Fabrication of Alginate-Based O/W Nanoemulsions for Transdermal Drug Delivery of Lidocaine: Influence of the Oil Phase and Surfactant
Abstract
1. Introduction
2. Results and Discussion
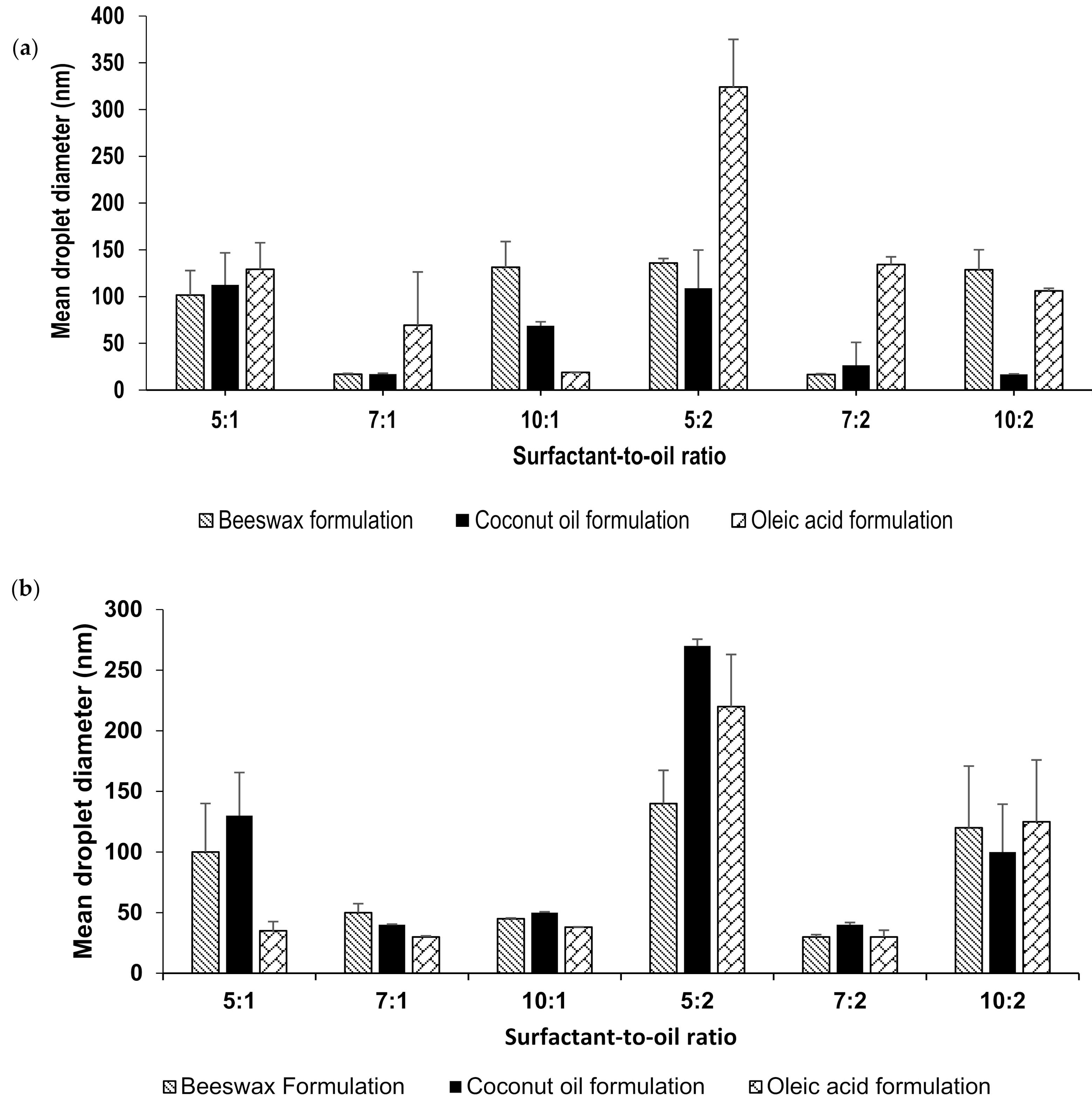
2.1. Preparation and Particle Size Measurement
2.2. Drug Content and Encapsulation Effeciency
2.3. Cryogenic Transmission Electron Microscopy
2.4. Differential Scanning Calorimetry of Nanoemulsions
2.5. Permeation Studies
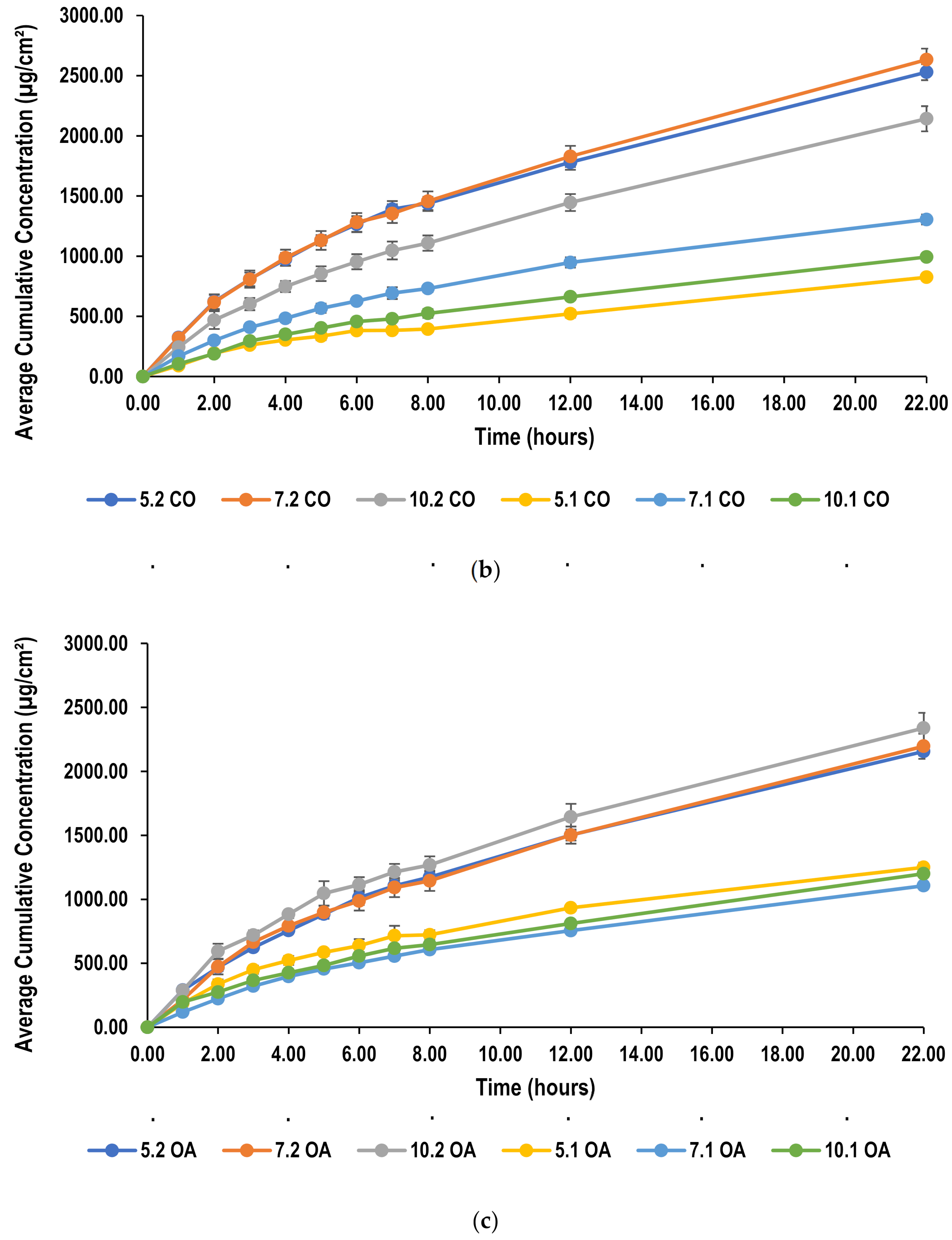
2.5.1. In Vitro Release and Permeation Studies from Polydimethylsiloxane (PDMS) Membrane
2.5.2. Effect of Oil Concentration on Permeation of Lidocaine through PDMS Membrane
2.5.3. Effect of Surfactant Concentration on Permeation of Lidocaine through PDMS Membrane
2.5.4. Effect of Oil Type on Permeation of Lidocaine through PDMS Membrane
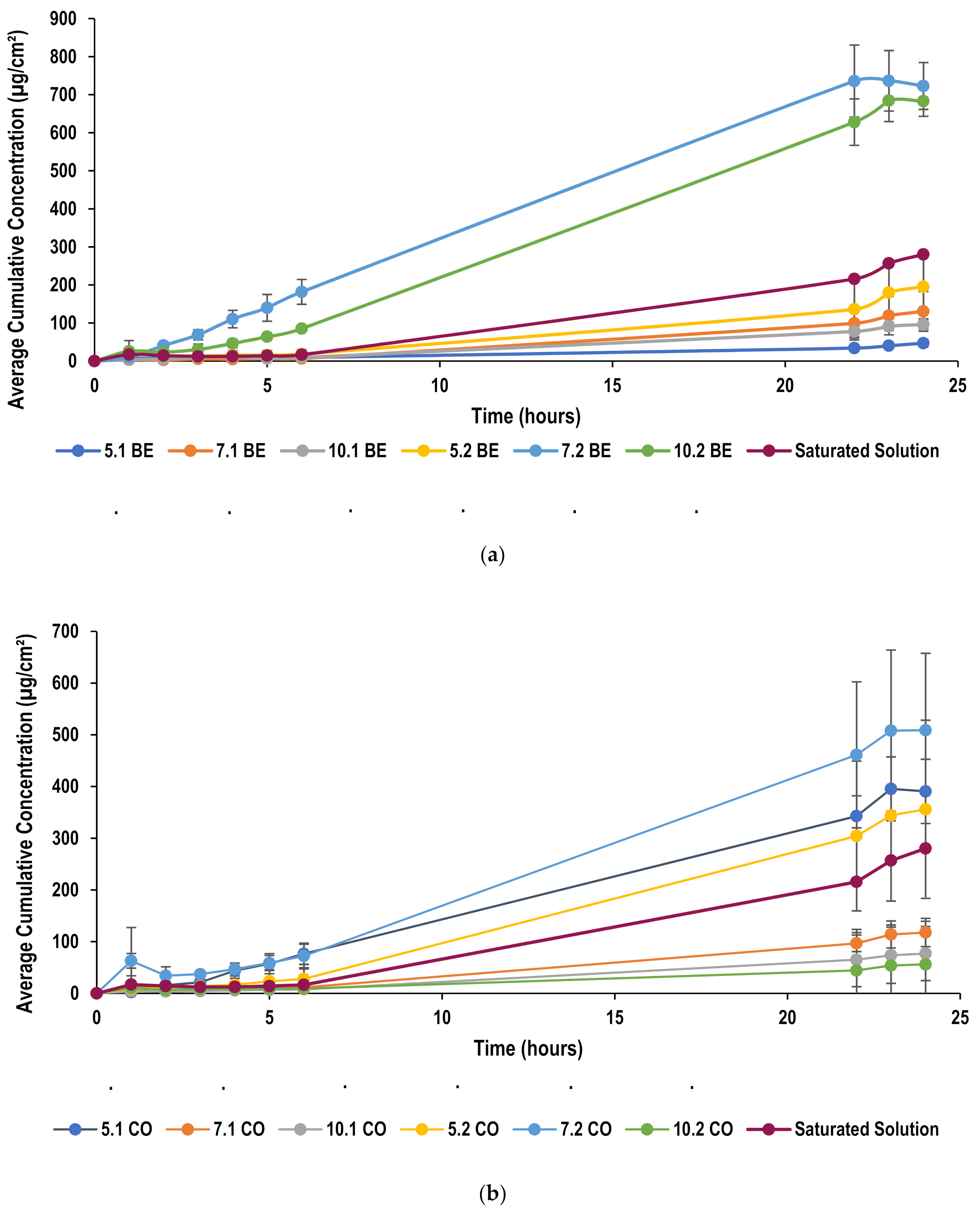
2.5.5. Skin Permeation Studies
2.5.6. Effect of Oil Concentration on Permeation through Pig Skin
2.5.7. Effect of Surfactant Concentration on Permeation through Pig Skin
2.5.8. Effect of Oil Type on Permeation through Pig Skin
2.6. Adsorption Studies
3. Materials and Methods
3.1. Materials
3.2. Preparation of Nanoemulsions
3.3. Particle Size Determination and Zeta Potential
3.4. Drug Content and Drug Entrapment Effeciency
3.5. Cryogenic Transmission Electron Microscopy (Cryo-TEM)
3.6. Lidocaine Quantification
3.7. Differential Scanning Calorimetry (DSC)
3.8. In Vitro Permeation Studies
3.8.1. Polydimethylsiloxane (PDMS) Permeation Studies
3.8.2. Pig Skin Permeation Studies
3.9. Adsorption Study
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Benson, H.A.; Watkinson, A. (Eds.) Topical and Transdermal Drug Delivery: Principles and Practice; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Ali, S.; Shabbir, M.; Shahid, N. The structure of skin and transdermal drug delivery system—A review. Res. J. Pharm. Technol. 2015, 8, 103–109. [Google Scholar] [CrossRef]
- Alkilani, A.Z.; McCrudden, M.T.C.; Donnelly, R.F. Transdermal drug delivery: Innovative pharmaceutical developments based on disruption of the barrier properties of the stratum corneum. Pharmaceutics 2015, 7, 438–470. [Google Scholar] [CrossRef] [PubMed]
- Mahato, R. Microneedles in Drug Delivery. In Emerging Nanotechnologies for Diagnostics, Drug Delivery and Medical Devices; Elsevier: Amsterdam, The Netherlands, 2017; pp. 331–353. [Google Scholar]
- Uchechi, O.; Ogbonna, J.D.N.; Attama, A.A. Nanoparticles for Dermal and Transdermal Drug Delivery. In Application of Nanotechnology in Drug Delivery; Intech: London, UK, 2014. [Google Scholar]
- Sharma, N.; Agarwal, G.; Rana, A.C.; Bhat, Z.A.; Kumar, D. A Review: Transdermal Drug Delivery System: A Tool For Novel Drug Delivery System. Int. J. Drug Dev. Res. 2011, 3, 70–84. [Google Scholar]
- Kathe, K.; Kathpalia, H. Film forming systems for topical and transdermal drug delivery. Asian J. Pharm. Sci. 2017, 12, 487–497. [Google Scholar] [CrossRef]
- Roohnikan, M.; Laszlo, E.; Babity, S.; Brambilla, D. A snapshot of transdermal and topical drug delivery research in Canada. Pharmaceutics 2019, 11, 256. [Google Scholar] [CrossRef] [PubMed]
- Prausnitz, M.R.; Langer, R. Transdermal drug delivery. Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Sonneville-Aubrun, O.; Simonnet, J.-T.; L’Alloret, F. Nanoemulsions: A new vehicle for skincare products. Adv. Colloid Interface Sci. 2004, 108–109, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Chime, S.A.; Kenechukwu, F.C.; Attama, A.A. Nanoemulsions—Advances in Formulation, Characterization and Applications in Drug Delivery. In Application of Nanotechnology in Drug Delivery; Intech: London, UK, 2014. [Google Scholar]
- Tadros, T.; Izquierdo, P.; Esquena, J.; Solans, C. Formation and stability of nano-emulsions. Adv. Colloid Interface Sci. 2004, 108–109, 303–318. [Google Scholar] [CrossRef]
- Kinyanjui, T.; Artz, W.E.; Mahungu, S. Emulsifiers|Organic Emulsifiers. In Encyclopedia of Food Sciences and Nutrition; Elsevier: Amsterdam, The Netherlands, 2003; pp. 2070–2077. [Google Scholar]
- Butterworth, J.F.; Strichartz, G.R. Molecular Mechanisms of Local Anesthesia. Anesthesiology 1990, 72, 711–734. [Google Scholar] [CrossRef]
- Taylor, A.; McLeod, G. Basic pharmacology of local anaesthetics. BJA Educ. 2020, 20, 34–41. [Google Scholar] [CrossRef]
- Weinberg, L. Pharmacokinetics and pharmacodynamics of lignocaine: A review. World J. Anesthesiol. 2015, 4, 17. [Google Scholar] [CrossRef]
- Stoicea, N.; Fiorda-Diaz, J.; Joseph, N.; Shabsigh, M.; Arias-Morales, C.; Gonzalez-Zacarias, A.A.; Mavarez-Martinez, A.; Marjoribanks, S.; Bergese, S.D. Advanced Analgesic Drug Delivery and Nanobiotechnology. Drugs 2017, 77, 1069–1076. [Google Scholar] [CrossRef][Green Version]
- Bhatia, G.; Banga, A.K. Effect of Modulated Alternating and Direct Current Iontophoresis on Transdermal Delivery of Lidocaine Hydrochloride. Biomed. Res. Int. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Wang, Y.; Su, W.; Li, Q.; Li, C.; Wang, H.; Li, Y.; Cao, Y.; Chang, J.; Zhang, L. Preparation and evaluation of lidocaine hydrochloride-loaded TAT-conjugated polymeric liposomes for transdermal delivery. Int. J. Pharm. 2013, 441, 748–756. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, S.; Liao, Z.; Wang, C.; Liu, Y.; Feng, S.; Jiang, X.; Chang, J. PEGlated magnetic polymeric liposome anchored with TAT for delivery of drugs across the blood-spinal cord barrier. Biomaterials 2010, 31, 6589–6596. [Google Scholar] [CrossRef]
- Yuan, J.S.; Ansari, M.; Samaan, M.; Acosta, E.J. Linker-based lecithin microemulsions for transdermal delivery of lidocaine. Int. J. Pharm. 2008, 349, 130–143. [Google Scholar] [CrossRef]
- Yuan, J.S.; Acosta, E.J. Extended release of lidocaine from linker-based lecithin microemulsions. Int. J. Pharm. 2009, 368, 63–71. [Google Scholar] [CrossRef]
- Yuan, J.S.; Yip, A.; Nguyen, N.; Chu, J.; Wen, X.Y.; Acosta, E.J. Effect of surfactant concentration on transdermal lidocaine delivery with linker microemulsions. Int. J. Pharm. 2010, 392, 274–284. [Google Scholar] [CrossRef]
- Zhu, X.-L.; Li, G.-F.; Zeng, K.; Cheng, Z.-L. Preparation of lidocaine nanoemulsion and its transdermal absorption by rat skin ex vivo. Nan Fang Yi Ke Da Xue Xue Bao 2010, 30, 451–454. [Google Scholar]
- Negi, P.; Singh, B.; Sharma, G.; Beg, S.; Katare, O.P. Biocompatible lidocaine and prilocaine loaded-nanoemulsion system for enhanced percutaneous absorption: QbD-based optimisation, dermatokinetics and in vivo evaluation. J. Microencapsul. 2015, 32, 419–431. [Google Scholar] [CrossRef]
- Sarheed, O.; Dibi, M.; Ramesh, K.V.R.N.S. Studies on the Effect of Oil and Surfactant on the Formation of Alginate-Based O/W Lidocaine Nanocarriers Using Nanoemulsion Template. Pharmaceutics 2020, 12, 1223. [Google Scholar] [CrossRef]
- Pierre, M.B.R.; Ricci, E.; Tedesco, A.C.; Bentley, M.V.L.B. Oleic Acid as Optimizer of the Skin Delivery of 5-Aminolevulinic Acid in Photodynamic Therapy. Pharm. Res. 2006, 23, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Hosny, K.M.; Alhakamy, N.A.; Sindi, A.M.; Khallaf, R.A. Coconut Oil Nanoemulsion Loaded with a Statin Hypolipidemic Drug for Management of Burns: Formulation and In Vivo Evaluation. Pharmaceutics 2020, 12, 1061. [Google Scholar] [CrossRef] [PubMed]
- Attama, A.; Schicke, B.; Mullergoymann, C. Further characterization of theobroma oil–beeswax admixtures as lipid matrices for improved drug delivery systems. Eur. J. Pharm. Biopharm. 2006, 64, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Osborne, D.W.; Ward, A.J.I.; O’Neill, K.J. Microemulsions as topical drug delivery vehicles: In-vitro transdermal studies of a model hydrophilic drug. J. Pharm. Pharmacol. 1991, 43, 451–454. [Google Scholar] [CrossRef]
- Saberi, A.H.; Fang, Y.; McClements, D.J. Fabrication of vitamin E-enriched nanoemulsions: Factors affecting particle size using spontaneous emulsification. J. Colloid Interface Sci. 2013, 391, 95–102. [Google Scholar] [CrossRef] [PubMed]
- McClements, D.J. Food Emulsions: Principles, Practices, and Techniques, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2015; ISBN 9780429154034. [Google Scholar]
- An, Y.; Yan, X.; Li, B.; Li, Y. Microencapsulation of capsanthin by self-emulsifying nanoemulsions and stability evaluation. Eur. Food Res. Technol. 2014, 239, 1077–1085. [Google Scholar] [CrossRef]
- Shukla, A.; Krause, A.; Neubert, R.H.H. Microemulsions as colloidal vehicle systems for dermal drug delivery. Part IV: Investigation of microemulsion systems based on a eutectic mixture of lidocaine and prilocaine as the colloidal phase by dynamic light scattering. J. Pharm. Pharmacol. 2003, 55, 741–748. [Google Scholar] [CrossRef]
- Shukla, A.; Janich, M.; Jahn, K.; Krause, A.; Kiselev, M.A.; Neubert, R.H.H. Investigation of pharmaceutical oil/water microemulsions by small-angle scattering. Pharm. Res. 2002, 19, 881–886. [Google Scholar] [CrossRef]
- Leung, R.; Shah, D.O. Solubilization and phase equilibria of water-in-oil microemulsions. J. Colloid Interface Sci. 1987, 120, 330–344. [Google Scholar] [CrossRef]
- Chuesiang, P.; Siripatrawan, U.; Sanguandeekul, R.; McLandsborough, L.; McClements, D.J. Optimization of cinnamon oil nanoemulsions using phase inversion temperature method: Impact of oil phase composition and surfactant concentration. J. Colloid Interface Sci. 2018, 514, 208–216. [Google Scholar] [CrossRef]
- Mohammadi, M.; Assadpour, E.; Jafari, S.M. Encapsulation of food ingredients by nanostructured lipid carriers (NLCs). In Lipid-Based Nanostructures for Food Encapsulation Purposes; Elsevier: Amsterdam, The Netherlands, 2019; pp. 217–270. [Google Scholar]
- Rizza, M.A.; Wijayanti, W.; Hamidi, N.; Wardana, I.N.G. Role of Intermolecular Forces on the Contact Angle of Vegetable Oil Droplets during the Cooling Process. Sci. World J. 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Sarheed, O.; Shouqair, D.; Ramesh, K.V.R.N.S.; Khaleel, T.; Amin, M.; Boateng, J.; Drechsler, M. Formation of stable nanoemulsions by ultrasound-assisted two-step emulsification process for topical drug delivery: Effect of oil phase composition and surfactant concentration and loratadine as ripening inhibitor. Int. J. Pharm. 2020, 576, 118952. [Google Scholar] [CrossRef]
- Sarheed, O.; Shouqair, D.; Ramesh, K.; Amin, M.; Boateng, J.; Drechsler, M. Physicochemical characteristics and in vitro permeation of loratadine solid lipid nanoparticles for transdermal delivery. Ther. Deliv. 2020, 11, 685–700. [Google Scholar] [CrossRef]
- Machado, A.H.E.; Lundberg, D.; Ribeiro, A.J.; Veiga, F.J.; Lindman, B.; Miguel, M.G.; Olsson, U. Preparation of Calcium Alginate Nanoparticles Using Water-in-Oil (W/O) Nanoemulsions. Langmuir 2012, 28, 4131–4141. [Google Scholar] [CrossRef]
- McClements, D.J. Nanoemulsions versus microemulsions: Terminology, differences, and similarities. Soft Matter 2012, 8, 1719–1729. [Google Scholar] [CrossRef]
- Deutch-Kolevzon, R.; Aserin, A.; Garti, N. Synergistic cosolubilization of omega-3 fatty acid esters and CoQ10 in dilutable microemulsions. Chem. Phys. Lipid. 2011, 164, 654–663. [Google Scholar] [CrossRef]
- Fisher, S.; Wachtel, E.J.; Aserin, A.; Garti, N. Solubilization of simvastatin and phytosterols in a dilutable microemulsion system. Coll. Surf. B Biointerface 2013, 107, 35–42. [Google Scholar] [CrossRef]
- Lidich, N.; Wachtel, E.J.; Aserin, A.; Garti, N. Water-dilutable microemulsions for transepithelial ocular delivery of riboflavin phosphate. J. Colloid Interface Sci. 2016, 463, 342–348. [Google Scholar] [CrossRef]
- Dalmazzone, C.; Noïk, C.; Clausse, D. Application of DSC for Emulsified System Characterization. Oil Gas Sci. Technol. Rev. IFP 2009, 64, 543–555. [Google Scholar] [CrossRef]
- Montenegro, L.; Campisi, A.; Sarpietro, M.G.; Carbone, C.; Acquaviva, R.; Raciti, G.; Puglisi, G. In vitro evaluation of idebenone-loaded solid lipid nanoparticles for drug delivery to the brain. Drug Dev. Ind. Pharm. 2011, 37, 737–746. [Google Scholar] [CrossRef]
- Spernath, A.; Aserin, A.; Garti, N. Phase transition induced by water dilution in phospholipid U-type food-grade microemulsions studied by DSC. J. Therm. Anal. Calorim. 2006, 83, 297–308. [Google Scholar] [CrossRef]
- Lidich, N.; Aserin, A.; Garti, N. Structural characteristics of oil-poor dilutable fish oil omega-3 microemulsions for ophthalmic applications. J. Colloid Interface Sci. 2016, 463, 83–92. [Google Scholar] [CrossRef]
- Mishra, V.; Bansal, K.; Verma, A.; Yadav, N.; Thakur, S.; Sudhakar, K.; Rosenholm, J. Solid Lipid Nanoparticles: Emerging Colloidal Nano Drug Delivery Systems. Pharmaceutics 2018, 10, 191. [Google Scholar] [CrossRef]
- Bhuiyan, A.K.M.M.H.; Waters, L.J. Permeation of pharmaceutical compounds through silicone membrane in the presence of surfactants. Coll. Surf. Physicochem. Eng. Asp. 2017, 516, 121–128. [Google Scholar] [CrossRef][Green Version]
- Waters, L.J.J.; Bhuiyan, A.K.M.M.H. Ionisation effects on the permeation of pharmaceutical compounds through silicone membrane. Coll. Surf. B Biointerface 2016, 141, 553–557. [Google Scholar] [CrossRef]
- Shahzad, Y.; Waters, L.J.; Barber, C. Solvent selection effects on the transport of compounds through silicone membrane. Coll. Surf. Physicochem. Eng. Asp. 2014, 458, 96–100. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Wang, X.; Song, Y.; Wang, X.; Hao, J. Design and Development of Lidocaine Microemulsions for Transdermal Delivery. AAPS PharmSciTech 2019, 20, 1–9. [Google Scholar] [CrossRef]
- Zhang, J.; Michniak-Kohn, B. Investigation of microemulsion microstructures and their relationship to transdermal permeation of model drugs: Ketoprofen, lidocaine, and caffeine. Int. J. Pharm. 2011, 421, 34–44. [Google Scholar] [CrossRef]
- Solans, C.; Izquierdo, P.; Nolla, J.; Azemar, N.; Garciacelma, M. Nano-emulsions. Curr. Opin. Colloid Interface Sci. 2005, 10, 102–110. [Google Scholar] [CrossRef]
- Waters, L.J.; Finch, C.V.; Bhuiyan, A.K.M.M.H.; Hemming, K.; Mitchell, J.C. Effect of plasma surface treatment of poly(dimethylsiloxane) on the permeation of pharmaceutical compounds. J. Pharm. Anal. 2017, 7, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Santi, P.; Catellani, P.L.; Colombo, P.; Ringard-Lefebvre, C.; Barthélémy, C.; Guyot-Hermann, A.-M. Partition and transport of verapamil and nicotine through artificial membranes. Int. J. Pharm. 1991, 68, 43–49. [Google Scholar] [CrossRef]
- Todo, H. Transdermal permeation of drugs in various animal species. Pharmaceutics 2017, 9, 33. [Google Scholar] [CrossRef]
- Barbero, A.M.; Frasch, H.F. Pig and guinea pig skin as surrogates for human in vitro penetration studies: A quantitative review. Toxicol. In Vitro 2009, 23, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Barber, E. A comparative study of the rates of in vitro percutaneous absorption of eight chemicals using rat and human skin*1. Fundam. Appl. Toxicol. 1992, 19, 493–497. [Google Scholar] [CrossRef]
- Gray, G.M.; Yardley, H.J. Lipid compositions of cells isolated from pig, human, and rat epidermis. J. Lipid Res. 1975, 16, 434–440. [Google Scholar] [CrossRef]
- Kim, M.-J.; Doh, H.-J.; Choi, M.-K.; Chung, S.-J.; Shim, C.-K.; Kim, D.-D.; Kim, J.S.; Yong, C.-S.; Choi, H.-G. Skin Permeation Enhancement of Diclofenac by Fatty Acids. Drug Deliv. 2008, 15, 373–379. [Google Scholar] [CrossRef]
- Bhuiyan, A. A Comparative Study of Chemical Based Skin Mimics with Pharmaceutical Applications. Ph.D. Thesis, University of Huddersfield, Huddersfield, UK, 2016. [Google Scholar]
- Esposito, E.; Drechsler, M.; Cortesi, R. Microscopy characterisation of micro- and nanosystems for pharmaceutical use. OA Drug Des. Deliv. 2013, 1. [Google Scholar] [CrossRef]
- Lee, P.J.; Ahmad, N.; Langer, R.; Mitragotri, S.; Prasad Shastri, V. Evaluation of chemical enhancers in the transdermal delivery of lidocaine. Int. J. Pharm. 2006, 308, 33–39. [Google Scholar] [CrossRef]
- Waters, L.J.; Jesney, H.; Molinari, M.; Shahzad, Y. Quantification of the adsorption of benzoates on poly(dimethylsiloxane) membrane. Eur. Polym. J. 2019, 118, 286–289. [Google Scholar] [CrossRef]











| SOR | Lipid Type | Flux (J) µg/h/cm2 | Permeability Coefficient cm/h |
|---|---|---|---|
| 5:1 | BE | 57.08 | 0.57 |
| CO | 55.14 | 0.27 | |
| OA | 55.70 | 0.56 | |
| 7:1 | BE | 69.47 | 0.69 |
| CO | 57.14 | 0.57 | |
| OA | 46.91 | 0.47 | |
| 10:1 | BE | 34.51 | 0.35 |
| CO | 42.53 | 0.38 | |
| OA | 48.93 | 0.49 | |
| 5:2 | BE | 132.16 | 1.32 |
| CO | 129.38 | 1.29 | |
| OA | 112.18 | 1.12 | |
| 7:2 | BE | 122.66 | 1.23 |
| CO | 128.55 | 1.29 | |
| OA | 96.56 | 0.97 | |
| 10:2 | BE | 113.94 | 1.14 |
| CO | 120.44 | 1.01 | |
| OA | 108.52 | 1.09 |
| SOR | Lipid Type | Flux (J) µg/h/cm2 | Permeability Coefficient cm/h |
|---|---|---|---|
| 5:1 | BE | 6.59 | 0.06 |
| CO | 17.59 | 0.18 | |
| OA | 12.05 | 0.12 | |
| 7:1 | BE | 15.67 | 0.16 |
| CO | 5.45 | 0.05 | |
| OA | 2.42 | 0.02 | |
| 10:1 | BE | 6.32 | 0.06 |
| CO | 5.33 | 0.05 | |
| OA | 8.35 | 0.08 | |
| 5:2 | BE | 29.71 | 0.30 |
| CO | 7.83 | 0.08 | |
| OA | 5.09 | 0.05 | |
| 7:2 | BE | 34.19 | 0.34 |
| CO | 16.12 | 0.16 | |
| OA | 17.03 | 0.17 | |
| 10:2 | BE | 17.34 | 0.17 |
| CO | 7.20 | 0.07 | |
| OA | 4.30 | 0.04 |
| Surfactant-to-Oil Ratio | Surfactant Amount (g) | Oil Phase Amount (g) |
|---|---|---|
| 5:1 | 0.75 | 0.15 |
| 7:1 | 1.05 | 0.15 |
| 10:1 | 1.50 | 0.15 |
| 5:2 | 0.75 | 0.30 |
| 7:2 | 1.05 | 0.30 |
| 10:2 | 1.50 | 0.30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarheed, O.; Dibi, M.; Ramesh, K.V.R.N.S.; Drechsler, M. Fabrication of Alginate-Based O/W Nanoemulsions for Transdermal Drug Delivery of Lidocaine: Influence of the Oil Phase and Surfactant. Molecules 2021, 26, 2556. https://doi.org/10.3390/molecules26092556
Sarheed O, Dibi M, Ramesh KVRNS, Drechsler M. Fabrication of Alginate-Based O/W Nanoemulsions for Transdermal Drug Delivery of Lidocaine: Influence of the Oil Phase and Surfactant. Molecules. 2021; 26(9):2556. https://doi.org/10.3390/molecules26092556
Chicago/Turabian StyleSarheed, Omar, Manar Dibi, Kanteti V. R. N. S. Ramesh, and Markus Drechsler. 2021. "Fabrication of Alginate-Based O/W Nanoemulsions for Transdermal Drug Delivery of Lidocaine: Influence of the Oil Phase and Surfactant" Molecules 26, no. 9: 2556. https://doi.org/10.3390/molecules26092556
APA StyleSarheed, O., Dibi, M., Ramesh, K. V. R. N. S., & Drechsler, M. (2021). Fabrication of Alginate-Based O/W Nanoemulsions for Transdermal Drug Delivery of Lidocaine: Influence of the Oil Phase and Surfactant. Molecules, 26(9), 2556. https://doi.org/10.3390/molecules26092556






