2.1.1. Monomers
The considered MX
molecules are characterized by a linear structure in which M–X bonds are formed by overlapping of the hybridized
sp orbital of the metal atom with one of the orbitals of X. Due to lower electronegativity of the metal atom, this atom is endowed with a partial positive charge (
Table 1), becoming electron-depleted and therefore a Lewis acid center.
The atomic charge values shown in
Table 1 confirm the known fact that they can be very significantly dependent on the method of obtaining them in the calculations [
173,
174,
175,
176]. The atomic charges obtained by the NBO and QTAIM methods seem to be greatly exaggerated. In the context of the presented results, however, it is more important that all the methods of obtaining atomic charges used here (i.e., Hirshfeld [
177,
178,
179], NBO [
180,
181], and QTAIM [
182,
183,
184]) show that in the set of MX
molecules for a given metal M the most positive charge on the M atom occurs when X = F. This is fully understandable due to the very high electronegativity of the fluorine atom. Conversely, the smallest positive charge on the M atom occurs when X = H. This result is not as expected, because, due to the positive inductive effect (+I) of the methyl group, one would expect the smallest positive atomic charge of M in MMe
. It is also seen that the Cl and Br atoms lead to similar atomic charges on M. Importantly, all the methods used show that the highest positive charge occurs in MgF
, and the lowest in ZnH
. If we refer to the most reliable [
175,
176] Hirshfeld atomic charges, then these values are 0.924 and 0.354 au, respectively. The former value suggests an extremely high polarization of the Mg-F bond, which practically becomes the Mg
F
ionic one. A practical consequence of this finding is that, assuming electrostatic reasoning, the MgF
molecule should be the best Lewis acid, and therefore it should theoretically form the strongest adducts with carbenes and CDPs.
With the values of the atomic charges obtained by various theoretical methods, it is interesting to see if there are clear relationships between them.
Figure 2 shows the relationships between the Hirshfeld charges and their equivalents obtained by the NBO or QTAIM method.
As can be clearly seen, the linear relationships between the Hirshfeld atomic charges and those obtained by the NBO or QTAIM method are very weak. Particularly in the case of the latter method, the obtained coefficient of determination is unacceptably low. This result shows that especially the atomic charges obtained by QTAIM should not be treated as reliable. This flaw of QTAIM-based atomic charges was attributed to irregular shapes of atomic basins, which give them multipolar moments.
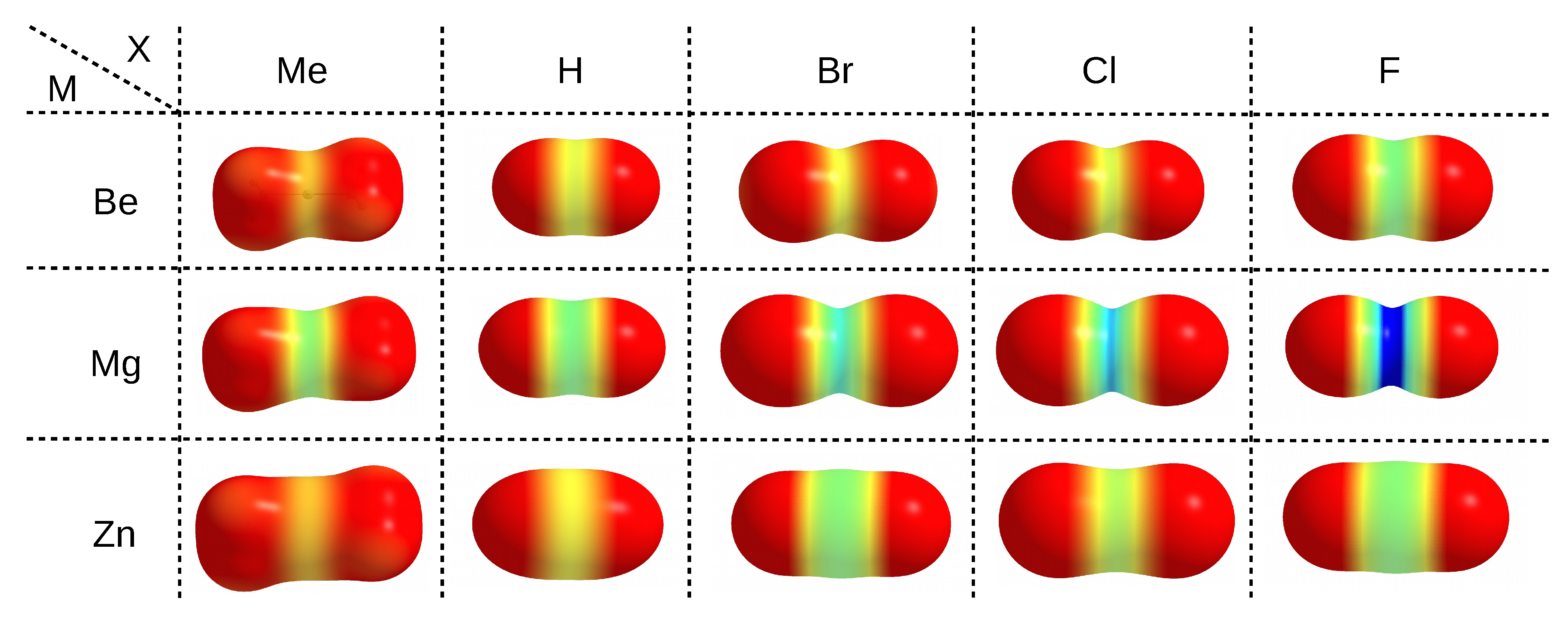
The electrophilic properties of a particular metal atom, which is an acidic center in the MX
molecule, can be nicely illustrated by means of maps of the distribution of the molecular electrostatic potential (MESP) projected onto the electron density isosurface, as shown in
Figure 3.
The use of same scale of the electrostatic potential values (from 0.0 au (red) to 0.2 (blue)) for all MX
molecules allows one to easily capture the existing relationships. It is clearly seen that, upon going in the series Me→H→Br→Cl→F, i.e., from left to right in
Figure 3, a belt of even more positive electrostatic potential develops around the central metal atom. This is of course confirmed by the corresponding values of the maximum electrostatic potential on M (
(M)), which are provided in the last column of
Table 1. For zinc compounds, these values (in au) increase in this series as follows: 0.037 < 0.050 < 0.065 < 0.071 < 0.093. Although the MESP maps for zinc molecules are very similar to those for beryllium, it is worth noting that in the former case, the corresponding MESP belt is wider and larger in diameter due to the larger atomic radius of Zn
(88 pm) compared to Be
(59 pm) [
185]. Therefore, compared to beryllium, the zinc atom should be more accessible. The more important result, however, is that, for a given X, the belts of positive MESP are most visible when the central metal atom is magnesium. The values of
(M) increase monotonically quite quickly in the order given earlier, reaching a maximum value of 0.243 au in MgF
(
Table 1). The fact that the
(M) values increase in this order, while
q(M) does not, suggests that
(M) is perhaps the more reliable parameter describing the acidic nature of the central metal atom in MX
molecules than
q(M). Although the linear relationship between the value of
(M) is not very good (
= 0.795) either when
q(M) is computed utilizing the Hirshfeld method, it is much better than in the case of NBO- and especially QTAIM-based charges (
Figure 4).
Imidazol-2-ylidene, imidazolidin-2-ylidene, tetrahydropyrymid-2-ylidene, cyclopropenylidene, and (NH
)
C have been used as model representatives of carbenes. In particular, the first two carbenes are often used in organic and organometallic chemistry and represent an important starting point in the syntheses of larger carbene compounds [
98,
102,
103]. The CDPs group is represented by (PH
)
C and its amino derivative (NH
)
C. Both are the starting molecules for more complex CDPs obtained by substituting hydrogen atoms in one or both of the -PH
or -NH
groups. It is worth mentioning here that the fully saturated phenyl derivative, i.e., (PPh
)
C was the first synthesized CDP [
154]. Some fundamental parameters characterizing the considered carbenes and CDPs are presented in
Table 2.
When analyzing the obtained values of the atomic charge on the carbon atom, one can easily notice their great diversity, even in terms of sign. In the case of carbenes, positive QTAIM atomic charges have been obtained. Additionally, this method has given (too) large variation in the negative values on the C atom in (PH
)
C and (NH
)
C (−2.261 and −0.179 au, respectively). Both of these findings strongly suggest that atomic charges of QTAIM are highly unreliable. A similar conclusion applies to the atomic charges of NBO, although the values themselves are not that large. It is worth mentioning that the value of the atomic charge on a carbon atom of −1.43 au in (PPh
)
C was used by Tonner et al. [
167] as an argument supporting the bonding scheme of CDPs presented in
Figure 1. However, taking into account large dependence of the atomic charge on the method used in calculations, it seems that this argument was perhaps not entirely correct. The more reliable [
175,
176] Hirshfeld atomic charges are negative in both carbenes and CDPs. Understandably, in the latter case they are much larger, which results from the role of the carbon atom as an acceptor in the R→C bonds (
Figure 1).
Further valuable information on the nucleophilic abilities of singlet carbenes and CDPs can be obtained from the values of the minimum electrostatic potential on C (the penultimate column in
Table 2) and the distribution of this potential around this atom (see
Figure 5).
The electrostatic potential maps clearly show the negative potential area around the C(2) atom in the carbenes or the C(0) atom in the CDPs. On the other hand, strong positive potential concerns mainly hydrogen atoms in strongly polar N-H bonds. While the characteristics of the negative potential distribution around the carbon atom are similar in carbenes (which is in line with the rather similar values of
(C);
Table 2), there is a clear difference between (PH
)
C and (NH
)
C. Specifically, in the latter case, this area in much clearer and much more spread around the carbon atom, which better emphasizes the great nucleophilic properties of this molecule. Both of these molecules also differ considerably in the value of
(C) (−0.067 and −0.109 au, respectively).
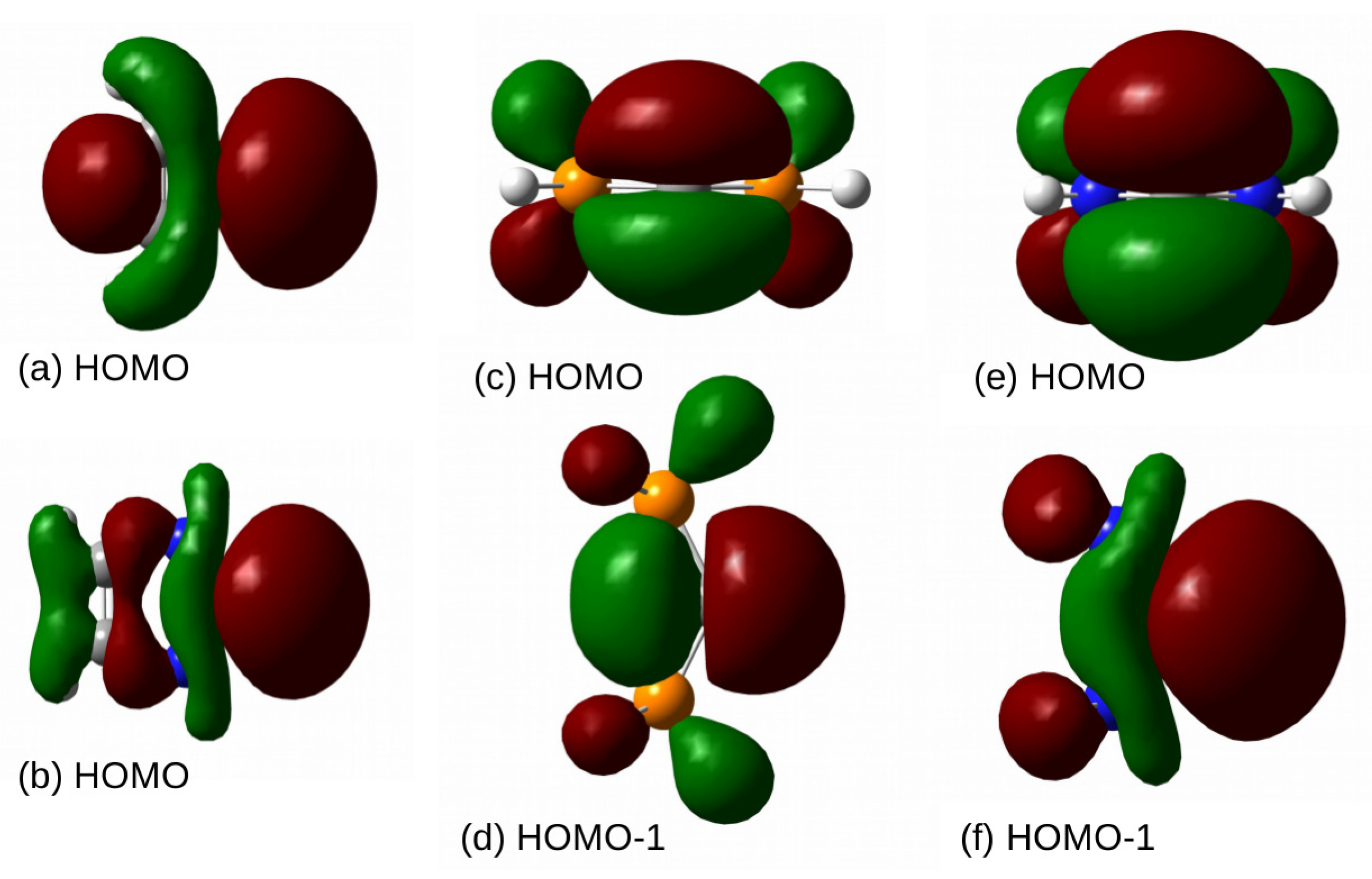
Further information on the reactivity of molecules can be obtained from the Frontier Molecular Orbital theory [
186,
187], which has found its mathematical support in the Klopman–Salem Equation [
188,
189]. According to it, the electron-donating properties of the molecule can be characterized by the energy of HOMO. These energies for carbenes and CDPs are shown in the last column of
Table 2. By far the least negative value of the HOMO energy obtained for (NH
)
C (−4.35 eV) confirms that this molecule should undoubtedly be the most reactive, willingly acting as a Lewis base. It should be noted, however, that the HOMO energy, like the LUMO energy, which is also often used in the Frontier Molecular Orbital theory, is a global quantity, i.e., resulting from the electronic structure of the entire molecule, and therefore it does not necessarily correctly assess the nucleophilic and electrophilic properties of a molecule, which are most often strongly local. Moreover, these energies do not necessarily correlate well with the parameters characterizing the dimer strength. For example, as shown by Martín-Sómer et al. [
24], LUMO energies correlate well with interaction energies (of some beryllium bonds) only when they are computed for acceptor molecules in their dimer geometries. For this reason, LUMO energy values for the fully optimized MX
molecules were not exposed in
Table 2. Moreover, in the case of MX
molecules, the LUMO energy strongly depends on the X-M-X angle (
), decreasing considerably with increasing deviation from the linearity of the molecule. In this way, Martín-Sómer et al. [
24] explained the large non-linearity of the BeH
X
(X = F, Cl, Br;
) molecules in their dimers with ammonia. Therefore, it seems that there is nothing to prevent the same cause of MX
bending also working for other Lewis bases, such as the carbenes and CDPs considered here. It is also worth mentioning that the electron lone pair, which in carbenes is HOMO (quite strongly delocalized), in the case of CDPs, i.e., (PH
)
C and (NH
)
C, becomes HOMO-1, while HOMO is the electron lone pair perpendicular to the plane of the molecule (
Figure 6).
2.1.2. Dimers
The previous subsection has shown that in MX
(M = Be, Mg, Zn; X = H, F, Cl, Br, Me) molecules, the metal atom is a relatively strong acid center, while the C(2) atoms in the carbenes and C(0) in the CDPs are strong basic regions. Moreover, these atoms are the only such regions in these molecules (see
Figure 3 and
Figure 5). Due to this alignment in electronic properties, it should be expected that the MX
molecules quite easily form a M⋯C bond to the C(2) carbon in carbenes or C(0) in CDPs. If so, it should lead to a particular type of beryllium, magnesium, or zinc (spodium) bond. As mentioned in the Introduction, the main purpose of this article is to describe these interactions. Nevertheless, the electrostatic potential distributions for MX
(
Figure 3) and carbenes and CDPs (
Figure 5) suggest that other interactions accompanying the leading M⋯C interaction may also be possible. In particular, some symptoms of the presence of a hydrogen bond of the N-H⋯X type (where X is a halogen atom, especially F) are to be expected. The geometries of the fully optimized dimers are shown in
Figure 7. It is convenient to describe the characteristics of the systems containing carbenes and CDPs separately.
The basic parameters characterizing the investigated carbene-containing dimers are shown in
Table 3.
Due to the simple structure of the cyclopropenylidene molecule, dimers containing this carbene will be discussed first. It should be noted that the plane of the slightly bent MX
molecule is oriented perpendicular to the plane of the cyclopropenylidene ring (
Figure 7). For this reason, the interaction between MX
and cyclopropenylidene is free from any other significant interactions than C⋯M. Although the earlier analysis of the values of atomic charges and electrostatic potentials on M and C suggests that the strongest C⋯M interaction should be present in the case of MgF
and the weakest in the case of ZnH
, this is not in line with the values of the distance C⋯M (
). Rather, these distances result from the radius of the metal atom, so in the case of beryllium,
is less than ca. 1.83 Å, while in the case of Mg and Zn, this distance is over 2 Å. The penultimate column in
Table 3 shows that cyclopropenylidene⋯MX
dimers are strongest (32–35 kcal/mol) when M is either Be or Mg and X is a halogen, especially Cl or Br. The lowest dissociation energy (10.3 kcal/mol) has been obtained in the case of ZnMe
. The weakest C⋯M in the presence of methyl groups has also been obtained in the case of M = Be or Mg and is in line with the weak +I character of the methyl group. Due to the C
symmetry, the following relations hold:
=
=
and
=
. The greatest elongation of the MX bond (0.093 Å) occurs in BeBr
. Along with a similar BeCl
, in this molecule, there is also the greatest deviation from linearity (
amounts to ca. 134
). Thus, the geometric characteristics of the MX
molecule itself and the obtained
values suggest that in the cyclopropenylidene⋯MX
dimers, the interaction should be strongest for BeBr
and BeCl
. It is interesting to see if similar finding also apply to dimers involving the other carbenes.
As
Figure 7 shows, the MX
molecule lies in the same plane as the backbone atoms of the carbene molecule. This arrangement is also characteristic for dimers involving CDPs, (PH
)
C and (NH
)
C. In at least some cases, the planar geometry of the dimer can be explained by additional beneficial interactions (as will be discussed). As was the case with cyclopropenylidene, the intermolecular distance C⋯M is much shorter for beryllium (ca. 1.76–1.85 Å) than for either magnesium (ca. 2.17–2.28 Å) or zinc (2.03–2.20 Å). However, this does not mean stronger C⋯M interactions. The calculated dissociation energy values clearly show (
Table 3) that, as was the case for cyclopropenylidene, the strongest intermolecular C⋯M interaction occurs for BeBr
and BeCl
. Although the bond strength of the former is ca. 47–48 kcal/mol, it reaches up to 53 kcal/mol when BeBr
interacts with tetrahydropyrymid-2-ylidene. On the other hand, similar to cyclopropenylidene, the C⋯M interaction is the weakest (but clearly stronger than that of cyclopropenylidene) when the MX
molecule is ZnMe
. Consequently, in the dimers considered here, the dissociation energies of C(2)⋯M have a wide range from 10 to 53 kcal/mol. This result is in full accord with the recent generalization given by Alkorta and Legon that beryllium and magnesium bonds (the current results show also include the zinc bonds) are generally much stronger than hydrogen bonds, halogen bonds, etc. [
29].
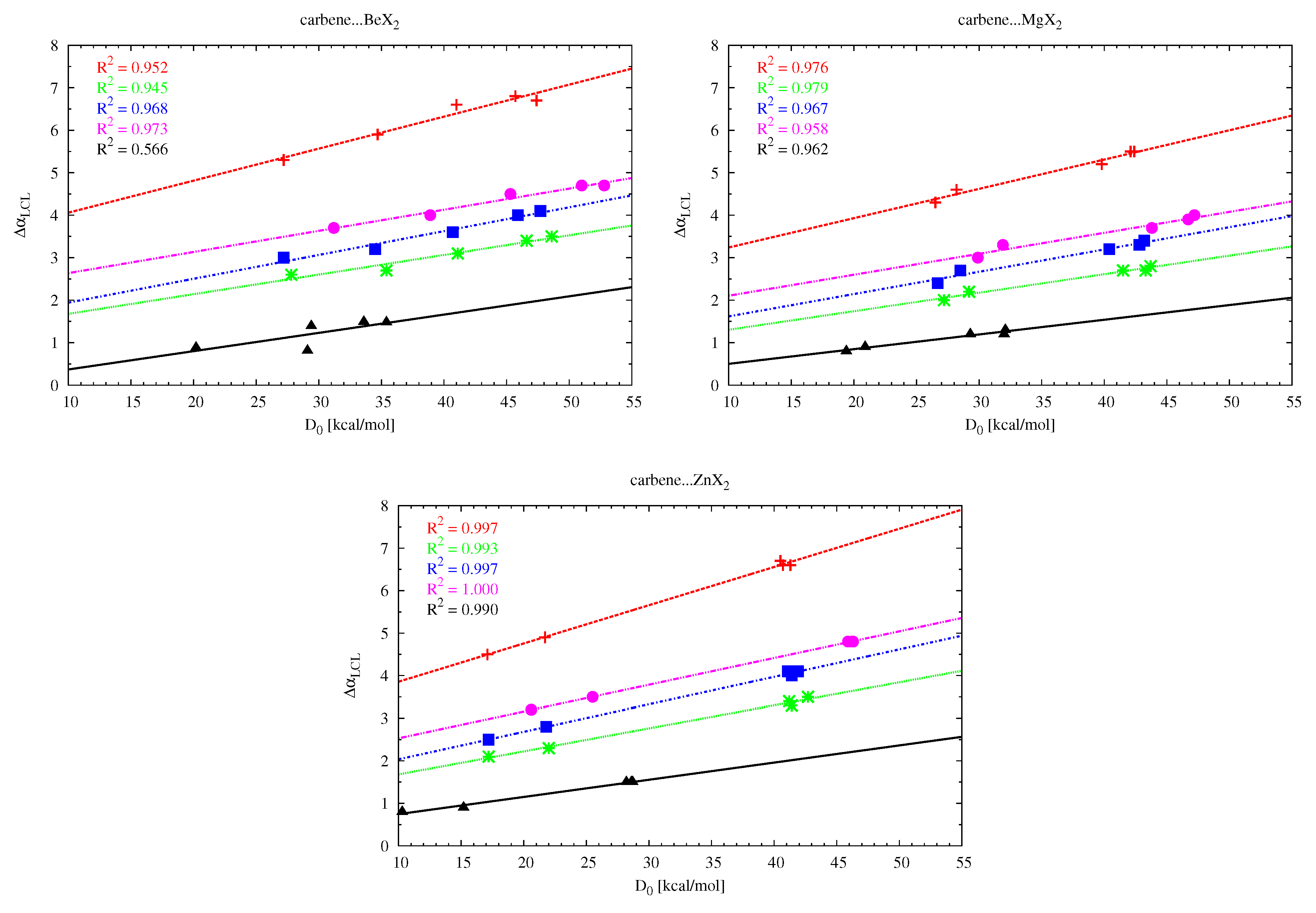
The LCL angle change (
) values show that the interaction between MX
and the carbene molecule leads to the opening of the latter molecule, with the effect being the greatest for (NH
)
C (e.g.,
= 6.8
for (NH
)
C⋯BeCl
). This shows that the
angle in (NH
)
C is more flexible than in cyclic and therefore more rigid imidazol-2-ylidene, imidazolidin-2-ylidene and tetrahydropyrymid-2-ylidene (
in cyclopropenylidene is negligible). Although in general the
angle-opening effect in the carbene molecule does not seem to be dependent on the strength of the interaction with MX
, such a relationship can be found when comparing systems with similar skeleton stiffness. Therefore, in the group of the aforementioned cyclic carbenes, the strongest effect occurs in tetrahydropyrymid-2-ylidene (4.8
). Excellent linear relationships have been found (see
Figure 8) between the change of the opening angle
and the dissociation energy of the carbene⋯MX
dimer as long as the carbenes and the MX
molecules are treated separately. Note that the greater sensitivity of the opening angle in the case of the (NH
)
C carbene is evident here by slightly larger slopes of the corresponding (red) lines. Moreover, the slopes of the linear fits for cyclic carbenes are similar to each other.
A characteristic effect that occurs during an interaction of the initially linear MX
molecule with a strong Lewis base is its significant bend [
23]. For example, Martín-Sómer et al. have reported XCX angles (
) of 134
–139
, (B3LYP/6-311+G(3df,2p)) for dimers of X-substituted (X = F, Cl, Br) BeX
derivatives with ammonia [
24]. This bending effect is much less (138
–149
,) in BeX
(X = H, F, Cl) dimers with ethylene or acetylene, being much weaker Lewis bases interacting via
bonds [
26]. The high sensitivity of the
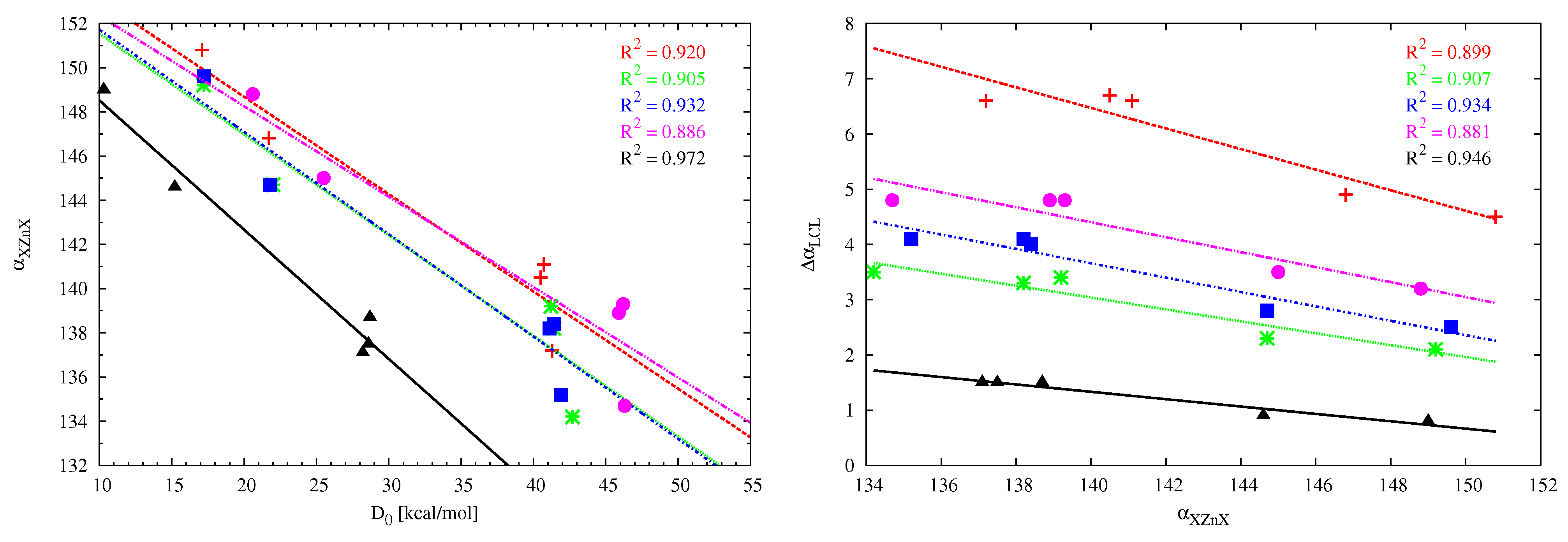
angle makes it particularly interesting to trace its values in the considered dimers. Due to the large number of the studied set of systems and their diversity (different acid centers M, different X substituents, different carbenes), a fairly wide range of
variability has been obtained, from 131 to 151
, i.e., as much as 20
. The bending effect is greatest for BeCl
and BeBr
and the smallest for ZnMe
. The linear correlation between the XMX angle and the dissociation energy is acceptable for ZnX
(
Figure 9, left) and the dimers of either imidazol-2-ylidene (
= 0.942) or imidazolidin-2-ylidene (
= 0.922) with BeX
(not shown). The fitting line for cyclopropenylidene has slightly different slope than the other four cases, which may result from different (perpendicular) orientation of the interacting molecules (
Figure 7). The weak linear correlation for the remaining cases of carbene⋯MX
(M = Be, Mg) dimers may, at least partly, result from the presence of additional interactions is some of the considered dimers, which should have some influence on the angle XMX. In the case of the dimers involving ZnX
, as a consequence of good linear relationships between
and
(
Figure 8) and
and
(
Figure 9, left), one also observes good linear relationships between
and
(
Figure 9, right).
Another effect observed during the formation of the carbene⋯MX
dimers is a significant elongation of both MX bonds. It should be clearly underlined here that, in general, both MX bond elongations are not necessarily of equal magnitude, so it is not necessarily true that
=
=
(
Table 3). These unsymmetrical elongations of MX result from the presence of certain accompanying interactions in some of the dimers studied here. Such cases are also clearly visible from different values of CMX1 and CMX2 angles (
and
, respectively) in
Table 3. In such cases, the smaller of these angles (
) takes a value roughly about the right angle.
As already mentioned, any significant additional interactions are impossible in cyclopropenylidene dimers. In this case, the effect of MX bond elongation is therefore symmetrical, which allows for straigtforward analysis of the obtained relationships. The greatest elongation of the MX bonds is for X = Cl or Br, but only when the M atom is either beryllium or zinc (up to 0.093 Å for BeBr). Hence, the elongation effect is not entirely consistent with the strength of C⋯M if measured by . On the contrary, the smallest elongation of the MX bond occurs in MgMe (0.030 Å) and MgF (0.031 Å). Although the relatively small magnitude of the effect in the former case can be explained by a relatively weak interaction (the largest distance C⋯M amounting to 2.288 Å and the smallest bending of 149.1), BeH is also characterized by a small MX bond elongation (0.039 Å), and this molecule forms the shortest contact with cyclopropenylidene, amounting to only 1.743 Å. For the latter molecule, i.e., MgF, the small effect of the MgF bond elongation can most likely be explained by a high polarity of the bond and therefore its considerable resistance to changes. It seems that the magnitude of the MX bond elongation does not clearly depend on or the interaction strength measured by .
As mentioned earlier, the asymmetry of the MX elongation effects in case of many dimers involving the remaining carbenes makes the analysis much more difficult, but mean value () provides some information. Regardless of carbene, this value for BeBr is always greater than 0.106 Å and reaches a maximum value of 0.111 Å for tetrahydropyrymid-2-ylidene, thus confirming that presumably the C⋯M interaction is the strongest in the tetrahydropyrymid-2-ylidene⋯BeBr dimer. The occurrence of the minimum values of appears to be more irregular. Although BeH is generally characterized by low values (ca. 0.048 Å), the lowest values (ca. 0.040 Å) are nevertheless found for MgMe interacting with either imidazol-2-ylidene or imidazolidin-2-ylidene.
The last column in
Table 3 shows values of charge transfer calculated by means of the most reliable [
175,
176] Hirshfeld atomic charges (CT
). First, it should be noted that the obtained values are negative, which means that the formation of the carbene⋯MX
dimer leads to an increase in the total charge on the MX
molecule. Secondly, the obtained values are very large. Suffice it to mention that the corresponding charge transfer values obtained (on the same level of theory) for dimers HOH⋯OH
and HOH⋯NH
are −0.098 and −0.122 au, respectively. Thus, even the weakest charge transfers obtained for the investigated dimers are over two times greater (e.g., −0.270 au for cyclopropenylidene⋯ZnMe
) and even reach almost four times higher values in some dimers with BeBr
(e.g., CT
amounts to ca. −0.46 au for imidazol-2-ylidene, imidazolidin-2-ylidene, and tetrahydropyrymid-2-ylidene). Undoubtedly, therefore, the carbene⋯MX
(M = Be, Mg, Zn) dimers considered here are characterized by a significant charge transfer, which is particularly high in the presence of highly polarizable halogen atoms in MX
, especially Br. This finding is also manifested by very good (
= 0.955) linear correlation between CT
and
when X = Br and only slightly worse for X = Cl (
= 0.917), while this correlation is very weak (
= 0.154) for much less polarizable fluorine (
Figure 10).
For the cyclopropenylidene⋯MX
dimers, there are also very good linear correlations between CT
and either
or D
(in the latter relationship, except in the case of M = Be) if only systems with different M atoms are treated separately (
Figure 11).
Unfortunately, similar relationships are generally much worse for other carbenes, which can be explained by the presence of additional intermolecular interactions in some of them, which to some extent affects the obtained values of the analyzed parameters.
The fundamental data characterizing CDP⋯MX
dimers are included in
Table 4.
Its penultimate column shows that the C(0)⋯M interactions in the dimers formed by (PH
)
C are comparable in strength to the C(2)⋯M bonds formed by the investigated carbenes, whereas those formed by (NH
)
C are much stronger. Again, the maximum value is found for BeBr
, reaching 84 kcal/mol, a value comparable to the energy of weaker covalent bonds [
190]. The other dimers with high values of
are (NH
)
C⋯BeCl
and (NH
)
C⋯ZnF
(ca. 79 kcal/mol). It is worth recalling here the theoretical research by Jabłoński and Palusiak [
108] on the ability of carbenes and CDPs to form hydrogen bonds. The results of those studies have shown that for the same Lewis acid (e.g., HCCH), the hydrogen bond to (NH
)
C is much stronger than to (PH
)
C (the MP2/aug-cc-pVTZ-based BSSE-corrected interaction energies amount to −9.16 and −5.31 kcal/mol, respectively, [
108]), which further confirms the greater basicity of the former molecule. Although the (NH
)
C⋯BeBr
and (NH
)
C⋯BeCl
dimers are characterized by short C⋯Be distances (1.643 and 1.661 Å, respectively), the short C⋯Be distance (1.655 Å) is also present in the (NH
)
C⋯BeH
dimer with much weaker interaction (64 kcal/mol).
Very high bond strength of C(0)⋯M in the (NH)C⋯MX dimers is in line with high values of charge transfer, which can even reach -0.610 au in the (NH)C⋯BeBr dimer. This value is more than six times greater than that of the water dimer and exactly five times greater than that of the water-ammonia dimer. A curiosity is the relatively low CT value (−0.287 au) obtained for the (NH)C⋯ZnF dimer with a simultaneous very high dissociation energy (78.8 kcal/mol). In the next subsection, however, it will be shown that this dimer is characterized by a highly advanced proton transfer from N to F, which results in the formation of the N⋯H-F hydrogen bond. The formation of the H-F bond requires some removal of the electron charge from the fluorine atom.
An interesting result is that, as in the case of carbenes (
Table 3), the interaction between MX
and (NH
)
C causes a significant opening of the
angle, whereas in the case of (PH
)
C, the change in the
angle is much smaller and may have different sign, most often being negative. This finding clearly differentiates the nitrogen atom from the phosphorus atom.
The comparison of the values of
and
as well as
and
shows a clear difference between the dimers with (PH
)
C and the dimers with (NH
)
C. Specifically, the former of them are characterized by the equality of both quantities, which indicates symmetry of these dimers with respect to the axis passing through the C and M atoms (see also
Figure 7). In the latter case, however, this symmetry is clearly broken in most of the dimers, which results from the presence of other interactions accompanying the leading contact C⋯M. As a result, the search for linear correlations between the parameters from
Table 4 for systems with (NH
)
C is pointless, while the search for such correlations for systems with (PH
)
C seems to be justified. Indeed, some reasonable linear correlations have been found, such as, for example, between
and
(see
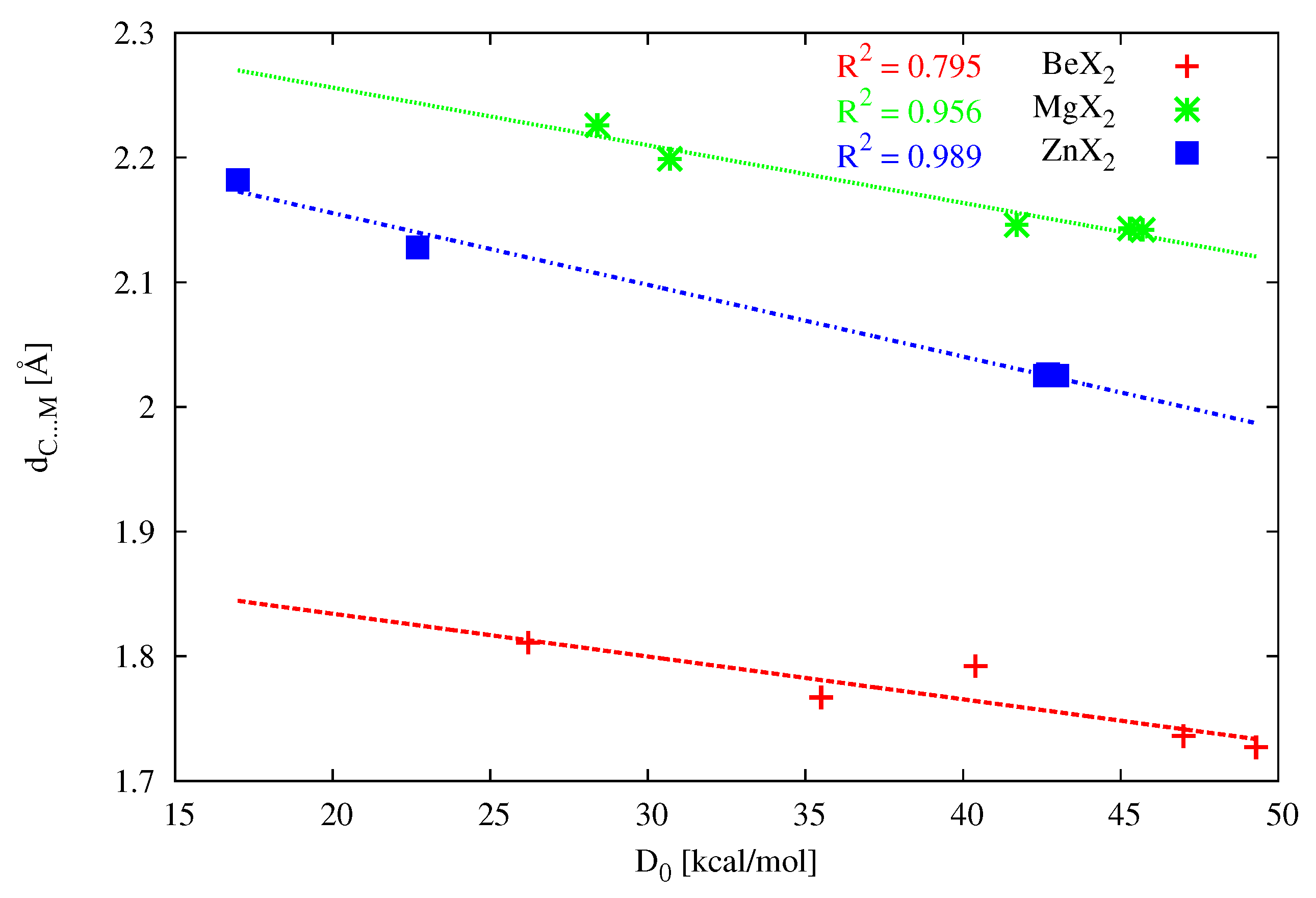
Figure 12) when the metal atom is either Mg (
= 0.956) or especially Zn (
= 0.989). On the other hand, when the acidic metal center is beryllium, the linear correlation clearly deteriorates (
= 0.795). At least in part, this may be due to the much shorter Be-X bond compared to the Mg-X or Zn-X bonds, and thus stronger, although still rather weak, intermolecular interactions of the type -(PH
)⋯X. Moreover, quite good linear relationships between CT
and
(
= 0.901),
(
= 0.911) or
(
= 0.930) have been found for the analyzed (PH
)
C⋯ZnX
dimers.