Inhibitory Effects of Coumarin Derivatives on Tyrosinase
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
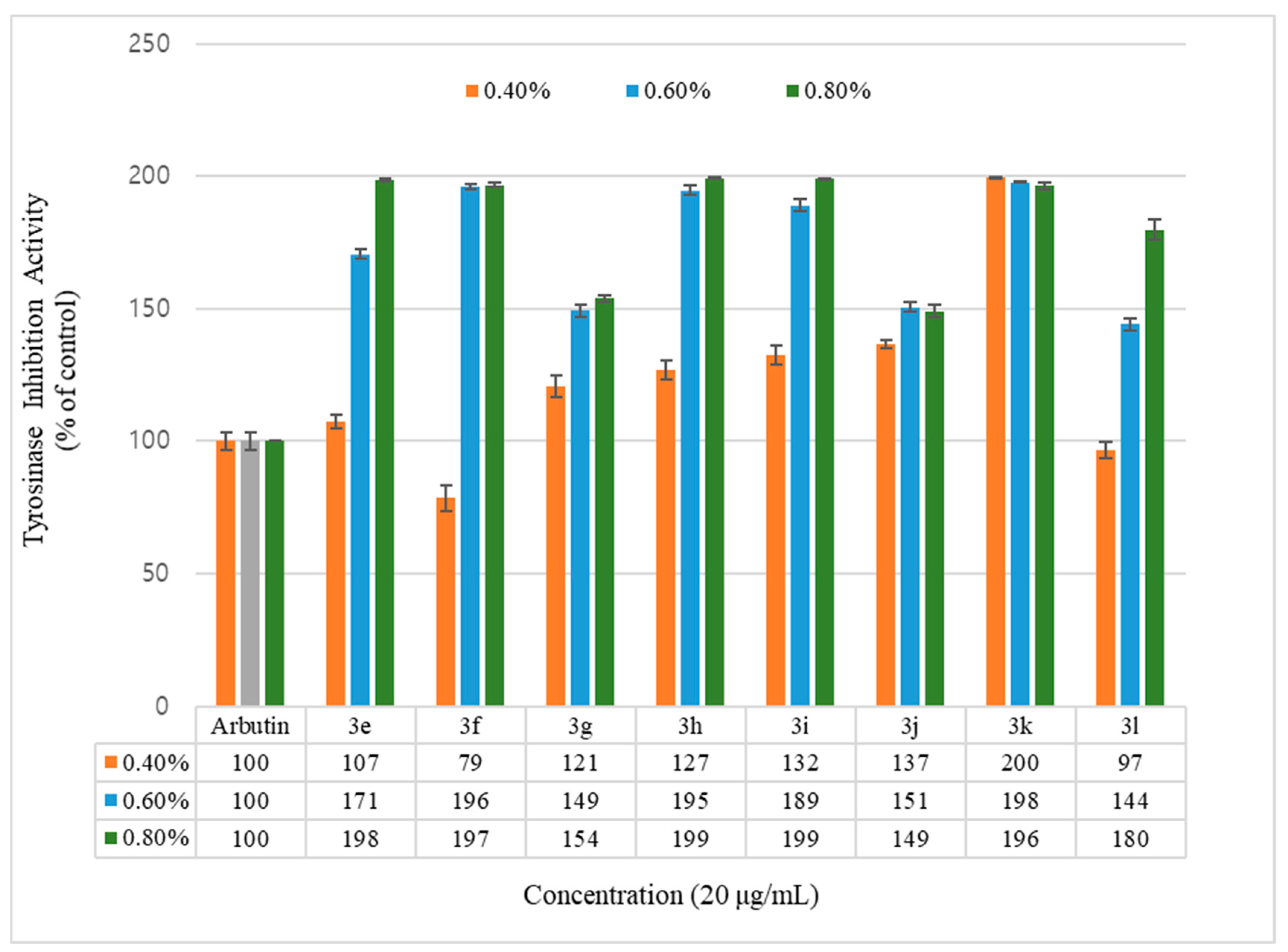
2.2. Tyrosinase Inhibitory Activity
3. Materials and Methods
3.1. Materials
3.2. Synthesis
3.3. Tyrosinase Inhibition Assay
3.4. Statistical Analysis
4. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
Sample Availability
References
- Khan, S.B.; Khan, M.T.H.; Jang, E.S.; Akhtar, K.; Seo, J.; Han, H. Tyrosinase inhibitory effect of benzoic acid derivatives and their structure-activity relationships. J. Enzyme Inhib. Med. Chem. 2010, 25, 812–817. [Google Scholar] [CrossRef]
- Ohguchi, K.; Tanaka, T.; Ito, T.; Iinuma, M.; Matsumoto, K.; Akao, Y.; Nozawa, Y. Inhibitory effects of resveratrol derivatives from dipterocarpaceae plants on tyrosinase activity. Biosci. Biotechnol. Biochem. 2003, 67, 1587–1589. [Google Scholar] [CrossRef] [Green Version]
- Pillaiyar, T.; Manickam, M.; Namasivayam, V. Skin whitening agents: Medicinal chemistry perspective of tyrosinase inhibitors. J. Enzyme Inhib. Med. Chem. 2017, 32, 403–425. [Google Scholar] [CrossRef] [Green Version]
- Briganti, S.; Camera, E.; Picardo, M. Chemical and instrumental approaches to treat hyperpigmentation. Pigment Cell Res. 2003, 16, 101–110. [Google Scholar] [CrossRef]
- Arndt, K.A.; Fitzpatrick, T.B. Topical Use of Hydroquinone as a Depigmenting Agent. JAMA J. Am. Med. Assoc. 1965, 194, 965–967. [Google Scholar] [CrossRef]
- Zhou, H.; Kepa, J.K.; Siegel, D.; Miura, S.; Hiraki, Y.; Ross, D. Benzene metabolite hydroquinone up-regulates chondromodulin-I and inhibits tube formation in human bone marrow endothelial cells. Mol. Pharmacol. 2009, 76, 579–587. [Google Scholar] [CrossRef] [Green Version]
- Cabanes, J.; Chazarra, S.; Carmona, F.G. Kojic acid, a Cosmetic Skin Whitening Agent, is a Slow-binding Inhibitor of Catecholase Activity of Tyrosinase. J. Pharm. Pharmacol. 1994, 46, 982. [Google Scholar] [CrossRef]
- Iwahori, A.; Hirota, Y.; Sampe, R. NII-Electronic Library Service. Chem. Pharm. Bull. 1970, 43, 2091. [Google Scholar]
- Shimogaki, H.; Tanaka, Y.; Tamai, H.; Masuda, M. In vitro and in vivo evaluation of ellagic acid on melanogenesis inhibition. Int. J. Cosmetic Sci. 2000, 22, 291–303. [Google Scholar] [CrossRef]
- Curto, E.V.; Kwong, C.; Hermersdörfer, H.; Glatt, H.; Santis, C.; Virador, V.; Hearing, V.J.; Dooley, T.P. Inhibitors of mammalian melanocyte tyrosinase: In vitro comparisons of alkyl esters of gentisic acid with other putative inhibitors. Biochem. Pharmacol. 1999, 57, 663–672. [Google Scholar] [CrossRef]
- Fujimoto, N.; Onodera, H.; Mitsumori, K.; Tamura, T.; Maruyama, S.; Ito, A. Changes in thyroid function during development of thyroid hyperplasia induced by kojic acid in F344 rats. Carcinogenesis. 1999, 20, 1567–1572. [Google Scholar] [CrossRef]
- Spínola, V.; Mendes, B.; Câmara, J.S.; Castilho, P.C. Effect of time and temperature on vitamin C stability in horticultural extracts. UHPLC-PDA vs. iodometric titration as analytical methods. LWT Food Sci. Technol. 2013, 50, 489–495. [Google Scholar] [CrossRef]
- Akak, C.M.; Djama, C.M.; Nkengfack, A.E.; Tu, P.F.; Lei, L. Di New coumarin glycosides from the leaves of Diospyros crassiflora (Hiern). Fitoterapia. 2010, 81, 873–877. [Google Scholar] [CrossRef]
- Ngo, N.T.N.; Nguyen, V.T.; Van Vo, H.; Vang, O.; Duus, F.; Ho, T.-D.H.; Pham, H.D.; Nguyen, L.-H.D. Cytotoxic Coumarins from the Bark of Mammea siamensis. Chem. Pharm. Bull. 2010, 58, 1487–1491. [Google Scholar] [CrossRef] [Green Version]
- Blahová, J.; Nka Svobodová, Z. Assessment of Coumarin Levels in Ground Cinnamon Available in the Czech Retail Market. Sci. World J. 2012. [Google Scholar] [CrossRef] [Green Version]
- Woehrlin, F.; Fry, H.; Abraham, K. Quantification of Flavoring Constituents in Cinnamon: High Variation of Coumarin in Cassia Bark from the German Retail Market and in Authentic Samples from Indonesia. J. Agric. Food Chem. 2010, 58, 10568–10575. [Google Scholar] [CrossRef]
- Rychlik, M. Quantification of Free Coumarin and Its Liberation from Glucosylated Precursors by Stable Isotope Dilution Assays Based on Liquid Chromatography-Tandem Mass Spectrometric Detection. J. Agric. Food Chem. 2008, 56, 796–801. [Google Scholar] [CrossRef]
- Ong, E.B.B.; Watanabe, N.; Saito, A.; Futamura, Y.; Abd El Galil, K.H.; Koito, A.; Najimudin, N.; Osada, H. Vipirinin, a coumarin-based HIV-1 Vpr inhibitor, interacts with a hydrophobic region of Vpr. J. Biol. Chem. 2011, 286, 14049–14056. [Google Scholar] [CrossRef] [Green Version]
- Pan, R.; Gao, X.H.; Li, Y.; Xia, Y.F.; Dai, Y. Anti-arthritic effect of scopoletin, a coumarin compound occurring in Erycibe obtusifolia Benth stems, is associated with decreased angiogenesis in synovium. Fundam. Clin. Pharmacol. 2010, 24, 477–490. [Google Scholar] [CrossRef]
- Bhattacharyya, S.S.; Paul, S.; Mandal, S.K.; Banerjee, A.; Boujedaini, N.; Khuda-Bukhsh, A.R. A synthetic coumarin (4-Methyl-7 hydroxy coumarin) has anti-cancer potentials against DMBA-induced skin cancer in mice. Eur. J. Pharmacol 2009, 614, 128–136. [Google Scholar] [CrossRef]
- Bucolo, C.; Ward, K.W.; Mazzon, E.; Cuzzocrea, S.; Drago, F. Protective effects of a coumarin derivative in diabetic rats. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3846–3852. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Wu, F.; Chen, L.; Zhao, L.; Zhao, Z.; Wang, M.; Lei, S. Biological evaluation of coumarin derivatives as mushroom tyrosinase inhibitors. Food Chem. 2012, 135, 2872–2878. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Nano, G.M.; Palmisano, G.; Tagliapietra, S. 4-Hydroxycoumarin and Related Systems: Sitoselectivity of the Mitsunobu Reaction with Prenyl Alcohols. Heterocycles 2003, 60, 1351–1358. [Google Scholar] [CrossRef]
- Wang, N.; Hebert, D.N. Tyrosinase maturation through the mammalian secretory pathway: Bringing color to life. Pigment Cell Res. 2006, 19, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Jeon, M.-J.; Kim, M.-H.; Jang, H.-J.; Lee, S.-W.; Kim, J.-H.; Kim, H.-S.; Lee, S.-H. Whitening Effect of Hizikia fusiformis Ethanol Extract and Its Fractions. J. Life Sci. 2012, 22, 889–896. [Google Scholar] [CrossRef] [Green Version]


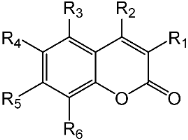
| Compound | R1 | R2 | R3 | R4 | R5 | R6 | Yield (%) |
|---|---|---|---|---|---|---|---|
| 3e | Geranyloxy | H | H | H | H | H | 87 |
| 3f | Ph | H | H | H | Geranyloxy | H | 92 |
| 3g | H | CH3 | H | H | Geranyloxy | H | 91 |
| 3h | H | CF3 | H | H | Geranyloxy | H | 90 |
| 3i | H | H | H | H | Geranyloxy | H | 94 |
| 3j | H | CH3 | H | Cl | Geranyloxy | H | 90 |
| 3k | H | CH3 | H | Geranyloxy | Geranyloxy | H | 86 |
| 3l | H | CH3 | H | Geranyloxy | H | H | 89 |

| Compound | R1 | R2 | R | R4 | R5 | R6 |
|---|---|---|---|---|---|---|
| 1e | OH | H | H | H | H | H |
| 1i | H | H | H | H | OH | H |
| 1j | H | CH3 | H | Cl | OH | H |
| 1k | H | CH3 | H | OH | OH | H |
| 1l | H | CH3 | H | OH | H | H |
| 1m | CN | H | H | H | OH | H |
| 1n | Cl | CH3 | H | H | OH | H |
| 1o | NO2 | OH | H | H | H | H |
| 1p | H | CH2COOH | H | H | OH | H |
| 1q | H | H | H | OH | H | H |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roh, E.-J. Inhibitory Effects of Coumarin Derivatives on Tyrosinase. Molecules 2021, 26, 2346. https://doi.org/10.3390/molecules26082346
Roh E-J. Inhibitory Effects of Coumarin Derivatives on Tyrosinase. Molecules. 2021; 26(8):2346. https://doi.org/10.3390/molecules26082346
Chicago/Turabian StyleRoh, Eon-Joo. 2021. "Inhibitory Effects of Coumarin Derivatives on Tyrosinase" Molecules 26, no. 8: 2346. https://doi.org/10.3390/molecules26082346





