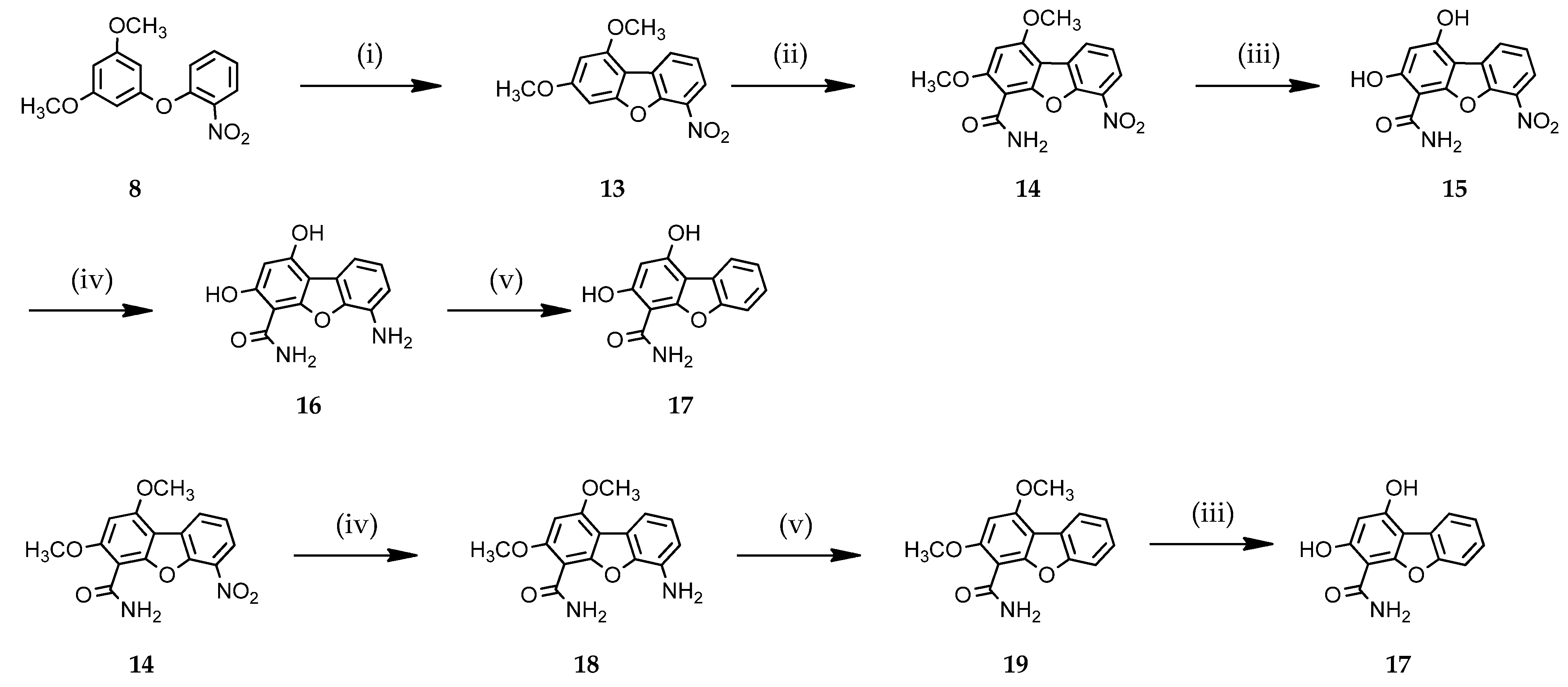
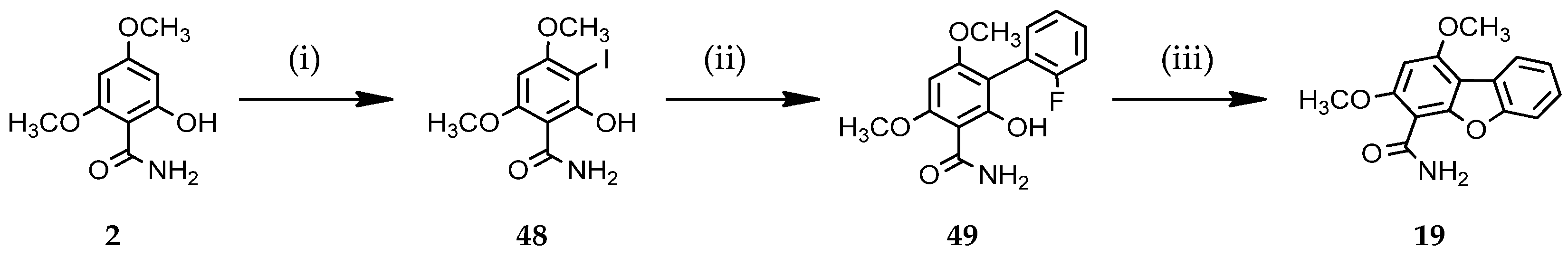3.1.2. Access to Dibenzofuran Derivatives 14–19 by Intramolecular C-C Bond Formation from Diaryl Ethers
2-Hydroxy-4,6-dimethoxybenzamide (2)
To a stirred suspension of 3,5-dimethoxyphenol 4 (1.00 g, 6.5 mmol) in acetonitrile (20 mL) at 0 °C under argon, chlorosulfonyl isocyanate (1.26 mL, 13 mmol) was added. The mixture was maintained at this temperature for 10 min and then the reaction mixture was quenched with HCl 5 M (20 mL). After 10 h at room temperature, the reaction was diluted with water (100 mL) and extracted with dichloromethane (3 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (8/2) as eluent to give compound 2 (576 mg, 45% yield) as a white powder. Rf 0.46 (cyHex/AcOEt 6:4); Mp: 154–155 °C; 1H NMR (400 MHz, DMSO-d6) δ 14.69 (s, 1H, OH), 8.04–7.86 (m, 2H, CONH2), 6.08 (d, J = 2.5 Hz, 1H, H5), 6.05 (d, J = 2.4 Hz, 1H, H3), 3.87 (s, 3H, CH3), 3.77 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 171.7 (C=O), 165.9 (C2), 163.7 (C6), 160.2 (C4), 96.6 (C1), 94.1 (C3), 90.1 (C5), 56.0 (CH3), 55.3 (CH3); IR (cm−1): 3429, 3209, 1620, 1587, 1492, 1415, 1355, 1315, 1294, 1217, 1199, 1159, 1109, 1041; MS (ESI) m/z (%): 198.1 (100) [M + H]+; UPLC purity = 97%, tR 1.93 min.
2,4-Dimethoxy-6-(2′-nitrophenoxy)benzamide (6)
A mixture of 2-hydroxy-4,6-dimethoxybenzamide 2 (0.50 g, 2.5 mmol) and potassium hydroxide (0.13 g, 2.5 mmol) was heated to 120 °C and stirred for 30 min, and then 1-iodo-2-nitrobenzene (0.64 g, 2.5 mmol) and copper powder (0.20 g, 3.13 mmol) were added, respectively. The resulting mixture was stirred at 170 °C for 24 h and then cooled down to room temperature. After the addition of water, the aqueous layer was extracted with ethyl acetate (100 mL) and washed with water (2 × 100 mL). The organic layers were washed with brine, dried over sodium sulfate, filtered, and evaporated to dryness. The resulting oil was purified by silica gel column chromatography using cyclohexane/ethyl acetate (8/2) as an eluent to give compound 6 (125 mg, 15% yield) as a brown powder. Rf 0.45 (cyHex/AcOEt 8:2); Mp: 209–210 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.51 (s, 1H, NH), 11.76 (s, 1H, NH), 8.60 (dd, J = 8.4, 1.5 Hz, 1H, H3′), 8.24 (dd, J = 8.4, 1.5 Hz, 1H, H6′), 7.85 (t, J = 7.9 Hz, 1H, H4′), 7.46 (t, J = 7.9 Hz, 1H, H5′), 6.31 (d, J = 2.3 Hz, 1H, H5), 6.24 (d, J = 2.3 Hz, 1H, H3), 4.10 (s, 3H, CH3), 3.89 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 168.5 (C=O), 165.7 (C4), 164.6 (C2), 159.9 (C6), 139.6 (C1′), 134.8 (C4′), 132.0 (C2′), 125.4 (C5′), 124.8 (C3′), 124.8 (C6′), 97.2 (C1), 94.7 (C5), 91.0 (C3), 56.5 (CH3), 55.6 (CH3); IR (cm−1): 3284, 1639, 1575, 1502, 1390, 1346, 1276, 1249, 1157, 1047; MS (ESI) m/z (%): 319.1 (100) [M + H]+; UPLC purity = 97%, tR 3.21 min.
1,3-Dibenzyloxy-5-(2′-nitrophenoxy)benzene (7)
A mixture of 3,5-dibenzyloxyphenol 3 (1.00 g, 3.26 mmol) and potassium hydroxide (0.18 g, 3.26 mmol) was heated to 120 °C and stirred for 30 min, and then 1-iodo-2-nitrobenzene (0.82 g, 3.26 mmol) and copper powder (0.40 g, 6.25 mmol) were added, respectively. The resulting mixture was stirred at 170 °C for 2 h and then cooled down to room temperature. After the addition of water, the aqueous layer was extracted with ethyl acetate (100 mL) and washed with water (2 × 100 mL). The organic layers were washed with brine, dried over sodium sulfate, filtered, and evaporated to dryness. The resulting oil was purified by silica gel column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent to give compound 7 (1.02 g, 73% yield) as a yellow powder. Rf 0.76 (cyHex/AcOEt 6:4); Mp: 148–149 °C; 1H NMR (400 MHz, CDCl3) δ 7.80 (dd, J = 8.1, 1.7 Hz, 1H, H3′), 7.35 (ddd, J = 8.5, 7.4, 1.7 Hz, 1H, H4′), 7.34–7.15 (m, 10H, HAr), 7.12 (ddd, J = 8.5, 7.4, 1.7 Hz, 1H, H5′), 6.93 (dd, J = 8.4, 1.3 Hz, 1H, H6′), 6.33 (t, J = 2.2 Hz, 1H, H2), 6.17 (s, 1H, H4), 6.17 (s, 1H, H6), 4.86 (s, 4H, 2CH2); 13C NMR (100 MHz, CDCl3) δ 160.9 (2C1,3), 157.7 (C5), 150.0 (C1′), 141.5 (C2′), 136.4 (2C), 134.2 (C4′), 128.6 (2C), 128.6 (2C), 128.1 (1C), 128.1 (1C), 127.6 (2C), 127.6 (2C), 125.7 (C4′), 123.5 (C3′), 121.3 (C6′), 98.7 (2C4,6), 98.2 (C2), 70.3 (2CH2); IR (cm−1): 1589, 1529, 1450, 1355, 1236, 1157, 1128; MS (ESI) m/z (%): 428.2 (100) [M + H]+; UPLC purity = 100%, tR 3.09 min.
1,3-Dimethoxy-5-(2′-nitrophenoxy)benzene (8)
A mixture of 3,5-dimethoxyphenol 4 (1.00 g, 6.5 mmol) and potassium hydroxide (0.18 g, 6.5 mmol) was heated to 120 °C and stirred for 30 min, and then 1-iodo-2-nitrobenzene (0.82 g, 6.5 mmol) and copper powder (1.00 g, 15.6 mmol) were added, respectively. The resulting mixture was stirred at 170 °C for 2 h and then cooled down to room temperature. After the addition of water, the aqueous layer was extracted with ethyl acetate (100 mL). The organic layers were washed with brine, dried over sodium sulfate, filtered, and evaporated to dryness. The resulting oil was purified by silica gel column chromatography using cyclohexane/ethyl acetate (8/2) as eluent to give compound 8 (1.4 g 78% yield) as a yellow oil. Rf 0.42 (cyHex/AcOEt 8:2); 1H NMR (400 MHz, DMSO-d6) δ 8.07 (dd, J = 8.2 Hz, 1.6 Hz, 1H, H3′), 7.77–7.63 (m, 1H, H5′), 7.38 (t, J = 7.8 Hz, 1H, H4′), 7.21 (d, J = 8.4 Hz, 1H, H6′), 6.38 (t, J = 2.3 Hz, 1H, H2), 6.24 (s, 2H, H4,6), 3.74 (s, 6H, 2CH3); 13C NMR (100 MHz, DMSO-d6) δ 161.4 (2C1,3), 157.2 (C5), 148.8 (C1′), 141.0 (C2′), 134.9 (C4′), 125.5 (C5′), 124.2 (C3′), 120.9 (C6′), 97.1 (2C4,6), 96.4 (C2), 55.4 (2CH3); IR (cm−1): 1585, 1523, 1471, 1346, 1234, 1203, 1149, 1126, 1049; MS (ESI) m/z (%): 276.1 (100) [M + H]+; UPLC purity = 97%, tR 3.02 min.
2-(3′,5′-Dibenzyloxyphenoxy)aniline (9)
To a solution of 1,3-dibenzyloxy-5-(2′-nitrophenoxy)benzene 7 (100 mg, 0.23 mmol) in methanol (10 mL) at room temperature, saturated aqueous ammonium chloride (5 mL) and zinc dust (75 mg, 1.15 mmol) were added sequentially. After stirring for 30 min at room temperature, additional zinc was added (75 mg, 1.15 mmol) and the reaction mixture was refluxed for 2 h. After the addition of water, the aqueous layer was extracted with dichloromethane (3 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent to give compound 9 (59 mg, 65% yield) as a brown powder. Rf 0.77 (cyHex/AcOEt 6:4); Mp: 217–218 °C; 1H NMR (400 MHz, CDCl3) δ 7.37–7.12 (m, 10H, HAr), 6.89 (td, J = 7.6, 1.5 Hz, 1H, H5), 6.80 (dd, J = 8.0, 1.5 Hz, 1H, H6), 6.68 (dt, J = 8.6, 2.6 Hz, 1H, H3), 6.65 (td, J = 7.6, 1.5 Hz, 1H, H4), 6.24 (s, 1H, H4′), 6.14 (s, 2H, H2′,6′), 4.87 (s, 4H, 2CH2); 13C NMR (100 MHz, CDCl3) δ 160.8 (2C3′,5′), 159.5 (C1′), 142.4 (C2), 138.9 (C1), 136.6 (2C), 128.6 (4C), 128.0 (2C), 127.6 (4C), 125.3 (C5), 120.9 (C4), 118.8 (C3), 116.6 (C6), 96.7 (2C2′,6′), 96.4 (C4′), 70.1 (2CH2); IR (cm−1): 2819, 1597, 1492, 1450, 1379, 1253, 1157, 1126, 1060; MS (ESI) m/z (%): 398.1 (100) [M + H]+; UPLC purity = 100%, tR 3.12 min.
2-(3′,5′-Dimethoxyphenoxy)aniline (10)
To a solution of 1,3-dimethoxy-5-(2′-nitrophenoxy)benzene 8 (250 mg, 0.91 mmol) in methanol (10 mL) at room temperature, saturated aqueous ammonium chloride (5 mL) and zinc dust (295 mg, 4.54 mmol) were added sequentially. After stirring for 30 min at room temperature, additional zinc was added (295 mg, 4.54 mmol) and the reaction mixture was refluxed for 2 h. After the addition of water, the aqueous layer was extracted with ethyl acetate (100 mL) and washed with water (2 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 7 (205 mg, 92% yield) as a brown oil. Rf 0.80 (cyHex/AcOEt 6:4); 1H NMR (400 MHz, CDCl3) δ 6.89 (td, J = 7.6, 1.5 Hz, 1H, H4), 6.82 (dd, J = 8.0, 1.4 Hz, 1H, H3), 6.71 (dd, J = 7.9, 1.6 Hz, 1H, H6), 6.62 (td, J = 7.6, 1.6 Hz, 1H, H5), 6.09 (t, J = 2.2 Hz, 1H, H4′), 6.06 (d, J = 2.3 Hz, 1H, 2H2′), 6.05 (d, J = 2.3 Hz, 2H, 2H6′), 3.64 (s, 3H, CH3), 3.60 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 161.2 (2C3′,5′), 159.4 (C1′), 141.0 (C2), 140.4 (C1), 125.1 (C4), 120.7 (C5), 116.4 (C3), 115.8 (C6), 95.1 (2C2′,6′), 94.0 (C4′), 55.0 (2CH3); IR (cm−1): 3373, 2964, 1697, 1595, 1500, 1473, 1458, 1427, 1301, 1269, 1197, 1149, 1130, 1051; MS (ESI) m/z (%): 246.1 (100) [M + H]+; UPLC purity = 100%, tR 2.51 min.
1,3-Dimethoxy-6-nitrodibenzo[b,d]furan (13)
1,3-Dimethoxy-5-(2′-nitrophenoxy)benzene 8 (1.00 g, 3.6 mmol) was dissolved in warm pivalic acid (50 °C, 60 g). Silver acetate (1.20 g, 7.2 mmol) and palladium (II) acetate (40 mg, 0.18 mmol) were added, and the reaction mixture was properly stirred at 130 °C for 4 h. After cooling to room temperature and the addition of water (100 mL), the aqueous layer was extracted with ethyl acetate (100 mL). The organic layers were washed with brine, dried over sodium sulfate, filtered, and evaporated to dryness. The resulting oil was purified by silica gel column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent to give compound 13 (0.71 g, 72% yield) as a yellow powder. Rf 0.64 (cyHex/AcOEt 6:4); Mp: 212–213 °C; 1H NMR (400 MHz, DMSO-d6) δ 8.27 (dd, J = 7.6 Hz, 1.1 Hz, 1H, H9), 8.18 (dd, J = 8.3, 1.1 Hz, 1H, H7), 7.55 (t, J = 8.0 Hz, 1H, H8), 7.13 (d, J = 1.8 Hz, 1H, H4), 6.66 (d, J = 1.8 Hz, 1H, H2), 4.03 (s, 3H, CH3), 3.90 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 162.2 (C3), 158.2 (C1), 155.5 (C), 147.1 (C), 132.9 (C6), 127.6 (C8), 126.9 (C), 123.7 (C9), 120.5 (C7), 104.4 (C), 95.3 (C2), 89.3 (C4), 56.1 (CH3), 56.1 (CH3); IR (cm−1): 3088, 2933, 1637, 1602, 1531, 1502, 1450, 1417, 1344, 1219, 1145, 1095; MS (ESI) m/z (%): 274.1 (100) [M + H]+; UPLC purity = 99%, tR 3.16 min.
1,3-Dimethoxy-6-nitrodibenzo[b,d]furan-4-carboxamide (14)
To a stirred suspension of 1,3-dimethoxy-6-nitrodibenzo[b,d]furan 13 (1.00 g, 3.7 mmol) in acetonitrile (20 mL) at 0 °C under argon, chlorosulfonyl isocyanate (0.64 mL, 7.4 mmol) was added. The mixture was maintained at room temperature for 12 h and then the reaction mixture was quenched with HCl 5 M (20 mL). After 6 h at room temperature, the reaction was diluted with water (100 mL) and extracted with ethyl acetate (100 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 14 (0.61 g, 53% yield) as a yellow powder. Rf 0.63 (CH2Cl2/MeOH 9:1); Mp: 298–299 °C; 1H NMR (400 MHz, DMSO-d6) δ 8.32 (d, J = 7.6 Hz, 1H, H9), 8.20 (d, J = 8.3 Hz, 1H, H7), 7.77 (s, 1H, NH), 7.67–7.54 (m, 2H, NH, H8), 6.82 (s, 1H, H2), 4.13 (s, 3H, CH3), 3.98 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 163.7 (C=O), 158.7 (C1), 155.8 (C3), 154.8 (C), 147.1 (C), 133.0 (C6), 127.7 (C8), 126.6 (C), 123.8 (C9), 120.9 (C7), 105.1 (C), 104.3 (C4), 92.2 (C2), 56.8 (CH3), 56.4 (CH3); IR (cm−1): 3136, 3091, 2846, 2140, 1977, 1643, 1606, 1521, 1427, 1355, 1323, 1217, 108; MS (ESI) m/z (%): 317.2 (100) [M + H]+; UPLC purity = 99%, tR 2.24 min; HRMS (TOF MS ES+): calcd. for C15H13N2O6 [M + H]+ 317.076813, found: 317.07664.
1,3-Dihydroxy-6-nitrodibenzo[b,d]furan-4-carboxamide (15)
A mixture of 1,3-dimethoxy-6-nitrodibenzo[b,d]furan-4-carboxamide 14 (50 mg, 0.16 mmol) in pyridine hydrochloride (3g) was irradiated using microwave heating at 200 °C for 15 min. After the addition of water, the aqueous layer was extracted with ethyl acetate (50 mL) and the resulting organic layer was washed with water (2 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 15 (23 mg, 51% yield) as a yellow powder. Rf 0.67 (CH2Cl2/MeOH 9:1); Mp > 300 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.80 (s, 1H, OH), 11.77 (s, 1H, OH), 8.54 (s, 1H, NH), 8.42–8.07 (m, 2H, NH, H9), 7.75–7.36 (m, 2H, H8,7), 6.39 (s, 1H, H2); 13C NMR (100 MHz, DMSO-d6) δ 169.6 (C=O), 165.1 (C1), 157.9 (C3), 155.7 (C), 146.3 (C), 133.0 (C6), 127.7 (C8), 126.5 (C), 124.5 (C9), 120.3 (C7), 103.1 (C4), 98.9 (C2), 92.7 (C); IR (cm−1): 3458, 3196, 1654, 1629, 1608, 1517, 1477, 1440, 1404, 1359, 1323, 1251, 1220, 1195, 1120, 1082; MS (ESI) m/z (%): 289.1 (100) [M + H]+; UPLC purity = 99%, tR 2.37 min; HRMS (TOF MS ES+): calcd. for C13H9N2O6 [M + H]+ 289.045512, found: 289.04558.
6-Amino-1,3-dihydroxydibenzo[b,d]furan-4-carboxamide (16)
To a solution of 1,3-dihydroxy-6-nitrodibenzo[b,d]furan-4-carboxamide 15 (50 mg, 0.17 mmol) in methanol (10 mL) at room temperature, saturated aqueous ammonium chloride (5 mL) and zinc dust (55 mg, 0.85 mmol) were added sequentially. After stirring for 30 min at room temperature, additional zinc was added (55 mg, 0.85 mmol) and the reaction mixture was refluxed for 2 h. After the addition of water, the aqueous layer was extracted with ethyl acetate (100 mL) and the resulting organic layer was washed with water (2 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 16 (28 mg, 62% yield) as a brown powder. Rf 0.34 (CH2Cl2/MeOH 9:1); Mp: 292-293 °C; 1H NMR (400 MHz, DMSO-d6) δ 14.19 (s, 1H, OH), 11.12 (s, 1H, OH), 8.19 (s, 1H, CONH2), 8.11 (s, 1H, CONH2), 7.12 (d, J = 7.5 Hz, 1H, H9), 7.03 (t, J = 7.6 Hz, 1H, H8), 6.63 (d, J = 7.8 Hz, 1H, H7), 6.25 (s, 1H, H2); 13C NMR (100 MHz, DMSO) δ 170.2 (C=O), 164.2 (C1), 157.7 (C3), 154.8 (C), 142.2 (C), 133.3 (C6), 124.4 (C8), 123.0 (C), 110.2 (C9), 108.1 (C7), 105.3 (C4), 97.8 (C2), 92.5 (C); IR (cm−1): 3294, 3192, 1492, 1427, 1261, 1190, 1085, 1053; MS (ESI) m/z (%): 259.2 (100) [M + H]+; UPLC purity = 99%, tR 1.67 min; HRMS (TOF MS ES+): calcd. for C13H11N2O4 [M + H]+ 259.071333, found: 259.07147.
1,3-Dihydroxydibenzo[b,d]furan-4-carboxamide (17)
Method A: To a solution of 6-amino-1,3-dihydroxydibenzo[b,d]furan-4-carboxamide 16 (100 mg, 0.38 mmol) in ethanol (10 mL) at room temperature, sulfuric acid (25%, 40 mL) was added under warming. The mixture was cooled to 0 °C, and a solution of sodium nitrite (54 mg, 0.76 mmol) in 5 mL of water was added dropwise with stirring for 30 min. Afterwards, the reaction mixture was stirred for 45 min at 80 °C and then the reaction was quenched with water. The aqueous layer was extracted with ethyl acetate (100 mL) and the organic layer was washed with brine, dried over sodium sulfate, and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 17 (49 mg, 52% yield) as a brown powder.
Method B: A mixture of 1,3-dimethoxydibenzo[b,d]furan-4-carboxamide 19 (see below) (100 mg, 0.37 mmol) in pyridine hydrochloride (3 g) was irradiated using microwave heating at 200 °C for 15 min. After the addition of water, the aqueous layer was extracted with ethyl acetate (50 mL) and the resulting organic layer was washed with water (2 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 17 (43 mg, 48% yield) as a brown powder.
Rf 0.20 (CH2Cl2); Mp: 219–220 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.93 (s, 1H, OH), 11.34 (s, 1H, OH), 8.28 (s, 1H, CONH2), 8.03–7.88 (m, 1H, H9), 7.85 (s, 1H, CONH2), 7.77–7.65 (m, 1H, H6), 7.57–7.25 (m, 2H, H7,8), 6.31 (s, 1H, H2); 13C NMR (100 MHz, DMSO-d6) δ 170.0 (C=O), 164.3 (C1), 157.7 (C3), 155.2 (C), 154.2 (C), 124.8 (C7), 123.7 (C8), 122.7 (C), 121.0 (C9), 111.3 (C6), 104.4 (C4), 98.0 (C2), 92.6 (C); IR (cm−1): 3456, 3132, 1604, 1446, 1409, 1348, 1315, 1286, 1230, 1195, 1103, 1053; MS (ESI) m/z (%): 244.1 (100) [M + H]+; UPLC purity = 98%, tR 2.14 min; HRMS (TOF MS ES+): calcd. for C13H10NO4 [M + H]+ 244.060434, found: 244.06010.
6-Amino-1,3-dimethoxydibenzo[b,d]furan-4-carboxamide (18)
To a solution of 1,3-dimethoxy-6-nitrodibenzo[b,d]furan-4-carboxamide 14 (50 mg, 0.16 mmol) in methanol (10 mL) at room temperature, saturated aqueous ammonium chloride (5 mL) and zinc dust (55 mg, 0.85 mmol) were added sequentially. After stirring for 30 min at room temperature, additional zinc was added (55 mg, 0.85 mmol) and the reaction mixture was refluxed for 2 h. After the addition of water, the aqueous layer was extracted with ethyl acetate (100 mL) and the resulting organic layer was washed with water (2 × 50 mL). The combined organic extracts were dried over sodium sulfate and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as eluent to give compound 18 (22 mg, 49% yield) as a brown powder. Rf 0.52 (CH2Cl2/MeOH 9:1); Mp: 293–294 °C; 1H NMR (400 MHz, DMSO-d6) δ 7.67 (s, 1H, NH), 7.44 (s, 1H, NH), 7.17 (d, J = 7.5 Hz, 1H, H9), 7.03 (t, J = 7.7 Hz, 1H, H8), 6.70 (d, J = 5.9 Hz, 1H, H7), 6.68 (s, 1H, H2), 5.25 (s, 2H, NH2), 4.05 (s, 3H, CH3), 3.93 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 164.6 (C=O), 157.2 (C1), 155.5 (C3), 153.9 (C), 142.9 (C), 133.1 (C6), 123.7 (C8), 122.7 (C), 111.2 (C9), 109.1 (C7), 107.0 (C4), 104.8 (C), 90.9 (C2), 56.6 (CH3), 56.0 (CH3); IR (cm−1): 3180, 1643, 1604, 1492, 1413, 1319, 1211, 1101; MS (ESI) m/z (%): 287.2 (100) [M + H]+; UPLC purity = 99%, tR 1.83 min; HRMS (TOF MS ES+): calcd. for C15H15N2O4 [M + H]+ 287.102633, found: 287.10220.
1,3-Dimethoxydibenzo[b,d]furan-4-carboxamide (19)
Method A: To a solution of 6-amino-1,3-dimethoxydibenzo[b,d]furan-4-carboxamide 18 (50 mg, 0.17 mmol) in ethanol (10 mL) at room temperature, sulfuric acid (25%, 20 mL) was added under warming. The mixture was cooled to 0 °C, and a solution of sodium nitrite (24 mg, 0.34 mmol) in 5 mL of water was added dropwise with stirring for 30 min. The reaction mixture was stirred for 45 min at 80 °C and then the reaction was quenched with water. The aqueous layer was extracted with ethyl acetate (100 mL) and the organic layer was washed with brine, dried over sodium sulfate, and filtered. The solvent was evaporated in vacuo and the residue was purified by silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to give compound 19 (22 mg, 46% yield) as a pale brown powder.
Method B: A mixture of 2’-fluoro-2-hydroxy-4,6-dimethoxybiphenyl-3-carboxamide 49 (see below) (50 mg, 0.17 mmol) and cesium carbonate (166 mg, 0.51 mmol) in acetonitrile was irradiated using microwave (80 W) heating at 150 °C for 1 h. Ethyl acetate (50 mL) was added to the mixture and the organic layer was washed with water (2 × 50 mL), dried over sodium sulfate, and filtered. The solvent was evaporated in vacuo and the residue was crystallized from diisopropyl ether to furnish the title compound 19 as a pale brown solid (28 mg, 61%)
Rf 0.41 (CH2Cl2/MeOH 9:1); Mp: 296–297 °C; 1H NMR (400 MHz, DMSO-d6) δ 7.95 (dd, J = 7.5, 1.5 Hz, 1H, H9), 7.69 (s, 1H, NH), 7.65 (d, J = 8.0 Hz, 1H, H6), 7.51 (s, 1H, NH), 7.38 (m, 2H, H7,8), 6.72 (s, 1H, H2), 4.09 (s, 3H, CH3), 3.95 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 164.5 (C=O), 157.6 (C1), 155.7 (C3), 154.8 (C), 154.4 (C), 125.5 (C7), 123.2 (C8), 122.5 (C), 121.3 (C9), 110.9 (C6), 105.9 (C4), 104.7 (C), 91.1 (C2), 56.7 (CH3), 56.1 (CH3); IR (cm−1): 3381, 3201, 2968, 1641, 1602, 1452, 1421, 1321, 1213; MS (ESI) m/z (%): 272.2 (100) [M + H]+; UPLC purity = 99%, tR 2.21 min; HRMS (TOF MS ES+): calcd. for C15H14NO4 [M + H]+ 272.091734, found: 272.09149.
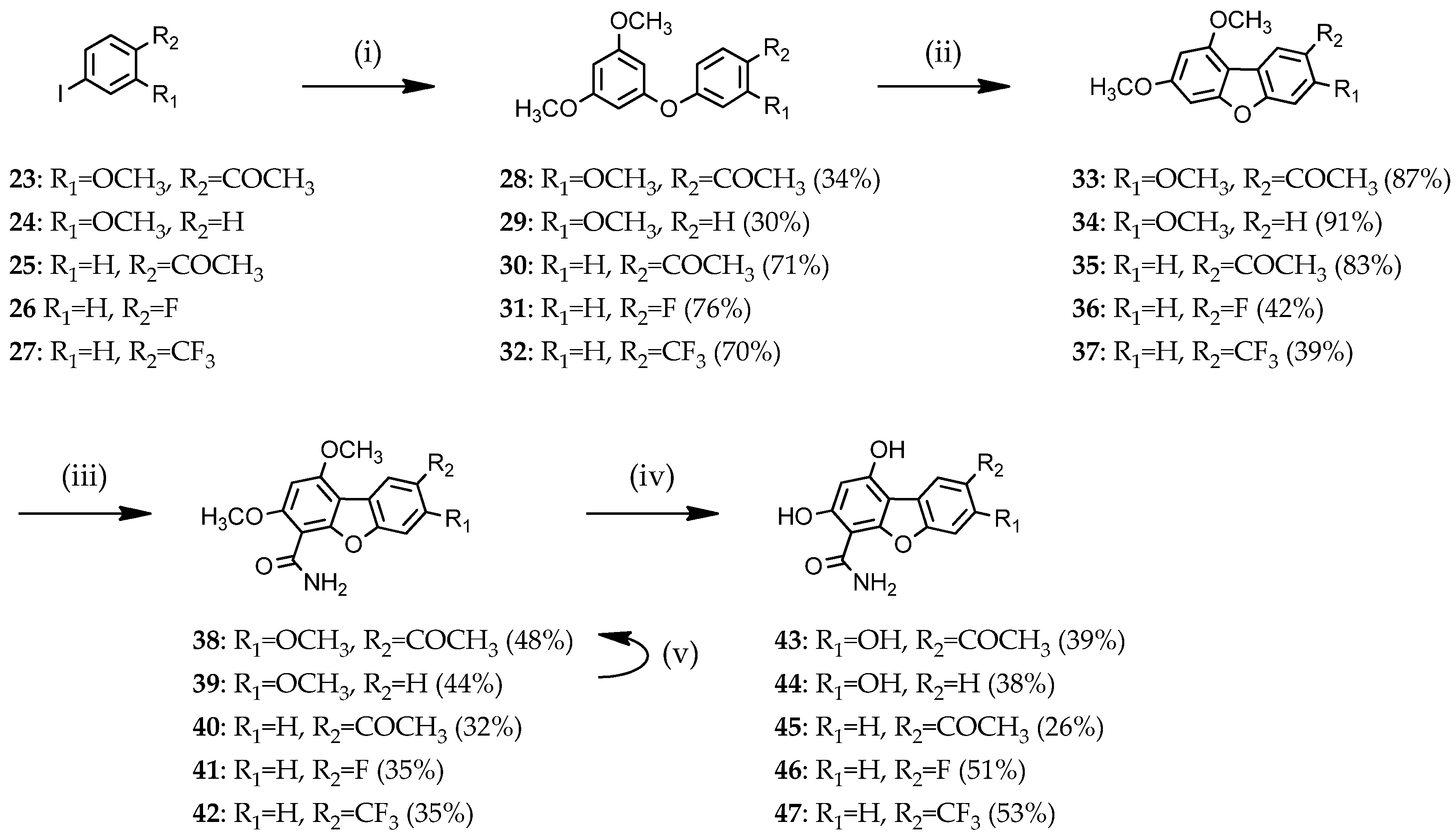
Access to dibenzofuran derivatives 38–47 was achieved by intramolecular C-C bond formation from diaryl ethers.
3.1.4. General Procedure for the Preparation of Diaryl Ethers 28–32
To a solution of 3,5-dimethoxyphenol 4 (1 equiv.) in N,N-dimethylformamide, cesium carbonate (2 equiv.), copper(1) iodide (0.1 equiv.), and iodo-aryl derivatives 23–27 (1.5 equiv.) were added. The reaction mixture was irradiated using microwave heating at 110 °C for 1 h. After cooling to room temperature, the mixture was diluted with ethyl acetate (50 mL) and water (100 mL). The suspension was filtered through a pad of Celite®. The two layers were separated, and the organic layer was washed with water (3 × 50 mL), dried over sodium sulfate, filtered, and evaporated to dryness. The crude product was then purified by chromatography to provide 28–32.
1-[4-(3′,5′-Dimethoxyphenoxy)-2-methoxyphenyl]ethanone (28)
The reaction was carried out using 3,5-dimethoxyphenol 4 (400 mg, 2.6 mmol), Cs2CO3 (1.69 g, 5.2 mmol), CuI (50 mg, 0.26 mmol), and 1-(4-iodo-2-methoxyphenyl)ethanone 23 (1.08 g, 3.9 mmol) in DMF (3 mL). Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 28 (266 mg, 34% yield) as a colorless oil. Rf 0.54 (cyHex/AcOEt 8:2); 1H NMR (400 MHz, CDCl3) δ 7.78 (d, J = 8.8 Hz, 1H, H6), 6.61 (d, J = 2.2 Hz, 1H, H3), 6.56 (dd, J = 8.7, 1.4 Hz, 1H, H5), 6.29 (t, J = 2.2 Hz, 1H, H4′), 6.22–6.21 (m, 2H, H2′,6′), 3.86 (s, 3H, CH3), 3.76 (s, 6H, 2CH3), 2.59 (s, 3H, COCH3); 13C NMR (100 MHz, CDCl3) δ 198.2 (C=O), 162.3 (C2), 161.9 (2C3′,5′), 161.0 (C4), 157.5 (C1′), 132.6 (C6), 123.0 (C1), 109.9 (C5), 101.9 (C3), 98.5 (2C2′,6′), 96.9 (C4′), 55.8 (OCH3), 55.6 (2OCH3), 31.9 (COCH3); IR (cm−1): 2839, 1644, 1579, 1462, 1419, 1357, 1257, 1195, 1149, 1128, 1051, 1028; MS (ESI) m/z (%): 303.2 (100) [M + H]+; UPLC purity = 98%, tR 2.98 min.
1,3-Dimethoxy-5-(3′-methoxyphenoxy)benzene (29)
The reaction was carried out using 3,5-dimethoxyphenol 4 (200 mg, 1.30 mmol), Cs2CO3 (847 mg, 2.60 mmol), CuI (25 mg, 0.13 mmol), and 1-iodo-3-methoxybenzene 24 (455 mg, 1.95 mmol) in DMF (3 mL). Purification by flash column chromatography using cyclohexane/ethyl acetate (9/1) as the eluent afforded compound 29 (103 mg, 30% yield) as a colorless oil. Rf 0.55 (cyHex/AcOEt 8:2); 1H NMR (400 MHz, CDCl3) δ 7.15 (t, J = 8.1 Hz, 1H, H5′), 6.64–6.49 (m, 3H, H2′,4′,6′), 6.15 (t, J = 2.2 Hz, 1H, H2), 6.11 (d, J = 2.4 Hz, 1H), 6.11 (d, J = 2.4 Hz, 1H), 3.71 (s, 3H, CH3), 3.67 (s, 6H, 2CH3).; 13C NMR (100 MHz, CDCl3) δ 161.5 (2C1,3), 160.9 (C3′), 158.9 (C1′), 157.9 (C5), 130.0 (C5), 111.3 (C6′), 109.1 (C2′), 105.1 (C4′), 97.4 (2C4,6), 95.6 (C2), 55.4 (2CH3), 55.3 (CH3); IR (cm−1): 2835, 1583, 1261, 1193, 1143, 1126, 1047; MS (ESI) m/z (%): 261.1 (100) [M + H]+; UPLC purity = 99%, tR 3.20 min.
1-[4-(3′,5′-Dimethoxyphenoxy)phenyl]ethanone (30)
The reaction was carried out using 3,5-dimethoxyphenol 4 (1.00 g, 6.5 mmol), Cs2CO3 (4.23g, 13 mmol), CuI (124 mg, 0.65 mmol), and 4-iodoacetophenone 25 (2.40 g, 9.7 mmol) in DMF (5 mL). Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 30 (1.20 g, 71% yield) as a brown powder. Rf 0.60 (cyHex/AcOEt 8:2); Mp: 85–86 °C; 1H NMR (400 MHz, CDCl3) δ 7.96 (mt, 2H, H2,6), 7.05 (mt, 2H, H3,5), 6.32 (t, J = 2.2 Hz, 1H, H4′), 6.24 (d, J = 2.3 Hz, 2H, H2′,6′), 3.79 (s, 6H, 2CH3), 2.60 (s, 3H, COCH3); 13C NMR (100 MHz, CDCl3) δ 196.7 (C=O), 161.7 (2C3′,5′), 161.5 (C4), 157.3 (C1′), 132.1 (C1), 130.5 (2C2,6), 117.6 (2C3,5), 98.5 (2C2′,6′), 96.7 (C4′), 55.4 (2CH3), 26.4 (COCH3); IR (cm−1): 1674, 1585, 1425, 1361, 1267, 1230, 1136, 1053; MS (ESI) m/z (%): 273.2 (100) [M + H]+; UPLC purity = 99%, tR 2.92 min.
1-(4′-Fluorophenoxy)-3,5-dimethoxybenzene (31)
The reaction was carried out using 3,5-dimethoxyphenol 4 (1.00 g, 6.5 mmol), Cs2CO3 (4.23 g, 13 mmol), CuI (124 mg, 0.65 mmol), and 1-fluoro-4-iodobenzene 26 (1.1 mL, 9.75 mmol) in DMF (5 mL). Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 31 (1.23 g, 76% yield) as a yellow oil. Rf 0.30 (cyHex/CH2Cl2 8:2); 1H NMR (400 MHz, DMSO-d6) δ 7.22 (t, J = 8.4 Hz, 2H, H3′,5′), 7.08 (dd, J = 8.4, 4.6 Hz 2H, H2′,6′), 6.28 (t, J = 2.4Hz, 1H, H4), 6.11 (d, J = 2.4Hz, 2H, H2,6), 3.70 (s, 6H, 2CH3); 13C NMR (100 MHz, DMSO-d6) δ 161.3 (2C3,5), 158.9 (C1), 158.3 (d, JCF = 238 Hz, C4′), 152.1 (d, JCF = 2 Hz, C1′), 120.8 (d, JCF = 8 Hz, 2C2′,6′), 116.4 (d, JCF = 24 Hz, 2C3′,5′), 96.6 (2C2,6), 95.3 (C4), 55.2 (2CH3); IR (cm−1): 2943, 2839, 1591, 1499, 1462, 1238, 1150, 824; MS (ESI) m/z (%): 249.1 (100) [M + H]+; UPLC purity = 100%, tR 3.55 min.
1,3-Dimethoxy-5-[4′-(trifluoromethyl)phenoxy]benzene (32)
The reaction was carried out using 3,5-dimethoxyphenol 4 (500 mg, 3.25 mmol), Cs2CO3 (2.12 g, 6.5 mmol), CuI (62 mg, 325 µmol), and 1-iodo-4-(trifluoromethyl)benzene 27 (720 µL, 4.87 mmol) in DMF (3 mL). Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 32 (678 mg, 70% yield) as a colorless oil. Rf 0.29 (cyHex/CH2Cl2 8:2); 1H NMR (400 MHz, CDCl3) δ 7.58 (d, J = 8.5 Hz, 2H, H3′,5′), 7.08 (d, J = 8.5 Hz, 2H, H2′,6′), 6.30 (t, J = 2.2 Hz, 1H, H2), 6.21 (d, J = 2.2 Hz, 2H, H4,6), 3.77 (s, 6H, 2CH3); 13C NMR (100 MHz, CDCl3) δ 161.9 (2C1,3), 160.1 (C5), 157.6 (C1′), 127.1 (q, JCF = 4 Hz, 2C3′,5′), 125.2 (q, JCF = 310 Hz, CF3), 122.8 (C4′), 118.2 (2C2′,6′), 98.3 (2C4,6), 96.6 (C2), 55.5 (2CH3); IR (cm−1): 2943, 1589, 1321, 1105, 835; MS (ESI) m/z (%): 299.1 (100) [M + H]+; UPLC purity = 99%, tR 3.42 min.
3.1.5. General Procedure for the Preparation of 1,3-dimethoxydibenzofurans 33–37
Diaryl ethers 28–32 (1 equiv.) were dissolved in warm pivalic acid (50 °C, excess). Silver acetate (5 equiv.) and palladium (II) acetate (0.1 equiv.) were added, and the reaction mixture was properly stirred at 130 °C for 4–24 h. After cooling to room temperature, the mixture was diluted with methanol (50 mL) and NaOH (1 M) was added until the pH reached 9. Elemental silver was removed by filtration through a pad of Celite®. The filtrate was extracted with ethyl acetate (100 mL). The organic layers were washed with brine (2 × 100 mL), dried over sodium sulfate, filtered, and evaporated to dryness. The crude product was then purified by chromatography to provide 33–37.
1-(3,7,9-Trimethoxydibenzo[b,d]furan-2-yl)ethanone (33)
The reaction was carried out using 1-[4-(3,5-dimethoxyphenoxy)-2-methoxyphenyl]ethanone 28 (100 mg, 0.33 mmol) and AgOAc (275 mg, 1.65 mmol), Pd(OAc)2 (9 mg, 0.03 mmol) in PivOH (5 g) for 4 h. Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 33 (86 mg, 87% yield) as a brown powder. Rf 0.48 (cyHex/AcOEt 8:2); Mp: 155–156 °C; 1H NMR (400 MHz, DMSO-d6) δ 8.10 (s, 1H, H1), 7.46 (s, 1H, H4), 6.92 (d, J = 1.9 Hz, 1H, H6), 6.57 (d, J = 1.9 Hz, 1H, H8), 3.99 (s, 3H, CH3), 3.98 (s, 3H, CH3), 3.86 (s, 3H, CH3), 2.58 (s, 3H, COCH3); 13C NMR (100 MHz, DMSO-d6) δ 198.0 (C=O), 160.6 (C7), 158.5 (C3), 158.0 (C), 157.9 (C), 154.9 (C9), 124.1 (C2), 121.9 (C1), 115.9 (C), 105.4 (C), 95.8 (C8), 94.5 (C6), 89.2 (C4), 56.3 (CH3), 55.9 (CH3), 55.8 (CH3), 31.6 (CH3); IR (cm−1): 1664, 1600, 1462, 1408, 1354, 1269, 1236, 1139, 1095; MS (ESI) m/z (%): 301.2 (100) [M + H]+; UPLC purity = 99%, tR 3.00 min.
1,3,7-Trimethoxydibenzo[b,d]furan (34)
The reaction was carried out using 1,3-dimethoxy-5-(3-methoxyphenoxy)benzene 29 (100 mg, 0.38 mmol), AgOAc (317 mg, 1.90 mmol), and Pd(OAc)2 (9 mg, 0.04 mmol) in PivOH (5 g) for 12 h. Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 34 (90 mg, 91% yield) as a brown powder. Rf 0.51 (cyHex/AcOEt 8:2); Mp: 153–154 °C; 1H NMR (400 MHz, CDCl3) δ 7.78 (d, J = 8.4 Hz, 1H, H9), 6.97 (d, J = 2.2 Hz, 1H, H6), 6.82 (dd, J = 8.4, 2.3 Hz, 1H, H8), 6.61 (d, J = 1.9 Hz, 1H, H4), 6.32 (d, J = 2.0 Hz, 1H, H2), 3.91 (s, 3H, CH3), 3.80 (s, 3H, CH3), 3.80 (s, 3H, CH3); 13C NMR (100 MHz, CDCl3) δ 160.0 (C3), 158.2 (C7), 158.1 (C), 156.6 (C), 155.2 (C1), 121.8 (C9), 117.0 (C), 110.4 (C8), 107.2 (C), 96.4 (C2), 93.8 (C4), 88.7 (C6), 55.8 (CH3), 55.7 (CH3), 55.6 (CH3); IR (cm−1): 2940, 2832, 1594, 1435, 1365, 1320, 1264, 1206, 1125, 1036; MS (ESI) m/z (%): 259.1 (100) [M + H]+; UPLC purity = 99%, tR 3.24 min.
1-(7,9-Dimethoxydibenzo[b,d]furan-2-yl)ethanone (35)
The reaction was carried out using 1-[4-(3,5-dimethoxyphenoxy)phenyl]ethanone 30 (1.00 g, 3.7 mmol), AgOAc (3.06 g, 18.5 mmol), and Pd(OAc)2 (80 mg, 0.37 mmol) in PivOH (50 g) for 24 h. Purification by flash column chromatography using cyclohexane/ethyl acetate (8/2) as the eluent afforded compound 35 (0.83 g, 83% yield) as a brown powder. Rf 0.34 (cyHex/AcOEt 8:2); Mp: 162–163 °C; 1H NMR (400 MHz, CDCl3) δ 8.60 (d, J = 1.9 Hz, 1H, H1), 8.02 (dd, J = 8.6, 2.0 Hz, 1H, H3), 7.52 (d, J = 8.6 Hz, 1H, H4), 6.74 (d, J = 1.9 Hz, 1H, H6), 6.46 (d, J = 1.9 Hz, 1H, H8), 4.06 (s, 3H, CH3), 3.92 (s, 3H, CH3), 2.73 (s, 3H, COCH3); 13C NMR (100 MHz, CDCl3) δ 197.6 (C=O), 161.6 (C7), 158.8 (C), 158.3 (C9), 156.1 (C), 132.7 (C2), 125.8 (C3), 124.1 (C), 122.6 (C1), 110.6 (C4), 106.7 (C), 94.4 (C8), 88.6 (C6), 55.8 (CH3), 55.7 (CH3), 26.8 (COCH3); IR (cm−1): 1674, 1637, 1618, 1581, 1510, 1425, 1408, 1361, 1315, 1290, 1253, 1215, 1197, 1151, 1093, 1045, 1010; MS (ESI) m/z (%): 271.2 (100) [M + H]+; UPLC purity = 98%, tR 2.94 min.
8-Fluoro-1,3-dimethoxydibenzo[b,d]furan (36)
The reaction was carried out using 1-(4-fluorophenoxy)-3,5-dimethoxybenzene 31 (1.00 g, 4.03 mmol), AgOAc (3.36 g, 20.15 mmol), and Pd(OAc)2 (90 mg, 0.40 mmol) in PivOH (50 g) for 24 h. Purification by flash column chromatography using cyclohexane/dichloromethane (8/2) as the eluent afforded compound 36 (414 mg, 42% yield) as a white powder. Rf 0.23 (cyHex/CH2Cl2 8:2); Mp: 137–138 °C; 1H NMR (400 MHz, CDCl3) δ 7.66 (dd, J = 8.5, 2.7 Hz, 1H, H9), 7.39 (dd, J = 8.9, 4.1 Hz, 1H, H6), 7.02 (dt, J = 8.9, 2.7 Hz, 1H, H7), 6.68 (d, J = 2.0 Hz, 1H, H2), 6.39 (d, J = 2.0 Hz, 1H, H4), 4.00 (s, 3H, CH3), 3.89 (s, 3H, CH3); 13C NMR (100 MHz, CDCl3) δ 161.6 (C3), 159.2 (C4a), 159.1 (d, JCF = 238 Hz, C8), 156.0 (C1), 151.5 (C9b), 124.7 (d, JCF = 11 Hz, C9a), 111.7 (d, JCF = 26 Hz, C7), 111.0 (d, JCF = 9 Hz, C6), 107.9 (d, JCF = 25 Hz, C9), 107.2 (d, JCF = 3 Hz, C5a), 93.9 (C4), 88.5 (C2), 55.8 (CH3), 55.7 (CH3); IR (cm−1): 1674, 1637, 1618, 1581, 1510, 1425, 1408, 1361, 1315, 1290, 1253, 1215, 1197, 1151, 1093, 1045, 1010; MS (ESI) m/z (%): 247.1 (100) [M + H]+; UPLC purity = 99%, tR 3.23 min.
1,3-Dimethoxy-8-(trifluoromethyl)dibenzo[b,d]furan (37)
The reaction was carried out using 1,3-dimethoxy-5-(4-(trifluoromethyl)phenoxy)benzene 32 (500 mg, 1.65 mmol), AgOAc (560 mg, 8.38 mmol), and Pd(OAc)2 (37 mg, 0.165 mmol) in PivOH (25 g) for 24 h. Purification by flash column chromatography using cyclohexane/dichloromethane (95/5) as the eluent afforded compound 37 (190 mg, 39% yield) as a white powder. Rf 0.21 (cyHex/CH2Cl2 99:1); Mp: 131–132 °C; 1H NMR (400 MHz, CDCl3) δ 8.25 (s, 1H, H9), 7.58 (dd, J = 8.4, 1.9 Hz, 1H, H7), 7.53 (d, J = 8.7 Hz, 1H, H6), 6.72 (d, J = 2.0 Hz, 1H, H2), 6.43 (d, J = 2.0 Hz, 1H, H4), 4.03 (s, 3H, CH3), 3.90 (s, 3H, CH3); 13C NMR (100 MHz, CDCl3) δ 161.9 (C3), 158.9 (C4a), 157.0 (C9b), 156.1 (C1), 126.1 (C8), 125.3 (q, JCF = 310 Hz, CF3), 124.2 (C9a), 121.9 (q, JCF = 4 Hz, C7), 119.2 (q, JCF = 4 Hz, C9), 110.8 (C6), 106.5 (C5a), 94.4 (C4), 88.6 (C2), 55.9 (CH3), 55.7 (CH3); IR (cm−1): 2947, 1620, 1321, 1151, 1094, 808; MS (ESI) m/z (%): 297.1 (100) [M + H]+; UPLC purity = 100%, tR 3.47 min.
3.1.6. General Procedure for the Preparation of 1,3-dimethoxydibenzofuran-4-carboxamides 38–42
To a stirred suspension of 1,3-dimethoxydibenzofurans 33–37 (1 equiv.) in acetonitrile at 0 °C under argon, chlorosulfonyl isocyanate (2 equiv.) was added. The mixture was maintained at room temperature for 6-12 h and then the reaction mixture was quenched with HCl 5 M (20 mL). After 6–12 h at room temperature, the reaction was diluted with water (100 mL) and extracted with ethyl acetate (100 mL). The combined organic extracts were washed with brine (2 × 100 mL), dried over sodium sulfate, filtered, and evaporated to dryness. The crude product was then purified by chromatography to provide 38–42.
8-Acetyl-1,3,7-trimethoxydibenzo[b,d]furan-4-carboxamide (38)
Method A: The reaction was carried out using 1-(3,7,9-trimethoxydibenzo[b,d]furan-2-yl)ethanone 33 (100 mg, 0.33 mmol), CSI (58 µL, 0.66 mmol) in CH3CN (10 mL) for 12 h and then 12 h for the hydrolysis step at 60 °C, as an exception. Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 38 (54 mg, 48% yield) as a white powder.
Method B: Anhydrous AlCl3 (111 mg, 0.83 mmol) was added to acetyl chloride (24 µL, 0.33 mmol) in 1,2-dichloroethane (10 mL). Then, 1,3,7-trimethoxydibenzo[b,d]furan-4-carboxamide 39 (see below) in 1,2-dichloroethane (20 mL) was added slowly and the mixture was left at room temperature for 1 h. After the reaction (TLC control) was completed, the reaction mixture was quenched with 1 N HCl and extracted with dichloromethane (2 × 25 mL). The combined organic extracts were washed with water, dried over anhydrous sodium sulfate, filtered, and concentrated in vacuum. The crude product obtained was purified over silica gel column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent to obtain the pure product 38 (42 mg, 74% yield).
Rf 0.59 (CH2Cl2/MeOH 9:1); Mp: 228–229 °C; 1H NMR (400 MHz, DMSO-d6) δ 8.13 (s, 1H, H9), 7.70 (s, 1H, CONH2), 7.54 (m, 2H, CONH2, H6), 6.72 (s, 1H, H2), 4.08 (s, 3H, CH3), 3.97 (s, 3H, CH3), 3.93 (s, 3H, CH3), 2.58 (s, 3H, COCH3); 13C NMR (100 MHz, DMSO-d6) δ 197.9 (COCH3), 164.2 (CONH2), 158.6 (C), 158.3 (C7), 157.1 (C), 155.1 (C1), 154.9 (C3), 124.2 (C8), 122.1 (C9), 115.4 (C), 105.5 (C), 105.0 (C4), 96.0 (C6), 91.6 (C2), 56.7 (CH3), 56.4 (CH3), 56.2 (CH3), 31.6 (COCH3); IR (cm−1): 3387, 3197, 1606, 1402, 1317, 1273, 1089; MS (ESI) m/z (%): 344.2 (100) [M + H]+; UPLC purity = 98%, tR 2.15 min; HRMS (TOF MS ES+): calcd. for C18H18NO6 [M + H]+ 344.112864, found: 344.11210.
1,3,7-Trimethoxydibenzo[b,d]furan-4-carboxamide (39)
The reaction was carried out using 1,3,7-trimethoxydibenzo[b,d]furan 34 (100 mg, 0.38 mmol) and CSI (67 µL, 0.77 mmol) in CH3CN (10 mL) for 6 h and then 6 h for the hydrolysis step. Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 39 (50 mg, 44% yield) as a white powder. Rf 0.67 (CH2Cl2/MeOH 9:1); Mp: 332–333 °C; 1H NMR (400 MHz, DMSO-d6) δ 7.79 (d, J = 8.5 Hz, 1H, H9), 7.67 (s, 1H, CONH2), 7.48 (s, 1H, CONH2), 7.30 (d, J = 2.3 Hz, 1H, H6), 6.95 (dd, J = 8.5, 2.3 Hz, 1H, H8), 6.69 (s, 1H, H2), 4.07 (s, 3H, CH3), 3.92 (s, 3H, CH3), 3.84 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 164.5 (C=O), 158.2 (C7), 156.5 (C), 156.1 (C), 154.8 (C1), 154.4 (C3), 121.4 (C9), 115.5 (C), 110.8 (C8), 106.1 (C4), 104.9 (C), 96.7 (C6), 91.2 (C2), 56.7 (CH3), 56.0 (CH3), 55.6 (CH3); IR (cm−1): 3377, 3192, 2970, 2839, 1643, 1610, 1454, 1431, 1375, 1321, 1267, 1209, 1107, 1026; MS (ESI) m/z (%): 302.1 (100) [M + H]+; UPLC purity = 99%, tR 2.26 min; HRMS (TOF MS ES+): calcd. for C16H16NO5 [M + H]+ 302.102299, found: 302.10192.
8-Acetyl-1,3-dimethoxydibenzo[b,d]furan-4-carboxamide (40)
The reaction was carried out using 1-(7,9-dimethoxydibenzo[b,d]furan-2-yl)ethanone 35 (200 mg, 0.74 mmol) and CSI (128 µL, 1.48 mmol) in CH3CN (20 mL) for 12 h and then 12 h for the hydrolysis step. Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 40 (74 mg, 32% yield) as a yellow powder. Rf 0.61 (CH2Cl2/MeOH 9:1); Mp: 237-238 °C; 1H NMR (400 MHz, DMSO-d6) δ 8.48 (s, 1H, H9), 8.07 (d, J = 8.8 Hz, 1H, H7), 7.90–7.43 (m, 3H, H6, CONH2), 6.78 (s, 1H, H2), 4.14 (s, 3H, CH3), 3.96 (s, 3H, CH3), 2.68 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 197.0 (COCH3), 164.1 (CONH2), 158.2 (C3), 157.5 (C1), 155.9 (C), 155.1 (C), 132.6 (C8), 126.6 (C7), 122.9 (C), 121.3 (C9), 110.9 (C6), 105.3 (C4), 104.8 (C), 91.6 (C2), 56.7 (CH3), 56.3 (CH3), 26.8 (COCH3); IR (cm−1): 3381, 1649, 1612, 1415, 1365, 1321, 1286, 1215, 1097; MS (ESI) m/z (%): 314.2 (100) [M + H]+; UPLC purity = 100%, tR 1.99 min; HRMS (TOF MS ES+): calcd. for C17H16NO5 [M + H]+ 314.102299, found: 314.10187.
8-Fluoro-1,3-dimethoxydibenzo[b,d]furan-4-carboxamide (41)
The reaction was carried out using 8-fluoro-1,3-dimethoxydibenzo[b,d]furan 36 (185 mg, 0.75 mmol) and CSI (130 µL, 1.5 mmol) in CH3CN (20 mL) for 12 h and then 12 h for the hydrolysis step. Purification by reverse phase column chromatography (C18) using water/acetonitrile (7/3) as the eluent afforded, after lyophilization, compound 41 (76 mg, 35% yield) as a white powder. Rf 0.26 (AcOEt); Mp: 230–231 °C; 1H NMR (400 MHz, DMSO-d6) δ 7.71–7.67 (s, 1H, NH), 7.68 (dd, J = 8.9, 4.1 Hz, 1H, H6), 7.64 (dd, J = 8.4, 2.7 Hz, 1H, H9), 7.51 (s, 1H, NH), 7.24 (dt, J = 8.9, 2.7 Hz, 1H, H7), 6.72 (s, 1H, H2), 4.09 (s, 3H, CH3), 3.95 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 164.3 (C=O), 158.5 (d, JCF = 235 Hz, C8), 158.2 (C3), 155.8 (C1), 155.5 (C4a), 151.1 (C9b), 123.6 (d, JCF = 11 Hz, C9a), 112.4 (d, JCF = 26 Hz, C7), 112.1 (d, JCF = 9 Hz, C6), 107.1 (d, JCF = 26 Hz, C9), 105.8 (C5a), 104.7 (C4), 91.2 (C2), 56.7 (CH3), 56.2 (CH3); IR (cm−1): 3381, 1649, 1612, 1415, 1365, 1321, 1286, 1215, 1097; MS (ESI) m/z (%): 290.1 (100) [M + H]+; UPLC purity = 99%, tR 2.23 min; HRMS (TOF MS ES+): calcd. for C15H13FNO4 [M + H]+ 290.075037, found: 290.07422.
1,3-Dimethoxy-8-(trifluoromethyl)dibenzo[b,d]furan-4-carboxamide (42)
The reaction was carried out using 1,3-dimethoxy-8-(trifluoromethyl)dibenzo[b,d]furan 37 (145 mg, 0.50 mmol) and CSI (85 µL, 1.0 mmol) in CH3CN (15 mL) for 12 h then 12 h for the hydrolysis step. Purification by flash column chromatography using ethyl acetate/methanol (95/5) as the eluent afforded compound 42 (58 mg, 35% yield) as a white powder. Rf 0.30 (AcOEt); Mp: 250–251 °C; 1H NMR (400 MHz, DMSO-d6) δ 8.22 (d, J = 1.8 Hz, 1H, H9), 7.90 (d, J = 8.7 Hz, 1H, H6), 7.78 (dd, J = 8.7, 1.8 Hz, 1H, H7), 7.74 (s, 1H, NH), 7.59 (s, 1H, NH), 6.81 (s, 1H, H2), 4.17 (s, 3H, CH3), 4.02 (s, 3H, CH3); 13C NMR (100 MHz, DMSO-d6) δ 164.1 (C=O), 158.6 (C3), 156.6 (C4a), 156.1 (C1), 155.3 (C9b), 125.9 (C8), 124.2 (q, JCF = 310 Hz, CF3), 123.2 (C9a), 122.5 (q, JCF = 4 Hz, C7), 118.1 (q, JCF = 4 Hz, C9), 111.8 (C6), 105.0 (C5a), 104.6 (C4), 91.6 (C2), 56.7 (CH3), 56.3 (CH3); IR (cm−1): IR: 3379, 3179, 2974, 1649, 1618, 1319, 1115, 814; MS (ESI) m/z (%): 340.1 (100) [M + H]+; UPLC purity = 98%, tR 2.64 min; HRMS (TOF MS ES+): calcd. for C16H13F3NO4 [M + H]+ 340.071843, found: 340.07112.
3.1.7. General Procedure for the Preparation of 1,3-hydroxydibenzofuran-4-carboxamide 43–47
A mixture of 1,3-dimethoxydibenzofuran-4-carboxamides 38-42 (50 mg) in warm pyridine hydrochloride (3g) was irradiated using microwave (100 W) heating at 200 °C for 15 min. After the addition of water, the mixture was extracted with ethyl acetate (100 mL). The organic layers were washed with brine, dried over sodium sulfate, filtered, and evaporated to dryness. The crude product was then purified by chromatography to provide 43–47.
8-Acetyl-1,3,7-trihydroxydibenzo[b,d]furan-4-carboxamide (43)
The reaction was carried out using 8-acetyl-1,3,7-trimethoxydibenzo[b,d]furan-4-carboxamide 38 (50 mg, 0.15 mmol) in warm pyridine hydrochloride (3g). Purification by flash column chromatography using dichloromethane as the eluent afforded compound 43 (17 mg, 39% yield) as a brown powder. Rf 0.53 (CH2Cl2/MeOH 9:1); Mp: 335–336 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.82 (s, 1H, OH), 12.44 (s, 1H, OH), 11.38 (s, 1H, OH), 8.30 (m, 2H, H9, CONH2), 7.81 (s, 1H, CONH2), 7.25 (s, 1H, H6), 6.32 (s, 1H, H2), 2.74 (s, 3H, COCH3); 13C NMR (100 MHz, DMSO-d6) δ 203.7 (COCH3), 169.7 (CONH2), 163.7 (C7), 160.1 (C3), 158.8 (C), 157.0 (C1), 155.7 (C), 123.3 (C9), 117.4 (C), 115.5 (C8), 103.5 (C4), 99.8 (C2), 98.5 (C6), 93.0 (C), 27.8 (COCH3); IR (cm−1): 3462, 3334, 3170, 1614, 1469, 1396, 1246, 1103, 1041; MS (ESI) m/z (%): 302.1 (100) [M + H]+; UPLC purity = 99%, tR 2.17 min; HRMS (TOF MS ES+): calcd. for C15H12NO6 [M + H]+ 302.065914, found: 302.06557.
1,3,7-Trihydroxydibenzo[b,d]furan-4-carboxamide (44)
The reaction was carried out using 1,3,7-trimethoxydibenzo[b,d]furan-4-carboxamide 39 (50 mg, 0.17 mmol) in warm pyridine hydrochloride (3g). Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 44 (16 mg, 38% yield) as a brown powder. Rf 0.67 (CH2Cl2/MeOH 9:1); Mp: 360–361 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.73 (s, 1H, OH), 11.09 (s, 1H, OH), 9.75 (s, 1H, OH), 8.20 (s, 1H, CONH2), 7.80 (s, 1H, CONH2), 7.68 (d, J = 8.5 Hz, 1H, H9), 7.10 (s, 1H, H6), 6.81 (d, J = 8.3 Hz, 1H, H8), 6.25 (s, 1H, H2); 13C NMR (100 MHz, DMSO-d6) δ 170.1 (C=O), 162.8 (C7), 156.7 (C3), 155.7 (C1), 155.5 (C), 154.9 (C), 121.1 (C9), 114.4 (C), 112.0 (C8), 104.7 (C4), 98.5 (C2), 97.8 (C6), 92.7 (C); IR (cm−1): 3423, 3321, 3242, 3219, 1664, 1556, 1502, 1446, 1288, 1257, 1190, 1107, 1029; MS (ESI) m/z (%): 260.1 (100) [M + H]+; UPLC purity = 100%, tR 1.64 min; HRMS (TOF MS ES+): calcd. for C13H10NO5 [M + H]+ 260.055349, found: 260.05591.
8-Acetyl-1,3-dihydroxydibenzo[b,d]furan-4-carboxamide (45)
The reaction was carried out using 8-acetyl-1,3-dimethoxydibenzo[b,d]furan-4-carboxamide 40 (50 mg, 0.16 mmol) in warm pyridine hydrochloride (3g). Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 45 (12 mg, 26% yield) as a yellow powder. Rf 0.56 (CH2Cl2/MeOH 9:1); Mp: 355–356 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.98 (s, 1H, OH), 11.54 (s, 1H, OH), 8.47 (d, J = 1.9 Hz, 1H, H9), 8.32 (s, 1H, CONH2), 8.06 (dd, J = 8.6, 2.0 Hz, 1H, H7), 7.88 (s, 1H, CONH2), 7.81 (d, J = 8.6 Hz, 1H, H6), 6.36 (s, 1H, H2), 2.68 (s, 3H, COCH3); 13C NMR (100 MHz, DMSO-d6) δ 197.0 (COCH3), 169.8 (CONH2), 164.8 (C3), 157.8 (C1), 156.8 (C), 156.0 (C), 133.0 (C8), 125.9 (C7), 123.2 (C), 121.0 (C9), 111.3 (C6), 103.9 (C4), 98.4 (C2), 92.8 (C), 26.8 (COCH3); IR (cm−1): 3479, 3190, 1600, 1415, 1359, 1284, 1249, 1190, 1045; MS (ESI) m/z (%): 286.1 (100) [M + H]+; UPLC purity = 99%, tR 2.04 min; HRMS (TOF MS ES+): calcd. for C15H12NO5 [M + H]+ 286.070999, found: 286.07079.
8-Fluoro-1,3-dihydroxydibenzo[b,d]furan-4-carboxamide (46)
The reaction was carried out using 8-fluoro-1,3-dimethoxydibenzo[b,d]furan-4-carboxamide 41 (50 mg, 0.17 mmol) in warm pyridine hydrochloride (3g). Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 46 (23 mg, 51% yield) as a beige powder. Rf 0.39 (cyHex/AcOEt 6:4); Mp: 342–343 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.99 (s, 1H, OH), 11.54 (s, 1H, OH), 8.29 (s, 1H, NH), 7.82 (s, 1H, NH), 7.71 (dd, J = 8.8, 4.0 Hz, 1H, H6), 7.60 (dd, J = 8.4, 2.8 Hz, 1H, H9), 7.21 (dt, J = 8.8, 2.8 Hz, 1H, H7), 6.33 (s, 1H, H2); 13C NMR (100 MHz, DMSO-d6) δ 170.0 (C=O), 165.0 (C3), 158.8 (d, JCF = 236 Hz, C8), 158.1 (C1), 156.4 (C4a), 150.4 (C9b), 124.0 (d, JCF = 11 Hz, C9a), 112.4 (d, JCF = 9 Hz, C6), 111.7 (d, JCF = 26 Hz, C7), 106.7 (d, JCF = 26 Hz, C9), 104.3 (d, JCF = 3 Hz, C5a), 98.1 (C4), 92.6 (C2); IR (cm−1): 3481, 3188, 3098, 1628, 1474, 1410, 1161, 802; MS (ESI) m/z (%): 262.1 (100) [M + H]+; UPLC purity = 99%, tR 2.28 min; HRMS (TOF MS ES+): calcd. for C13H9FNO4 [M + H]+ 262.043737, found: 262.04354.
1,3-Dihydroxy-8-(trifluoromethyl)dibenzo[b,d]furan-4-carboxamide (47)
The reaction was carried out using 1,3-dimethoxy-8-(trifluoromethyl)dibenzo[b,d]furan-4-carboxamide 42 (50 mg, 0.15 mmol) in warm pyridine hydrochloride (3g). Purification by flash column chromatography using cyclohexane/ethyl acetate (6/4) as the eluent afforded compound 47 (24 mg, 53% yield) as a beige powder. Rf 0.40 (cyHex/AcOEt 6:4); Mp: 347–348 °C; 1H NMR (400 MHz, DMSO-d6) δ 13.99 (s, 1H, OH), 11.98 (s, 1H, OH), 8.33 (s, 1H, NH), 8.14 (d, J = 1.6 Hz, 1H, H9), 7.90 (d, J = 8.8 Hz, 1H, H6), 7.87 (s, 1H, NH), 7.74 (dd, J = 8.8, 1.6 Hz, 1H, H7), 6.53 (s, 1H, H2); 13C NMR (100 MHz, DMSO-d6) δ 169.8 (C=O), 165.1 (C3), 158.3 (C1), 156.1 (C4a), 155.9 (C9b), 125.9 (C8), 124.6 (q, JCF = 310 Hz, CF3), 123.6 (C9a), 121.9 (q, JCF = 4 Hz, C7), 117.6 (q, JCF = 4 Hz, C9), 112.3 (C6), 103.6 (C5a), 98.7 (C4), 92.6 (C2); IR (cm−1): 3474, 3217, 1632, 1308, 1109, 887, 818; MS (ESI) m/z (%): 312.1 (100) [M + H]+; UPLC purity = 99%, tR 2.58 min; HRMS (TOF MS ES+): calcd. for C14H9F3NO4 [M + H]+ 312.040543, found: 312.04098.