Abstract
Angucyclinones are aromatic polyketides that possess impressive structural diversity and significant biological activities. The structural diversity of these natural products is attributed to various enzymatic or nonenzymatic modifications on their tetracyclic benz(a)anthracene skeleton. Previously, we discovered an unusual phenylamine-incorporated angucyclinone (1) from a marine Streptomyces sp. PKU-MA00218, and identified that it was produced from the nonenzymatic conversion of a C-ring-cleaved angucyclinone (2) with phenylamine. In this study, we tested the nonenzymatic conversion of 2 with more phenylamine analogues, to expand the utility of this feasible conversion in unusual angucyclinones generation. The (3-ethynyl)phenylamine and disubstituted analogues including (3,4-dimethyl)phenylamine, (3,4-methylenedioxy)phenylamine, and (4-bromo-3-methyl)phenylamine were used in the conversion of 2, which was isolated from the fermentation of Streptomyces sp. PKU-MA00218. All four phenylamine analogues were incorporated into 2 efficiently under mild conditions, generating new compounds 3–6. The activation of 3–6 on nuclear factor erythroid 2-related factor 2 (Nrf2) transcription were tested, which showed that 4 possessing a dimethyl-substitution gave most potent activity. These results evidenced that disubstitutions on phenylamine can be roughly tolerated in the nonenzymatic reactions with 2, suggesting extended applications of more disubstituted phenylamines incorporation to generate new bioactive angucyclinones in the future.
1. Introduction
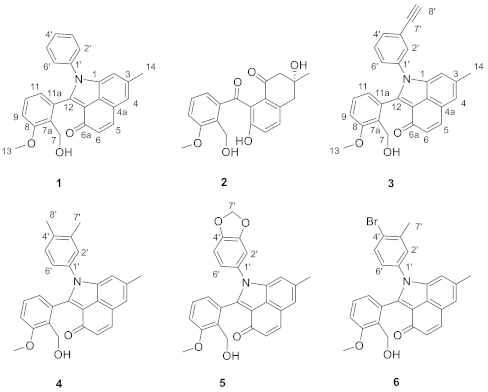
Angucyclinones are aromatic polyketides produced by type II polyketide synthases (PKSs) from actinomycetes exclusively [1]. The structures of these natural products feature a tetracyclic benz(a)anthracene skeleton, which can be modified by enzymatic or nonenzymatic reactions to achieve impressive structural diversity. As a part of our ongoing project for natural product discovery and biosynthesis from marine bacteria [2,3,4,5,6], we previously discovered an unusual phenylamine-incorporated angucyclinone (1) featuring 1-phenylbenzo(cd)indol-3(1H)-one moiety from a marine Streptomyces sp. PKU-MA00218 [2]. Based on genome sequencing, bioinformatics analysis, heterologous expression, gene deletion, and nonenzymatic conversion in vitro, we identified that 1 was produced from the nonenzymatic conversion of a C-ring-cleaved angucyclinone (2) with phenylamine, and generated a series of derivatives by the incorporation of monosubstituted phenylamine analogues into 2. These derivatives represented a new group of angucyclinones and showed different degrees of activation activities on nuclear factor erythroid 2-related factor 2 (Nrf2) transcriptions in HepG2 cells, highlighting the powerful combination of biosynthesis and nonenzymatic reactions in the generation of structurally unusual and bioactive molecules. To further expand the structural diversity of this group of phenylamine analogues-incorporated angucyclinones, we report herein the generation of derivative 3 by the incorporation of (3-ethynyl)phenylamine, and 4–6 by the incorporation of disubstituted phenylamines into 2 (Figure 1). The generation of 3–6 can be efficiently achieved under mild conditions, and the activation of 3–6 on Nrf2 transcriptions were subsequently tested.
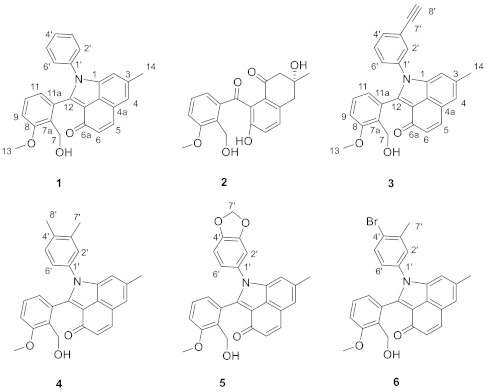
Figure 1.
The structure of compounds 1–6.
2. Results and Discussion
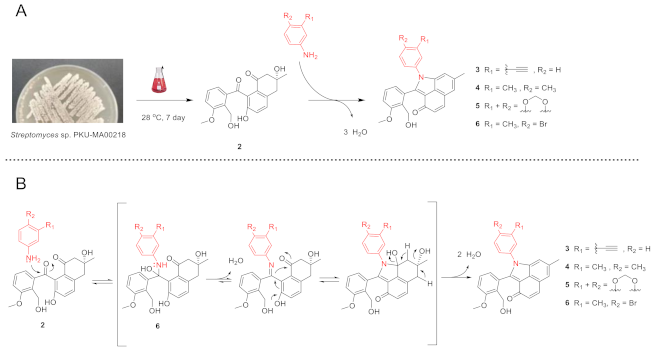
2.1. Generation of 3–6 by Nonenzymatic Incorporation of Phenylamine Analogues into C-Ring-Cleaved Angucyclinone 2
Monosubstituted phenylamine analogues have been efficiently incorporated into 2 under mild conditions, previously [2]. To test the efficiency of other phenylamine analogues in the incorporation, we selected four analogues with distinct substitutions on the phenylamine. The (3-ethynyl)phenylamine contains an ethynyl moiety that is never used previously and may facilitate the biological targets identification through a click reaction with chemical probes; the (3,4-dimethyl)phenylamine, (3,4-methylenedioxy)phenylamine and (4-bromo-3-methyl)phenylamine contain groups that affect the electron density of phenyl moiety in different ways. The four phenylamine analogues were individually incubated with 2, which was isolated with a combination of chromatographic methods from a large-scale fermentation of Streptomyces sp. PKU-MA00218 (Materials and Methods). Four new angucyclinone products 3–6 were efficiently produced under the similar mild conditions used previously (Scheme 1A and Supplementary Materials). The conversion mechanisms of 2 to 3–6 were similarly proposed as that of 2 to 1 (Scheme 1B) [2]. Compounds 3–6 were purified by semipreparative HPLC, and their structures were elucidated with UV, IR, NMR, and high resolution electrospray ionization mass spectroscopy (HR-ESI-MS) analyses (Supplementary Materials).
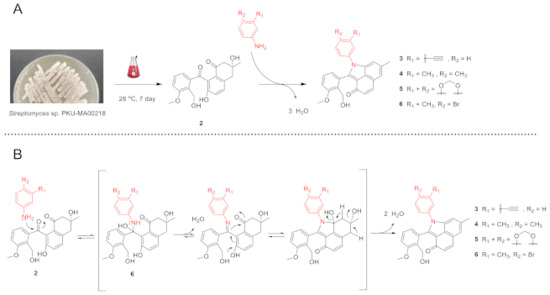
Scheme 1.
The conversion of 2 to 3–6 and proposed conversion mechanism. (A) The conversion of 2 to 3–6. (B) Proposed conversion mechanism of 2 to 3–6.
2.2. Structural Elucidation of 3–6
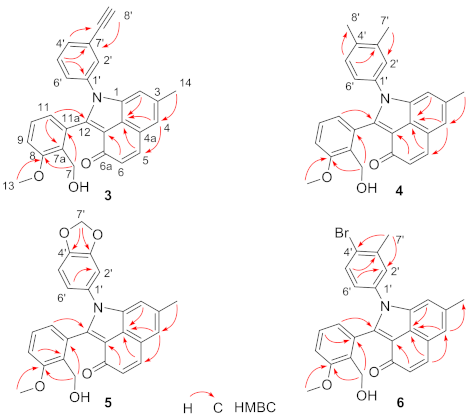
Compound 3 was obtained as green-yellow solid, UV (MeOH) λmax (log ε): 272 (5.17), 358 (5.13), 415 (5.06) nm. IR (KBr) νmax: 3287, 2919, 1636, 1594, 1582, 1460, 1296, 1263, 1028 cm−1. HR-ESI-MS analysis afforded an [M + H]+ ion at m/z 420.1600 (calcd for C28H22NO3, 420.1594) giving the molecular formula of 3 as C28H21NO3. 1H-NMR (600 MHz, DMSO-d6) and 13C-NMR (150 MHz, DMSO-d6) data, Table 1. The 1H-NMR and 13C-NMR spectra of 3 resembled that of 1, except that the resonances at δH 7.54 (H-2′/H-6′), 7.45 (H-3′/H-5′), 7.41 (H-4′) and δC 136.0 (C-1′), 127.6 (C-2′/C-6′), 129.3 (C-3′/C-5′), 128.3 (C-4′) in 1 were replaced by δH 7.70 (H-2′), 7.50 (H-4′), 7.45 (H-5′), 7.29 (H-6′) and δC 136.4 (C-1′), 130.7 (C-2′), 122.7 (C-3′), 131.8 (C-4′), 129.7 (C-5′), 128.6 (C-6′) in 3. Besides, compound 3 contains additional signals attributed to an ethynyl moiety at δH 4.31 (s, H-8′) and δC 82.4 (C-7′), 82.2 (C-8′). These results suggested the difference between 3 and 1 was that 3 contained a (3-ethynyl)phenylamino moiety instead of a phenylamino moiety. The (3-ethynyl)phenylamino moiety in 3 was further confirmed by HMBC experiment (Figure 2 and Supplementary Materials) and comparison of 1H-NMR and 13C-NMR data with published data of (3-ethynyl)phenylamino moiety in literatures [7,8]. Thus, compound 3 was identified as a new (3-ethynyl)phenylamine-incorporated angucyclinone analogue.

Table 1.
The 1H-NMR (600 MHz) and 13C-NMR (150 MHz) data of compounds 1 and 3–6 in DMSO-d6.
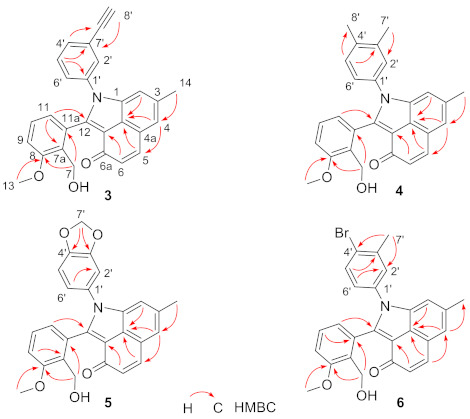
Figure 2.
Key HMBC correlations of compounds 3–6.
Compound 4 was obtained as green–yellow solid, UV (MeOH) λmax (log ε): 271 (5.99), 359 (6.02), 415 (5.99) nm. IR (KBr) νmax: 3375, 2919, 1635, 1596, 1504, 1458, 1055, 1028, 1007, 821, 759 cm−1. HR-ESI-MS analysis afforded an [M + H]+ ion at m/z 424.1907 (calcd for C28H26NO3, 424.1907) giving the molecular formula of 4 as C28H25NO3. 1H-NMR (600 MHz, DMSO-d6) data, Table 1; 13C-NMR (150 MHz, DMSO-d6) data, Table 1. The 1H-NMR and 13C-NMR spectra of 4 resembled that of 1, except that the resonances at δH 7.54 (H-2′/H-6′), 7.45 (H-3′/H-5′), 7.41 (H-4′) and δC 136.0 (C-1′), 127.6 (C-2′/C-6′), 129.3 (C-3′/C-5′), 128.3 (C-4′) in 1 were replaced by δH 7.36 (H-2′), 7.16 (H-5′), 7.19 (H-6′) and δC 137.4 (C-1′), 128.3 (C-2′), 136.7 (C-3′), 133.6 (C-4′), 130.1 (C-5′), 124.9 (C-6′) as well as two additional methyl groups signals at δH 2.28 (s, 3H, H-7′), 2.21 (s, 3H, H-8′) and δC 19.4 (C-7′), 19.0 (C-8′) in 4. These results suggested the only difference between 4 and 1 was that 4 contained a (3,4-dimethyl)phenylamino moiety instead of a phenylamino moiety. The (3,4-dimethyl)phenylamino moiety in 4 was further confirmed by HMBC experiment (Figure 2 and Supplementary Materials) and comparison of 1H-NMR and 13C-NMR signals with published data of (3,4-dimethyl)phenylamino moiety in literature [9]. Thus, compound 4 was identified as a new (3,4-dimethyl)phenylamine-incorporated angucyclinone analogue.
Compound 5 was obtained as green-yellow solid, UV (MeOH) λmax (log ε): 273 (5.99), 355 (5.98), 415 (5.95) nm. IR (KBr) νmax: 3346, 2917, 1635, 1596, 1502, 1489, 1460, 1263, 1239, 1029 cm−1. HR-ESI-MS analysis afforded an [M + H]+ ion at m/z 440.1490 (calcd for C27H22NO5, 440.1492) giving the molecular formula of 5 as C27H21NO5. 1H-NMR (600 MHz, DMSO-d6) and 13C-NMR (150 MHz, DMSO-d6) data, Table 1. The 1H-NMR and 13C-NMR spectra of 5 resembled that of 1, except that the resonances at δH 7.54 (H-2′/H-6′), 7.45 (H-3′/H-5′), 7.41 (H-4′) and δC 136.0 (C-1′), 127.6 (C-2′/C-6′), 129.3 (C-3′/C-5′), 128.3 (C-4′) in 1 were replaced by δH 7.18 (H-2′), 7.04 (H-5′), 6.98 (H-6′) and δC 129.7 (C-1′), 108.2 (C-2′), 147.1 (C-3′), 147.5 (C-4′), 108.7 (C-5′), 121.6 (C-6′) as well as additional methylenedioxy signals at δH 6.10 (br s, 1H, H-7′a), 6.08 (br s, 1H, H-7′b), and δC 101.9 (C-7′) in 5. These results suggested the only difference between 5 and 1 was that 5 contained a (3,4-methylenedioxy)phenylamino moiety instead of a phenylamino moiety. The (3,4-methylenedioxy)phenylamino moiety in 5 was further confirmed by HMBC experiment (Figure 2 and Supplementary Materials) and comparison of 1H-NMR and 13C-NMR signals with published data of (3,4-methylenedioxy)phenylamino moiety in literatures [10,11]. Thus, compound 5 was identified as a new (3,4-methylenedioxy)phenylamine-incorporated angucyclinone analogue.
Compound 6 was obtained as green-yellow solid, UV (MeOH) λmax (log ε): 271 (6.27), 356 (6.26), 415 (6.20) nm. IR (KBr) νmax: 3343, 2917, 2850, 1635, 1584, 1477, 1463, 1296, 1263, 1152, 1053, 1029 cm−1. HR-ESI-MS analysis afforded an [M + H]+ ion at m/z 488.0862 (calcd for C27H23BrNO3, 488.0856) giving the molecular formula of 6 as C27H22BrNO3. The 1H NMR (600 MHz, DMSO-d6) and 13C NMR (150 MHz, DMSO-d6) data, Table 1. The 1H-NMR and 13C-NMR spectra of 6 resembled that of 1, except that the resonances at δH 7.54 (H-2′/H-6′), 7.45 (H-3′/H-5′), 7.41 (H-4′) and δC 136.0 (C-1′), 127.6 (C-2′/C-6′), 129.3 (C-3′/C-5′), 128.3 (C-4′) in 1 were replaced by δH 7.61 (H-2′), 7.62 (H-5′), 7.28 (H-6′) and δC 135.5 (C-1′), 130.1 (C-2′), 138.6 (C-3′), 124.0 (C-4′), 132.8 (C-5′), 127.0 (C-6′) as well as an additional methyl group signals at δH 2.32 (s, 3H, H-7′), and δC 22.5 (C-7′) in 6. These results suggested the difference between 6 and 1 was that 6 contained a (4-bromo-3-methyl)phenylamino moiety instead of a phenylamino moiety. The (4-bromo-3-methyl)phenylamino moiety in 6 was further confirmed by HMBC experiment (Figure 2 and Supplementary Materials) and comparison of 1H-NMR and 13C-NMR signals with published data of (4-bromo-3-methyl)phenylamino moiety in literatures [12,13]. Thus, compound 6 was identified as a new (4-bromo-3-methyl)phenylamine-incorporated angucyclinone analogue.
2.3. Biological Activity Assays
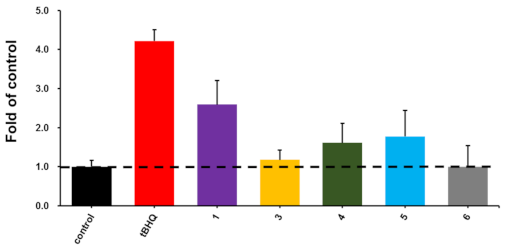
According to previous procedures, the activation of 3–6 on nuclear factor erythroid 2-related factor 2 (Nrf2) transcription in HepG2 cells was evaluated by using a luciferase reporter assay with tertiary butylhydroquinone (tBHQ) as the positive control [14]. Compound 3–5 showed Nrf2 transcription activation levels 1.18, 1.61, and 1.78 folds to that of the DMSO-treated negative control (Figure 3). Compound 1 showed better activity than 3–5, suggesting the substitutions on the phenylamine moieties in 3–5 impair the Nrf2 transcription activation. More assays need to be carried out to assess the potential biological activities of these unusual angucyclinones.
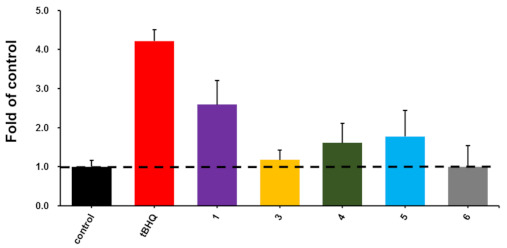
Figure 3.
The activation activities on Nrf2 transcription of compounds 1 and 3–6 at 10 μM, with tertiary butylhydroquinone (tBHQ) as the positive control. The activation potency of compounds were shown by the folds of the transcription activation level when cells were treated with DMSO.
3. Materials and Methods
3.1. General Experimental Procedures
Optical rotations were measured on an Autopol III automatic polarimeter (Rudolph Research Analytical, Hackettstown, NJ, USA). UV spectra were collected on a NanoDrop 2000C spectrophotometer (Thermo Scientific, Waltham, MA, USA). IR spectra were recorded with a NICOLET iS50 FT-IR (Thermo Scientific). 1H and 13C-NMR spectra were collected on a Bruker Avance-600 NMR spectrometer (Bruker Corporation, Billerica, MA, USA). HR-ESI-MS spectra were collected on a Waters Xevo G2 Q-TOF spectrometer equipped with an ESI ion source (Waters, Milford, MA, USA). The mass instrument was operated in positive ion mode with a capillary potential at 3000 V and sampling cone potential at 20 V. The m/z range was recorded from 105 to 1000 Da, sample concentration was between 0.005 and 0.05 mg/mL, and all mass spectra were externally calibrated using a sodium formate solution. HPLC analysis was performed on an Agilent 1260 series (Agilent Technologies, Santa Clara, CA, USA) with a C18 RP-column (Eclipse XDB C18, 250 × 4.6 mm, 5 μm, Agilent Technologies, Santa Clara, CA, USA). Semi-preparative HPLC was performed on a SSI 23201 system (Scientific Systems Inc., State College, PA, USA) with a YMC Pack ODS-A column (250 × 10 mm, 5 μm, YMC Co., LTD. Shimogyo-ku, Kyoto, Japan). Medium performance liquid chromatography (MPLC) was performed on a LC 3000 series (Beijing Tong Heng Innovation Technology, Beijing, China) with a ClaricepTM Flash i-series C18 cartridge (20–35 μm, 40 g, Bonna-Agela, Wilmington, DE, USA). Size exclusion chromatography was carried out using a Sephadex LH-20 (GE Healthcare, Chicago, IL, USA) column. All fermentations were carried out in MQD-B1R shakers (Minquan Instrument Co., Ltd., Shanghai, China).
3.2. The Fermentation and Isolation of 2
The spores of Streptomyces sp. PKU-MA00218 was inoculated in 250 mL Erlenmeyer flasks containing 50 mL seed medium (yeast extract 1 g, peptone 5 g, beef extract 1 g, FePO4 0.01 g, agar 18 g, and sea salts 33 g in 1.0 L distilled H2O, pH 7.4) at 28 °C, 220 rpm in a rotary shaker for three days. Then each of the seed cultures (16 mL) was inoculated into autoclaved 2 L Erlenmeyer flasks containing 400 mL of production medium (soluble starch 10 g, casein 0.3 g, KNO3 2 g, K2HPO4∙3H2O 2 g, MgSO4∙7H2O 0.05 g, CaCO3 0.02 g, FeSO4∙7H2O 0.01 g, and sea salts 33 g in 1.0 L distilled H2O, pH 7.0). The fermentations (total 24 L) were continued at 28 °C, 220 rpm for seven days. The D101 resins (5 g/100 mL) were added 12 h before the fermentation finished.
The resins and cell mass were harvested by centrifugation and extracted with MeOH. The MeOH extract was concentrated, resuspended in H2O, and extracted with EtOAc for three times. The EtOAc extract was fractionated by MPLC, using a gradient elution program from 5% MeOH in H2O to 100% MeOH over 30 min, with the flowrate of 15 mL/min under the UV detection at 254 nm, to give 15 fractions (a1–a15). Fraction a10–a15 were combined and subjected to Sephadex LH-20 chromatography eluting with MeOH to give 5 fractions (b1–b5). Fraction b4 was further purified by semi-preparative HPLC eluting with CH3CN/H2O (45/55, v/v) with the flowrate of 2 mL/min under the UV detection at 254 nm, to afford compound 2 (16 mg).
3.3. The Conversion of 2 to 3–6
Each of the four phenylamine analogues was incubated with 2 (~3 mg) (molar ratio of 1 to phenylamine analogue is 1 to 10) in 500 μL CH3CN/HAc (4/1, v/v) at 50 °C for 3 h, respectively. The reaction products were purified by semi-preparative HPLC using CH3CN/H2O (60/40, v/v) as the mobile phase, with a flow rate of 2 mL/min with UV detection at 320 nm, to afford compound 3 (1.4 mg, yield: 59%), 4 (1.7 mg, yield: 71%), 5 (1.8 mg, yield: 73%), 6 (1.3 mg, yield: 47%).
3.4. Biological Activity Assays
The assay of Nrf2 transcription activation of 3–6 were carried out following the reported protocols [15]. Human hepatocellular carcinoma (HepG2) cells were stably transfected with Nrf2/ARE luciferase reporter to generate the HepG2-ARE-C8 cells. The HepG2-ARE-C8 cells were plated in 96-well plates and exposed with tested compounds at 10 μM, with tertiary butylhydroquinone (tBHQ) as the positive control and DMSO as the negative control. After 6 h treatment, the cells were harvested in the luciferase cell culture lysis reagent and used for luciferase activity determination. The luciferase activities were measured using the Luciferase Assay System (Promega, WI, USA) on a Centro LB 960 microplate luminometer (Berthold, Germany). The transcription activities of tested compounds were obtained in triplicates and showed as the folds of the transcription level when cells were treated with DMSO [16].
4. Conclusions
In this study, four unusual phenylamine analogues-incorporated angucyclinones 3–6 containing benzo(cd)indol-3(1H)-one moieties were efficiently generated, expanding the structural diversity of angucyclinones. The generation of 3, which contains an ethynyl moiety, may facilitate the biological targets (not limited to Nrf2 transcription activation) identification through a click reaction with chemical probes in the future. The high efficient incorporation of the disubstituted phenylamine analogues in the generation of 4–6 further evidenced the high reactivity of 2 when reacting with phenylamine analogues, which may not be limited by substitution groups on the phenylamine. These results set the stage for the incorporation of more diverse phenylamine analogues, such as dihalogen-substituted phenylamines, to further expand the structural diversity of this unusual group of angucyclinones. The new introduced chemical groups can imbed new biological activities into the angucyclinone analogues, and more biological activities need to be assayed in our next steps.
Supplementary Materials
The following are available online. Figure S1: HPLC analysis of reactions of the phenylamine analogues with 2. Figures S2–S6: NMR, HR-ESI-MS, and IR spectra of compound 3, Figures S7–S11: NMR, HRESIMS, and IR spectra of compound 4, Figures S12–S16: NMR, HRESIMS, and IR spectra of compound 5, Figures S17–S21: NMR, HRESIMS, and IR spectra of compound 6.
Author Contributions
Conceptualization, M.M.; methodology, H.X., G.W., Z.W., Y.K.; formal analysis, H.X., G.W., Y.K., M.Y., D.Y., M.M.; investigation, H.X., G.W., Z.W., Y.K., J.S., J.J.; writing—original draft preparation, G.W., H.X.; writing—review and editing, M.M.; project administration, D.Y., M.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded in part by the National Key Research and Development Program of China (2019YFC0312502), the National Natural Science Foundation of China (grant numbers 21877002, 81991525, 22077007, 81741148, 81673332), the key project at central government level: the ability establishment of sustainable use for valuable Chinese medicine resources (2060302-1903-03), and China Postdoctoral Science Foundation (grant number 2018M641123).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We thank Qin Li and Fen Liu of the State Key Laboratory of Natural and Biomimetic Drugs, School of Pharmaceutical Sciences, Peking University, for NMR data collection.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kharel, M.K.; Pahari, P.; Shepherd, M.D.; Tibrewal, N.; Nybo, S.E.; Shaaban, K.A.; Rohr, J. Angucyclines: Biosynthesis, mode-of-action, new natural products, and synthesis. Nat. Prod. Rep. 2012, 29, 264–325. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Jin, J.; Yang, X.Y.; Song, J.; Yu, J.H.; Geng, T.T.; Zhang, Z.Y.; Ma, X.Y.; Wang, G.Y.; Xiao, H.; et al. Discovery of a phenylamine-incorporated Angucyclinone from marine Streptomyces sp. PKU-MA00218 and Generation of derivatives with phenylamine analogues. Org. Lett. 2019, 21, 2813–2817. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.J.; Wang, G.Y.; Jin, J.; Liu, T.; Ma, X.Y.; Zhang, Z.Y.; Geng, T.T.; Song, J.; Ma, X.J.; Zhang, Y.T.; et al. Discovery and biosynthesis of pepticinnamins G-M featuring three enzymes-catalyzed nonproteinogenic amino acid formation. J. Org. Chem. 2020, 85, 8673–8682. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.C.; Liu, D.; Guan, X.; Yan, Y.C.; Zhang, J.P.; Zhang, Y.P.; Yang, D.H.; Ma, M.; Lin, W.H. Piperazine ring formation by a single-module NRPS and cleavage by an α-KG-dependent nonheme iron dioxygenase in brasiliamide biosynthesis. Appl. Microbiol. Biotechnol. 2020, 104, 6149–6159. [Google Scholar] [CrossRef]
- Jin, J.; Yang, X.Y.; Liu, T.; Xiao, H.; Wang, G.Y.; Zhou, M.J.; Liu, F.W.; Zhang, Y.T.; Liu, D.; Chen, M.H.; et al. Fluostatins M-Q featuring a 6-5-6-6 ring skeleton and high oxidized A-rings from marine Streptomyces sp. PKU-MA00045. Mar. Drugs 2018, 16, 87. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.J.; Liu, F.W.; Yang, X.Y.; Jin, J.; Dong, X.; Zeng, K.W.; Liu, D.; Zhang, Y.T.; Ma, M.; Yang, D.H. Bacillibactin and bacillomycin analogues with cytotoxicities against human cancer cell lines from marine Bacillus sp. PKU-MA00093 and PKU-MA00092. Mar. Drugs 2018, 16, 22. [Google Scholar] [CrossRef]
- Brusilowskij, B.; Schalley, C.A. Multidentate pyridyl-based ligands in the coordination-driven self-assembly of palladium metallo-macrocycles. Eur. J. Org. Chem. 2011, 2011, 469–477. [Google Scholar] [CrossRef]
- Lai, B.B.; Huang, Z.P.; Jia, Z.F.; Bai, R.X.; Gu, Y.L. Silica supported metal acetylacetonate catalysts with a robust and flexible linker constructed by using butoxy 3,4 dihydropyrans as dual anchoring reagents and ligand donors. Catal. Sci. Technol. 2016, 6, 1810–1820. [Google Scholar] [CrossRef]
- Mukhopadhyay, C.; Tapaswi, P.K. Highly efficient and simple catalytic system for the N-Arylation of some hindered Aza-heterocycles in water. Synth. Commun. 2012, 42, 2217–2228. [Google Scholar] [CrossRef]
- Reem, A.K.A.; Marwa, A.M.S.; Samir, Y.A. Synthesis and anticancer activity of bis-benzo[d][1,3]dioxol-5-yl thiourea derivatives with molecular docking study. Bioorg. Chem. 2019, 90, 103088–103096. [Google Scholar]
- Beresneviiute, K.; Beresnevicius, Z.; Mikulskiene, G.; Kihlberg, J.; Broddefalk, J. 13C NMR study of dihydropyrimidinedione and dihydropyrimidine-2-thione derivatives. Magn. Reson. Chem. 1997, 35, 553–555. [Google Scholar] [CrossRef]
- Liu, Y.X.; Yan, Y.G.; Xue, D.; Wang, Z.F.; Xiao, J.L.; Wang, C. Highly efficient binuclear copper-catalyzed oxidation of N,N-dimethylanilines with O2. ChemCatChem 2020, 12, 2221–2225. [Google Scholar] [CrossRef]
- Hambleton, P.T.; Hedgecock, C.J.R.; Kay, D.P.; Kuo, E.A.; Tully, W.R. Preparation of 2-Cyano-2-(cyclopropylcarbonyl)acetanilides and Analogs as Anti-Inflammatories. Patent PCT EP484223 A2 19920506, 6 May 1992. [Google Scholar]
- Lee, J.M.; Johnson, J.A. An important role of Nrf2-ARE pathway in the cellular defense mechanism. J. Biochem. Mol. Biol. 2004, 37, 139–143. [Google Scholar] [CrossRef]
- Xi, M.Y.; Jia, J.M.; Sun, H.P.; Sun, Z.Y.; Jiang, J.W.; Wang, Y.J.; Zhang, M.Y.; Zhu, J.F.; Xu, L.L.; Jiang, Z.Y.; et al. 3-aroylmethylene-2,3,6,7-tetrahydro-1H-pyrazino[2,1-a]isoquinolin-4(11bH)-ones as potent Nrf2/ARE inducers in human cancer cells and AOM-DSS treated mice. J. Med. Chem. 2013, 56, 7925–7938. [Google Scholar] [CrossRef]
- Ji, S.; Li, R.; Wang, Q.; Miao, W.J.; Li, Z.W.; Si, L.L.; Qiao, X.; Yu, S.W.; Zhou, D.M.; Ye, M. Anti-H1N1 virus, cytotoxic and Nrf2 activation activities of chemical constituents from Scutellaria baicalensis. J. Ethnopharmacol. 2015, 176, 475–484. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).