New Cocrystals of Antipsychotic Drug Aripiprazole: Decreasing the Dissolution through Cocrystallization
Abstract
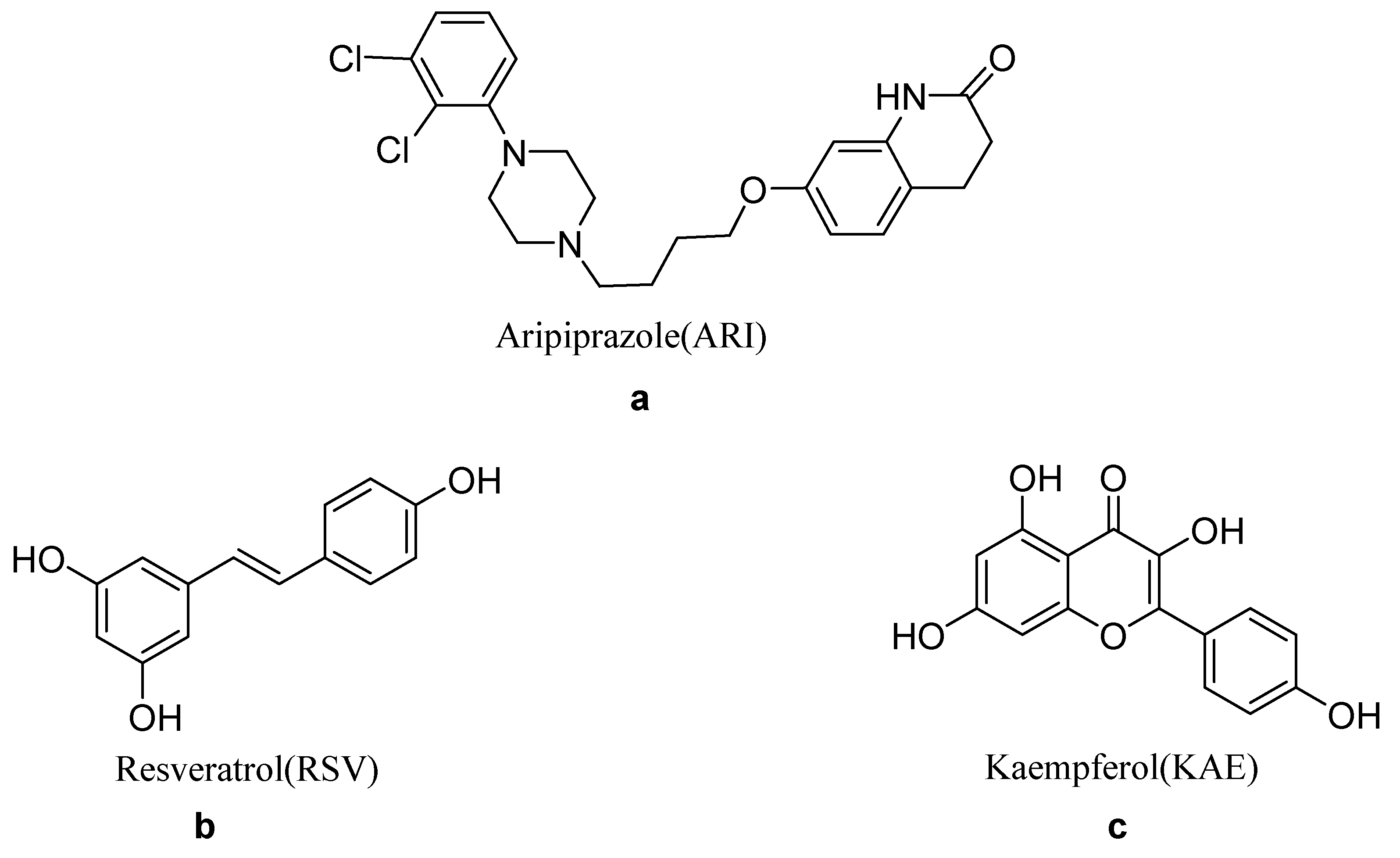
:1. Introduction
2. Results and Discussion
2.1. Single Crystal X-ray Diffraction Analysis (SCXRD)
2.2. Powder X-ray Diffraction Analysis (PXRD)
2.3. Thermal Analysis
2.4. Attenuated Total Reflection Fourier’s Transform Infrared Spectroscopy (ATR-FTIR)
2.5. Dissolution Rate
3. Materials and Methods
3.1. Materials
3.2. Preparation of Cocrystal
3.3. Characterization Method
3.3.1. Single-Crystal X-ray Diffraction
3.3.2. Powder X-ray Diffraction
3.3.3. Thermogravimetric Analysis
3.3.4. Differential Scanning Calorimetry
3.3.5. Attenuated Total Reflection Fourier’s Transform Infrared Spectroscopy
3.4. Powder Dissolution
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Kumar, A.; Kumar, S.; Nanda, A. A Review about Regulatory Status and Recent Patents of Pharmaceutical Co-Crystals. Adv. Pharm. Bull. 2018, 8, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Kuminek, G.; Cao, F.; da Rocha, A.B.D.; Cardoso, S.G.; Rodriguez-Hornedo, N. Cocrystals to facilitate delivery of poorly soluble compounds beyond-rule-of-5. Adv. Drug Deliv. Rev. 2016, 101, 143–166. [Google Scholar] [CrossRef] [Green Version]
- Thipparaboina, R.; Kumar, D.; Chavan, R.B.; Shastri, N.R. Multidrug co-crystals: Towards the development of effective therapeutic hybrids. Drug Discov. Today 2016, 21, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Almarsson, O.; Peterson, M.L.; Zaworotko, M. The A to Z of pharmaceutical cocrystals: A decade of fast-moving new science and patents. Pharm. Pat. Anal. 2012, 1, 313–327. [Google Scholar] [CrossRef]
- Aitipamula, S.; Banerjee, R.; Bansal, A.K.; Biradha, K.; Cheney, M.L.; Choudhury, A.R.; Desiraju, G.R.; Dikundwar, A.G.; Dubey, R.; Duggirala, N.; et al. Polymorphs, Salts, and Cocrystals: What’s in a Name? Cryst. Growth Des. 2012, 12, 2147–2152. [Google Scholar] [CrossRef]
- Ranjan, S.; Devarapalli, R.; Kundu, S.; Vangala, V.R.; Ghosh, A.; Reddy, C.M. Three new hydrochlorothiazide cocrystals: Structural analyses and solubility studies. J. Mol. Struct. 2017, 1133, 405–410. [Google Scholar] [CrossRef]
- Tao, Q.; Chen, J.-M.; Ma, L.; Lu, T.-B. Phenazopyridine Cocrystal and Salts That Exhibit Enhanced Solubility and Stability. Cryst. Growth Des. 2012, 12, 3144–3152. [Google Scholar] [CrossRef]
- Sugandha, K.; Kaity, S.; Mukherjee, S.; Isaac, J.; Ghosh, A. Solubility Enhancement of Ezetimibe by a Cocrystal Engineering Technique. Cryst. Growth Des. 2014, 14, 4475–4486. [Google Scholar] [CrossRef]
- Martin, F.A.; Pop, M.M.; Borodi, G.; Filip, X.; Kacso, I. Ketoconazole Salt and Co-crystals with Enhanced Aqueous Solubility. Cryst. Growth Des. 2013, 13, 4295–4304. [Google Scholar] [CrossRef]
- Zhu, B.; Zhang, Q.; Wang, J.-R.; Mei, X. Cocrystals of Baicalein with Higher Solubility and Enhanced Bioavailability. Cryst. Growth Des. 2017, 17, 1893–1901. [Google Scholar] [CrossRef]
- Chen, Y.; Li, L.; Yao, J.; Ma, Y.-Y.; Chen, J.-M.; Lu, T.-B. Improving the Solubility and Bioavailability of Apixaban via Apixaban–Oxalic Acid Cocrystal. Cryst. Growth Des. 2016, 16, 2923–2930. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, W.; Sun, W.-J.; Lu, T.; Tong, H.H.Y.; Sun, C.C.; Zheng, Y. Resveratrol cocrystals with enhanced solubility and tabletability. Int. J. Pharm. 2016, 509, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Hickey, M.B.; Peterson, M.L.; Scoppettuolo, L.A.; Morrisette, S.L.; Vetter, A.; Guzmán, H.; Remenar, J.F.; Zhang, Z.; Tawa, M.D.; Haley, S.; et al. Performance comparison of a co-crystal of carbamazepine with marketed product. Eur. J. Pharm. Biopharm. 2007, 67, 112–119. [Google Scholar] [CrossRef] [PubMed]
- McNamara, D.P.; Childs, S.L.; Giordano, J.; Iarriccio, A.; Cassidy, J.; Shet, M.S.; Mannion, R.; O’Donnell, E.; Park, A. Use of a Glutaric Acid Cocrystal to Improve Oral Bioavailability of a Low Solubility API. Pharm. Res. 2006, 23, 1888–1897. [Google Scholar] [CrossRef]
- Chen, A.M.; Ellison, M.E.; Peresypkin, A.; Wenslow, R.M.; Variankaval, N.; Savarin, C.G.; Natishan, T.K.; Mathre, D.J.; Dormer, P.G.; Euler, D.H.; et al. Development of a pharmaceutical cocrystal of a monophosphate salt with phosphoric acid. Chem. Commun. 2007, 419–421. [Google Scholar] [CrossRef]
- Variankaval, N.; Wenslow, R.; Murry, J.; Hartman, R.; Helmy, R.; Kwong, E.; Clas, S.-D.; Dalton, C.; Santos, I. Preparation and Solid-State Characterization of Nonstoichiometric Cocrystals of a Phosphodiesterase-IV Inhibitor and l-Tartaric Acid. Cryst. Growth Des. 2006, 6, 690–700. [Google Scholar] [CrossRef]
- Trask, A.V.; Motherwell, W.D.S.; Jones, W. Pharmaceutical Cocrystallization: Engineering a Remedy for Caffeine Hydration. Cryst. Growth Des. 2005, 5, 1013–1021. [Google Scholar] [CrossRef]
- Basavoju, S.; Boström, D.; Velaga, S.P. Indomethacin–Saccharin Cocrystal: Design, Synthesis and Preliminary Pharmaceutical Characterization. Pharm. Res. 2008, 25, 530–541. [Google Scholar] [CrossRef]
- Bak, A.; Gore, A.; Yanez, E.; Stanton, M.; Tufekcic, S.; Syed, R.; Akrami, A.; Rose, M.; Surapaneni, S.; Bostick, T.; et al. The Co-Crystal Approach to Improve the Exposure of a Water-Insoluble Compound: AMG 517 Sorbic Acid Co-Crystal Characterization and Pharmacokinetics. J. Pharm. Sci. 2008, 97, 3942–3956. [Google Scholar] [CrossRef]
- Geng, N.; Chen, J.-M.; Li, Z.-J.; Jiang, L.; Lu, T.-B. Approach of Cocrystallization to Improve the Solubility and Photostability of Tranilast. Cryst. Growth Des. 2013, 13, 3546–3553. [Google Scholar] [CrossRef]
- Vangala, V.R.; Chow, P.S.; Tan, R.B.H. Co-Crystals and Co-Crystal Hydrates of the Antibiotic Nitrofurantoin: Structural Studies and Physicochemical Properties. Cryst. Growth Des. 2012, 12, 5925–5938. [Google Scholar] [CrossRef]
- Krishna, G.R.; Shi, L.; Bag, P.P.; Sun, C.C.; Reddy, C.M. Correlation Among Crystal Structure, Mechanical Behavior, and Tabletability in the Co-Crystals of Vanillin Isomers. Cryst. Growth Des. 2015, 15, 1827–1832. [Google Scholar] [CrossRef]
- Bandari, S.; Dronamraju, V.; Eedara, B.B. Development and preliminary characterization of levofloxacin pharmaceutical cocrystals for dissolution rate enhancement. J. Pharm. Investig. 2017, 47, 583–591. [Google Scholar] [CrossRef]
- Stavropoulos, K.; Johnston, S.C.; Zhang, Y.; Rao, B.G.; Hurrey, M.; Hurter, P.; Topp, E.M.; Kadiyala, I. Cocrystalline Solids of Telaprevir with Enhanced Oral Absorption. J. Pharm. Sci. 2015, 104, 3343–3350. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.-S.; Kim, J.-S.; Kim, M.-S.; Alhalaweh, A.; Cho, W.; Hwang, S.-J.; Velaga, S.P. Bioavailability of indomethacin-saccharin cocrystals. J. Pharm. Pharmacol. 2010, 62, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- Sanphui, P.; Devi, V.K.; Clara, D.; Malviya, N.; Ganguly, S.; Desiraju, G.R. Cocrystals of Hydrochlorothiazide: Solubility and Diffusion/Permeability Enhancements through Drug–Coformer Interactions. Mol. Pharm. 2015, 12, 1615–1622. [Google Scholar] [CrossRef]
- Saikia, B.; Bora, P.; Khatioda, R.; Sarma, B. Hydrogen Bond Synthons in the Interplay of Solubility and Membrane Permeability/Diffusion in Variable Stoichiometry Drug Cocrystals. Cryst. Growth Des. 2015, 15, 5593–5603. [Google Scholar] [CrossRef]
- Yan, Y.; Chen, J.-M.; Lu, T.-B. Simultaneously enhancing the solubility and permeability of acyclovir by crystal engineering approach. CrystEngComm 2013, 15, 6457–6460. [Google Scholar] [CrossRef]
- López-Cedrún, J.; Videla, S.; Burgueño, M.; Juárez, I.; Aboul-Hosn, S.; Martín-Granizo, R.; Grau, J.; Puche, M.; Gil-Diez, J.-L.; Hueto, J.-A.; et al. Co-crystal of Tramadol–Celecoxib in Patients with Moderate to Severe Acute Post-surgical Oral Pain: A Dose-Finding, Randomised, Double-Blind, Placebo- and Active-Controlled, Multicentre, Phase II Trial. Drugs R&D 2018, 18, 137–148. [Google Scholar]
- Gascon, N.; Almansa, C.; Merlos, M.; Vela, J.M.; Encina, G.; Morte, A.; Smith, K.; Plata-Salaman, C. Co-crystal of tramadol-celecoxib: Preclinical and clinical evaluation of a novel analgesic. Expert Opin. Investig. Drugs 2019, 28, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Aitipamula, S.; Chow, P.S.; Tan, R.B.H. Trimorphs of a pharmaceutical cocrystal involving two active pharmaceutical ingredients: Potential relevance to combination drugs. Crystengcomm 2009, 11, 1823–1827. [Google Scholar] [CrossRef]
- Cheney, M.L.; Weyna, D.R.; Shan, N.; Hanna, M.; Wojtas, L.; Zaworotko, M.J. Coformer Selection in Pharmaceutical Cocrystal Development: A Case Study of a Meloxicam Aspirin Cocrystal That Exhibits Enhanced Solubility and Pharmacokinetics. J. Pharm. Sci. 2011, 100, 2172–2181. [Google Scholar] [CrossRef]
- Swainston Harrison, T.; Perry, C.M. Aripiprazole: A review of its use in schizophrenia and schizoaffective disorder. Drugs 2004, 64, 1715–1736. [Google Scholar] [CrossRef]
- Ardiana, F.; Lestari, M.L.; Indrayanto, G. Aripiprazole. Profiles Drug Subst. Excip. Relat. Methodol. 2013, 38, 35–85. [Google Scholar] [PubMed]
- Brittain, H.G. Aripiprazole: Polymorphs and solvatomorphs. Profiles Drug Subst. Excip. Relat. Methodol. 2012, 37, 1–29. [Google Scholar] [PubMed]
- Nanubolu, J.B.; Sridhar, B.; Babu, V.; Jagadeesh, B.; Ravikumar, K. Sixth polymorph of aripiprazole—An antipsychotic drug. Crystengcomm 2012, 14, 4677–4685. [Google Scholar] [CrossRef]
- Braun, D.E.; Gelbrich, T.; Kahlenberg, V.; Tessadri, R.; Wieser, J.; Griesser, U.J. Conformational polymorphism in aripiprazole: Preparation, stability and structure of five modifications. J. Pharm. Sci. 2009, 98, 2010–2026. [Google Scholar] [CrossRef]
- Braun, D.E.; Gelbrich, T.; Kahlenberg, V.; Tessadri, R.; Wieser, J.; Griesser, U.J. Stability of Solvates and Packing Systematics of Nine Crystal Forms of the Antipsychotic Drug Aripiprazole. Cryst. Growth Des. 2009, 9, 1054–1065. [Google Scholar] [CrossRef]
- Delaney, S.P.; Pan, D.; Yin, S.X.; Smith, T.M.; Korter, T.M. Evaluating the Roles of Conformational Strain and Cohesive Binding in Crystalline Polymorphs of Aripiprazole. Cryst. Growth Des. 2013, 13, 2943–2952. [Google Scholar] [CrossRef]
- Delaney, S.P.; Smith, T.M.; Pan, D.; Yin, S.X.; Korter, T.M. Low-Temperature Phase Transition in Crystalline Aripiprazole Leads to an Eighth Polymorph. Cryst. Growth Des. 2014, 14, 5004–5010. [Google Scholar] [CrossRef]
- Zeidan, T.A.; Trotta, J.T.; Tilak, P.A.; Oliveira, M.A.; Chiarella, R.A.; Foxman, B.M.; Almarsson, O.; Hickey, M.B. An unprecedented case of dodecamorphism: The twelfth polymorph of aripiprazole formed by seeding with its active metabolite. Crystengcomm 2016, 18, 1486–1488. [Google Scholar] [CrossRef]
- Cho, M.-Y.; Kim, P.; Kim, G.-Y.; Lee, J.-Y.; Song, K.-H.; Lee, M.-J.; Yoon, W.; Yun, H.; Choi, G.J. Preparation and Characterization of Aripiprazole Cocrystals with Coformers of Multihydroxybenzene Compounds. Cryst. Growth Des. 2017, 17, 6641–6652. [Google Scholar] [CrossRef]
- Nanubolu, J.B.; Ravikumar, K. Correlating the melting point alteration with the supramolecular structure in aripiprazole drug cocrystals. CrystEngComm 2016, 18, 1024–1038. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, B.; Jia, L.; Wang, Y.; Wang, M.; Yang, H.; Qiao, Y.; Gong, J.; Tang, W. Tuning Physicochemical Properties of Antipsychotic Drug Aripiprazole with Multicomponent Crystal Strategy Based on Structure and Property Relationship. Cryst. Growth Des. 2020, 20, 3747–3761. [Google Scholar] [CrossRef]
- Raoufinia, A.; Baker, R.A.; Eramo, A.; Nylander, A.G.; Landsberg, W.; Kostic, D.; Larsen, F. Initiation of aripiprazole once-monthly in patients with schizophrenia. Curr. Med Res. Opin. 2015, 31, 583–592. [Google Scholar] [CrossRef] [Green Version]
- Bastianetto, S.; Ménard, C.; Quirion, R. Neuroprotective action of resveratrol. Biochim. Biophys. Acta 2015, 1852, 1195–1201. [Google Scholar] [CrossRef] [Green Version]
- Magaji, M.G.; Iniaghe, L.O.; Abolarin, M.; Abdullahi, O.I.; Magaji, R.A. Neurobehavioural evaluation of resveratrol in murine models of anxiety and schizophrenia. Metab. Brain Dis. 2017, 32, 437–442. [Google Scholar] [CrossRef]
- He, H.; Zhang, Q.; Li, M.; Wang, J.-R.; Mei, X. Modulating the Dissolution and Mechanical Properties of Resveratrol by Cocrystallization. Cryst. Growth Des. 2017, 17, 3989–3996. [Google Scholar] [CrossRef]
- Ren, J.; Lu, Y.; Qian, Y.; Chen, B.; Wu, T.; Ji, G. Recent progress regarding kaempferol for the treatment of various diseases. Exp. Ther. Med. 2019, 18, 2759–2776. [Google Scholar] [CrossRef] [Green Version]
- He, H.; Huang, Y.; Zhang, Q.; Wang, J.-R.; Mei, X. Zwitterionic Cocrystals of Flavonoids and Proline: Solid-State Characterization, Pharmaceutical Properties, and Pharmacokinetic Performance. Cryst. Growth Des. 2016, 16, 2348–2356. [Google Scholar] [CrossRef]
- Yin, H.-M.; Wu, N.; Zhou, B.-J.; Hong, M.-H.; Zhu, B.; Qi, M.-H.; Ren, G.-B. Slow-Release Drug–Drug Cocrystals of Oxaliplatin with Flavonoids: Delaying Hydrolysis and Reducing Toxicity. Cryst. Growth Des. 2021, 21, 75–85. [Google Scholar] [CrossRef]
- Sheldrick, G. A Short History of ShelX. Acta Crystallogr. Sect. A Found. Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]











| Compounds | ARI2-RSV1·MeOH | ARI-KAE·EtOH | ARI-KAE·IPA |
|---|---|---|---|
| Empirical formula | C61H70Cl4N6O8 | C40H43Cl2N3O9 | C41H45Cl2N3O9 |
| Formula weight | 1157.03 | 780.67 | 794.7 |
| Temperature/K | 296.15 | 100.00 (10) | 296.15 |
| Crystal system | monoclinic | triclinic | triclinic |
| Space group | Pn | P-1 | P-1 |
| a/Å | 14.837 (4) | 11.1969 (2) | 9.219 (3) |
| b/Å | 10.201 (2) | 13.9621 (3) | 10.708 (4) |
| c/Å | 21.184 (5) | 14.0547 (3) | 20.176 (7) |
| α/° | 90 | 61.715 (2) | 94.863 (5) |
| β/° | 109.556 (3) | 86.352 (2) | 92.821 (4) |
| γ/° | 90 | 79.081 (2) | 97.687 (5) |
| Volume/Å3 | 3021.3 (12) | 1898.96 (7) | 1963.0 (11) |
| Z | 2 | 2 | 2 |
| ρcalcg/cm3 | 1.272 | 1.365 | 1.344 |
| μ/mm−1 | 0.254 | 2.039 | 0.225 |
| Radiation | MoKα (λ = 0.71073) | CuKα (λ = 1.54184) | MoKα (λ = 0.71073) |
| 2θ/° | 3.992 to 55.138 | 7.146 to 147.818 | 3.854 to 54.746 |
| Goodness-of-fit on F2 | 1.032 | 1.054 | 1.031 |
| Final R indexes [I ≥ 2σ (I)] | R1 = 0.0799, wR2 = 0.1792 | R1 = 0.0438, wR2 = 0.1233 | R1 = 0.0498, wR2 = 0.1225 |
| Final R indexes [all data] | R1 = 0.1366, wR2 = 0.2097 | R1 = 0.0451, wR2 = 0.1244 | R1 = 0.0740, wR2 = 0.1385 |
| CCDC NO. | 2072710 | 2072711 | 2072712 |
| Compounds | D-H⋯A | d(D-H)/Å | d(H⋯A)/Å | d(D⋯A)/Å | ∠D-H⋯A/° | Symmetry Code |
|---|---|---|---|---|---|---|
| ARI2-RSV1·MeOH | O5-H5A⋯N5 | 0.82 | 1.93 | 2.702(9) | 157.1 | |
| O6-H6B⋯O4 | 0.82 | 1.98 | 2.765(8) | 161.4 | −3/2 + X, 1 − Y, −1/2 + Z | |
| O7-H7⋯N | 0.82 | 1.99 | 2.660(9) | 138.9 | −1 + X, + Y, + Z | |
| O8A-H8AA⋯O7 | 0.82 | 2.07 | 2.89(2) | 176 | −1 + X, + Y, + Z | |
| N3-H3⋯O4 | 0.86 | 2.02 | 2.884(8) | 177.8 | −2 + X, + Y, −1 + Z | |
| N6-H6A⋯O2 | 0.86 | 2 | 2.863(9) | 177.2 | 2 + X, + Y,1 + Z | |
| ARI-KAE·EtOH | O2-H2A⋯O9 | 0.82 | 1.88 | 2.6801(18) | 166.6 | −1 + X, 1 + Y, −1 + Z |
| O3-H3⋯O7 | 0.82 | 1.87 | 2.6755(19) | 168 | −X, 1 − Y, 1 − Z | |
| O6-H6⋯O9 | 0.82 | 2.05 | 2.7633(16) | 145.1 | 1 − X, 1 − Y, 1 − Z | |
| O7-H7A⋯N3 | 0.82 | 2.02 | 2.813(2) | 163.8 | ||
| O8-H8⋯O4 | 0.82 | 1.91 | 2.6325(16) | 147.1 | ||
| N1-H1⋯O4 | 0.86 | 1.95 | 2.7728(18) | 160.1 | 1 − X, 1 − Y, 1 − Z | |
| ARI-KAE·IPA | N3-H3⋯O6 | 0.86 | 1.98 | 2.839(2) | 172.3 | 1 + X, +Y, +Z |
| O9-H9⋯N2 | 0.82 | 1.96 | 2.782(3) | 175.2 | 2 − X, 1 − Y, 1 − Z | |
| O3-H3A⋯O9 | 0.82 | 1.82 | 2.631(2) | 172.5 | ||
| O4-H4A⋯O6 | 0.82 | 1.9 | 2.628(2) | 147.5 | ||
| O7-H7⋯O2 | 0.82 | 1.99 | 2.691(2) | 143.5 | −1 + X, +Y, +Z | |
| O8-H8⋯O2 | 0.82 | 1.9 | 2.711(2) | 170.3 | 3 − X, 2 − Y, 2 − Z |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, W.; Ma, R.; Liang, F.; Duan, C.; Zhang, G.; Chen, Y.; Hao, C. New Cocrystals of Antipsychotic Drug Aripiprazole: Decreasing the Dissolution through Cocrystallization. Molecules 2021, 26, 2414. https://doi.org/10.3390/molecules26092414
Liu W, Ma R, Liang F, Duan C, Zhang G, Chen Y, Hao C. New Cocrystals of Antipsychotic Drug Aripiprazole: Decreasing the Dissolution through Cocrystallization. Molecules. 2021; 26(9):2414. https://doi.org/10.3390/molecules26092414
Chicago/Turabian StyleLiu, Wenwen, Ru Ma, Feifei Liang, Chenxin Duan, Guisen Zhang, Yin Chen, and Chao Hao. 2021. "New Cocrystals of Antipsychotic Drug Aripiprazole: Decreasing the Dissolution through Cocrystallization" Molecules 26, no. 9: 2414. https://doi.org/10.3390/molecules26092414







