Neuroprotective and Antioxidant Enhancing Properties of Selective Equisetum Extracts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Obtaining Extracts
2.2. Semi-Quantitative and Qualitative Determination of the Polyphenolic Compounds (UHPLC-DAD)
2.3. In Vitro Evaluation of the Antioxidant Activity of the Plant Extracts Obtained from the Three Species of the Equisetum Genus
2.3.1. The Chelating Capacity of the Ferrous Ion Determination
2.3.2. Determination of the Lipoxygenase Inhibition Capacity
2.3.3. Determination of the Scavenger Action of the Hydroxyl Radical
2.3.4. Determination of the Scavenger Capacity of the Superoxide Anion
2.4. In Vivo Evaluation of the Neuroprotective Activity of the Plant Extracts Obtained from the Three Species of Equisetum
2.4.1. Novel Tank Diving Test (NTT)
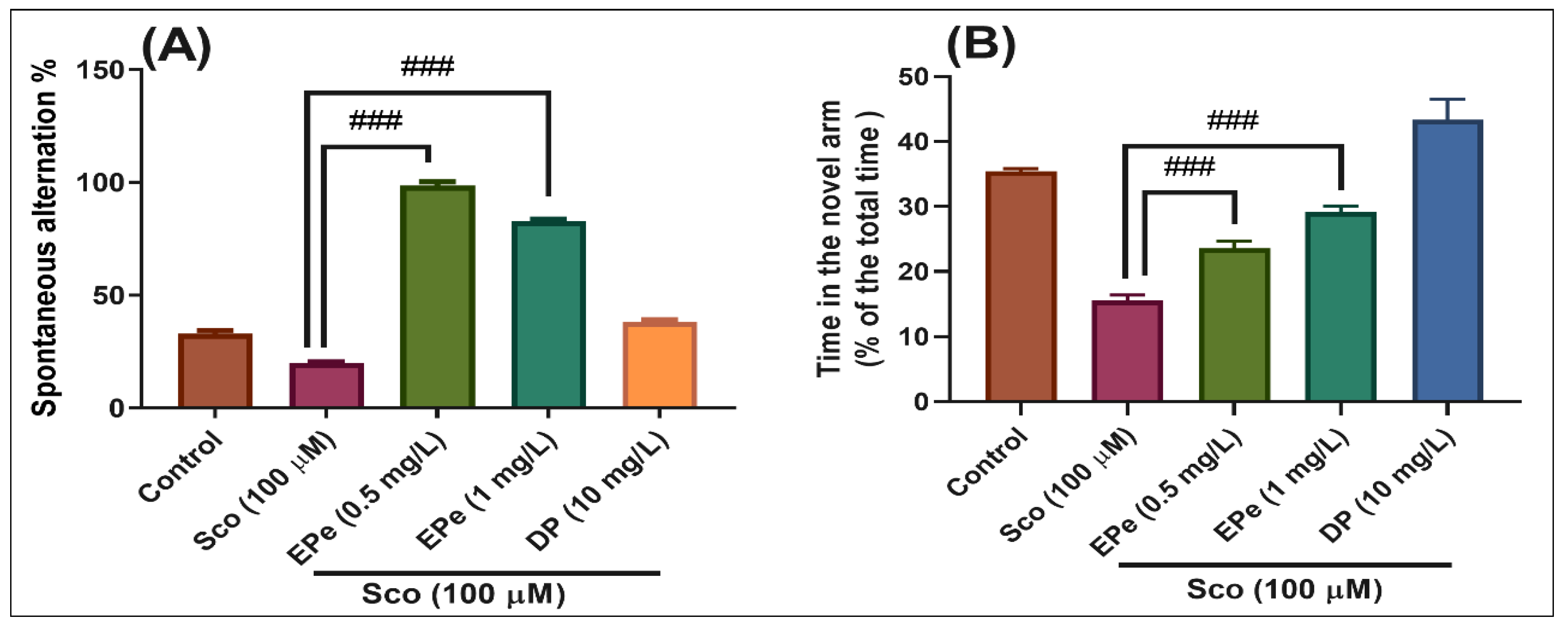
2.4.2. Y-Maze Test
3. Materials and Methods
3.1. Plant Materials
3.2. UHPLC Analysis of Phenolic Acids and Flavonoids
3.3. Antioxidant Capacity Assay
3.4. Animals and Group Division
3.5. Behavioral Assays
3.5.1. Novel Tank Diving Test (NTT)
3.5.2. Y-Maze Test
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Feoktistov, D.; Gureeva, I. Anatomical features of cross-sections of the genus Equisetum members. Ukr. J. Ecol. 2018, 8, 210–216. [Google Scholar] [CrossRef]
- Elgorriaga, A.; Escapa, I.H.; Rothwell, G.W.; Tomescu, A.M.F.; Rubén Cúneo, N. Origin of Equisetum: Evolution of horsetails (Equisetales) within the major euphyllophyte clade Sphenopsida. Am. J. Bot. 2018, 105, 1286–1303. [Google Scholar] [CrossRef]
- Husby, C. Biology and Functional Ecology of Equisetum with Emphasis on the Giant Horsetails. Bot. Rev. 2013, 79, 147–177. [Google Scholar] [CrossRef]
- Augustyniak, A.; Bartosz, G.; Čipak, A.; Duburs, G.; Horáková, L.; Łuczaj, W.; Majekova, M.; Odysseos, A.D.; Rackova, L.; Skrzydlewska, E.; et al. Natural and synthetic antioxidants: An updated overview. Free Radic. Res. 2010, 44, 1216–1262. [Google Scholar] [CrossRef]
- Pallag, A.; Filip, G.A.; Olteanu, D.; Clichici, S.; Baldea, I.; Jurca, T.; Micle, O.; Vicaş, L.; Marian, E.; Soriţău, O.; et al. Equisetum arvense L. Extract Induces Antibacterial Activity and Modulates Oxidative Stress, Inflammation, and Apoptosis in Endothelial Vascular Cells Exposed to Hyperosmotic Stress. Oxid. Med. Cell. Longev. 2018, 2018, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Snafi, A.E. The pharmacology of Equisetum arvense- A review. IOSR J. Pharm. 2017, 07, 31–42. [Google Scholar] [CrossRef]
- Mimica-Dukic, N.; Simin, N.; Cvejic, J.; Jovin, E.; Orcic, D.; Bozin, B. Phenolic Compounds in Field Horsetail (Equisetum arvense L.) as Natural Antioxidants. Molecules 2008, 13, 1455–1464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masella, R.; Di Benedetto, R.; Varì, R.; Filesi, C.; Giovannini, C. Novel mechanisms of natural antioxidant compounds in biological systems: Involvement of glutathione and glutathione-related enzymes. J. Nutr. Biochem. 2005, 16, 577–586. [Google Scholar] [CrossRef]
- Calliste, C.A.; Trouillas, P.; Allais, D.P.; Simon, A.; Duroux, J.L. Free radical scavenging activities measured by electron spin resonance spectroscopy and B16 cell antiproliferative behaviors of seven plants. J. Agric. Food Chem. 2001, 49, 3321–3327. [Google Scholar] [CrossRef]
- Štajner, D.; Popović, B.M.; Čanadanović-Brunet, J.; Boža, P. Free radical scavenging activity of three Equisetum species from Fruška gora mountain. Fitoterapia 2006, 77, 601–604. [Google Scholar] [CrossRef]
- Burton, G.J.; Jauniaux, E. Oxidative stress. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 287–299. [Google Scholar] [CrossRef] [Green Version]
- Dröge, W. Free Radicals in the Physiological Control of Cell Function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef] [PubMed]
- Milovanović, V.; Radulović, N.; Todorović, Z.; Stanković, M.; Stojanović, G. Antioxidant, antimicrobial and genotoxicity screening of hydro-alcoholic extracts of five Serbian Equisetum species. Plant Foods Hum. Nutr. 2007, 62, 113–119. [Google Scholar] [CrossRef]
- Glasauer, A.; Chandel, N.S. ROS. Curr. Biol. 2013, 23, R100–R102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lipinski, B. Hydroxyl Radical and Its Scavengers in Health and Disease. Oxid. Med. Cell. Longev. 2011, 2011, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2015; ISBN 9780198717478. [Google Scholar]
- Čanadanović-Brunet, J.M.; Ćetković, G.S.; Djilas, S.M.; Tumbas, V.T.; Savatović, S.S.; Mandić, A.I.; Markov, S.L.; Cvetković, D.D. Radical scavenging and antimicrobial activity of horsetail (Equisetum arvense L.) extracts. Int. J. Food Sci. Technol. 2009, 44, 269–278. [Google Scholar] [CrossRef]
- Neha, K.; Haider, M.R.; Pathak, A.; Yar, M.S. Medicinal prospects of antioxidants: A review. Eur. J. Med. Chem. 2019, 178, 687–704. [Google Scholar] [CrossRef] [PubMed]
- Elufioye, T.O.; Berida, T.I.; Habtemariam, S. Plants-Derived Neuroprotective Agents: Cutting the Cycle of Cell Death through Multiple Mechanisms. Evidence-Based Complement. Altern. Med. 2017, 2017, 1–27. [Google Scholar] [CrossRef] [Green Version]
- Hritcu, L.; Foyet, H.S.; Stefan, M.; Mihasan, M.; Asongalem, A.E.; Kamtchouing, P. Neuroprotective effect of the methanolic extract of Hibiscus asper leaves in 6-hydroxydopamine-lesioned rat model of Parkinson’s disease. J. Ethnopharmacol. 2011, 137, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Kim, S.-K. Neuroprotective Properties of Chitosan and Its Derivatives. Mar. Drugs 2010, 8, 2117–2128. [Google Scholar] [CrossRef]
- Venditti, E.; Bacchetti, T.; Tiano, L.; Carloni, P.; Greci, L.; Damiani, E. Hot vs. cold water steeping of different teas: Do they affect antioxidant activity? Food Chem. 2010, 119, 1597–1604. [Google Scholar] [CrossRef]
- Eid, R.; Arab, N.T.T.; Greenwood, M.T. Iron mediated toxicity and programmed cell death: A review and a re-examination of existing paradigms. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 399–430. [Google Scholar] [CrossRef]
- Rackova, L.; Oblozinsky, M.; Kostalova, D.; Kettmann, V.; Bezakova, L. Free radical scavenging activity and lipoxygenase inhibition of Mahonia aquifolium extract and isoquinoline alkaloids. J. Inflamm. 2007, 4, 15. [Google Scholar] [CrossRef] [Green Version]
- Kuhn, H.; Banthiya, S.; van Leyen, K. Mammalian lipoxygenases and their biological relevance. Biochim. Biophys. Acta - Mol. Cell Biol. Lipids 2015, 1851, 308–330. [Google Scholar] [CrossRef] [Green Version]
- Alam, M.N.; Bristi, N.J.; Rafiquzzaman, M. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm. J. 2013, 21, 143–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, H.; Kim, D.H.; Cho, J.H.; Kim, Y.C. Hepatoprotective and free radical scavenging activities of phenolic petrosins and flavonoids isolated from Equisetum arvense. J. Ethnopharmacol. 2004, 95, 421–424. [Google Scholar] [CrossRef]
- Bencan, Z.; Sledge, D.; Levin, E.D. Buspirone, chlordiazepoxide and diazepam effects in a zebrafish model of anxiety. Pharmacol. Biochem. Behav. 2009, 94, 75–80. [Google Scholar] [CrossRef] [Green Version]
- Vieira, R.; Venâncio, C.; Félix, L. Teratogenic, Oxidative Stress and Behavioural Outcomes of Three Fungicides of Natural Origin (Equisetum arvense, Mimosa tenuiflora, Thymol) on Zebrafish (Danio rerio). Toxics 2021, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Kaur, S.; Bedi, P.M.S.; Kaur, D. Anxiolytic effects of Equisetum arvense Linn. extracts in mice. Indian J. Exp. Biol. 2011, 49, 352–356. [Google Scholar]
- Sarris, J.; McIntyre, E.; Camfield, D.A. Plant-based medicines for anxiety disorders, part 1: A review of preclinical studies. CNS Drugs 2013, 27, 207–219. [Google Scholar] [CrossRef]
- Navarrete-Yañez, V.; Garate-Carrillo, A.; Rodriguez, A.; Mendoza-Lorenzo, P.; Ceballos, G.; Calzada-Mendoza, C.; Hogan, M.C.; Villarreal, F.; Ramirez-Sanchez, I. Effects of (-)-epicatechin on neuroinflammation and hyperphosphorylation of tau in the hippocampus of aged mice. Food Funct. 2020, 11, 10351–10361. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Oh, J.H.; Yoo, H.S.; Lee, Y.M.; Lee, M.K.; Hong, J.T.; Oh, K.W. (-)-Epigallocatechin-3-O-gallate (EGCG) reverses caffeine-induced anxiogenic-like effects. Neurosci. Lett. 2010, 481, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Vignes, M.; Maurice, T.; Lanté, F.; Nedjar, M.; Thethi, K.; Guiramand, J.; Récasens, M. Anxiolytic properties of green tea polyphenol (-)-epigallocatechin gallate (EGCG). Brain Res. 2006, 1110, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Gadotti, V.M.; Zamponi, G.W. Anxiolytic effects of the flavonoid luteolin in a mouse model of acute colitis. Mol. Brain 2019, 12, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crupi, R.; Paterniti, I.; Ahmad, A.; Campolo, M.; Esposito, E.; Cuzzocrea, S. Effects of Palmitoylethanolamide and Luteolin in an Animal Model of Anxiety/Depression. CNS Neurol. Disord. Drug Targets 2013, 12, 989–1001. [Google Scholar] [CrossRef]
- Ishisaka, M.; Kakefuda, K.; Yamauchi, M.; Tsuruma, K.; Shimazawa, M.; Tsuruta, A.; Hara, H. Luteolin shows an antidepressant-like effect via suppressing endoplasmic reticulum stress. Biol. Pharm. Bull. 2011, 34, 1481–1486. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.L.; Liu, M.; Cui, W.; Yang, L.; Zhang, C.N. Quercetin affects shoaling and anxiety behaviors in zebrafish: Involvement of neuroinflammation and neuron apoptosis. Fish Shellfish Immunol. 2020, 105, 359–368. [Google Scholar] [CrossRef]
- Lee, B.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.H. Protective Effects of Quercetin on Anxiety-Like Symptoms and Neuroinflammation Induced by Lipopolysaccharide in Rats. Evidence-based Complement. Altern. Med. 2020, 2020. [Google Scholar] [CrossRef]
- Kosari-Nasab, M.; Shokouhi, G.; Ghorbanihaghjo, A.; Mesgari-Abbasi, M.; Salari, A.A. Quercetin mitigates anxiety-like behavior and normalizes hypothalamus-pituitary-adrenal axis function in a mouse model of mild traumatic brain injury. Behav. Pharmacol. 2019, 30, 282–289. [Google Scholar] [CrossRef]
- Samad, N.; Saleem, A.; Yasmin, F.; Shehzad, M.A. Quercetin protects against stress-induced anxiety- and depression- like behavior and improves memory in male mice. Physiol. Res. 2018, 67, 795–808. [Google Scholar] [CrossRef]
- Salgueiro, J.B.; Ardenghi, P.; Dias, M.; Ferreira, M.B.C.; Izquierdo, I.; Medina, J.H. Anxiolytic natural and synthetic flavonoid ligands of the central benzodiazepine receptor have no effect on memory tasks in rats. Pharmacol. Biochem. Behav. 1997, 58, 887–891. [Google Scholar] [CrossRef]
- Ahmad, H.; Rauf, K.; Zada, W.; McCarthy, M.; Abbas, G.; Anwar, F.; Shah, A.J. Kaempferol facilitated extinction learning in contextual fear conditioned rats via inhibition of fatty-acid amide hydrolase. Molecules 2020, 25, 4683. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Tsuji, M.; Miyamoto, J.; Masuya, J.; Iimori, M.; Matsumiya, T. Caffeic acid produces antidepressive- and/or anxiolytic-like effects through indirect modulation of the α1A-adrenoceptor system in mice. Neuroreport 2003, 14, 1067–1070. [Google Scholar] [CrossRef]
- Lorigooini, Z.; Nouri, A.; Mottaghinia, F.; Balali-Dehkordi, S.; Bijad, E.; Dehkordi, S.H.; Soltani, A.; Amini-Khoei, H. Ferulic acid through mitigation of NMDA receptor pathway exerts anxiolytic-like effect in mouse model of maternal separation stress. J. Basic Clin. Physiol. Pharmacol. 2021, 32. [Google Scholar] [CrossRef]
- Nonnis, S.; Angiulli, E.; Maffioli, E.; Frabetti, F.; Negri, A.; Cioni, C.; Alleva, E.; Romeo, V.; Tedeschi, G.; Toni, M. Acute environmental temperature variation affects brain protein expression, anxiety and explorative behaviour in adult zebrafish. Sci. Rep. 2021, 11, 2521. [Google Scholar] [CrossRef]
- Dos Santos Junior, J.G.; Do Monte, F.H.M.; Blanco, M.M.; Do Nascimento Bispo Lanziotti, V.M.; Maia, F.D.; De Almeida Leal, L.K. Cognitive enhancement in aged rats after chronic administration of Equisetum arvense L. with demonstrated antioxidant properties in vitro. Pharmacol. Biochem. Behav. 2005, 81, 593–600. [Google Scholar] [CrossRef]
- Itoh, A.; Komatsuzaki, Y.; Lukowiak, K.; Saito, M. Epicatechin increases the persistence of long-term memory formed by conditioned taste aversion in Lymnaea. J. Exp. Biol. 2021, 224, jeb238055. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Wang, Z.; Oteiza, P.I. (-)-Epicatechin mitigates high fat diet-induced neuroinflammation and altered behavior in mice. Food Funct. 2020, 11, 5065–5076. [Google Scholar] [CrossRef]
- Diaz, A.; Treviño, S.; Pulido-Fernandez, G.; Martínez-Muñoz, E.; Cervantes, N.; Espinosa, B.; Rojas, K.; Pérez-Severiano, F.; Montes, S.; Rubio-Osornio, M.; et al. Epicatechin reduces spatial memory deficit caused by amyloid-β25-35 toxicity modifying the heat shock proteins in the CA1 region in the hippocampus of rats. Antioxidants 2019, 8, 113. [Google Scholar] [CrossRef] [Green Version]
- Tan, X.; Yang, Y.; Xu, J.; Zhang, P.; Deng, R.; Mao, Y.; He, J.; Chen, Y.; Zhang, Y.; Ding, J.; et al. Luteolin exerts neuroprotection via modulation of the p62/Keap1/Nrf2 pathway in intracerebral hemorrhage. Front. Pharmacol. 2020, 10, 1551. [Google Scholar] [CrossRef]
- Richetti, S.K.; Blank, M.; Capiotti, K.M.; Piato, A.L.; Bogo, M.R.; Vianna, M.R.; Bonan, C.D. Quercetin and rutin prevent scopolamine-induced memory impairment in zebrafish. Behav. Brain Res. 2011, 217, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Sang, Z.; Wang, K.; Shi, J.; Cheng, X.; Zhu, G.; Wei, R.; Ma, Q.; Yu, L.; Zhao, Y.; Tan, Z.; et al. Apigenin-rivastigmine hybrids as multi-target-directed liagnds for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2020, 187, 111958. [Google Scholar] [CrossRef] [PubMed]
- Hussein, R.M.; Mohamed, W.R.; Omar, H.A. A neuroprotective role of kaempferol against chlorpyrifos-induced oxidative stress and memory deficits in rats via GSK3β-Nrf2 signaling pathway. Pestic. Biochem. Physiol. 2018, 152, 29–37. [Google Scholar] [CrossRef]
- Hermawati, E.; Arfian, N.; Mustofa, M.; Partadiredja, G. Chlorogenic acid ameliorates memory loss and hippocampal cell death after transient global ischemia. Eur. J. Neurosci. 2020, 51, 651–669. [Google Scholar] [CrossRef]
- Deshmukh, R.; Kaundal, M.; Bansal, V. Samardeep Caffeic acid attenuates oxidative stress, learning and memory deficit in intra-cerebroventricular streptozotocin induced experimental dementia in rats. Biomed. Pharmacother. 2016, 81, 56–62. [Google Scholar] [CrossRef]
- Yu, C.; Pan, S.; Zhang, J.; Li, X.; Niu, Y. Ferulic acid exerts Nrf2-dependent protection against prenatal lead exposure-induced cognitive impairment in offspring mice. J. Nutr. Biochem. 2021, 91, 108603. [Google Scholar] [CrossRef]
- Pallag, A.; Jurca, T.; Pasca, B.; Sirbu, V.; Honiges, A.N.A.; Costuleanu, M. Analysis of Phenolic Compounds Composition by HPLC and Assessment of Antioxidant Capacity in Equisetum arvense L. Extracts. Rev. Chem. 2016, 67, 1623–1627. [Google Scholar]
- Oniszczuk, A.; Podgórski, R.; Oniszczuk, T.; Zukiewicz-Sobczak, W.; Nowak, R.; Waksmundzka-Hajnos, M. Extraction methods for the determination of phenolic compounds from Equisetum arvense L. herb. Ind. Crops Prod. 2014, 61, 377–381. [Google Scholar] [CrossRef]
- Trouillas, P.; Calliste, C.-A.; Allais, D.-P.; Simon, A.; Marfak, A.; Delage, C.; Duroux, J.-L. Antioxidant, anti-inflammatory and antiproliferative properties of sixteen water plant extracts used in the Limousin countryside as herbal teas. Food Chem. 2003, 80, 399–407. [Google Scholar] [CrossRef]
- Zhang, H.; Li, N.; Li, K.; Li, P. Effect of ethanol root extract of equisetum arvense (L) on urinary bladder activity in rats and analysis of principal plant constituents. Trop. J. Pharm. Res. 2015, 14, 1451–1458. [Google Scholar] [CrossRef] [Green Version]
- Dinis, T.C.P.; Madeira, V.M.C.; Almeida, L.M. Action of Phenolic Derivatives (Acetaminophen, Salicylate, and 5-Aminosalicylate) as Inhibitors of Membrane Lipid Peroxidation and as Peroxyl Radical Scavengers. Arch. Biochem. Biophys. 1994, 315, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Malterud, K.E.; Rydland, K.M. Inhibitors of 15-Lipoxygenase from Orange Peel. J. Agric. Food Chem. 2000, 48, 5576–5580. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.B.; Chul Hong, S.; Jin Jeong, H. 3,4-Dihydroxybenzaldehyde purified from the barley seeds (Hordeum vulgare) inhibits oxidative DNA damage and apoptosis via its antioxidant activity. Phytomedicine 2009, 16, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Luo, D. Antioxidant activities of different fractions of polysaccharides purified from Gynostemma pentaphyllum Makino. Carbohydr. Polym. 2007, 1, 54–58. [Google Scholar] [CrossRef]
- Cachat, J.; Stewart, A.; Grossman, L.; Gaikwad, S.; Kadri, F.; Chung, K.M.; Wu, N.; Wong, K.; Roy, S.; Suciu, C.; et al. Measuring behavioral and endocrine responses to novelty stress in adult zebrafish. Nat. Protoc. 2010, 5, 1786–1799. [Google Scholar] [CrossRef]
- Rosemberg, D.B.; Rico, E.P.; Mussulini, B.H.M.; Piato, Â.L.; Calcagnotto, M.E.; Bonan, C.D.; Dias, R.D.; Blaser, R.E.; Souza, D.O.; de Oliveira, D.L. Differences in Spatio-Temporal Behavior of Zebrafish in the Open Tank Paradigm after a Short-Period Confinement into Dark and Bright Environments. PLoS ONE 2011, 6, e19397. [Google Scholar] [CrossRef] [Green Version]
- Cognato, G.d.P.; Bortolotto, J.W.; Blazina, A.R.; Christoff, R.R.; Lara, D.R.; Vianna, M.R.; Bonan, C.D. Y-Maze memory task in zebrafish (Danio rerio): The role of glutamatergic and cholinergic systems on the acquisition and consolidation periods. Neurobiol. Learn. Mem. 2012, 98, 321–328. [Google Scholar] [CrossRef]
- Zanandrea, R.; Abreu, M.S.; Piato, A.; Barcellos, L.J.G.; Giacomini, A.C.V.V. Lithium prevents scopolamine-induced memory impairment in zebrafish. Neurosci. Lett. 2018, 664, 34–37. [Google Scholar] [CrossRef]
- Dumitru, G.; El-Nashar, H.A.S.; Mostafa, N.M.; Eldahshan, O.A.; Boiangiu, R.S.; Todirascu-Ciornea, E.; Hritcu, L.; Singab, A.N.B. Agathisflavone isolated from Schinus polygamus (Cav.) Cabrera leaves prevents scopolamine-induced memory impairment and brain oxidative stress in zebrafish (Danio rerio). Phytomedicine 2019, 58, 152889. [Google Scholar] [CrossRef]









| Compound Expressed in mg/g Plant Product * | Sample (70% Methanolic Extract) | Sample (70% Ethanolic Extract) | ||||
|---|---|---|---|---|---|---|
| E. sylvaticum | E. pratense | E. telmateia | E. sylvaticum | E. pratense | E. telmateia | |
| epicatechin | 0.9010 ± 0.021 | 0.5925 ± 0.001 | 1.1967 ± 0.001 | 0.1586 ± 0.002 | 0.0263 ± 0.003 | 0.7079 ± 0.011 |
| quercetin-3-glucoside | 1.6995 ± 0.001 | 8.2442 ± 0.001 | 23.4765 ± 0.033 | 41.9429 ± 0.022 | 3.8440 ± 0.001 | 13.6175 ± 0.022 |
| luteolin-7-glucoside | 11.0456 ± 0.011 | 1.8562 ± 0.001 | 5.6447 ± 0.021 | 3.7656 ± 0.019 | 0.0378 ± 0.011 | 1.2254 ± 0.003 |
| apigenin-7-glucoside | 2.3249 ± 0.013 | 2.2947 ± 0.021 | 27.7463 ± 0.009 | 7.8806 ± 0.003 | 1.4626 ± 0.004 | 21.0042 ± 0.021 |
| luteolin | 0.6150 ± 0.011 | 0.0344 ± 0.003 | 0.4214 ± 0.009 | 0.4069 ± 0.011 | 0.0076 ± 0.021 | 0.1455 ± 0.011 |
| quercetin | 0.0658 ± 0.001 | 0.0059 ± 0.002 | 0.4013 ± 0.022 | 0.0255 ± 0.009 | 0.0071 ± 0.001 | 0.1369 ± 0.005 |
| apigenin | 0.0716 ± 0.002 | 0.6260 ± 0.009 | 0.9571 ± 0.009 | 0.0591 ± 0.001 | 0.1359 ± 0.002 | 0.3758 ± 0.001 |
| kaempferol | 1.4997 ± 0.021 | 0.9781 ± 0.009 | 1.4417 ± 0.009 | 0.0216 ± 0.009 | 0.2022 ± 0.001 | 0.7609 ± 0.004 |
| Compound Expressed in mg/g Plant Product * | Sample (70% Methanolic Extract) | Sample (70% Ethanolic Extract) | ||||
|---|---|---|---|---|---|---|
| E. sylvaticum | E. pratense | E. telmateia | E. sylvaticum | E. pratense | E. telmateia | |
| neochlorogenic acid | 0.2639 ± 0.001 | 0.0730 ± 0.001 | 0.6381 ± 0.005 | 0.4838 ± 0.002 | 0.1879 ± 0.002 | 0.3059 ± 0.002 |
| chlorogenic acid | 4.3322 ± 0.001 | 0.7491 ± 0.002 | 10.1162 ± 0.002 | 3.6744 ± 0.001 | 0.7362 ± 0.003 | 14.3814 ± 0.004 |
| caffeic acid | 0.1468 ± 0.002 | 0.2363 ± 0.002 | 0.8590 ± 0.003 | 0.6624 ± 0.002 | 0.1922 ± 0.001 | 0.3244 ± 0.002 |
| ferulic acid | 0.6414 ± 0.002 | 0.2295 ± 0.005 | 1.0245 ± 0.001 | 5.7962 ± 0.004 | 0.5559 ± 0.001 | 1.0439 ± 0.003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batir-Marin, D.; Boev, M.; Cioanca, O.; Mircea, C.; Burlec, A.F.; Beppe, G.J.; Spac, A.; Corciova, A.; Hritcu, L.; Hancianu, M. Neuroprotective and Antioxidant Enhancing Properties of Selective Equisetum Extracts. Molecules 2021, 26, 2565. https://doi.org/10.3390/molecules26092565
Batir-Marin D, Boev M, Cioanca O, Mircea C, Burlec AF, Beppe GJ, Spac A, Corciova A, Hritcu L, Hancianu M. Neuroprotective and Antioxidant Enhancing Properties of Selective Equisetum Extracts. Molecules. 2021; 26(9):2565. https://doi.org/10.3390/molecules26092565
Chicago/Turabian StyleBatir-Marin, Denisa, Monica Boev, Oana Cioanca, Cornelia Mircea, Ana Flavia Burlec, Galba Jean Beppe, Adrian Spac, Andreia Corciova, Lucian Hritcu, and Monica Hancianu. 2021. "Neuroprotective and Antioxidant Enhancing Properties of Selective Equisetum Extracts" Molecules 26, no. 9: 2565. https://doi.org/10.3390/molecules26092565








