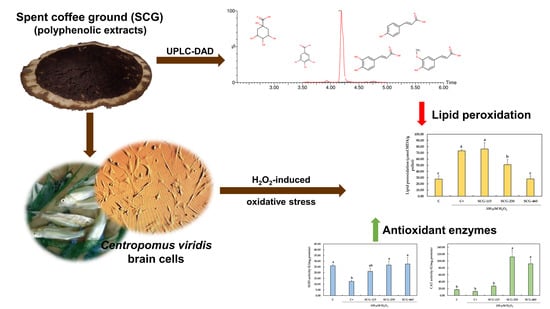
Polyphenolic Extracts from Spent Coffee Grounds Prevent H2O2-Induced Oxidative Stress in Centropomus viridis Brain Cells
Abstract
:1. Introduction
2. Results
2.1. Phenolic Compounds and Antioxidant Capacity of Polyphenolic Extracts from SCG
2.2. Non-Toxic Concentrations of Polyphenolic Extracts from SCG on C. viridis Brain Cells
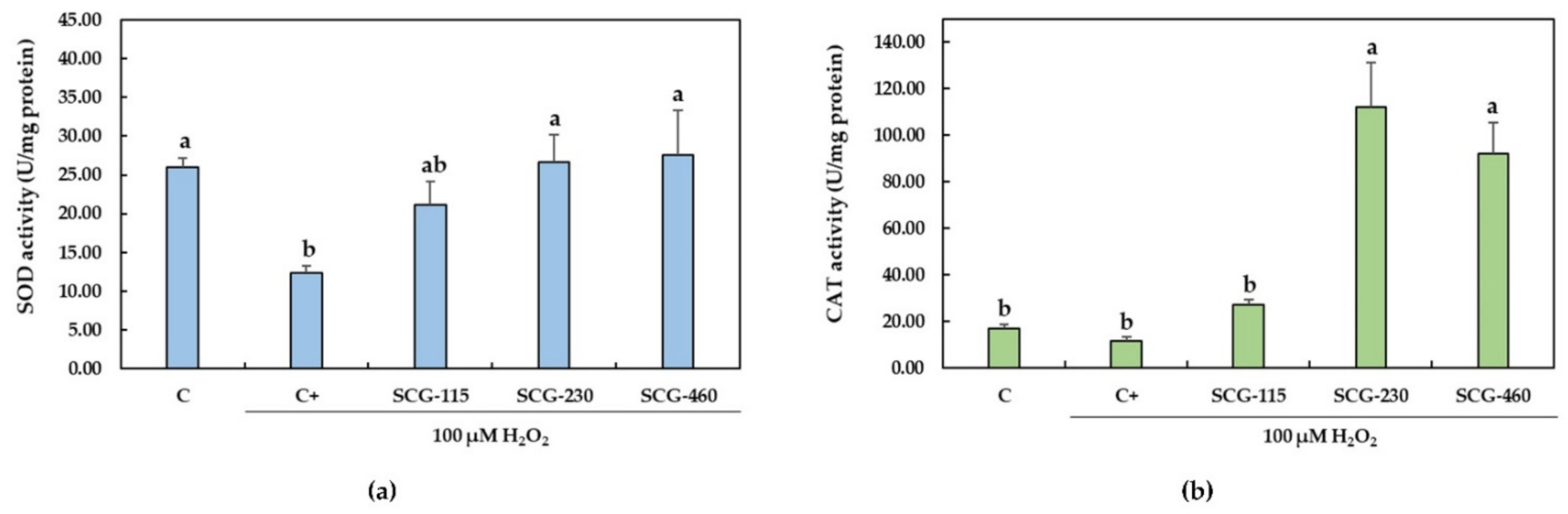
2.3. Protective Effect of Polyphenolic Extracts from SCG against H2O2-Induced Oxidative Stress
3. Discussion
4. Materials and Methods
4.1. Collection of Spent Coffe Grounds (SCG) and Preparation of the Polyphenolic Extracts
4.2. Total Phenolic Content (TPC) Determination
4.3. Quantification of Individual Phenolic Compounds in Spent Coffee Grounds by Ultraperformance Liquid Chromatography (UPLC)
4.4. Antioxidant Capacity of Polyphenolic Extracts from SCG
4.4.1. 2,2-Diphenyl-1-picrylhydrazyl Free Radical (DPPH•) Scavenging Activity Assay
4.4.2. Ferric Ion Reducing Antioxidant Power (FRAP) Assay
4.4.3. Oxygen Radical Absorbance Capacity (ORAC) Assay
4.5. Cell Culture
4.6. Determination of the Non-Toxic Concentrations of Spent Coffee Ground Polyphenolic Extracts. Thiazolyl Blue Tetrazolium Bromide (MTT) Cytotoxicity Assay
4.7. H2O2-Induced Oxidative Stress Trial
- C: control, non-treated cells.
- C+: cells treated with 100 µM H2O2 alone.
- SCG: cells pretreated with the spent coffee ground polyphenolic extracts at 115, 230 and 460 µg/mL, and treated with 100 µM H2O2.
4.8. Assay for Lipid Peroxidation
4.9. Assay for Antioxidant Enzymes
4.9.1. Superoxide Dismutase Activity
4.9.2. Catalase Activity
4.9.3. Concentration of Protein
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- FAO. The State of World Fisheries and Aquaculture 2020—Sustainability in Action; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020; p. 224. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2016—Contributing to Food Security and Nutrition for All; Food and Agriculture Organization of the United Nations: Rome, Italy, 2016; p. 200. [Google Scholar]
- Birnie-Gauvin, K.; Costantini, D.; Cooke, S.J.; Willmore, W.G. A comparative and evolutionary approach to oxidative stress in fish: A review. Fish Fish. 2017, 18, 928–942. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef] [Green Version]
- Lushchak, V.I. Environmentally induced oxidative stress in aquatic animals. Aquat. Toxicol. 2011, 101, 13–30. [Google Scholar] [CrossRef]
- Leyva-López, N.; Lizárraga-Velázquez, C.E.; Hernández, C.; Sánchez-Gutiérrez, E.Y. Exploitation of agro-industrial waste as potential source of bioactive compounds for aquaculture. Foods 2020, 9, 843. [Google Scholar] [CrossRef]
- Stevenson, D.E.; Hurst, R.D. Polyphenolic phytochemicals—Just antioxidants or much more? Cell. Mol. Life Sci. 2007, 64, 2900–2916. [Google Scholar] [CrossRef]
- Xu, J.; Gan, S.; Li, J.; Wand, D.-B.; Chen, Y.; Hu, X.; Yang, G.-Z. Garcinia xanthochymus extract protects PC12 cells from H2O2-induced apoptosis through modulation of PI3K/AKT and NRF2/HO-1 pathways. Chin. J. Nat. Med. 2017, 15, 825–833. [Google Scholar] [CrossRef]
- Cheng, D.; Wang, G.; Wang, X.; Tang, J.; Yu, Q.; Zhang, X.; Wang, S. Neuro-protection of chlorogenic acid against Al-induced apoptosis in PC12 cells via modulation of Al metabolism and Akt/GSK-3β pathway. J. Funct. Foods 2020, 70, 103984. [Google Scholar] [CrossRef]
- Santana, P.A.; Jara-Gutiérrez, C.; Mellado, M.; Forero, J.C.; Guzmán, F.; Barriga, A.; Albericio, F.; Álvarez, C.A. Effects of elderflower extract enriched with polyphenols on antioxidant defense of salmon leukocytes. Electron. J. Biotechnol. 2021, 52, 13–20. [Google Scholar] [CrossRef]
- Belščak-Cvitanović, A.; Komes, D. Chapter 4—Extraction and formulation of bioactive compounds. In Handbook of Coffee Processing By-Products, 1st ed.; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 93–140. [Google Scholar] [CrossRef]
- Choi, B.; Koh, E. Spent coffee as a rich source of antioxidative compounds. Food Sci. Biotechnol. 2017, 26, 921–927. [Google Scholar] [CrossRef]
- Arai, K.; Terashima, H.; Aizawa, S.; Taga, A.; Yamamoto, A.; Tsutsumiuchi, K.; Kodama, S. Simultaneous determination of trigonelline, caffeine, chlorogenic acid and their related compounds in instant coffee samples by HPLC using an acidic mobile phase containing octanesulfonate. Anal. Sci. 2015, 31, 831–835. [Google Scholar] [CrossRef] [Green Version]
- Ekambaram, P.; Narayanan, M.; Jayachandran, T. Changes in oxidative stress and antioxidant status in stressed fish brain. IJSR 2014, 3, 164–170. [Google Scholar]
- Bagnyukova, T.V.; Lushchak, O.V.; Storey, K.B.; Lushchak, V.I. Oxidative stress and antioxidant defense responses by goldfish tissues to acute change of temperature from 3 to 23 °C. J. Therm. Biol. 2007, 32, 227–234. [Google Scholar] [CrossRef]
- Clemens, J.A.; Panetta, J.A. 5—Free Radicals in Central Nervous System Diseases. In Immunopharmacology of Free Radical Species; Blake, D., Winyard, P.G., Eds.; Academic Press: London, UK, 1995; pp. 73–83. [Google Scholar] [CrossRef]
- Arriaga-Hernández, D.; Hernández, C.; Martínez-Montaño, E.; Ibarra-Castro, L.; Lizárraga-Velázquez, E.; Leyva-López, N.; Chávez-Sánchez, M.C. Fish meal replacement by soybean products in aquaculture feeds for white snook, Centropomus viridis: Effect on growth, diet digestibility, and digestive capacity. Aquaculture 2021, 530, 735823. [Google Scholar] [CrossRef]
- Freshney, R.I. Culture of specific cell types. In Culture of Animal Cells: A Manual of Basic Technique and Specialized Applications, 7th ed.; Freshney, R.I., Ed.; Wiley, John & Sons, Inc: Hoboken, NJ, USA, 2005; pp. 535–570. [Google Scholar]
- Avalos-Soriano, A.; De la Cruz-Cordero, R.; López-Martínez, F.J.; Rosado, J.L.; Duarte-Vázquez, M.Á.; Garcia-Gasca, T. Effect of a β-hydroxyphosphonate analogue of L-carnitine on insulin-sensitive and insulin-resistant 3T3-L1 adipocytes. Pharmacology 2015, 96, 99–106. [Google Scholar] [CrossRef]
- Castro, J.S.; Braz-Mota, S.; Campos, D.F.; Souza, S.S.; Val, A.L. High temperature, pH, and hypoxia cause oxidative stress and impair the spermatic performance of the amazon fish Colossoma macropomum. Front. Physiol. 2020, 11, 772. [Google Scholar] [CrossRef]
- Biller, J.D.; Takahashi, L.S. Oxidative stress and fish immune system: Phagocytosis and leukocyte respiratory burst activity. An. Acad. Bras. Cienc. 2018, 90, 3403–3414. [Google Scholar] [CrossRef] [Green Version]
- Mariotti-Celis, M.S.; Martínez-Cifuentes, M.; Huamán-Castilla, N.; Vargas-González, M.; Pedreschi, F.; Pérez-Correa, J.R. The antioxidant and safety properties of spent coffee ground extracts impacted by the combined hot pressurized liquid extraction–resin purification process. Molecules 2018, 23, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramón-Gonçalves, M.; Gómez-Mejía, E.; Rosales-Conrado, N.; León-González, M.E.; Madrid, Y. Extraction, identification and quantification of polyphenols from spent coffee grounds by chromatographic methods and chemometric analyses. Waste Manag. 2019, 96, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.; Juániz, I.; Monente, C.; Caemmerer, B.; Kroh, L.W.; De Peña, M.P.; Cid, C. Evaluation of spent coffee obtained from the most common coffeemakers as a source of hydrophilic bioactive compounds. J. Agric. Food Chem. 2012, 60, 12565–12573. [Google Scholar] [CrossRef] [PubMed]
- Osorio-Arias, J.; Delgado-Arias, S.; Cano, L.; Zapata, S.; Quintero, M.; Nuñez, H.; Ramírez, C.; Simpson, R.; Vega-Castro, O. Sustainable management and valorization of spent coffee grounds through the optimization of thin layer hot air-drying process. Waste Biomass Valoriz. 2020, 11, 5015–5026. [Google Scholar] [CrossRef]
- Campos-Vega, R.; Loarca-Piña, G.; Vergara-Castañeda, H.A.; Oomah, B.D. Spent coffee grounds: A review on current research and future prospects. Trends Food Sci. Technol. 2015, 45, 24–36. [Google Scholar] [CrossRef]
- Martini, D.; Del Bo’, C.; Tassotti, M.; Riso, P.; Del Rio, D.; Brighenti, F.; Porrini, M. Coffee consumption and oxidative stress: A review of human intervention studies. Molecules 2016, 21, 979. [Google Scholar] [CrossRef] [Green Version]
- Wei, F.; Tanokura, M. Chapter 17—Organic compounds in green coffee beans. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 149–162. [Google Scholar] [CrossRef]
- Angeloni, G.; Masella, P.; Guerrini, L.; Innocenti, M.; Bellumori, M.; Parenti, A. Application of a screening design to recover phytochemicals from spent coffee grounds. Food Bioprod. Process. 2019, 118, 50–57. [Google Scholar] [CrossRef]
- Magalhães, L.M.; Segundo, M.A.; Reis, S.; Lima, J.L.F.C. Methodological aspects about in vitro evaluation of antioxidant properties. Anal. Chim. Acta 2008, 613, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L. Oxygen radical absorbance capacity (ORAC): New horizons in relating dietary antioxidants/bioactives and health benefits. J. Funct. Foods 2015, 18, 797–810. [Google Scholar] [CrossRef]
- Abramovič, H. Chapter 93—Antioxidant properties of hydroxycinnamic acid derivatives: A focus on biochemistry, physicochemical parameters, reactive species, and biomolecular interactions. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 843–852. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Ransy, C.; Vaz, C.; Lombès, A.; Bouillaud, F. Use of H2O2 to cause oxidative stress, the catalase issue. Int. J. Mol. Sci. 2020, 21, 9149. [Google Scholar] [CrossRef]
- Desai, S.N.; Farris, F.F.; Ray, S.D. Lipid Peroxidation. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Academic Press: London, UK, 2014; pp. 89–93. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.-W.; Bai, J.-P.; Zhang, Q.; Hu, X.-L.; Tian, X.; Zhu, J.; Liu, J.; Meng, W.-H.; Zhao, Q.-C. Caffeoylquinic acid derivatives protect SH-SY5Y neuroblastoma cells from hydrogen peroxide-induced injury through modulating oxidative status. Cell. Mol. Neurobiol. 2017, 37, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.-W.; Bai, J.-P.; Zhang, Q.; Hu, X.-L.; Tian, X.; Zhu, J.; Liu, J.; Meng, W.-H.; Zhao, Q.-C. Erratum to: Caffeoylquinic acid derivatives protect SH-SY5Y neuroblastoma cells from hydrogen peroxide-induced injury through modulating oxidative status. Cell. Mol. Neurobiol. 2017, 37, 511–512. [Google Scholar] [CrossRef] [Green Version]
- Brewer, M.S. Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Bienert, G.P.; Møller, A.L.B.; Kristiansen, K.A.; Schulz, A.; Møller, I.M.; Schjoerring, J.K.; Jahn, T.P. Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes *. J. Biol. Chem. 2007, 282, 1183–1192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marinho, H.S.; Real, C.; Cyrne, L.; Soares, H.; Antunes, F. Hydrogen peroxide sensing, signaling and regulation of transcription factors. Redox Biol. 2014, 2, 535–562. [Google Scholar] [CrossRef] [Green Version]
- Rojo, A.I.; Sagarra, M.R.D.; Cuadrado, A. GSK-3β down-regulates the transcription factor Nrf2 after oxidant damage: Relevance to exposure of neuronal cells to oxidative stress. J. Neurochem. 2008, 105, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Taqvi, S.; Ahmed Bhat, E.; Sajjad, N.; Sabir, J.S.M.; Qureshi, A.; Rather, I.A.; Rehman, S. Protective effect of vanillic acid in hydrogen peroxide-induced oxidative stress in D.Mel-2 cell line. Saudi J. Biol. Sci. 2021, 28, 1795–1800. [Google Scholar] [CrossRef]
- Motohashi, H.; Yamamoto, M. Nrf2–Keap1 defines a physiologically important stress response mechanism. Trends. Mol. Med. 2004, 10, 549–557. [Google Scholar] [CrossRef]
- Boettler, U.; Sommerfeld, K.; Volz, N.; Pahlke, G.; Teller, N.; Somoza, V.; Lang, R.; Hofmann, T.; Marko, D. Coffee constituents as modulators of Nrf2 nuclear translocation and ARE (EpRE)-dependent gene expression. J. Nutr. Biochem. 2011, 22, 426–440. [Google Scholar] [CrossRef]
- Acevedo, F.; Rubilar, M.; Scheuermann, E.; Cancino, B.; Uquiche, E.; Garcés, M.; Inostroza, K.; Shene, C. Spent coffee grounds as a renewable source of bioactive compounds. J. Biobased Mater. Bioenergy 2013, 7, 420–428. [Google Scholar] [CrossRef]
- González-Gómez, D.; Cardoso, V.; Bohoyo, D.; Ayuso, M.C.; Delgado-Adamez, J. Application of experimental design and response surface methodology to optimize the procedure to obtain a bactericide and highly antioxidant aqueous extract from orange peels. Food Control 2014, 35, 252–259. [Google Scholar] [CrossRef]
- Swain, T.; Hillis, W.E. The phenolic constituents of Prunus domestica. I.—The quantitative analysis of phenolic constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- Gutiérrez-Grijalva, E.P.; Angulo-Escalante, M.A.; León-Félix, J.; Heredia, J.B. Effect of in vitro digestion on the total antioxidant capacity and phenolic content of 3 species of oregano (Hedeoma patens, Lippia graveolens, Lippia palmeri). J. Food Sci. 2017, 82, 2832–2839. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [Green Version]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Prior, R.L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Ferrari, M.; Fornasiero, M.C.; Isetta, A.M. MTT colorimetric assay for testing macrophage cytotoxic activity in vitro. J. Immunol. Methods 1990, 131, 165–172. [Google Scholar] [CrossRef]
- Chang, H.-T.; Jan, C.-R.; Liang, W.-Z. Protective effects of a phenolic glycoside compound curculigoside on H2O2-induced oxidative stress and cytotoxicity in normal human breast epithelial cells. J. Funct. Foods 2018, 41, 171–182. [Google Scholar] [CrossRef]
- Da-Wool, J.; Chi Heung, C.; Jong Suk, L.; Seung Hwan, L.; Taewan, K. Deastringent peel extracts of persimmon (Diospyros kaki Thunb. cv. Cheongdo-Bansi) protect neuronal PC-12 and SH-SY5Y cells against oxidative stress. J. Microbiol. Biotechnol. 2018, 28, 1094–1104. [Google Scholar] [CrossRef]
- Lizárraga-Velázquez, C.E.; Hernández, C.; González-Aguilar, G.A.; Heredia, J.B. Effect of dietary intake of phenolic compounds from mango peel extract on growth, lipid peroxidation and antioxidant enzyme activities in zebrafish (Danio rerio). Lat. Am. J. Aquat. Res. 2019, 47, 602–611. [Google Scholar] [CrossRef] [Green Version]
- Solé, M.; Potrykus, J.; Fernández-Díaz, C.; Blasco, J. Variations on stress defences and metallothionein levels in the Senegal sole, Solea senegalensis, during early larval stages. Fish Physiol. Biochem. 2005, 30, 57–66. [Google Scholar] [CrossRef]
- Aebi, H. Catalase. In Methods of Enzymatic Analysis, 2nd ed.; Bergmeyer, H.U., Ed.; Academic Press: Cambridge, MA, USA, 1974; Volume 2, pp. 673–684. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]


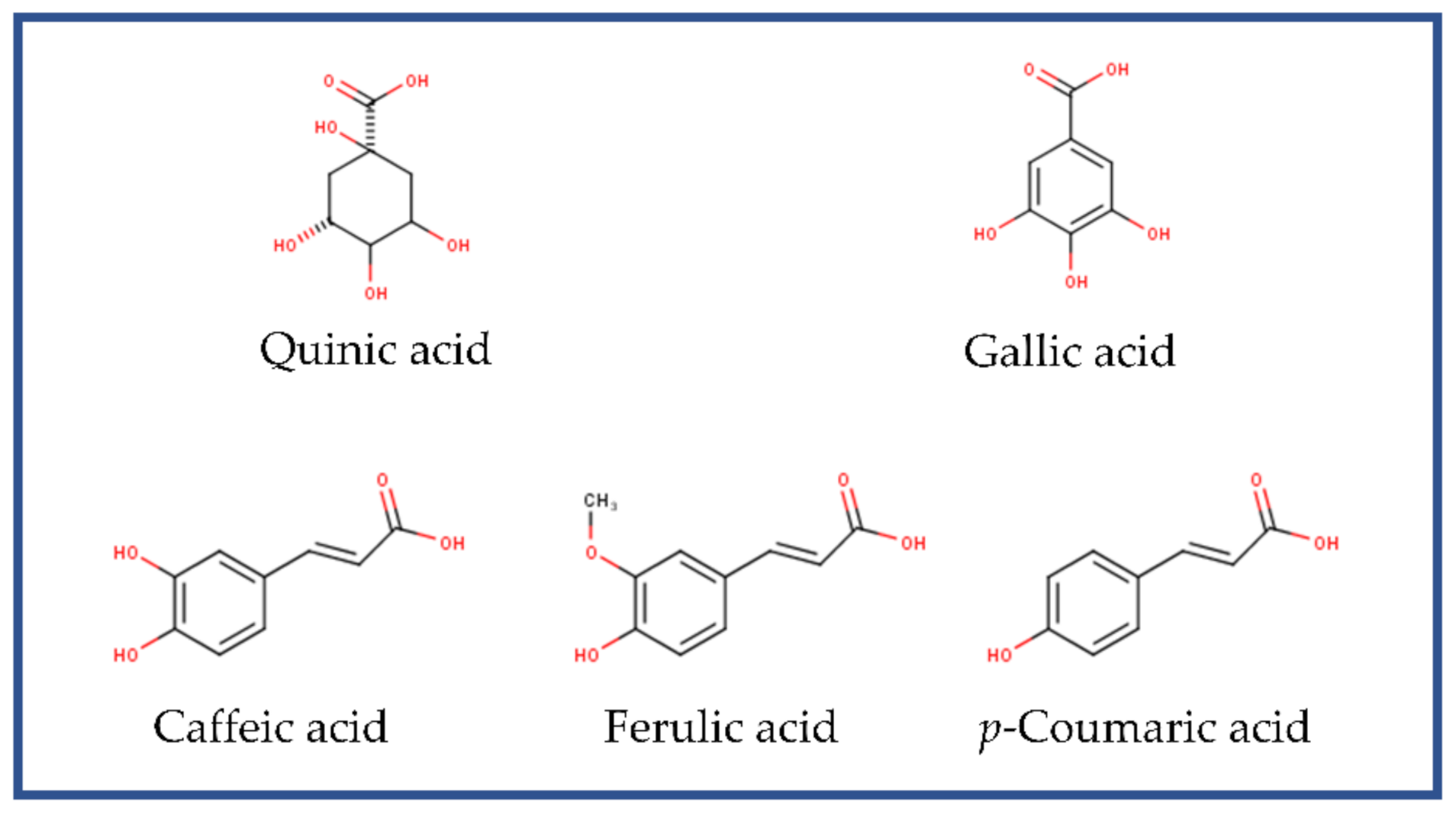
| Compound | Rt * | Concentration (mg/100 g D.W.) | Contribution (%) |
|---|---|---|---|
| Quinic acid | 0.9 | 656.43 ± 47.92 | 73.59 ± 5.37 |
| Gallic acid | 1.59 | 6.63 ± 0.80 | 0.74 ± 0.09 |
| Caffeic acid | 4.19 | 36.79 ± 10.06 | 4.12 ± 1.13 |
| Coumaric acid | 5.09 | 1.28 ± 0.33 | 0.14 ± 0.04 |
| Ferulic acid | 5.53 | 37.37 ± 3.64 | 4.19 ± 0.41 |
| DPPH | FRAP | ORAC |
|---|---|---|
| 3672.39 ± 602.12 1 | 428.29 ± 17.53 1 | 2417.42 ± 149.37 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leyva-López, N.; Peraza-Arias, M.; Avalos-Soriano, A.; Hernández, C.; Lizárraga-Velázquez, C.E.; Heredia, J.B. Polyphenolic Extracts from Spent Coffee Grounds Prevent H2O2-Induced Oxidative Stress in Centropomus viridis Brain Cells. Molecules 2021, 26, 6195. https://doi.org/10.3390/molecules26206195
Leyva-López N, Peraza-Arias M, Avalos-Soriano A, Hernández C, Lizárraga-Velázquez CE, Heredia JB. Polyphenolic Extracts from Spent Coffee Grounds Prevent H2O2-Induced Oxidative Stress in Centropomus viridis Brain Cells. Molecules. 2021; 26(20):6195. https://doi.org/10.3390/molecules26206195
Chicago/Turabian StyleLeyva-López, Nayely, Melissa Peraza-Arias, Anaguiven Avalos-Soriano, Crisantema Hernández, Cynthia E. Lizárraga-Velázquez, and J. Basilio Heredia. 2021. "Polyphenolic Extracts from Spent Coffee Grounds Prevent H2O2-Induced Oxidative Stress in Centropomus viridis Brain Cells" Molecules 26, no. 20: 6195. https://doi.org/10.3390/molecules26206195