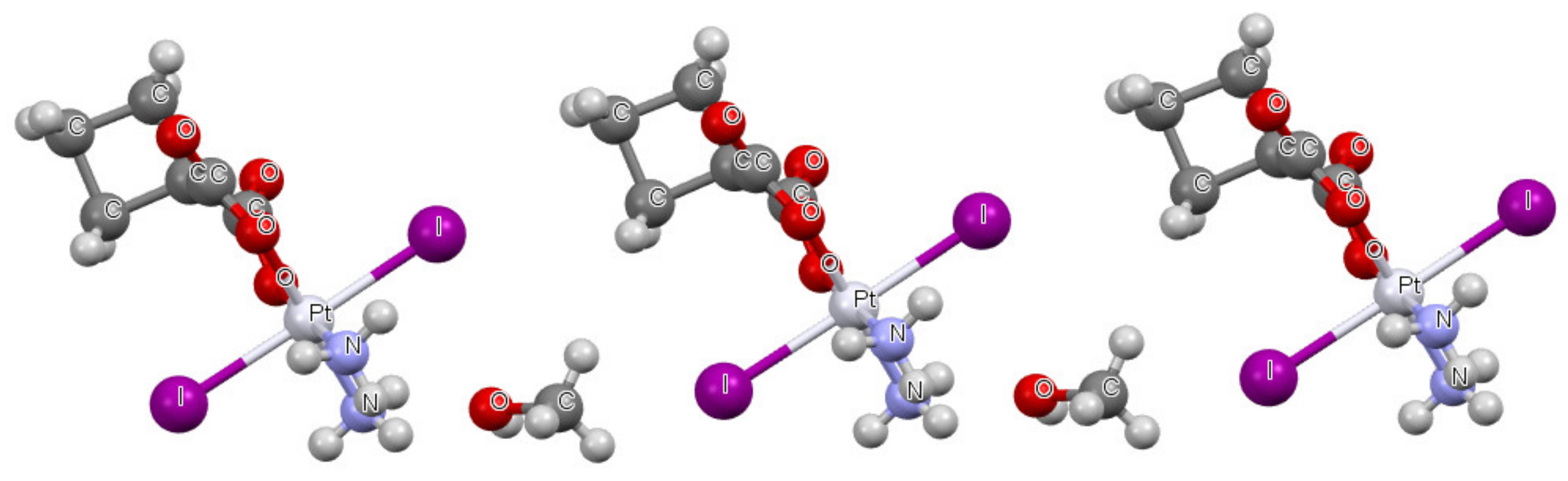

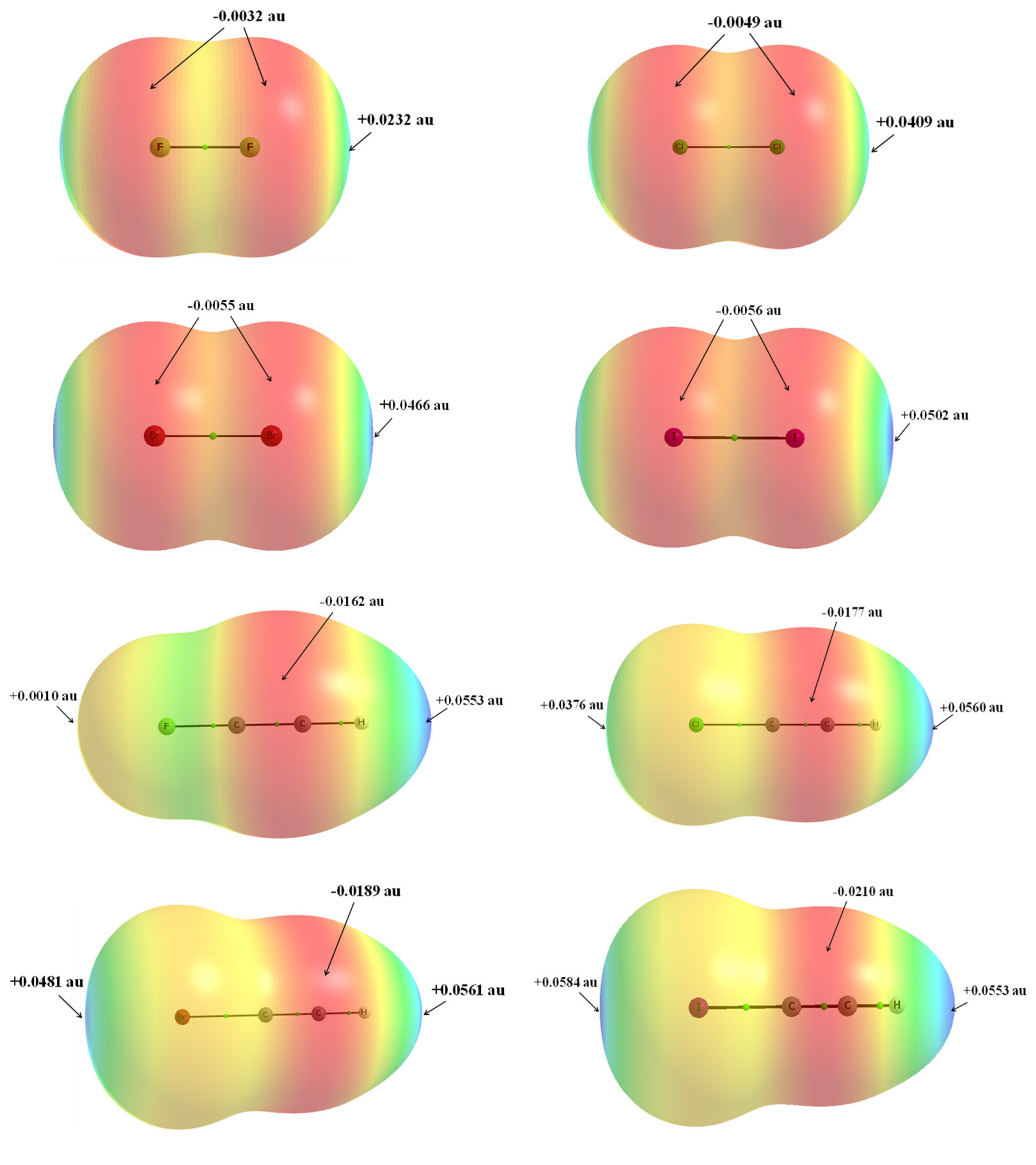
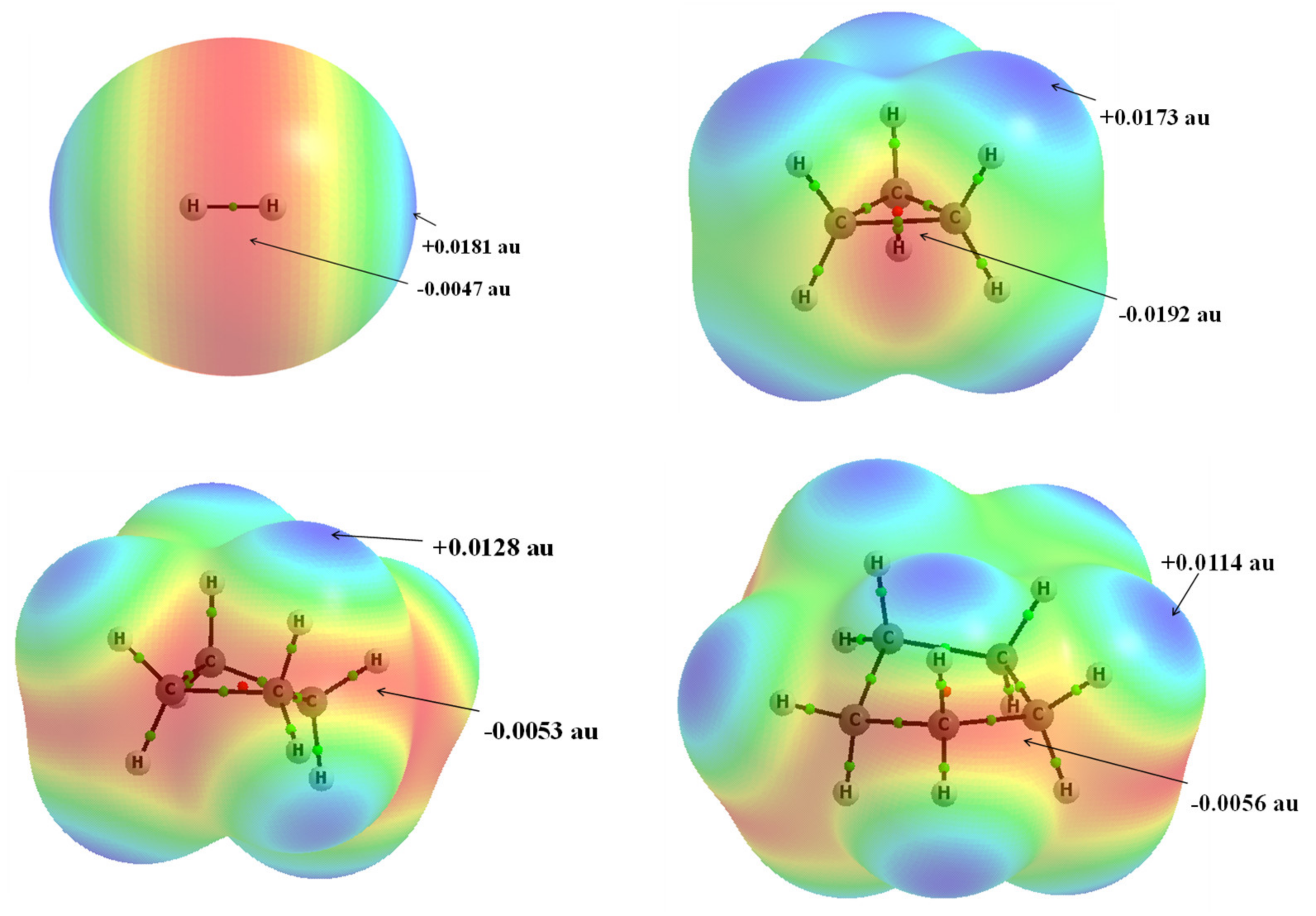

| Refcode | Max e.s.d. | A–X | X···σ | α | β |
|---|---|---|---|---|---|
| Dihydrogen as an electron donor | |||||
| NOLJIH | 0.01 | B-F | 2.432 | 86.4 | 113.7 |
| NOLJIH01 | 0.01 | B-F | 2.539 | 83.8 | 125.7 |
| NOLJIH02 | 0.01 | B-F | 2.432 | 86.4 | 113.6 |
| XAXMIT | 0.01 | B-F | 2.467 | 87.6 | 111.6 |
| Cyclopropane ring as an electron donor | |||||
| HEDQAL | 0.005 | C-F | 2.941 | 85.3 | 149.5 |
| HEDQIT | 0.005 | C-F | 2.940 | 85.3 | 149.5 |
| HIGNAO02 | 0.03 | Al-Br | 3.410 | 87.5 | 145.5 |
| KIKLIA | 0.01 | C-Br | 3.332 | 89.6 | 159.7 |
| OMIREI | 0.03 | C-Br | 3.430 | 89.4 | 157.1 |
| PILSEK | 0.005 | C-F | 3.038 | 89.9 | 127.1 |
| QUSREC | 0.01 | C-F | 3.038 | 89.9 | 148.6 |
| RUNQEW | 0.005 | C–Cl | 3.262 | 88.7 | 150.1 |
| SUTVOU | 0.005 | C-Br | 3.372 | 89.2 | 165.9 |
| UFIYEP | 0.005 | C-F | 2.982 | 87.0 | 126.4 |
| Cyclobutane ring as an electron donor | |||||
| DEZDUH | 0.01 | C-F | 3.025 | 89.9 | 133.5 |
| EQIXAF | 0.03 | P-F | 3.012 | 89.9 | 123.8 |
| EQIXIN | 0.03 | P-F | 2.989 | 89.9 | 125.5 |
| GEGDUU | 0.01 | C-F | 3.054 | 89.6 | 159.7 |
| GOGXOP | 0.005 | C-F | 2.993 | 88.4 | 147.7 |
| HEXYIV | 0.005 | C-F | 2.971 | 89.4 | 122.7 |
| PUFSOA | 0.005 | C-F | 3.011 | 89.6 | 155.8 |
| QASKAX | 0.03 | Pt-I | 3.551 | 89.1 | 189.4 |
| QUBGOJ | 0.01 | B-F | 3.012 | 90.0 | 165.4 |
| SITZOK | 0.005 | C-F | 3.022 | 89.8 | 153.5 |
| TOHDAY | 0.005 | C–Cl | 3.265 | 89.1 | 165.1 |
| TORPOX | 0.005 | C-F | 3.004 | 89.1 | 146.5 |
| YAMGAU01 | 0.005 | C-F | 3.001 | 88.6 | 151.0 |
| Cyclopentane ring as an electron donor | |||||
| XACHOC | 0.005 | C-F | 3.020 | 89.3 | 153.2 |
| HAFQEN | 0.005 | C-F | 3.024 | 89.6 | 149.4 |
| NARBEQ | 0.005 | C–Cl | 3.364 | 89.9 | 170.9 |
| VANNUT | 0.01 | B-F | 3.036 | 90.0 | 148.0 |
| Complex | Eint1 | Eint2 | X···σ | α | β |
|---|---|---|---|---|---|
| H2-FCCH | −0.09 | −0.10 | 3.482 | 86.0 | 179.6 |
| H2-ClCCH | −0.23 | −0.33 | 3.474 | 90.0 | 180.0 |
| H2-BrCCH | −0.38 | −0.48 | 3.295 | 89.9 | 179.9 |
| H2-ICCH | −0.66 | −0.62 | 3.248 | 87.2 | 177.3 |
| H2-F2 | −0.13 | −0.18 | 3.240 | 89.7 | 179.6 |
| H2-Cl2 | −0.31 | −0.48 | 3.134 | 90.0 | 180.0 |
| H2-Br2 | −0.52 | −0.57 | 3.110 | 89.9 | 179.8 |
| H2-I2 | −0.72 | −0.60 | 3.223 | 89.8 | 180.0 |
| C3H6-FCCH | −0.37 | −0.48 | 3.486 | 90.0 | 180.0 |
| C3H6-ClCCH | −1.16 | −1.63 | 3.394 | 90.0 | 180.0 |
| C3H6-BrCCH | −1.86 | −2.14 | 3.377 | 90.0 | 180.0 |
| C3H6-ICCH | −2.79 | −2.79 | 3.445 | 90.0 | 180.0 |
| C3H6-F2 | −0.57 | −0.89 | 3.165 | 89.9 | 179.9 |
| C3H6-Cl2 | −1.64 | −2.15 | 3.193 | 90.0 | 179.6 |
| C3H6-Br2 | −2.36 | −2.63 | 3.241 | 89.8 | 179.3 |
| C3H6-I2 | −2.97 | −2.97 | 3.399 | 89.4 | 179.4 |
| C4H8-FCCH | −0.45 | −0.59 | 3.455 | 90.0 | 179.9 |
| C4H8-ClCCH | −0.85 | −1.30 | 3.539 | 90.0 | 180.0 |
| C4H8-BrCCH | −1.30 | −1.62 | 3.525 | 90.0 | 179.9 |
| C4H8-ICCH | −1.92 | −1.99 | 3.610 | 89.9 | 179.8 |
| C4H8-F2 | −0.45 | −0.64 | 3.390 | 87.2 | 165.5 |
| C4H8-Cl2 | −0.98 | −1.53 | 3.409 | 88.3 | 170.9 |
| C4H8-Br2 | −1.47 | −1.83 | 3.449 | 89.9 | 179.4 |
| C4H8-I2 | −1.99 | −2.07 | 3.600 | 89.9 | 179.3 |
| C5H10-ClCCH | −0.89 | −1.32 | 3.571 | 89.7 | 179.9 |
| C5H10-BrCCH | −1.36 | −1.64 | 3.564 | 87.8 | 171.4 |
| C5H10-ICCH | −2.01 | −2.03 | 3.643 | 86.9 | 171.0 |
| C5H10-Cl2 | −1.04 | −1.54 | 3.470 | 85.5 | 164.9 |
| C5H10-Br2 | −1.55 | −1.85 | 3.522 | 84.8 | 164.9 |
| C5H10-I2 | −2.10 | −2.13 | 3.642 | 85.5 | 168.0 |
| Complex | QLb | σHH/CC→σAX * | ρBCP |
|---|---|---|---|
| H2-FCCH | 0.00 | 0 | 0.001 |
| H2-ClCCH | 0.00 | 0.11 | 0.002 |
| H2-BrCCH | 0.00 | 0.29 | 0.004 |
| H2-ICCH | 0.00 | 0.57 | 0.006 |
| H2-F2 | 0.00 | 0.06 | 0.001 |
| H2-Cl2 | 0.00 | 0.34 | 0.004 |
| H2-Br2 | 0.01 | 0.55 | 0.006 |
| H2-I2 | 0.01 | 0.1 | 0.006 |
| C3H6-FCCH | 0.00 | 0 | 0.002 |
| C3H6-ClCCH | 0.00 | 0.06 | 0.005 |
| C3H6-BrCCH | 0.01 | 0.12 | 0.006 |
| C3H6-ICCH | 0.01 | 0.26 | 0.007 |
| C3H6-F2 | 0.01 | 0.07 | 0.004 |
| C3H6-Cl2 | 0.02 | 0.29 | 0.007 |
| C3H6-Br2 | 0.03 | 0.41 | 0.008 |
| _C3H6-I2 | 0.02 | 0.37 | 0.008 |
| Complex | ΔEint | ΔEPauli | ΔEelstat | ΔEorb | ΔEdisp | % ΔEelstat | % ΔEorb | % ΔEdisp |
|---|---|---|---|---|---|---|---|---|
| C3H6-BrCCH | −2.23 | 2.97 | −1.79 | −1.26 | −2.16 | 34 | 24 | 41 |
| C3H6-ICCH | −3.47 | 4.13 | −2.53 | −2.23 | −2.83 | 33 | 29 | 37 |
| C3H6-Cl2 | −2.41 | 3.48 | −2.06 | −2.01 | −1.83 | 35 | 34 | 31 |
| C3H6-Br2 | −3.32 | 4.34 | −2.57 | −2.64 | −2.45 | 34 | 34 | 32 |
| C3H6-I2 | −4.08 | 4.88 | −2.88 | −3.10 | −2.97 | 32 | 35 | 33 |
| C4H8-ICCH | −2.60 | 2.83 | −1.17 | −1.49 | −2.76 | 22 | 27 | 51 |
| C4H8-Br2 | −1.93 | 2.60 | −1.06 | −1.16 | −2.31 | 23 | 26 | 51 |
| C4H8-I2 | −2.78 | 3.01 | −1.25 | −1.70 | −2.84 | 22 | 29 | 49 |
| C5H10-ICCH | −2.73 | 2.95 | −1.23 | −1.58 | −2.88 | 22 | 28 | 51 |
| C5H10-Br2 | −2.11 | 2.76 | −1.15 | −1.37 | −2.35 | 24 | 28 | 48 |
| C5H10-I2 | −3.00 | 3.24 | −1.36 | −1.93 | −2.95 | 22 | 31 | 47 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grabowski, S.J. A–X⋯σ Interactions—Halogen Bonds with σ-Electrons as the Lewis Base Centre. Molecules 2021, 26, 5175. https://doi.org/10.3390/molecules26175175
Grabowski SJ. A–X⋯σ Interactions—Halogen Bonds with σ-Electrons as the Lewis Base Centre. Molecules. 2021; 26(17):5175. https://doi.org/10.3390/molecules26175175
Chicago/Turabian StyleGrabowski, Sławomir J. 2021. "A–X⋯σ Interactions—Halogen Bonds with σ-Electrons as the Lewis Base Centre" Molecules 26, no. 17: 5175. https://doi.org/10.3390/molecules26175175






