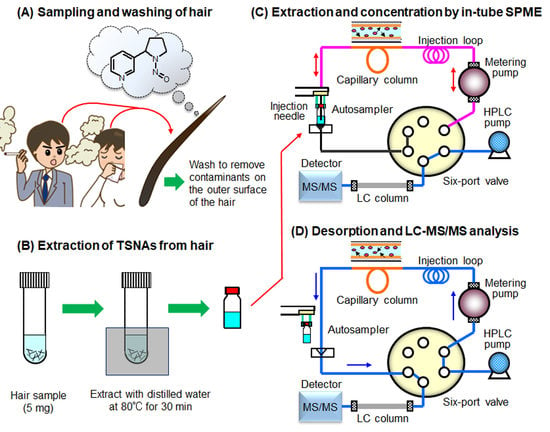
Online In-Tube Solid-Phase Microextraction Coupled to Liquid Chromatography–Tandem Mass Spectrometry for the Determination of Tobacco-Specific Nitrosamines in Hair Samples
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of In-Tube Solid-Phase Microextraction and Desorption of TSNAs
2.2. LC–MS/MS Analysis of TSNAs and Their Stable Isotope-Labeled Compounds
2.3. Analytical Method Validation
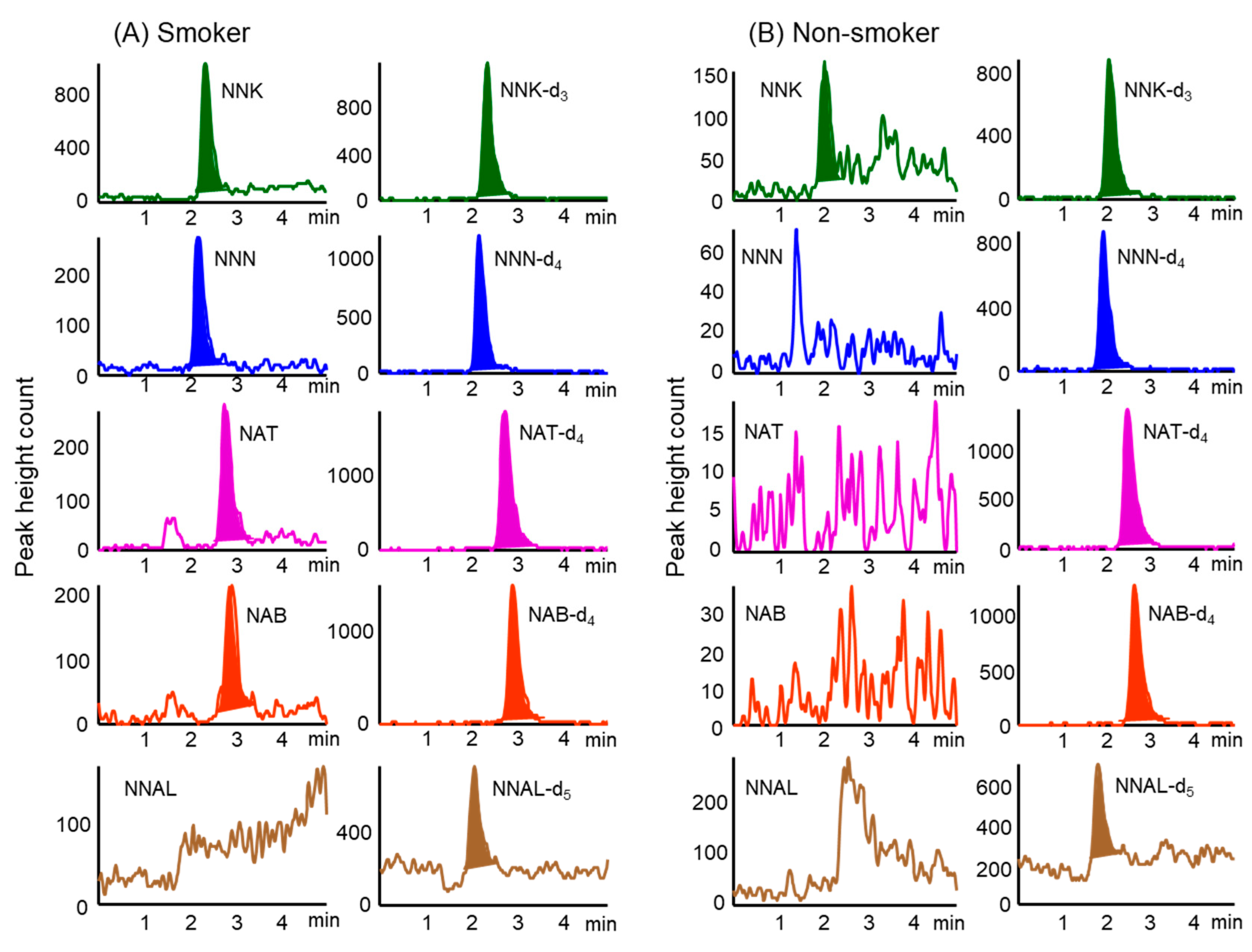
2.4. Application to the Analysis of Hair Samples
3. Materials and Methods
3.1. Reagents and Standard Solutions
3.2. Preparation of Hair Samples
3.3. LC–MS/MS Analysis
3.4. In-Tube SPME
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Hu, Q.; Hou, H. (Eds.) Tobacco Smoke Exposure Biomarkers; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2015. [Google Scholar]
- Mattes, W.; Yang, X.; Orr, M.S.; Richter, P.; Mendrick, D.L. Biomarkers of tobacco smoke exposure. Adv. Clin. Chem. 2014, 67, 1–45. [Google Scholar] [PubMed]
- World Health Organization [WHO]. Tobacco Fact Sheet. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco (accessed on 1 March 2021).
- Omare, M.O.; Kibet, J.K.; Cherutoi, J.K.; Kengara, F.O. A review of tobacco abuse and its epidemiological consequences. J. Public Health 2021. [Google Scholar] [CrossRef] [PubMed]
- Hori, M.; Tanaka, H.; Wakai, K.; Sasazuki, S.; Katanoda, K. Secondhand smoke exposure and risk of lung cancer in Japan: A systematic review and meta-analysis of epidemiologic studies. Jpn. J. Clin. Oncol. 2016, 46, 942–951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avila-Tang, E.; Al-Delaimy, W.K.; Ashley, D.L.; Benowitz, N.; Bernert, J.T.; Kim, S.; Samet, J.M.; Hecht, S.S. Assessing secondhand smoke using biological markers. Tob. Control. 2013, 22, 164–171. [Google Scholar] [CrossRef]
- Torres, S.; Merino, C.; Paton, B.; Correig, X.; Ramírez, N. Biomarkers of exposure to secondhand and thirdhand tobacco smoke: Recent advances and future perspectives. Int. J. Environ. Res. Public Health. 2018, 15, 2693. [Google Scholar] [CrossRef] [Green Version]
- Hecht, S.S.; Stepanov, I.; Carmella, S.G. Exposure and metabolic activation biomarkers of carcinogenic tobacco-specific nitrosamines. Acc. Chem. Res. 2016, 49, 106–114. [Google Scholar] [CrossRef] [Green Version]
- Yuan, J.-M.; Butler, L.M.; Stepanov, I.; Hecht, S.S. Urinary tobacco smoke–constituent biomarkers for assessing risk of lung cancer. Cancer Res. 2014, 74, 401–411. [Google Scholar] [CrossRef] [Green Version]
- Konstantinou, E.; Fotopoulou, F.; Drosos, A.; Dimakopoulou, N.; Zagoriti, Z.; Niarchos, A.; Makrynioti, D.; Kouretas, D.; Farsalinos, K.; Lagoumintzis, G.; et al. Tobacco-specific nitrosamines: A literature review. Food Chem. Toxicol. 2018, 118, 198–203. [Google Scholar] [CrossRef]
- Veena, S.; Rashmi, S. A review on mechanism of nitrosamine formation, metabolism and toxicity in vivo. Int. J. Toxicol. Pharmacol. Res. 2014, 6, 86–96. [Google Scholar]
- Gupta, A.K.; Tulsyan, S.; Bharadwaj, M.; Mehrotra, R. Grass roots approach to control levels of carcinogenic nitrosamines, NNN and NNK in smokeless tobacco products. Food Chem. Toxicol. 2019, 124, 359–366. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer [IARC]. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2012; Volume 100E, Available online: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono100E.pdf (accessed on 1 March 2021).
- Kintz, P.; Salomone, A.; Vincenti, M. (Eds.) Hair Analysis in Clinical and Forensic Toxicology; Academic Press: London, UK, 2015. [Google Scholar]
- Boumba, V.A.; Ziavrou, K.S.; Vougiouklakis, T. Hair as a biological indicator of drug use, drug abuse or chronic exposure to environmental toxicants. Int. J. Toxicol. 2006, 25, 143–163. [Google Scholar] [CrossRef] [PubMed]
- Schramm, K.W. Hair-biomonitoring of organic pollutants. Chemosphere 2008, 72, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Appenzeller, B.M.; Tsatsakis, A.M. Hair analysis for biomonitoring of environmental and occupational exposure to organic pollutants: State of the art, critical review and future needs. Toxicol. Lett. 2012, 210, 119–140. [Google Scholar] [CrossRef]
- Al-Delaimy, W.K. Hair as a biomarker for exposure to tobacco smoke. Tob. Control. 2012, 11, 176–182. [Google Scholar] [CrossRef] [Green Version]
- Vogliardi, S.; Tucci, M.; Stocchero, G.; Ferrara, S.D.; Donata Favretto, D. Sample preparation methods for determination of drugs of abuse in hair samples: A review. Anal. Chim. Acta 2015, 857, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb. Exp. Pharmacol. 2009, 192, 29–60. [Google Scholar]
- Pérez-Ortuño, R.; Martínez-Sánchez, J.M.; Fu, M.; Fernández, E.; Pascual, J.A. Evaluation of tobacco specific nitrosamines exposure by quantification of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) in human hair of non-smokers. Sci. Rep. 2016, 6, 25043. [Google Scholar] [CrossRef] [Green Version]
- Yao, L.; Yang, J.; Guan, Y.F.; Liu, B.Z.; Zheng, S.J.; Wang, W.M.; Zhu, X.I.; Zhang, Z.D. Development, validation, and application of a liquid chromatography-tandem mass spectrometry method for the determination of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol in human hair. Anal. Bioanal. Chem. 2012, 404, 2259–2266. [Google Scholar] [CrossRef]
- Clemens, M.M.; Cardenas, V.M.; Fischbach, L.A.; Cen, R.; Siegel, E.R.; Eswaran, H.; Ekanem, U.S.; Policherla, A.; Moody, H.L.; Magann, E.F.; et al. Use of electronic nicotine delivery systems by pregnant women II: Hair biomarkers for exposures to nicotine and tobacco-specific nitrosamines. Tob. Induc. Dis. 2019, 17, 50. [Google Scholar] [CrossRef]
- Ishizaki, A.; Kataoka, H. A sensitive method for the determination of tobacco-specific nitrosamines in mainstream and sidestream smokes of combustion cigarettes and heated tobacco products by online in in-tube solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry. Anal. Chim. Acta 2019, 1075, 98–105. [Google Scholar]
- Kataoka, H. Automated sample preparation using in-tube solid-phase microextraction and its application—A review. Anal. Bioanal. Chem. 2002, 373, 31–45. [Google Scholar] [CrossRef]
- Kataoka, H.; Ishizaki, A.; Saito, K. Recent progress in solid-phase microextraction and its pharmaceutical and biomedical applications. Anal. Methods 2016, 8, 5773–5788. [Google Scholar] [CrossRef]
- Kataoka, H. In-tube solid-phase microextraction: Current trends and future perspectives. J. Chromatogr. A 2021, 1636, 461787. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, H.; Inoue, T.; Saito, K.; Kato, H.; Masuda, K. Analysis of heterocyclic amines in hair by on-line in-tube solid-phase microextraction coupled with liquid chromatography–tandem mass spectrometry. Anal. Chim. Acta 2013, 786, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Ishizaki, A.; Kataoka, H. Biomonitoring method for the determination of polycyclic aromatic hydrocarbons in hair by online in-tube solid-phase microextraction coupled with high performance liquid chromatography and fluorescence detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 1000, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Inukai, T.; Kaji, S.; Kataoka, H. Analysis of nicotine and cotinine in hair by on-line in-tube solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry as biomarkers of exposure to tobacco smoke. J. Pharm. Biomed. Anal. 2018, 156, 272–277. [Google Scholar] [CrossRef] [PubMed]
- ICH Harmonized Tripartite Guideline, ICH Q2 (R1). Validation of Analytical Procedures: Text and Methodology. In Proceedings of the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use.

| TSNA | Linearity | LOD 2 (pg mL–1) | LOQ 3 (pg mg–1) | ||
|---|---|---|---|---|---|
| Range (pg mL–1) | Linearity 1 (R2) | Direct Injection | In-Tube SPME | In-Tube SPME | |
| NNK | 0.5–100 | 0.9999 | 1 | 0.05 | 0.03 |
| NNN | 0.5–100 | 1 | 1.8 | 0.04 | 0.03 |
| NAT | 0.5–100 | 0.9999 | 1.1 | 0.02 | 0.02 |
| NBT | 0.5–100 | 0.9998 | 3.5 | 0.07 | 0.04 |
| NNAL | 5–1000 | 0.9999 | 12.9 | 1.14 | 0.75 |
| TSNA | Nominal Concentration (pg mL–1) | Precision (CV 1%) (n = 5) | Accuracy (%) (n = 5) | ||
|---|---|---|---|---|---|
| Intra-Day | Inter-Day | Intra-Day | Inter-Day | ||
| NNK | 2 | 3.6 | 6.4 | 100.5 | 102.5 |
| 20 | 2.7 | 3.7 | 100.7 | 100.3 | |
| NNN | 2 | 7.3 | 9.2 | 104.5 | 104.5 |
| 20 | 2.7 | 4.6 | 104.7 | 104.2 | |
| NAT | 2 | 4.4 | 5.2 | 113.5 | 112.0 |
| 20 | 2.1 | 3.0 | 103.9 | 105.2 | |
| NBT | 2 | 3.1 | 8.0 | 114.5 | 118.5 |
| 20 | 3.0 | 7.7 | 94.2 | 99.3 | |
| NNAL | 20 | 4.2 | 7.6 | 95.0 | 96.3 |
| 200 | 4.2 | 7.0 | 94.3 | 99.2 | |
| TSNA | Concentration (pg mg–1 Hair) | Recovery | |
|---|---|---|---|
| Spiked | Mean ± SD (n = 3) | (%) | |
| NNK | 0 | ND 1 | |
| 2 | 1.85 ± 0.12 | 92.3 | |
| 20 | 20.0 ± 0.4 | 100.2 | |
| NNN | 0 | ND | - |
| 2 | 1.95 ± 0.09 | 97.3 | |
| 20 | 19.0 ± 0.8 | 94.9 | |
| NAT | 0 | ND | - |
| 2 | 1.97 ± 0.07 | 98.4 | |
| 20 | 20.1 ± 0.2 | 100.4 | |
| NAB | 0 | ND | - |
| 2 | 1.92 ± 0.05 | 96.2 | |
| 20 | 19.7 ± 0.2 | 98.3 | |
| NNAL | 0 | ND | - |
| 20 | 18.7 ± 0.9 | 93.7 | |
| 200 | 205.4 ± 0.8 | 102.7 | |
| TSNA | Smokers (n = 24) | Non-Smokers (n = 29) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Detection Frequency (%) | Content (pg mg–1 Hair) | Detection Frequency (%) | Content (pg mg–1 Hair) | |||||||
| Mean ± SD | Min.1 | Med. 1 | Max. 1 | Mean ± SD | Min. | Med. | Max. | |||
| NNK | 100 | 0.95 ± 0.96 2 | 0.08 | 0.68 | 3.97 | 34 | 0.05 ± 0.08 2 | 0.00 | 0.00 | 0.25 |
| NNN | 100 | 0.43 ± 0.85 3 | 0.02 | 0.22 | 4.44 | 14 | 0.02 ± 0.06 3 | 0.00 | 0.00 | 0.24 |
| NAT | 68 | 0.09 ± 0.09 | 0.00 | 0.08 | 0.35 | 0 | ND | 0.00 | 0.00 | 0.00 |
| NBT | 88 | 0.13 ± 0.27 | 0.00 | 0.06 | 1.09 | 0 | ND | 0.00 | 0.00 | 0.00 |
| NNAL | 0 | ND 4 | ND | ND | ND | 0 | ND | 0.00 | 0.00 | 0.00 |
| Total | 100 | 1.61 ±1.55 5 | 0.11 | 1.08 | 6.72 | 34 | 0.08 ± 0.11 5 | 0.00 | 0.00 | 0.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishizaki, A.; Kataoka, H. Online In-Tube Solid-Phase Microextraction Coupled to Liquid Chromatography–Tandem Mass Spectrometry for the Determination of Tobacco-Specific Nitrosamines in Hair Samples. Molecules 2021, 26, 2056. https://doi.org/10.3390/molecules26072056
Ishizaki A, Kataoka H. Online In-Tube Solid-Phase Microextraction Coupled to Liquid Chromatography–Tandem Mass Spectrometry for the Determination of Tobacco-Specific Nitrosamines in Hair Samples. Molecules. 2021; 26(7):2056. https://doi.org/10.3390/molecules26072056
Chicago/Turabian StyleIshizaki, Atsushi, and Hiroyuki Kataoka. 2021. "Online In-Tube Solid-Phase Microextraction Coupled to Liquid Chromatography–Tandem Mass Spectrometry for the Determination of Tobacco-Specific Nitrosamines in Hair Samples" Molecules 26, no. 7: 2056. https://doi.org/10.3390/molecules26072056