Fluorescent Liquid Tetrazines
Abstract
1. Introduction
2. Materials and Methods
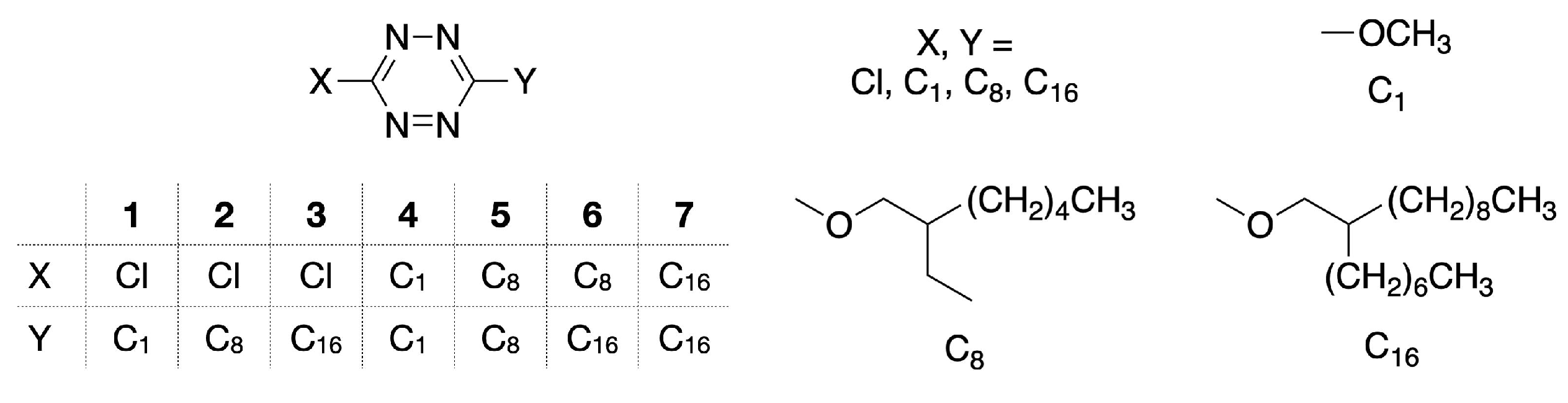
2.1. Synthesis and Characterization
2.2. Rheometry
2.3. Steady State Spectra
2.4. Time-Resolved Fluorescence
2.5. Nanosecond Time-Resolved Spectroscopy
2.6. Picosecond Time-Resolved Absorption
2.7. Computation
3. Results
3.1. Rheology
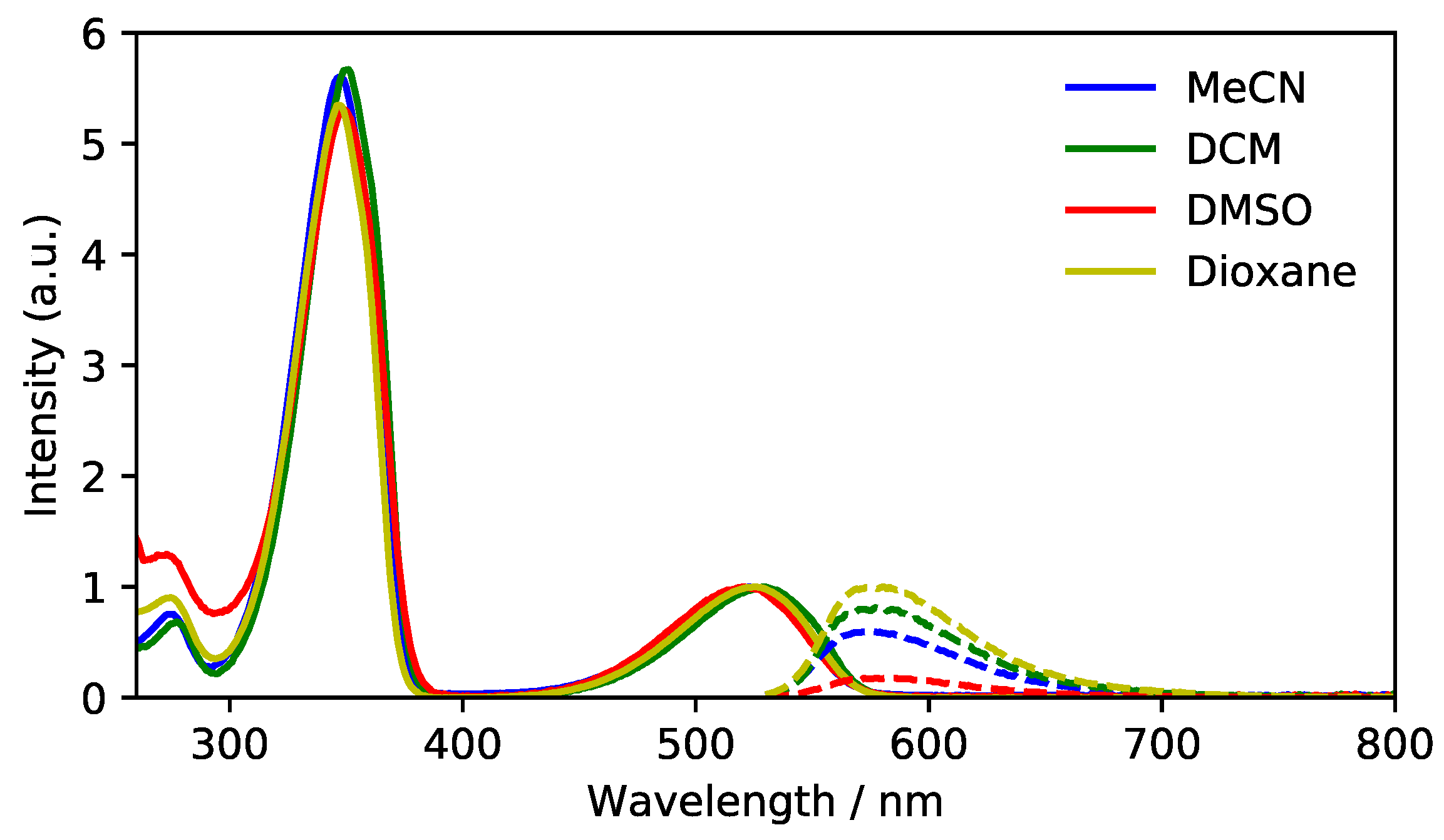
3.2. Absorption and Fluorescence Spectroscopy
3.3. Nanosecond Transient Absorption
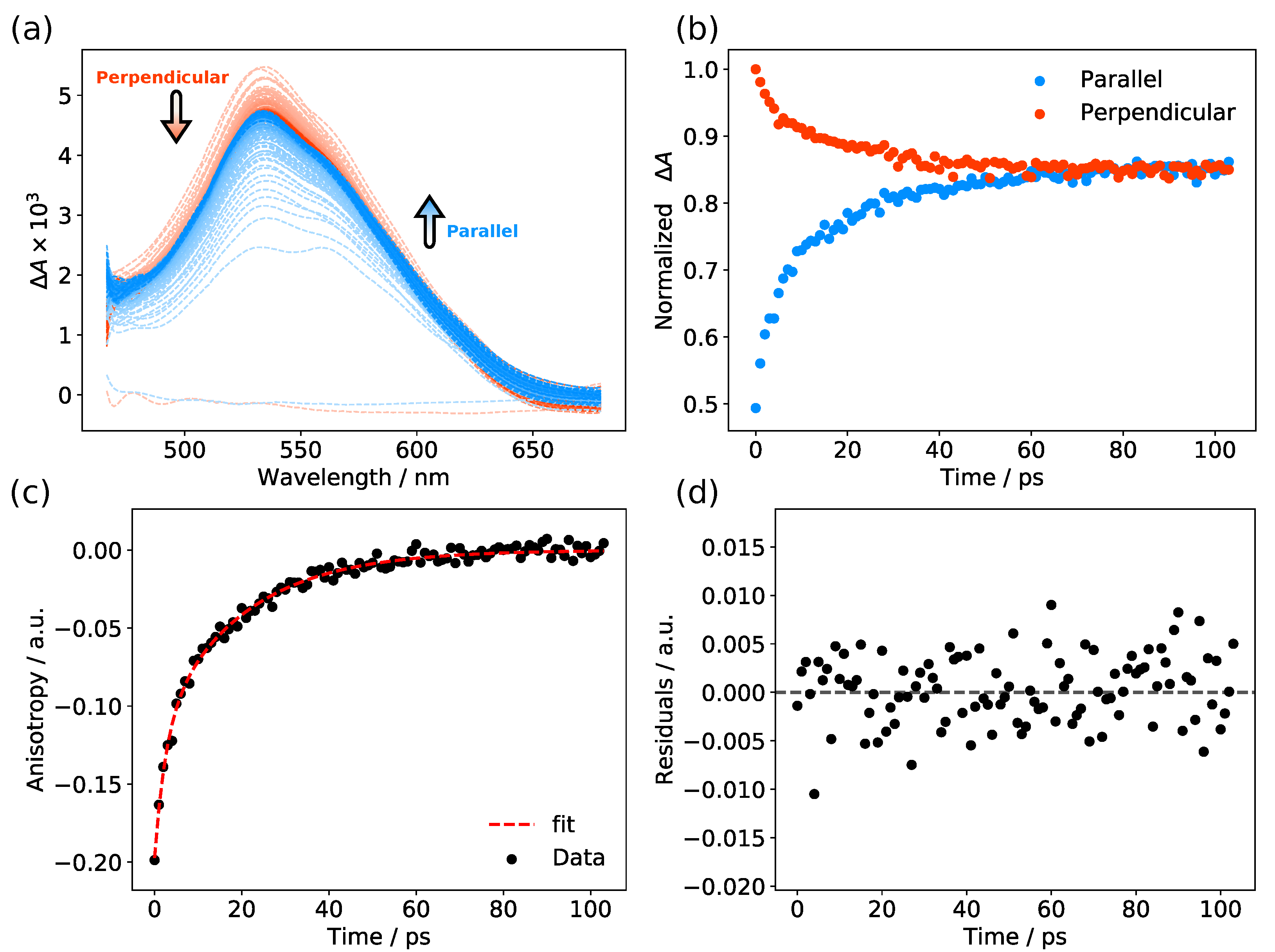
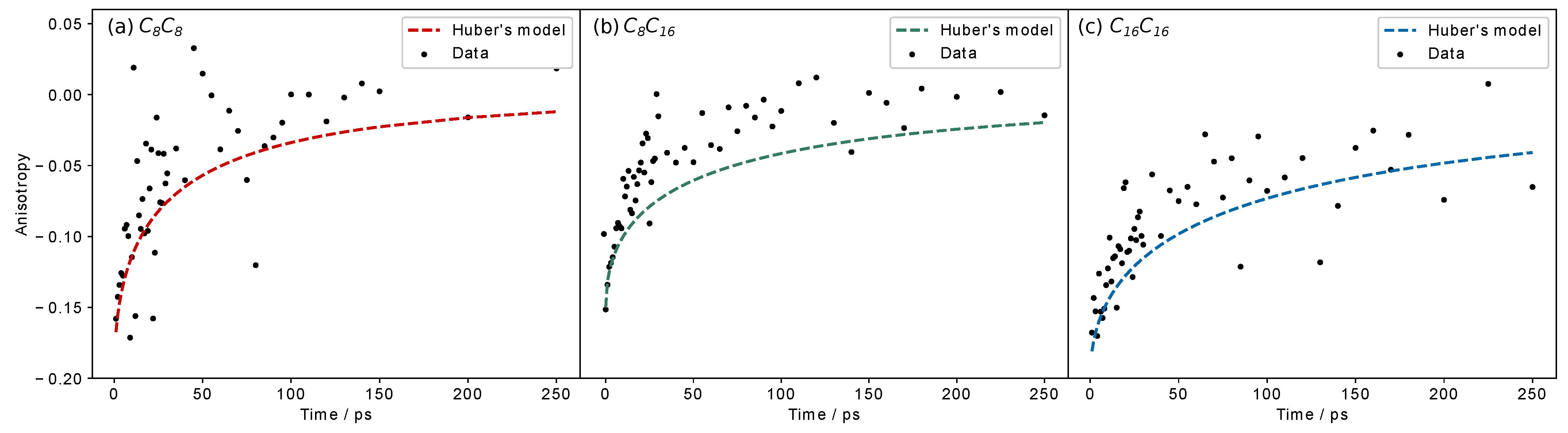
3.4. Picosecond Transient Absorption
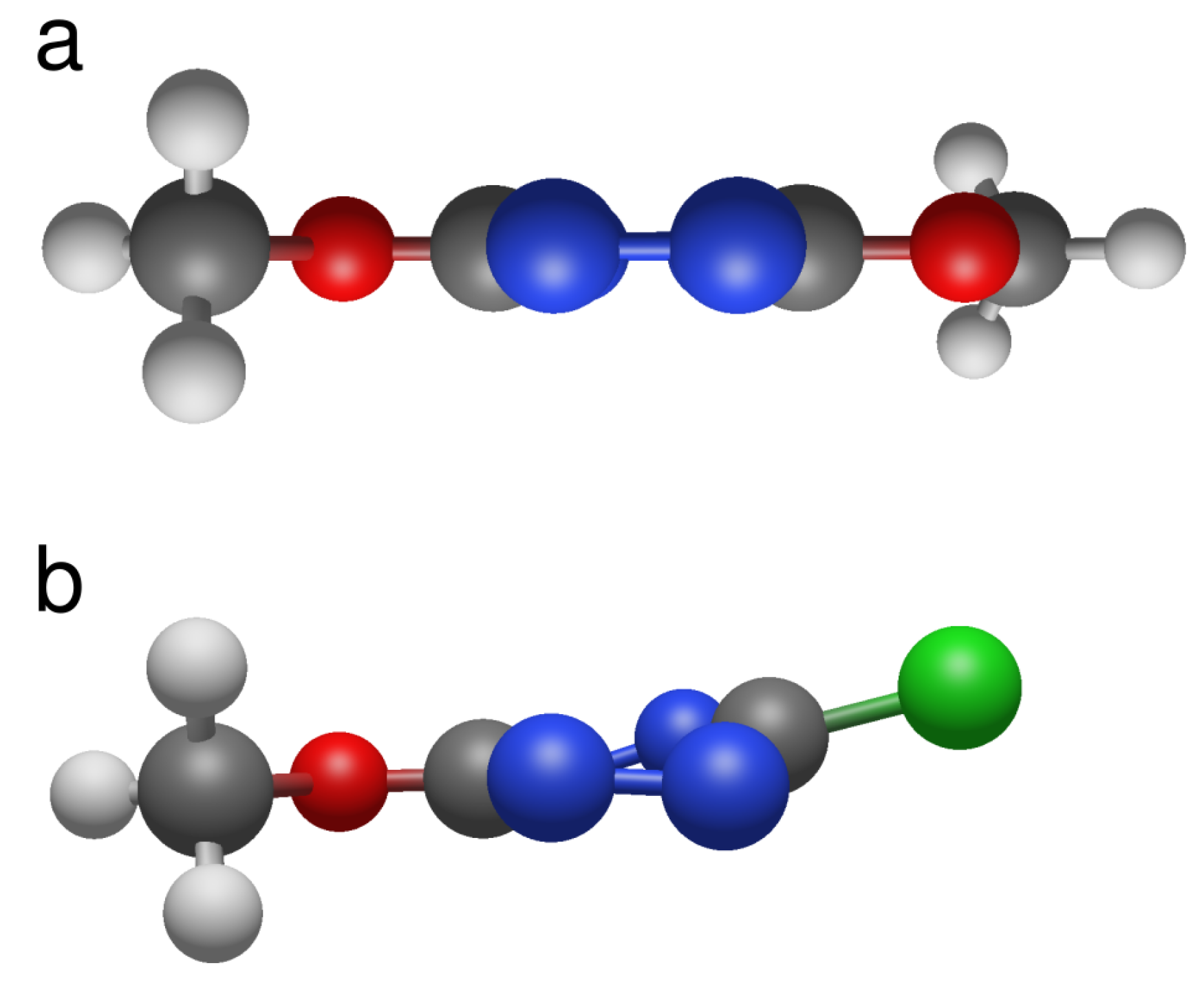
3.5. Computation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Babu, S.S.; Aimi, J.; Ozawa, H.; Shirahata, N.; Saeki, A.; Seki, S.; Ajayaghosh, A.; Möhwald, H.; Nakanishi, T. Solvent-Free Luminescent Organic Liquids. Angew. Chem. Int. Ed. 2012, 51, 3391–3395. [Google Scholar] [CrossRef]
- Ghosh, A.; Nakanishi, T. Frontiers of solvent-free functional molecular liquids. Chem. Commun. 2017, 53, 10344–10357. [Google Scholar] [CrossRef]
- Lu, F.; Nakanishi, T. Solvent-Free Luminous Molecular Liquids. Adv. Opt. Mater. 2019, 7, 1900176. [Google Scholar] [CrossRef]
- Varghese, S.; Das, S. Role of Molecular Packing in Determining Solid-State Optical Properties of π-Conjugated Materials. J. Phys. Chem. Lett. 2011, 2, 863–873. [Google Scholar] [CrossRef]
- Kushwaha, K.; Yu, L.; Stranius, K.; Singh, S.; Hultmark, S.; Iqbal, M.; Eriksson, L.; Johnston, E.; Erhart, P.; Müller, C.; et al. A Record Chromophore Density in High-Entropy Liquids of Two Low-Melting Perylenes: A New Strategy for Liquid Chromophores. Adv. Sci. 2019, 6, 1801650. [Google Scholar] [CrossRef]
- Schäfer, C.; Mony, J.; Olsson, T.; Börjesson, K. Entropic Mixing Allows Monomeric-Like Absorption in Neat BODIPY Films. Chem. Eur. J. 2020, 26, 14295–14299. [Google Scholar] [CrossRef]
- Gierschner, J.; Mack, H.G.; Oelkrug, D.; Waldner, I.; Rau, H. Modeling of the Optical Properties of Cofacial Chromophore Pairs: Stilbenophane. J. Phys. Chem. A 2004, 108, 257–263. [Google Scholar] [CrossRef]
- Diana, R.; Panunzi, B. Zinc (II) and AIEgens: The “Clip Approach” for a Novel Fluorophore Family. A Review. Molecules 2021, 26, 4176. [Google Scholar] [CrossRef]
- Würthner, F. Aggregation-Induced Emission (AIE): A Historical Perspective. Angew. Chem. Int. Ed. 2020, 59, 14192–14196. [Google Scholar] [CrossRef]
- Gong, Y.H.; Miomandre, F.; Méallet-Renault, R.; Badré, S.; Galmiche, L.; Tang, J.; Audebert, P.; Clavier, G. Synthesis and Physical Chemistry of s-Tetrazines: Which Ones are Fluorescent and Why? Eur. J. Org. Chem. 2009, 2009, 6121–6128. [Google Scholar] [CrossRef]
- Allain, C.; Piard, J.; Brosseau, A.; Han, M.; Paquier, J.; Marchandier, T.; Lequeux, M.; Boissière, C.; Audebert, P. Fluorescent and Electroactive Low-Viscosity Tetrazine-Based Organic Liquids. ACS Appl. Mater. Interfaces 2016, 8, 19843–19846. [Google Scholar] [CrossRef]
- Petrova, D.; Weber, B.; Allain, C.; Audebert, P.; Bonn, D.; Brouwer, A.M. Fast 3D Microscopy Imaging of Contacts Between Surfaces Using a Fluorescent Liquid. ACS Appl. Mater. Interfaces 2018, 10, 40973–40977. [Google Scholar] [CrossRef]
- Petrova, D.; Weber, B.; Allain, C.; Audebert, P.; Venner, C.H.; Brouwer, A.M.; Bonn, D. Fluorescence microscopy visualization of the roughness-induced transition between lubrication regimes. Sci. Adv. 2019, 5, eaaw4761. [Google Scholar] [CrossRef]
- Chowdhury, M.; Goodman, L. Fluorescence of s-Tetrazine. J. Chem. Phys. 1962, 36, 548–549. [Google Scholar] [CrossRef]
- Meyling, J.; Van Der Werf, R.; Wiersma, D. Excited state geometry of and radiationless processes in the lowest B3u(nπ*) singlet state of s-tetrazine. Chem. Phys. Lett. 1974, 28, 364–372. [Google Scholar] [CrossRef][Green Version]
- King, D.S.; Denny, C.T.; Hochstrasser, R.M.; Smith, A.B. The photochemical decomposition of 1,4-s-tetrazine-15N2. J. Am. Chem. Soc. 1977, 99, 271–273. [Google Scholar] [CrossRef]
- Gückel, F.; Maki, A.; Neugebauer, F.; Schweitzer, D.; Vogler, H. Spectroscopic investigations of the lowest triplet state of s-tetrazines. Chem. Phys. 1992, 164, 217–227. [Google Scholar] [CrossRef]
- Ben Châabane, R.; Guermazi, R.; Clavier, G.; Audebert, P.; Hedhli, A. Synthesis and characterization of new fluorinated tetrazines displaying a high fluorescence yield. Dyes Pigm. 2014, 108, 64–69. [Google Scholar] [CrossRef]
- Ghosh, S.; Chowdhury, M. S1 (n,π*), T1 (n,π*) and S2(n,π*) emissions in 3,6-diphenyl-s-tetrazine. Chem. Phys. Lett. 1982, 85, 233–238. [Google Scholar] [CrossRef]
- Plugge, M.; Alain-Rizzo, V.; Audebert, P.; Brouwer, A.M. Excited state dynamics of 3,6-diaryl-1,2,4,5-tetrazines. Experimental and theoretical studies. J. Photochem. Photobiol. A Chem. 2012, 234, 12–20. [Google Scholar] [CrossRef]
- Savastano, M.; García-Gallarín, C.; López de la Torre, M.D.; Bazzicalupi, C.; Bianchi, A.; Melguizo, M. Anion-π and lone pair-π interactions with s-tetrazine-based ligands. Coord. Chem. Rev. 2019, 397, 112–137. [Google Scholar] [CrossRef]
- Telfer, S.G.; McLean, T.M.; Waterland, M.R. Exciton coupling in coordination compounds. Dalton Trans. 2011, 40, 3097–3108. [Google Scholar] [CrossRef] [PubMed]
- Clavier, G.; Audebert, P. s-Tetrazines as Building Blocks for New Functional Molecules and Molecular Materials. Chem. Rev. 2010, 110, 3299–3314. [Google Scholar] [CrossRef] [PubMed]
- Würth, C.; Grabolle, M.; Pauli, J.; Spieles, M.; Resch-Genger, U. Relative and absolute determination of fluorescence quantum yields of transparent samples. Nat. Protoc. 2013, 8, 1535–1550. [Google Scholar] [CrossRef] [PubMed]
- Szabelski, M.; Ilijev, D.; Sarkar, P.; Luchowski, R.; Gryczynski, Z.; Kapusta, P.; Erdmann, R.; Gryczynski, I. Collisional Quenching of Erythrosine B as a Potential Reference Dye for Impulse Response Function Evaluation. Appl. Spectrosc. 2009, 63, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Snellenburg, J.J.; Laptenok, S.P.; Seger, R.; Mullen, K.M.; Stokkum, I.H.M.v. Glotaran: A Java-Based Graphical User Interface for the R Package TIMP. J. Stat. Softw. 2012, 49, 1–22. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian˜16 Revision C.01; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Aquilante, F.; Autschbach, J.; Baiardi, A.; Battaglia, S.; Borin, V.A.; Chibotaru, L.F.; Conti, I.; De Vico, L.; Delcey, M.; Fdez. Galván, I.; et al. Modern quantum chemistry with [Open] Molcas. J. Chem. Phys. 2020, 152, 214117. [Google Scholar] [CrossRef]
- He, T.; Ren, C.; Luo, Y.; Wang, Q.; Li, J.; Lin, X.; Ye, C.; Hu, W.; Zhang, J. Water-soluble chiral tetrazine derivatives: Towards the application of circularly polarized luminescence from upper-excited states to photodynamic therapy. Chem. Sci. 2019, 10, 4163–4168. [Google Scholar] [CrossRef]
- Miomandre, F.; Allain, C.; Clavier, G.; Audibert, J.F.; Pansu, R.B.; Audebert, P.; Hartl, F. Coupling thin layer electrochemistry with epifluorescence microscopy: An expedient way of investigating electrofluorochromism of organic dyes. Electrochem. Commun. 2011, 13, 574–577. [Google Scholar] [CrossRef]
- Birch, D.J.S.; Yip, P. Nanometrology. In Fluorescence Spectroscopy and Microscopy; Engelborghs, Y., Visser, A.J., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2014; Volume 1076, pp. 279–302. [Google Scholar]
- Marazzi, M.; Mai, S.; Roca-Sanjuán, D.; Delcey, M.G.; Lindh, R.; González, L.; Monari, A. Benzophenone Ultrafast Triplet Population: Revisiting the Kinetic Model by Surface-Hopping Dynamics. J. Phys. Chem. Lett. 2016, 7, 622–626. [Google Scholar] [CrossRef]
- Baba, M. Intersystem Crossing in the 1nπ* and 1ππ* States. J. Phys. Chem. A 2011, 115, 9514–9519. [Google Scholar] [CrossRef] [PubMed]
- Marian, C.M. Understanding and Controlling Intersystem Crossing in Molecules. Annu. Rev. Phys. Chem. 2021, 72, 617–640. [Google Scholar] [CrossRef] [PubMed]
- Huber, D.L. Dynamics of Incoherent Transfer. In Laser Spectroscopy of Solids; Yen, W.M., Selzer, P.M., Eds.; Topics in Applied Physics; Springer: Berlin/Heidelberg, Germany, 1981; pp. 83–111. [Google Scholar]
- Valeur, B.; Berberan-Santos, M.N. Molecular Fluorescence: Principles and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2021. [Google Scholar]
- Braslavsky, S.E.; Fron, E.; Rodriguez, H.B.; San Román, E.; Scholes, G.D.; Schweitzer, G.; Valeur, B.; Wirz, J. Pitfalls and limitations in the practical use of Förster’s theory of resonance energy transfer. Photochem. Photobiol. Sci. 2008, 7, 1444–1448. [Google Scholar] [CrossRef] [PubMed]

| Liquid | Viscosity [mPa·s] |
|---|---|
| Solvent | ||||||
|---|---|---|---|---|---|---|
| DCM | 0.069 c | 33 | 34 | 28 | ||
| neat | 0.08 d | 38 | 2.1 d | 24 d | ||
| neat | 0.068 e | 33 e | 2.1 e | 28e | ||
| DCM | 0.080 c | 36 | 35 | 26 | ||
| neat | 0.09 d | 43 | 2.1 d | |||
| DCM | 0.33 e | 158e | 2.1 e | 4.2 e | ||
| neat | 0.021 e | 4.6 e | 4.6 e | 213 e | ||
| DCM | 0.068 c | 32 | 37 | 29 | ||
| MeCN | 0.052 c | 31 | 31 | |||
| Dioxane | 0.089 c | 45 | 20 | |||
| neat | 0.11 d | 52 | 2.1 d | 17 d | ||
| DCM | 0.37 c | 151 | ||||
| MeCN | 0.34 c | |||||
| Dioxane | 0.40 c | |||||
| neat | 0.10 d | 48 | 2.1 d | 21 d |
| 32 | 44 | 1.08 | |
| 30 | 40 | 0.95 | |
| 27 | 25 | 0.73 | |
| 124 | 9 | 7.6 |
| 529 | 572 | 507 | |
| 529 | 572 | 507 | |
| 529 | 572 | 507 | |
| 521 | 527 | 494 |
| Solvent | Å | ||||
|---|---|---|---|---|---|
| DCM | 383 | 0.04 | |||
| neat | 3.65 | ||||
| DCM | 530 | 0.06 | |||
| neat | 4.57 | ||||
| DCM | 677 | 0.07 | |||
| Dioxane | 0.20 | ||||
| neat | 6.06 | ||||
| DCM | 388 | 0.04 |
| 0.2 | 9.7 | 0.0 | |
| 0.2 | 9.7 | 0.0 | |
| 1.1 | 12.5 | 0.0 | |
| 0.2 | 7.2 | 2.9 | |
| 0.2 | 7.2 | 3.3 | |
| 0.8 | 12.0 | 4.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paradiz Dominguez, M.; Demirkurt, B.; Grzelka, M.; Bonn, D.; Galmiche, L.; Audebert, P.; Brouwer, A.M. Fluorescent Liquid Tetrazines. Molecules 2021, 26, 6047. https://doi.org/10.3390/molecules26196047
Paradiz Dominguez M, Demirkurt B, Grzelka M, Bonn D, Galmiche L, Audebert P, Brouwer AM. Fluorescent Liquid Tetrazines. Molecules. 2021; 26(19):6047. https://doi.org/10.3390/molecules26196047
Chicago/Turabian StyleParadiz Dominguez, Maximilian, Begüm Demirkurt, Marion Grzelka, Daniel Bonn, Laurent Galmiche, Pierre Audebert, and Albert M. Brouwer. 2021. "Fluorescent Liquid Tetrazines" Molecules 26, no. 19: 6047. https://doi.org/10.3390/molecules26196047
APA StyleParadiz Dominguez, M., Demirkurt, B., Grzelka, M., Bonn, D., Galmiche, L., Audebert, P., & Brouwer, A. M. (2021). Fluorescent Liquid Tetrazines. Molecules, 26(19), 6047. https://doi.org/10.3390/molecules26196047








