Insect Protein Content Analysis in Handcrafted Fitness Bars by NIR Spectroscopy. Gaussian Process Regression and Data Fusion for Performance Enhancement of Miniaturized Cost-Effective Consumer-Grade Sensors
Abstract
:1. Introduction
2. Discussion
2.1. Sensor Suite
2.2. Spectra Pretreatment and Chemometrics
2.3. NIR Spectra of Intact and Prepared Insect Protein Bars
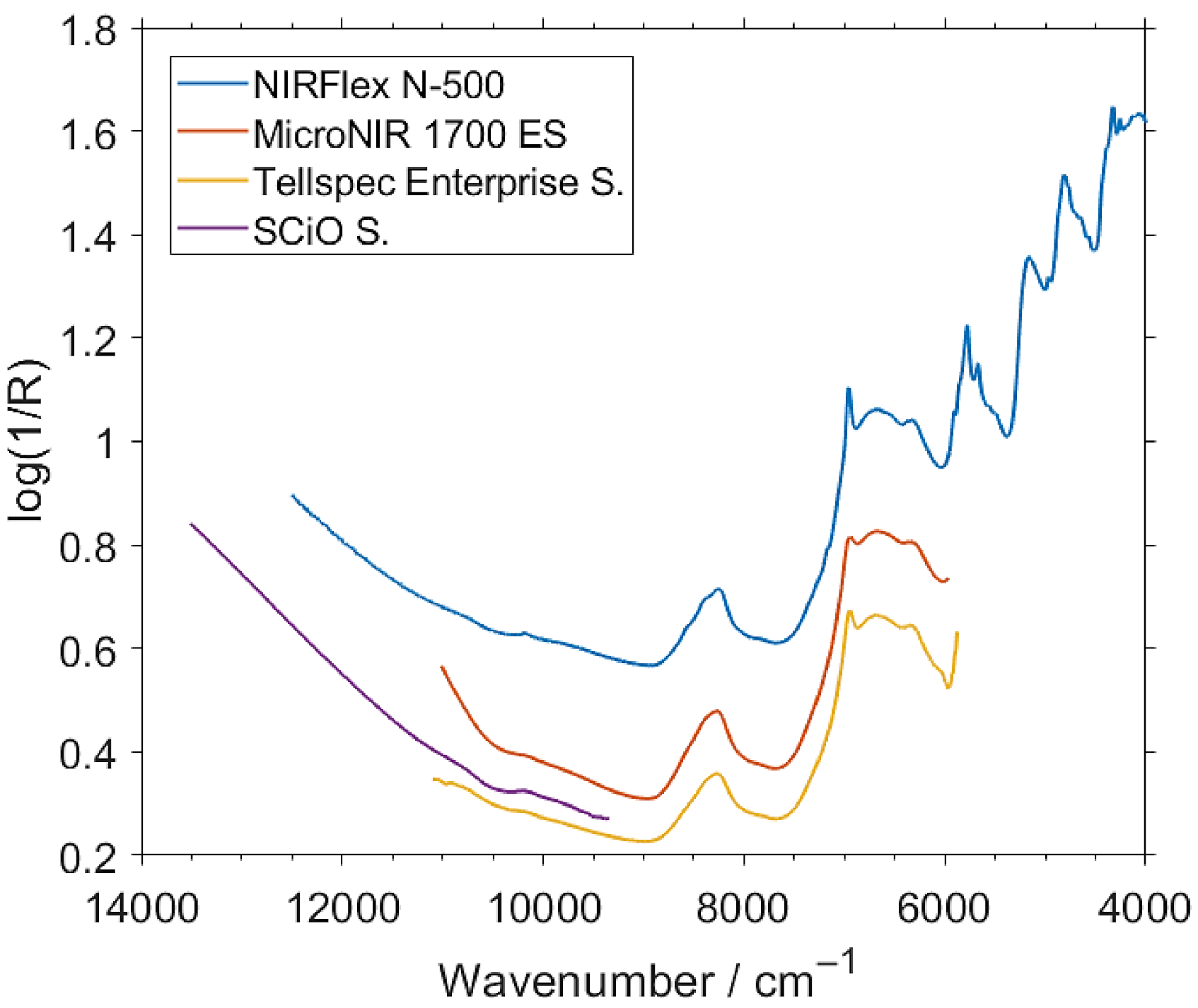
2.3.1. NIR Spectra of Intact Samples
2.3.2. NIR Spectra of Milled Samples
2.4. Comparison of the Analytical Performance of the Benchtop and Miniaturized NIR Spectrometers in the Prediction of the Total Protein Content in Insect Protein Bars
2.4.1. Intact Samples
2.4.2. Milled Samples
2.5. Performance Enhancement of the Cost-Effective Miniaturized NIR Spectrometers by Data Fusion
3. Materials and Methods
4. Summary
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Melgar-Lalanne, G.; Hernández-Álvarez, A.-J.; Salinas-Castro, A. Edible insects processing: Traditional and innovative technologies. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1166–1191. [Google Scholar] [CrossRef] [Green Version]
- De Castro, R.J.S.; Ohara, A.; Aguilar, J.G.d.S.; Domingues, M.A.F. Nutritional, functional and biological properties of insect proteins: Processes for obtaining, consumption and future challenges. Trends Food Sci. Technol. 2018, 76, 82–89. [Google Scholar] [CrossRef]
- Hermans, W.J.H.; Senden, J.M.; Churchward-Venne, T.A.; Paulussen, K.J.M.; Fuchs, C.J.; Smeets, J.S.J.; van Loon, J.J.A.; Verdijk, L.B.; van Loon, L.J.C. Insects are a viable protein source for human consumption: From insect protein digestion to postprandial muscle protein synthesis in vivo in humans: A double-blind randomized trial. Am. J. Clin. Nutr. 2021, 114, 934–944. [Google Scholar] [CrossRef] [PubMed]
- Gravel, A.; Doyen, A. The use of edible insect proteins in food: Challenges and issues related to their functional properties. Innov. Food Sci. Emerg. Technol. 2020, 59, 102272. [Google Scholar] [CrossRef]
- Gossner, C.M.-E.; Schlundt, J.; Embarek, P.B.; Hird, S.; Lo-Fo-Wong, D.; Beltran, J.J.O.; Teoh, K.N.; Tritscher, A. The melamine incident: Implications for international food and feed safety. Environ. Health Perspect. 2009, 117, 1803–1808. [Google Scholar] [CrossRef] [Green Version]
- Moore, J.C.; DeVries, J.W.; Lipp, M.; Griffiths, J.C.; Abernethy, D.R. Total protein methods and their potential utility to reduce the risk of food protein adulteration. Compr. Rev. Food Sci. F 2010, 9, 330–357. [Google Scholar] [CrossRef]
- Sharma, K.; Paradakar, M. The melamine adulteration scandal. Food Sec. 2010, 2, 97–107. [Google Scholar] [CrossRef]
- Ornelas-Soto, N.; Barbosa-García, O.; Lopez-de-Alba, P.L. Procedures of Food Quality Control: Analysis Methods, Sampling and Sample Pretreatment. In Quality Control of Herbal Medicines and Related Areas; Shoyama, Y., Ed.; IntechOpen: Rijeka, Croatia, 2011. [Google Scholar] [CrossRef] [Green Version]
- Truong, V.K.; Dupont, M.; Elbourne, A.; Gangadoo, S.; Pathirannahalage, P.R.; Cheeseman, S.; Chapman, J.; Cozzolino, D. From academia to reality check: A theoretical framework on the use of chemometric in food sciences. Foods 2019, 8, 164. [Google Scholar] [CrossRef] [Green Version]
- Luning, P.A.; Marcelis, W.J. Food Quality Management. Technological and Managerial Principles and Practices; Wageningen Academic: Wageningen, The Netherlands, 2020. [Google Scholar] [CrossRef]
- Kim, T.-K.; Yong, H.I.; Kim, Y.-B.; Kim, H.-W.; Choi, Y.-S. Edible insects as a protein source: A review of public perception, processing technology, and research trends. Food Sci. Anim. Resour. 2019, 39, 521–540. [Google Scholar] [CrossRef] [Green Version]
- Collins, C.M.; Vaskou, P.; Kountouris, Y. Insect food products in the western world: Assessing the potential of a new ‘green’ market. Ann. Entomol. Soc. Am. 2019, 112, 518–528. [Google Scholar] [CrossRef]
- Wade, M.; Hoelle, J. A review of edible insect industrialization: Scales of production and implications for sustainability. Environ. Res. Lett. 2020, 15, 123013. [Google Scholar] [CrossRef]
- Fanzo, J.; Bellows, A.L.; Spiker, M.L.; Thorne-Lyman, A.L.; Bloem, M.W. The importance of food systems and the environment for nutrition. Am. J. Clin. Nutr. 2021, 113, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.J.; Power, A.; Chapman, J.; Chandra, S.; Cozzolino, D. Vibrational spectroscopy methods for agro-food product analysis. Compr. Anal. Chem. 2018, 80, 51–68. [Google Scholar] [CrossRef]
- Sun, D.-W. Infrared Spectroscopy for Food Quality Analysis and Control; Academic Press; Elsevier: New York, NY, USA, 2009. [Google Scholar] [CrossRef]
- Manley, M.; Williams, P.J. Applications: Food: Science. In Near-Infrared Spectroscopy; Ozaki, Y., Huck, C.W., Tsuchikawa, S., Engelsen, S.B., Eds.; Springer Nature: Singapore, 2021; pp. 347–359. [Google Scholar] [CrossRef]
- Ozaki, Y.; Huck, C.W.; Beć, K.B. Near-IR spectroscopy and its applications. In Molecular and Laser Spectroscopy. Advances and Applications; Gupta, V.P., Ed.; Elsevier: San Diego, CA, USA, 2018; pp. 11–38. [Google Scholar] [CrossRef]
- Beć, K.B.; Grabska, J.; Huck, C.W. Physical principles of infrared spectroscopy. In Comprehensive Analytical Chemistry; Cozzolino, D., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; Volume 95. [Google Scholar] [CrossRef]
- Beć, K.B.; Grabska, J.; Huck, C.W. Biomolecular and bioanalytical applications of infrared spectroscopy—A review. Anal. Chim. Acta 2020, 1133, 150–177. [Google Scholar] [CrossRef] [PubMed]
- Cozzolino, D. The role of near-infrared sensors to measure water relationships in crops and plants. Appl. Spectrosc. Rev. 2017, 52, 837–849. [Google Scholar] [CrossRef]
- Beć, K.B.; Huck, C.W. Breakthrough potential in near-infrared spectroscopy: Spectra simulation. A review of recent developments. Front. Chem. 2019, 7, 48. [Google Scholar] [CrossRef] [Green Version]
- Cozzolino, D.; Roumeliotis, S.; Eglinton, J. Exploring the use of near infrared (NIR) reflectance spectroscopy to predict starch pasting properties in whole grain barley. Food Biophys. 2013, 8, 256–261. [Google Scholar] [CrossRef]
- Fassio, A.S.; Restaino, E.A.; Cozzolino, D. Determination of oil content in whole corn (Zea mays L.) seeds by means of near infrared reflectance spectroscopy. Comput. Electron. Agric. 2015, 110, 171–175. [Google Scholar] [CrossRef]
- Power, A.; Truong, V.K.; Chapman, J.; Cozzolino, D. From the laboratory to the vineyard—evolution of the measurement of grape composition using NIR spectroscopy towards high-throughput analysis. High-Throughput 2019, 8, 21. [Google Scholar] [CrossRef] [Green Version]
- Bobasa, E.M.; Netzel, M.E.; Cozzolino, D.; Phan, A.D.T.; Sultanbawa, Y. Measurement of total soluble solids and moisture in puree and dry powder of Kakadu plum (Terminalia ferdinanadiana) samples using hand-held near infrared spectroscopy. J. Near Infrared Spectrosc. 2021. [Google Scholar] [CrossRef]
- Nicolai, B.M.; Beullens, K.; Bobelyn, E.; Peirs, A.; Saeys, W.; Theron, K.I.; Lammertyn, J. Nondestructive measurement of fruit and vegetable quality by means of NIR spectroscopy: A review. Postharvest Biol. Technol. 2007, 46, 99–118. [Google Scholar] [CrossRef]
- Beghi, R.; Giovenzana, V.; Tugnolo, A.; Guidetti, R. Application of visible/near infrared spectroscopy to quality control of fresh fruits and vegetables in large-scale mass distribution channels: A preliminary test on carrots and tomatoes. J. Sci. Food Agric. 2018, 98, 2729–2734. [Google Scholar] [CrossRef] [PubMed]
- Szulc, J.; Gozdecka, G.; Poćwiardowski, W. The application of NIR spectroscopy in moisture determining of vegetable seeds. Czech J. Food Sci. 2020, 38, 131–136. [Google Scholar] [CrossRef]
- Burestan, N.F.; Sayyah, A.H.A.; Taghinezhad, E. Prediction of some quality properties of rice and its flour by near-infrared spectroscopy (NIRS) analysis. Food Sci. Nutr. 2021, 9, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Teye, E.; Amuah, C.L.Y.; cGrath, T.; Elliott, C. Innovative and rapid analysis for rice authenticity using hand-held NIR spectrometry and chemometrics. Spectrochim. Acta A 2019, 217, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Risoluti, R.; Gulliafa, G.; Materazi, S. Assessing the quality of milk using a multicomponent analytical platform MicroNIR/chemometric. Front. Chem. 2020, 8, 614718. [Google Scholar] [CrossRef] [PubMed]
- Riu, J.; Gorla, G.; Chakif, D.; Boque, R.; Giussani, B. Rapid Analysis of milk using low-cost pocket-size NIR spectrometers and multivariate analysis. Foods 2020, 9, 1090. [Google Scholar] [CrossRef]
- Holroyd, S.E. The use of NIRS in the dairy industry: New trends and applications. NIR News 2017, 28, 22–25. [Google Scholar] [CrossRef]
- Cozzolino, D.; Restaino, E.; La Manna, A.; Fernandez, E.; Fassio, A. Usefulness of near infrared reflectance (NIR) spectroscopy and chemometrics to discriminate between fishmeal, meat meal and soya meal samples. Cienc. E Investig. Agrar. 2009, 36, 209–214. [Google Scholar] [CrossRef]
- Prieto, N.; Pawluczyk, O.; Dugan, M.E.R.; Aalhus, J.L. A Review of the principles and applicationsof near-infrared spectroscopy to characterize meat, fat, and meat products. Appl. Spectrosc. 2017, 71, 1403–1426. [Google Scholar] [CrossRef] [Green Version]
- Simeone, M.L.; Parella, R.A.C.; Schaffert, R.E.; Damasceno, C.M.B.; Leal, M.C.B.; Paquini, C. Near infrared spectroscopy determination of sucrose, glucose andfructose in sweet sorghum juice. Microchem. J. 2017, 134, 125–130. [Google Scholar] [CrossRef] [Green Version]
- Kington, L.R.; Jones, T.M. Application for NIR analysis of beverages. In Handbook of Near-Infrared Analysis, 3rd ed.; Burns, D.A., Ciurczak, E.W., Eds.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Wang, L.; Sun, D.-W.; Cheng, J.-H. Quality analysis, classification, and authentication of liquid foods by near-infrared spectroscopy: A review of recent research developments. Crit. Rev. Food Sci. Nutr. 2017, 57, 1524–1538. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, S.; Schwab, A.; Henn, R.; Huck, C.W. Food fraud: An exploratory study for measuring consumer perception towards mislabeled food products and influence on self-authentication intentions. Trends Food Sci. Technol. 2016, 50, 211–218. [Google Scholar] [CrossRef]
- Beć, K.B.; Grabska, J.; Siesler, H.W.; Huck, C.W. Handheld near-infrared spectrometers: Where are we heading? NIR News 2020, 31, 28–35. [Google Scholar] [CrossRef] [Green Version]
- Beć, K.B.; Grabska, J.; Huck, C.W. Principles and applications of miniaturized near-infrared (NIR) spectrometers. Chem. A Eur. J. 2021, 27, 1514–1532. [Google Scholar] [CrossRef]
- Portable Ftir/Ftnir Spectrometer Interspec 300-X. Available online: https://interspectrum.ee/ftir-portable/portable-ftirft-nir-spectrometer-interspec-300-x/ (accessed on 5 July 2021).
- Mayr, S.; Schmelzer, J.; Kirchler, C.G.; Pezzei, C.K.; Beć, K.B.; Grabska, J.; Huck, C.W. Theae nigrae folium: Comparing the analytical performance of benchtop and handheld near-infrared spectrometers. Talanta 2020, 221, 121165. [Google Scholar] [CrossRef] [PubMed]
- Mayr, S.; Beć, K.B.; Grabska, J.; Schneckenreiter, E.; Huck, C.W. Near-infrared spectroscopy in quality control of Piper nigrum: A Comparison of performance of benchtop and handheld spectrometers. Talanta 2020, 223, 121809. [Google Scholar] [CrossRef]
- Mayr, S.; Beć, K.B.; Grabska, J.; Wiedemair, V.; Pürgy, V.; Popp, M.A.; Bonn, G.K.; Huck, C.W. Challenging handheld NIR spectrometers with moisture analysis in plant matrices: Performance of PLSR vs. GPR vs. ANN modelling. Spectrochim. Acta A 2021, 249, 119342. [Google Scholar] [CrossRef]
- Yang, J.; Lee, J. Application of sensory descriptive analysis and consumer studies to investigate traditional and authentic foods: A review. Foods 2019, 8, 54. [Google Scholar] [CrossRef] [Green Version]
- Food Processing. Available online: https://www.foodprocessing.com/articles/2015/artisanal-foods-increase-in-popularity/ (accessed on 5 July 2021).
- Lutz, O.M.D.; Bonn, G.K.; Rode, B.M.; Huck, C.W. Reproducible quantification of ethanol in gasoline via a customized mobile near-infrared spectrometer. Anal. Chim. Acta 2014, 826, 61–68. [Google Scholar] [CrossRef]
- MicroNIR Handheld Products. Available online: https://www.viavisolutions.com/en-us/osp/products/micronir-spectrometers/micronir-handheld-products (accessed on 5 July 2021).
- Norris, K.H. Why Log(1/R) for composition analysis with NIR? NIR News 2005, 16, 10–13. [Google Scholar] [CrossRef]
- Kessler, W. Multivariate Datenanalyse für die Pahrma-, Bio- und Prozessanalytik; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2007. [Google Scholar]
- Peng, Y.; Wu, P.; Siesler, H.W. Two-dimensional/ATR infrared correlation spectroscopic study on water diffusion in a poly(epsilon-caprolactone) matrix. Biomacromolecules 2003, 4, 1041–1044. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Fearn, T. Comparison of partial least squares regression, least squares support vector machines, and Gaussian process regression for a near infrared calibration. J. Near Infrared Spectrosc. 2017, 25, 5–14. [Google Scholar] [CrossRef]
- Fearn, T. Chemometrics: An enabling tool for NIR. NIR News 2013, 16, 17–19. [Google Scholar] [CrossRef]
- Redaelli, R.; Berardo, N. Evaluation of groat percentage in oats (Avena sativa L.) by near infrared spectroscopy. J. Near Infrared Spectrosc. 2002, 10, 103–109. [Google Scholar] [CrossRef]
- Wang, N.; Zhang, X.; Yu, Z.; Li, G.; Zhou, B. Quantitative analysis of adulterations in oat flour by FT-NIR spectroscopy, incomplete unbalanced randomized block design, and partial least squares. J. Anal. Methods Chem. 2014, 2014, 393596. [Google Scholar] [CrossRef]
- Mutlu, A.C.; Boyaci, I.H.; Genis, H.E.; Ozturk, R.; Basaran-Akgul, N.; Sanal, T.; Evlice, A.K. Prediction of wheat quality parameters using near-infrared spectroscopy and artificial neural networks. Eur. Food Res. Technol. 2011, 233, 267–274. [Google Scholar] [CrossRef]
- Barbin, D.F.; Felicio, A.L.s.S.M.; Sun, D.-W.; Nixdorf, S.L.; Hirooka, E.Y. Application of infrared spectral techniques on quality and compositional attributes of coffee: An overview. Food Res. Int. 2014, 61, 23–32. [Google Scholar] [CrossRef] [Green Version]
- Xiao, Z.; Lai, K.; Shen, Y.; Sun, X.; Pan, Y.; Rasco, B.A.; Huang, Y. Fat and moisture content in chinese fried bread sticks: Assessment and rapid near-infrared spectroscopic method development. J. Spectrosc. 2013, 2013, 973623. [Google Scholar] [CrossRef]
- Beć, K.B.; Grabska, J.; Kirchler, C.G.; Huck, C.W. NIR spectra simulation of thymol for better understanding of the spectra forming factors, phase and concentration effects and PLS regression features. J. Mol. Liq. 2018, 268, 895–902. [Google Scholar] [CrossRef]
- Workman, J.J., Jr.; Weyer, L. Practical Guide to Interpretive near Infrared Spectroscopy; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Bradstreet, R.B. Kjeldahl method for organic nitrogen. Anal. Chem. 1954, 26, 185–187. [Google Scholar] [CrossRef]



| Spectrometer (Vendor) | Key Components | Spectral Region | Resolution [nm] | Connectivity (Data Transfer) | Dimensions [cm] | Weight [g] | |||
|---|---|---|---|---|---|---|---|---|---|
| Source | Wavelength Selector | Detector | [nm] | [cm−1] | |||||
| NIRFlex N-500 (Büchi) | Tungsten halogen (duplicated) | Polarization interferometer (TiO2 wedges) | InGaAs (single-element, thermoelectric cooling) | 800–2500 | 12,500–4000 | ~2 | Ethernet | 45 × 35 × 25 | ca. 35,000 |
| MicroNIR 1700 ES (VIAVI) | Tungsten halogen (duplicated) | LVF | InGaAs (array; 128 elements) | 908–1676 | 11,013–5967 | 12.5 | USB—control and power delivery | 5.0 × 4.6 (Ø) | 58 |
| Enterprise Scanner NIR-S-G1 (Tellspec) | Tungsten halogen (duplicated) | Stationary dispersive grating and MEMS DMD | InGaAs (single-element) | 900–1700 | 11,111–5882 | 10 | Bluetooth (Cloud service) | 8.2 × 6.3 × 4.0 | 136 |
| SCiO (Consumer Physics) | LED | Bandpass filter | Si photodiode (CMOS) array (12 elements) | 740–1070 | 13,514–9346 | Not disclosed | Bluetooth (Cloud service) | 6.8 × 3.9 × 1.5 | 35 |
| PLSR | ||||
|---|---|---|---|---|
| Benchtop | Miniaturized | |||
| NIRFlex N-500 | MicroNIR 1700 ES | Tellspec Enterprise Sensor | SCiO Sensor | |
| Pretreatment | SG2 (29 SP) | SNV, SG2 (3 SP) | SNV, SG2 (25 SP) | SNV, SG2 (25 SP) |
| R2 (Cal) | 0.43 | 0.57 | 0.38 | 0.55 |
| R2 (CV) | 0.35 | 0.47 | 0.30 | 0.38 |
| RMSEC [%] | 0.641 | 0.557 | 0.668 | 0.568 |
| RMSECV [%] | 0.687 | 0.624 | 0.716 | 0.671 |
| R2 (TSV) | 0.49 | 0.46 | 0.40 | 0.59 |
| RMSEP [%] | 0.611 | 0.620 | 0.659 | 0.545 |
| GPR | ||||
| Benchtop | Miniaturized | |||
| NIRFlex N-500 | MicroNIR 1700 ES | Tellspec Enterprise Sensor | SCiO Sensor | |
| Pretreatment | SG2 (29 SP) | SNV, SG2 (3 SP) | SNV, SG2 (25 SP) | SNV, SG2 (25 SP) |
| R2 (Cal) | 0.99 | 1.00 | 0.54 | 1.00 |
| R2 (CV) | 0.42 | 0.53 | 0.33 | 0.52 |
| RMSEC [%] | 0.083 | 0.00015 | 0.579 | 0.00014 |
| RMSECV [%] | 0.65 | 0.59 | 0.70 | 0.60 |
| R2 (TSV) | 0.65 | 0.68 | 0.56 | 0.54 |
| RMSEP [%] | 0.506 | 0.482 | 0.578 | 0.580 |
| PLSR | ||||
|---|---|---|---|---|
| Benchtop | Miniaturized | |||
| NIRFlex N-500 | MicroNIR 1700 ES | Tellspec Enterprise Sensor | SCiO Sensor | |
| Pretreatment | SG (5 SP) | SG1 (7 SP) | SG1 (11 SP) | SG1 (11 SP) |
| R2 (Cal) | 0.96 | 0.89 | 0.65 | 0.54 |
| R2 (CV) | 0.88 | 0.80 | 0.52 | 0.46 |
| RMSEC [%] | 0.182 | 0.286 | 0.505 | 0.581 |
| RMSECV [%] | 0.309 | 0.382 | 0.591 | 0.630 |
| R2 (TSV) | 0.94 | 0.62 | 0.55 | 0.55 |
| RMSEP [%] | 0.210 | 0.525 | 0.571 | 0.568 |
| GPR | ||||
| Benchtop | Miniaturized | |||
| NIRFlex N-500 | MicroNIR 1700 ES | Tellspec Enterprise Sensor | SCiO Sensor | |
| Pretreatment | SG (5 SP) | SG1 (7 SP) | SG1 (11 SP) | SG1 (11 SP) |
| R2 (Cal) | 1 | 0.99 | 0.99 | 0.99 |
| R2 (CV) | 0.87 | 0.99 | 0.84 | 0.93 |
| RMSEC [%] | 0.0011 | 0.0006 | 0.0002 | 0.0003 |
| RMSECV [%] | 0.3150 | 0.0782 | 0.3397 | 0.2248 |
| R2 (TSV) | 0.91 | 0.94 | 0.87 | 0.84 |
| RMSEP [%] | 0.266 | 0.230 | 0.326 | 0.338 |
| Intact | Milled | |||
|---|---|---|---|---|
| PLSR | GPR | PLSR | GPR | |
| Pretreatment | SNV, SG2 (25 SP) | SNV, SG2 (25 SP) | SG1 (11 SP) | SG1 (11 SP) |
| R2 (Cal) | 0.41 | 0.9 | 0.53 | 0.99 |
| R2 (CV) | 0.28 | 0.55 | 0.48 | 0.9 |
| RMSEC [%] | 0.654 | 0.272 | 0.580 | 0.0002 |
| RMSECV [%] | 0.723 | 0.574 | 0.620 | 0.263 |
| R2 (TSV) | 0.38 | 0.64 | 0.51 | 0.89 |
| RMSEP [%] | 0.671 | 0.517 | 0.596 | 0.295 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beć, K.B.; Grabska, J.; Plewka, N.; Huck, C.W. Insect Protein Content Analysis in Handcrafted Fitness Bars by NIR Spectroscopy. Gaussian Process Regression and Data Fusion for Performance Enhancement of Miniaturized Cost-Effective Consumer-Grade Sensors. Molecules 2021, 26, 6390. https://doi.org/10.3390/molecules26216390
Beć KB, Grabska J, Plewka N, Huck CW. Insect Protein Content Analysis in Handcrafted Fitness Bars by NIR Spectroscopy. Gaussian Process Regression and Data Fusion for Performance Enhancement of Miniaturized Cost-Effective Consumer-Grade Sensors. Molecules. 2021; 26(21):6390. https://doi.org/10.3390/molecules26216390
Chicago/Turabian StyleBeć, Krzysztof B., Justyna Grabska, Nicole Plewka, and Christian W. Huck. 2021. "Insect Protein Content Analysis in Handcrafted Fitness Bars by NIR Spectroscopy. Gaussian Process Regression and Data Fusion for Performance Enhancement of Miniaturized Cost-Effective Consumer-Grade Sensors" Molecules 26, no. 21: 6390. https://doi.org/10.3390/molecules26216390









