Antioxidant and Antisteatotic Activities of a New Fucoidan Extracted from Ferula hermonis Roots Harvested on Lebanese Mountains
Abstract
1. Introduction
2. Results
2.1. Extraction Yield and Chemical Composition
2.2. Spectroscopic Characterization
2.3. Radical Scavenging Capacity
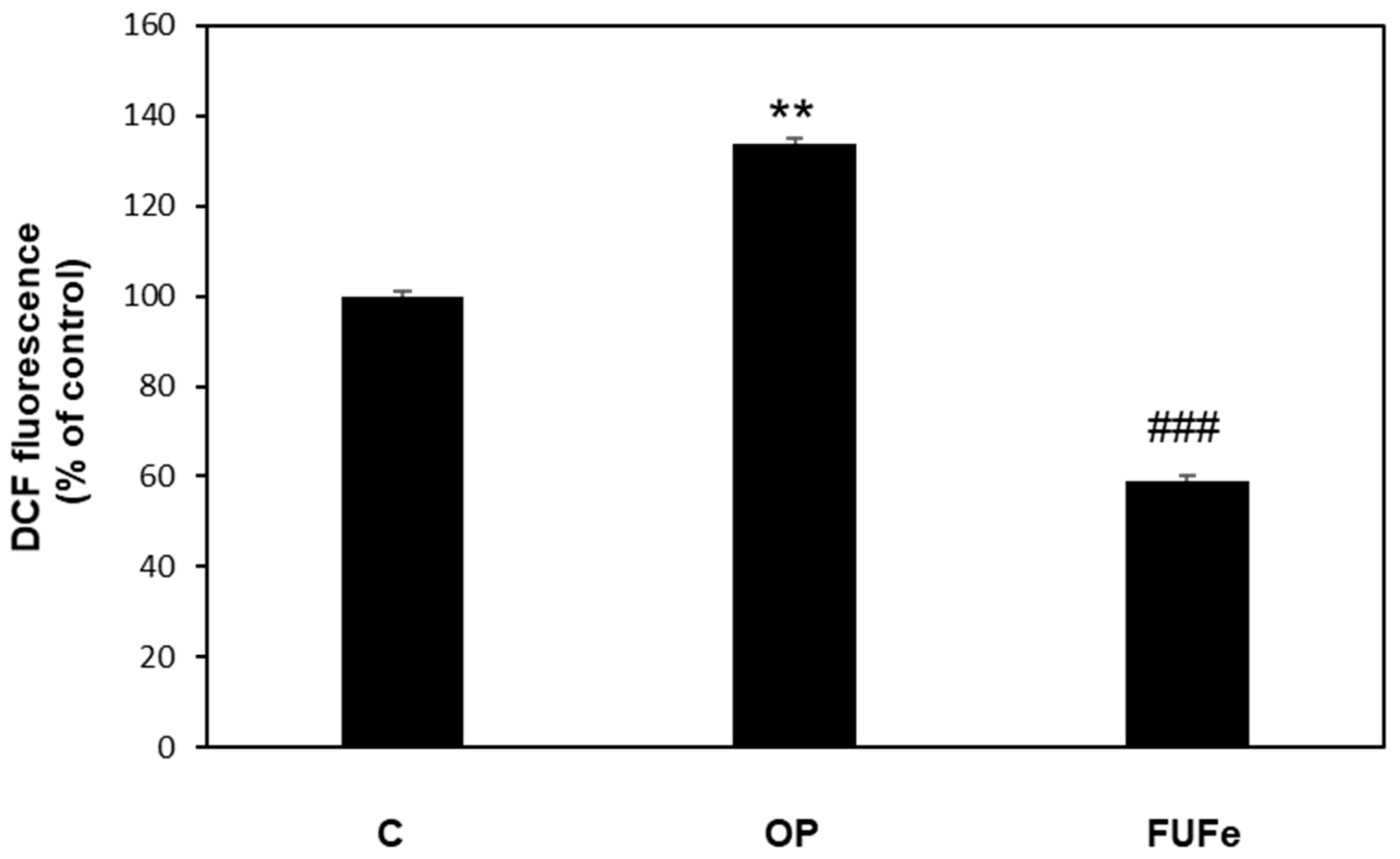
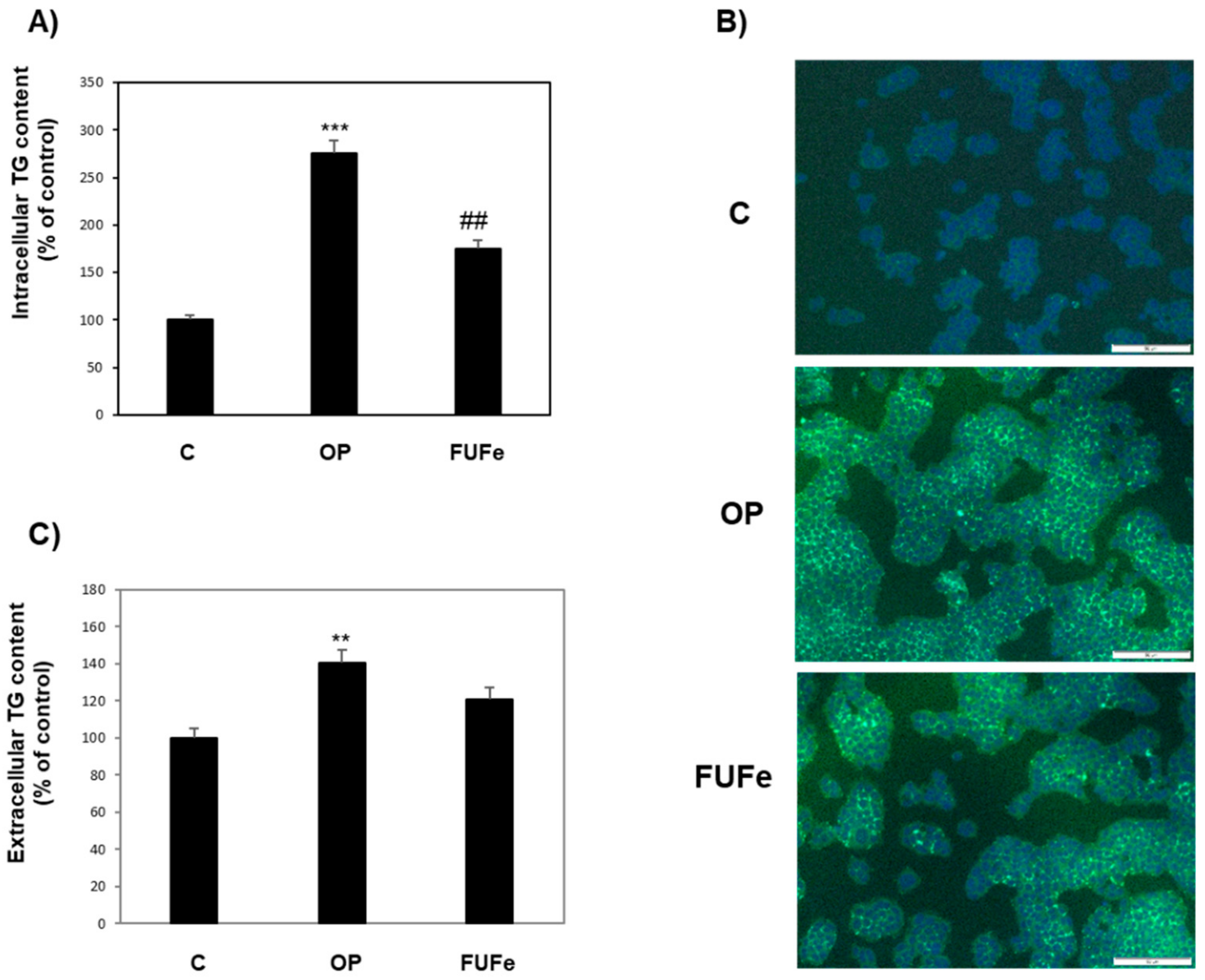
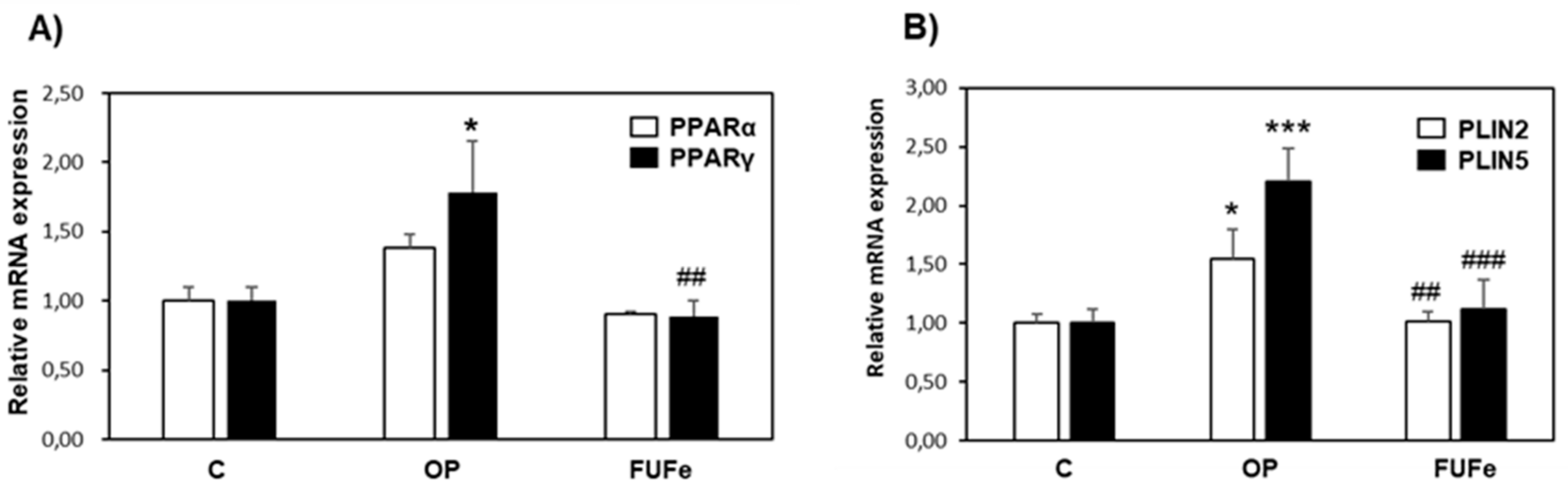
2.4. Antioxidant and Antisteatotic Activities of FUFe in a Cellular Model of Hepatic Steatosis
2.5. Effects of FUFe on Steatotic Endothelial Cells
3. Discussion
4. Materials and Methods
4.1. Biological Material and Chemicals
4.2. Extraction and Purification of Fucoidan
4.3. Chemical Characterization
4.4. Radical Scavenging Capacity
4.4.1. DPPH (2,2-diphenyl-1-picrylhydrazyl radical) Assay
4.4.2. ABTS (2,2-azinobis-3-ethylbenzothiazoline-6-sulfonic acid) Assay
4.4.3. FRAP (Ferric Reducing Antioxidant Power) Assay
4.5. Cell Culture and Treatments
4.6. ROS Production
4.7. Quantification of Triglycerides (TGs)
4.8. Nitric Oxide (NO) Production
4.9. RNA Extraction and Quantitative Real-Time PCR
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Petrovska, B.B. Historical review of medicinal plants’ usage. Pharmacogn. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef]
- Nediani, C.; Giovannelli, L. Oxidative Stress and Inflammation as Targets for Novel Preventive and Therapeutic Approches in Non Communicable Diseases. Antioxidants 2020, 9, 290. [Google Scholar] [CrossRef] [PubMed]
- Fitton, J.H.; Stringer, D.N.; Karpiniec, S.S. Therapies from Fucoidan: An Update. Mar. Drugs 2015, 13, 5920–5946. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xing, M.; Cao, Q.; Ji, A.; Liang, H.; Song, S. Biological Activities of Fucoidan and the Factors Mediating Its Therapeutic Effects: A Review of Recent Studies. Mar. Drugs 2019, 17, 183. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef] [PubMed]
- Haddad, M.; Zein, S.; Shahrour, H.; Hamadeh, K.; Karaki, N.; Kanaan, H. Antioxidant activity of water-soluble polysaccharide extracted from Eucalyptus cultivated in Lebanon. Asian Pac. J. Trop. Biomed. 2017, 7, 157–160. [Google Scholar] [CrossRef]
- Sattar, Z.; Iranshahi, M. Phytochemistry and pharmacology of Ferula persica Boiss.: A. review. Iran J. Basic Med. Sci. 2017, 20, 1–8. [Google Scholar] [CrossRef]
- Abdel-Kader, M.M.; Kassem, S.S.; Danial, E.N.; El Raouf, A.A.; Hanafi, E.M. Evaluation of Ferula Hermonis Extract as a growth promoter. Int. J. Acad. Res. 2011, 3, 8. [Google Scholar]
- Saboural, P.; Chaubet, F.; Rouzet, F.; Al-Shoukr, F.; Azzouna, R.B.; Bouchemal, N.; Picton, L.; Louedec, L.; Maire, M.; Rolland, L.; et al. Purification of a low molecular weight fucoidan for SPECT molecular imaging of myocardial infarction. Mar. Drugs 2014, 12, 4851–4867. [Google Scholar] [CrossRef]
- Myoung, L.C.; Boo, Y.L.; SangGua, Y. Relationship between Oversulfation and Conformation of Low and High Molecular Weight Fucoidans and Evaluation of Their in Vitro Anticancer Activity. Molecules 2011, 16, 8. [Google Scholar]
- Somasundaram, S.N.; Shanmugam, S.; Subramanian, B.; Jaganathan, R. Cytotoxic effect of fucoidan extracted from Sargassum cinereum on colon cancer cell line HCT-15. Int. J. Biol. Macromol. 2016, 91, 1215–1223. [Google Scholar] [CrossRef]
- Alwarsamy, M.; Gooneratne, R.; Ravichandran, R. Effect of fucoidan from Turbinaria conoides on human lung adenocarcinoma epithelial (A549) cells. Carbohydr. Polym. 2016, 152, 207–213. [Google Scholar] [CrossRef]
- Ajisaka, K.; Yokoyama, T.; Matsuo, K. Structural Characteristics and Antioxidant Activities of Fucoidans from Five Brown Seaweeds. J. Appl. Glycosci. 2016, 63, 31–37. [Google Scholar] [CrossRef]
- Haddad, M.; Zein, S.; Hazimeh, G.; Karaki, R.; Krivoruchko, E.; Mackhour, Y.; Kassem, Z.; Kanaan, H. Antitumor and Antioxidant Properties of Polysaccharides isolated from the brown algae Stypopodium schimperi growing on the Lebanese coast. Adv. Res. J. Multidiscip. Discov. 2017, 17, 8. [Google Scholar]
- Zein, S.; Haddad, M.; Krivoruchko, E.; Sobolev, A.P.; Azar, S.; Kanaan, H. A New Molecule of Water-Soluble Polysaccharide Isolated from Eucalyptus Growing in Lebanon. EJPMR 2018, 15, 7. [Google Scholar]
- Laeliocattleya, R.A.; Yunianta; Suloi, A.F.; Gayatri, P.P.; Putri, N.A.; Anggraeni, Y.C. Fucoidan Content from Brown Seaweed (Sargassum filipendula) and Its Potential as Radical Scavenger. J. Phys. Conf. Ser. 2020, 1430, 23–53. [Google Scholar] [CrossRef]
- Zou, C.; Du, Y.M.; Li, Y.; Yang, J.H.; Feng, T.; Zhang, L.; Kennedy, J.F. Preparation of lacquer polysaccharide sulfates and their antioxidant activity in vitro. Carbohydr. Polym. 2008, 73, 2. [Google Scholar] [CrossRef]
- Grasselli, E.; Canesi, L.; Portincasa, P.; Voci, A.; Vergani, L.; Demori, I. Models of non-Alcoholic Fatty Liver Disease and Potential Translational Value: The Effects of 3,5-L-diiodothyronine. Ann. Hepatol. 2017, 16, 707–719. [Google Scholar] [CrossRef] [PubMed]
- Grasselli, E.; Voci, A.; Canesi, L.; Goglia, F.; Ravera, S.; Panfoli, I.; Gallo, G.; Vergani, L. Non-receptor-mediated actions are responsible for the lipid-lowering effects of iodothyronines in FaO rat hepatoma cells. J. Endocrinol. 2011, 210, 59–69. [Google Scholar] [CrossRef]
- Souza-Mello, V. Peroxisome proliferator-activated receptors as targets to treat non-alcoholic fatty liver disease. World J. Hepatol. 2015, 7, 1012–1019. [Google Scholar] [CrossRef]
- Kimmel, A.R.; Brasaemle, D.L.; McAndrews-Hill, M.; Sztalryd, C.; Londos, C. Adoption of PERILIPIN as a unifying nomenclature for the mammalian PAT-family of intracellular lipid storage droplet proteins. J. Lipid Res. 2010, 51, 468–471. [Google Scholar] [CrossRef]
- Schadinger, S.E.; Bucher, N.L.; Schreiber, B.M.; Farmer, S.R. PPARgamma2 regulates lipogenesis and lipid accumulation in steatotic hepatocytes. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E1195–E1205. [Google Scholar] [CrossRef]
- Wolins, N.E.; Quaynor, B.K.; Skinner, J.R.; Tzekov, A.; Croce, M.A.; Gropler, M.C.; Varma, V.; Yao-Borengasser, A.; Rasouli, N.; Kern, P.A.; et al. OXPAT/PAT-1 is a PPAR-induced lipid droplet protein that promotes fatty acid utilization. Diabetes 2006, 55, 3418–3428. [Google Scholar] [CrossRef] [PubMed]
- Vergani, L.; Vecchione, G.; Baldini, F.; Grasselli, E.; Voci, A.; Portincasa, P.; Ferrari, P.F.; Aliakbarian, B.; Casazza, A.A.; Perego, P. Polyphenolic extract attenuates fatty acid-induced steatosis and oxidative stress in hepatic and endothelial cells. Eur. J. Nutr. 2018, 57, 1793–1805. [Google Scholar] [CrossRef] [PubMed]
- Turan, G. Determination of the Seasonal Yields of Total Fucose and Fucoidan Yields in Brown Seaweeds (Order Fucales) Distributed along the Coast of Urla (izmir, Turkey). Aquac. Fish. 2017, 1, 1–4. [Google Scholar] [CrossRef]
- García-Ríos, V.; Ríos-Leal, E.; Robledo, D.; Freile-Pelegrin, Y. Polysaccharides composition from tropical brown seaweeds. Phycol. Res. 2012, 60, 305–315. [Google Scholar] [CrossRef]
- Costa, L.S.; Fidelis, G.P.; Telles, C.B.; Dantas-Santos, N.; Camara, R.B.; Cordeiro, S.L.; Costa, M.S.; Almeida-Lima, J.; Melo-Silveira, R.F.; Oliveira, R.M.; et al. Antioxidant and antiproliferative activities of heterofucans from the seaweed Sargassum filipendula. Mar. Drugs 2011, 9, 952–966. [Google Scholar] [CrossRef]
- Rioux, L.E.; Turgeon, S.L.; Beaulieu, M. Characterization of polysaccharides extracted from brown seaweeds. Carbohydr. Polym. 2007, 69, 530–537. [Google Scholar] [CrossRef]
- Zhao, Y.; Zheng, Y.; Wang, J.; Ma, S.; Yu, Y.; White, W.L.; Yang, S.; Yang, F.; Lu, J. Fucoidan Extracted from Undaria pinnatifida: Source for Nutraceuticals/Functional Foods. Mar. Drugs 2018, 16, 321. [Google Scholar] [CrossRef]
- Jiao, G.; Yu, G.; Wang, W.; Zhao, X.; Zhang, J.; Ewart, S.H. Properties of polysaccharides in several seaweeds from Atlantic Canada and their potential anti-influenza viral activities. J. Ocean Univ. China 2012, 11, 205–212. [Google Scholar] [CrossRef]
- Kochhar, S.P.; Rossell, J.B. Detection, Estimation and Evaluation of Antioxidants in Food Systems. In Food Antioxidants, 1st ed.; Hudson, B.J.F., Ed.; Springer: Dordrecht, The Netherlands, 1990; pp. 19–64. [Google Scholar] [CrossRef]
- Adams, L.A.; Anstee, Q.M.; Tilg, H.; Targher, G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 2017, 66, 1138–1153. [Google Scholar] [CrossRef]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef] [PubMed]
- Rashed, Z.E.; Grasselli, E.; Khalifeh, H.; Canesi, L.; Demori, I. Brown-Algae Polysaccharides as Active Constituents against Nonalcoholic Fatty Liver Disease. Planta Med. 2020. [Google Scholar] [CrossRef]
- Masarone, M.; Rosato, V.; Dallio, M.; Gravina, A.G.; Aglitti, A.; Loguercio, C.; Federico, A.; Persico, M. Role of Oxidative Stress in Pathophysiology of Nonalcoholic Fatty Liver Disease. Oxid. Med. Cell. Longev. 2018, 2018, 9547613. [Google Scholar] [CrossRef] [PubMed]
- Pettinelli, P.; Videla, L.A. Up-regulation of PPAR-gamma mRNA expression in the liver of obese patients: An additional reinforcing lipogenic mechanism to SREBP-1c induction. J. Clin. Endocrinol. Metab. 2011, 96, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Rahimian, R.; Masih-Khan, E.; Lo, M.; van Breemen, C.; McManus, B.M.; Dube, G.P. Hepatic over-expression of peroxisome proliferator activated receptor gamma2 in the ob/ob mouse model of non-insulin dependent diabetes mellitus. Mol. Cell. Biochem. 2001, 224, 29–37. [Google Scholar] [CrossRef]
- Bickel, P.E.; Tansey, J.T.; Welte, M.A. PAT proteins, an ancient family of lipid droplet proteins that regulate cellular lipid stores. Biochim. Biophys. Acta 2009, 1791, 419–440. [Google Scholar] [CrossRef]
- Connolly, M.K.; Bedrosian, A.S.; Malhotra, A.; Henning, J.R.; Ibrahim, J.; Vera, V.; Cieza-Rubio, N.E.; Hassan, B.U.; Pachter, H.L.; Cohen, S.; et al. In hepatic fibrosis, liver sinusoidal endothelial cells acquire enhanced immunogenicity. J. Immunol. 2010, 185, 2200–2208. [Google Scholar] [CrossRef]
- Grandl, G.; Wolfrum, C. Hemostasis, endothelial stress, inflammation, and the metabolic syndrome. Semin. Immunopathol. 2018, 40, 215–224. [Google Scholar] [CrossRef]
- Luiking, Y.C.; Engelen, M.P.; Deutz, N.E. Regulation of nitric oxide production in health and disease. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 97–104. [Google Scholar] [CrossRef]
- Ermakova, S.; Men’shova, R.; Vishchuk, O.; Kim, S.-M.; Um, B.-H.; Isakov, V.; Zvyagintseva, T. Water-soluble polysaccharides from the brown alga Eisenia bicyclis: Structural characteristics and antitumor activity. Algal Res. 2013, 2, 51–58. [Google Scholar] [CrossRef]
- Imbs, T.I.; Shevchenko, N.M.; Sukhoverkhov, S.V.; Semenova, T.L.; Skriptsova, A.V.; Zvyagintseva, T.N. Seasonal Variations of the Composition and Structural Characteristics of Polysaccharides from the Brown Algae Costaria costata. Chem. Nat. Compd. 2009, 45, 8. [Google Scholar] [CrossRef]
- Jackson, S.G.; McCandless, E.L. Simple, rapid, turbidometric determination of inorganic sulfate and/or protein. Anal. Biochem. 1978, 90, 802–808. [Google Scholar] [CrossRef]
- Miller, N.J.; Rice-Evans, C.A. Factors influencing the antioxidant activity determined by the ABTS.+ radical cation assay. Free Radic. Res. 1997, 26, 195–199. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Lupidi, G.; Nabissi, M.; Petrelli, R.; Ngahang Kamte, S.L.; Cappellacci, L.; Fiorini, D.; Sut, S.; Dall’Acqua, S.; et al. The crop-residue of fiber hemp cv. Futura 75: From a waste product to a source of botanical insecticides. Environ. Sci. Pollut. Res. Int. 2018, 25, 10515–10525. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Halliwell, B.; Whiteman, M. Measuring reactive species and oxidative damage in vivo and in cell culture: How should you do it and what do the results mean? Br. J. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef]
- Lambruschini, C.; Demori, I.; El Rashed, Z.; Rovegno, L.; Canessa, E.; Cortese, K.; Grasselli, E.; Moni, L. Synthesis, Photoisomerization, Antioxidant Activity, and Lipid-Lowering Effect of Ferulic Acid and Feruloyl Amides. Molecules 2020, 26, 89. [Google Scholar] [CrossRef]
- Grasselli, E.; Voci, A.; Canesi, L.; Salis, A.; Damonte, G.; Compalati, A.D.; Goglia, F.; Gallo, G.; Vergani, L. 3,5-diiodo-L-thyronine modifies the lipid droplet composition in a model of hepatosteatosis. Cell. Physiol. Biochem. 2014, 33, 344–356. [Google Scholar] [CrossRef] [PubMed]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Grasselli, E.; Voci, A.; Demori, I.; Vecchione, G.; Compalati, A.D.; Gallo, G.; Goglia, F.; De Matteis, R.; Silvestri, E.; Vergani, L. Triglyceride Mobilization from Lipid Droplets Sustains the Anti-Steatotic Action of Iodothyronines in Cultured Rat Hepatocytes. Front. Physiol. 2015, 6, 418. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]

| Sulfate | Fucose | Glucose | Galactose | Mannose | Proteins |
|---|---|---|---|---|---|
| 1.8 ± 1.52 | 31.5 ± 6.1 | 29.3 ± 6.32 | 2.6 ± 5.82 | 1.2 ± 4.95 | 0.46 ± 7.6 |
| FUFe (µg/mL) | RSA% |
|---|---|
| 50 | 18.1 ± 4.2 |
| 75 | 26.6 ± 9.2 |
| 100 | 36.4 ± 9.1 |
| 200 | 60.5 ± 11.2 |
| 300 | 72.8 ± 5.1 |
| 400 | 75.7 ± 4.9 |
| 500 | 75.7 ± 4.2 |
| IC50 = 157.6 ± 3.3 µg/mL | |
| ABTS | FRAP | ||
|---|---|---|---|
| TEAC (μmol TE/g) | IC50 (μg/mL) | TEAC (μmol TE/g) | |
| Fucoidan from F. hermonis (FUFe) | 410.0 ± 14.0 | 44.26 ± 2.35 | 81.30 ± 7.20 |
| Trolox | 4.53 ± 1.53 | ||
| FU Source | Species | RSA at 500 µg/mL | Reference |
|---|---|---|---|
| Terrestrial plant | Ferula hermonis | 75.7% | - |
| Eucalyptus globulus | 95.6% | [6] | |
| Brown algae | Stypopodium schimperi | 44.13% | [14] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Rashed, Z.; Lupidi, G.; Kanaan, H.; Grasselli, E.; Canesi, L.; Khalifeh, H.; Demori, I. Antioxidant and Antisteatotic Activities of a New Fucoidan Extracted from Ferula hermonis Roots Harvested on Lebanese Mountains. Molecules 2021, 26, 1161. https://doi.org/10.3390/molecules26041161
El Rashed Z, Lupidi G, Kanaan H, Grasselli E, Canesi L, Khalifeh H, Demori I. Antioxidant and Antisteatotic Activities of a New Fucoidan Extracted from Ferula hermonis Roots Harvested on Lebanese Mountains. Molecules. 2021; 26(4):1161. https://doi.org/10.3390/molecules26041161
Chicago/Turabian StyleEl Rashed, Zeinab, Giulio Lupidi, Hussein Kanaan, Elena Grasselli, Laura Canesi, Hala Khalifeh, and Ilaria Demori. 2021. "Antioxidant and Antisteatotic Activities of a New Fucoidan Extracted from Ferula hermonis Roots Harvested on Lebanese Mountains" Molecules 26, no. 4: 1161. https://doi.org/10.3390/molecules26041161
APA StyleEl Rashed, Z., Lupidi, G., Kanaan, H., Grasselli, E., Canesi, L., Khalifeh, H., & Demori, I. (2021). Antioxidant and Antisteatotic Activities of a New Fucoidan Extracted from Ferula hermonis Roots Harvested on Lebanese Mountains. Molecules, 26(4), 1161. https://doi.org/10.3390/molecules26041161








