Dual-Template Magnetic Molecularly Imprinted Polymer for Simultaneous Determination of Spot Urine Metanephrines and 3-Methoxytyramine for the Diagnosis of Pheochromocytomas and Paragangliomas
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Material
2.2. Patients
2.3. Synthesis of MMIP and Magnetic Non-Imprinted Polymer
2.4. Performance of d-SPE
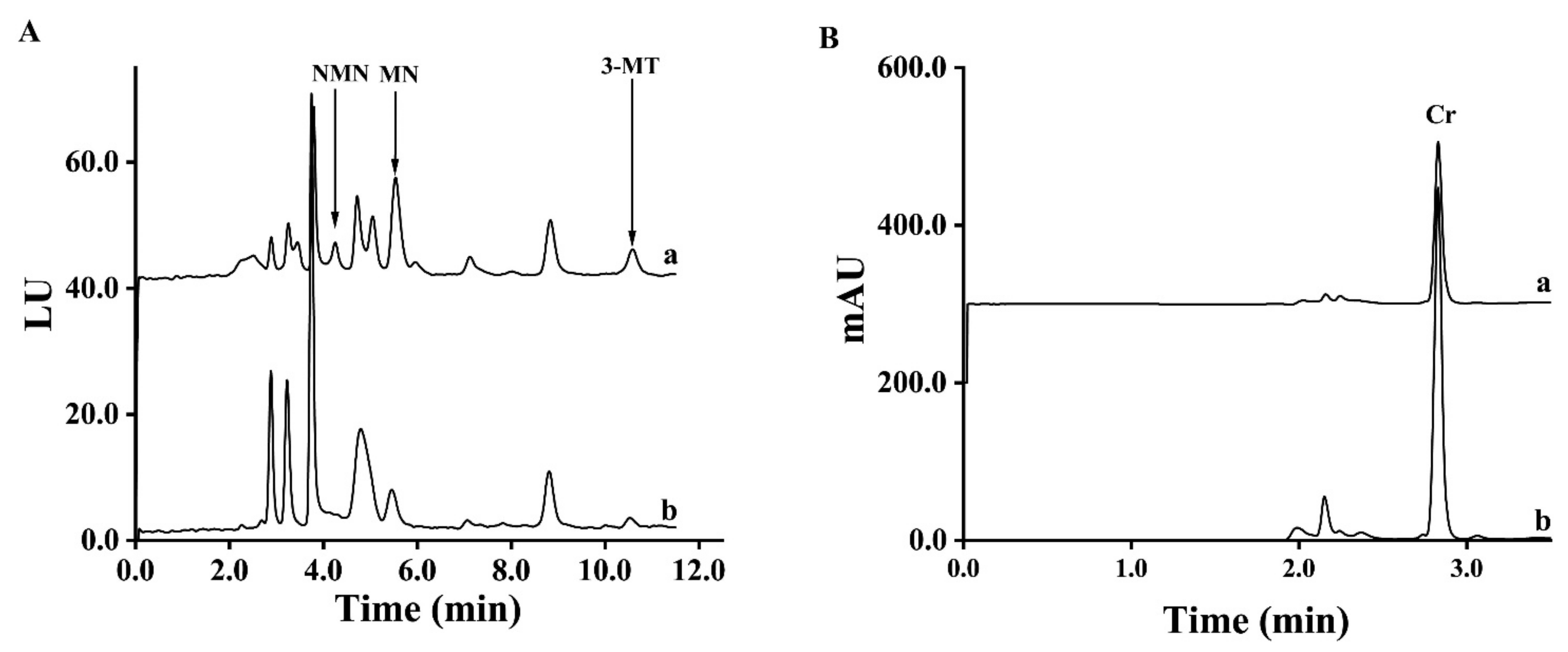
2.5. Chromatographic Conditions
3. Results and Discussion
3.1. MMIP Synthesis
3.2. Optimization of MMIP Synthesis
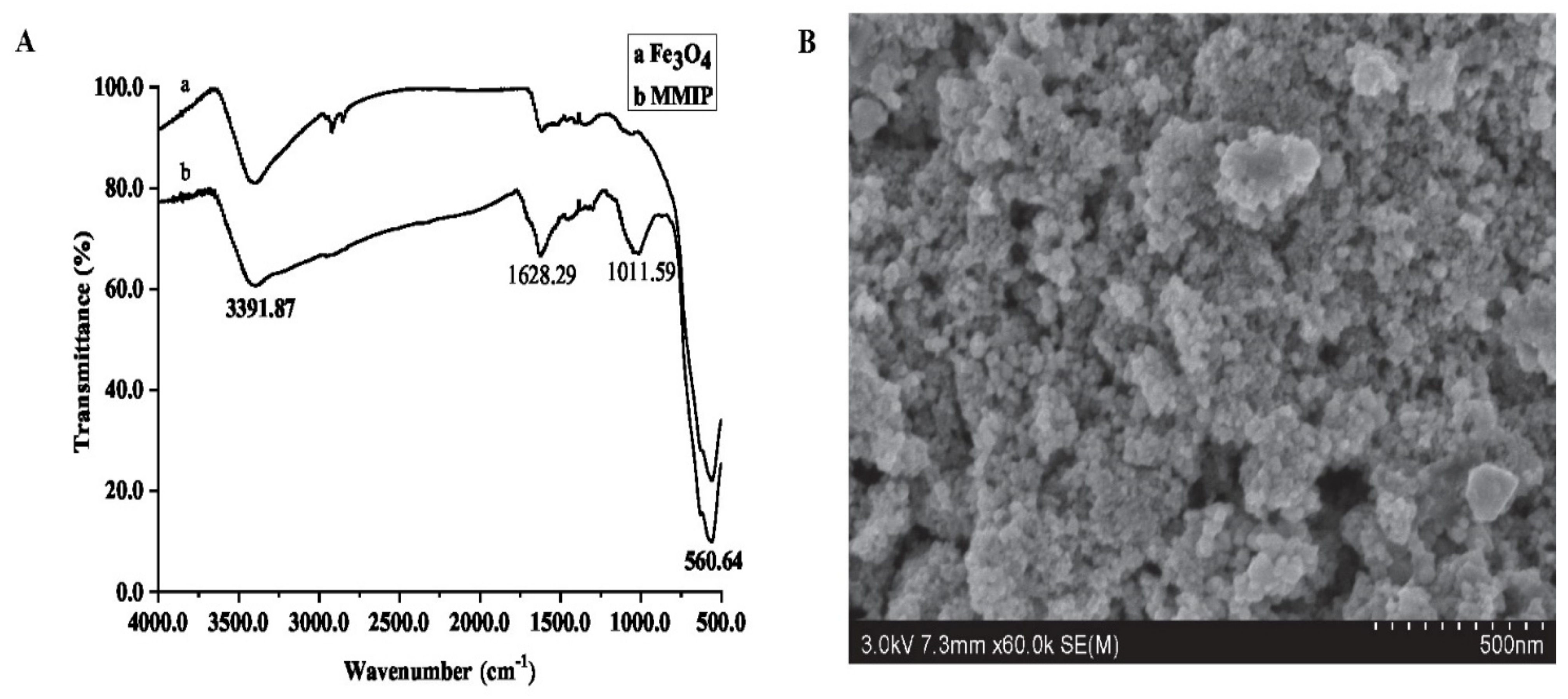
3.3. The Characteristics of MMIP
3.4. The Optimization of d-SPE Conditions
3.5. Precision, Usage Count and Stability of MMIP
3.6. Recoveries and Calibration Curves
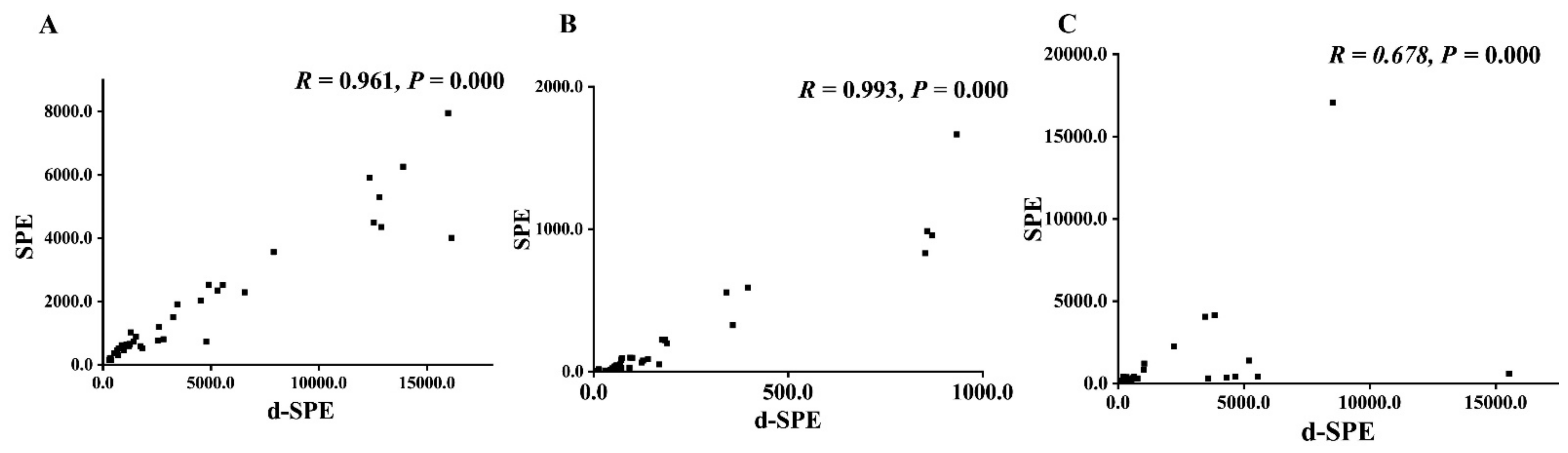
3.7. The Methods Comparison between SPE and d-SPE
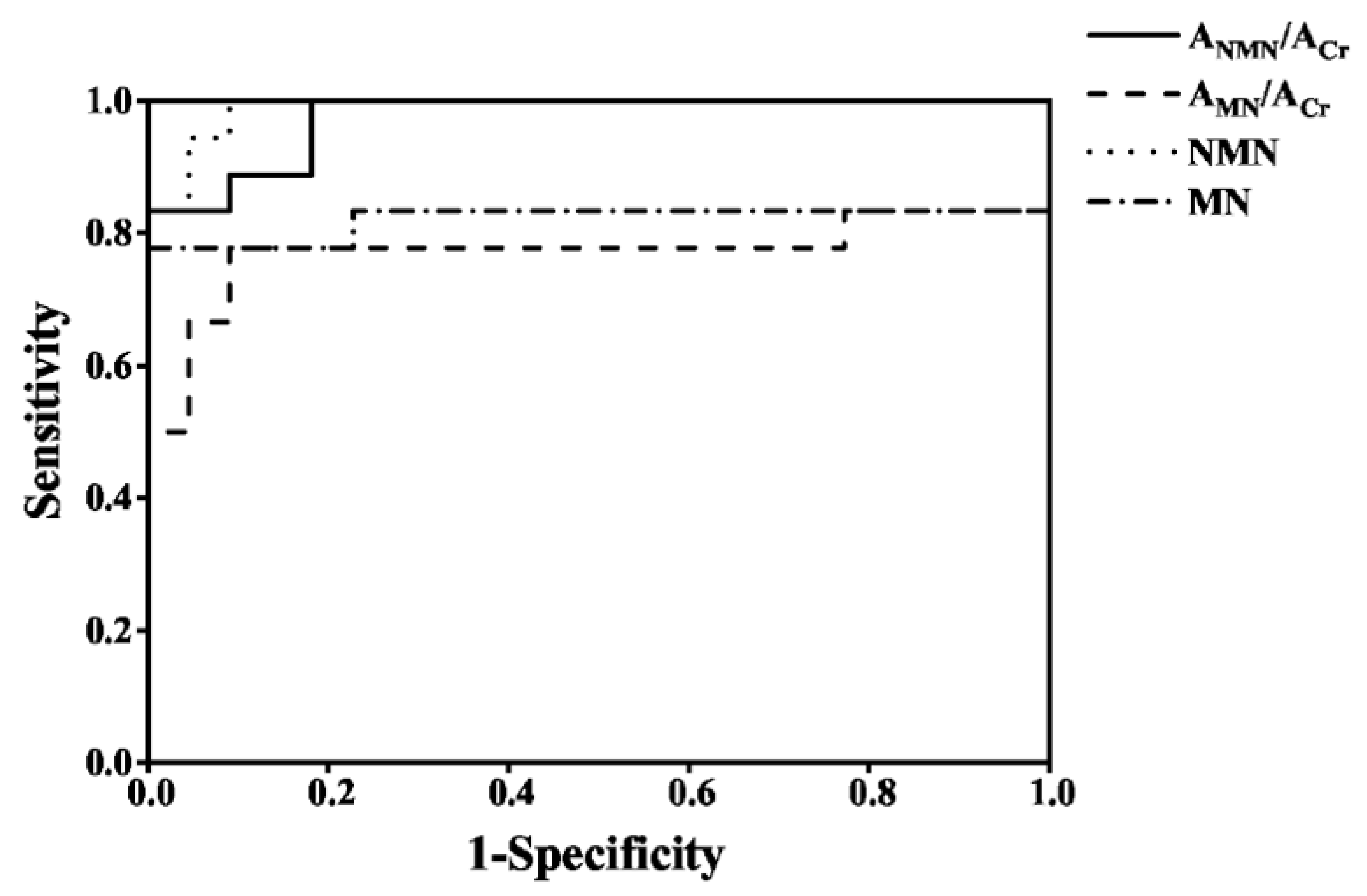
3.8. The Diagnosis of PPGLs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Lenders, J.W.M.; Eisenhofer, G.; Mannelli, M.; Pacak, K. Phaeochromocytoma. Lancet 2005, 366, 665–675. [Google Scholar] [CrossRef]
- Prejbisz, A.; Lenders, J.; Eisenhofer, G.; Januszewicz, A. Cardiovascular manifestations of phaeochromocytoma. J. Hypertens. 2011, 29, 2049–2060. [Google Scholar] [CrossRef] [PubMed]
- Manger, W. The protean manifestations of pheochromocytoma. Horm. Metab. Res. 2009, 41, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Manger, W. The vagaries of pheochromocytomas. Am. J. Hypertens. 2005, 18, 1266–1270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lenders, J.W.M.; Duh, Q.-Y.; Eisenhofer, G.; Gimenez-Roqueplo, A.-P.; Grebe, S.K.G.; Murad, M.H.; Naruse, M.; Pacak, K.; Young, W.F., Jr. Pheochromocytoma and Paraganglioma: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2014, 99, 1915–1942. [Google Scholar] [CrossRef] [PubMed]
- Van Duinen, N.; Corssmit, E.P.M.; De Jong, W.H.A.; Brookman, D.; Kema, I.P.; Romijn, J.A. Plasma levels of free metanephrines and 3-methoxytyramine indicate a higher number of biochemically active HNPGL than 24-h urinary excretion rates of catecholamines and metabolites. Eur. J. Endocrinol. 2013, 169, 377–382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Xie, W.; Song, Z.; Wang, T.; Li, X.; Gao, Y.; Li, Y.; Zhang, J.; Guo, X. Addition of 3-methoxytyramine or chromogranin A to plasma free metanephrines as the initial test for pheochromocytoma and paraganglioma: Which is the best diagnostic strategy. Clin. Endocrinol. 2022, 96, 132–138. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Lenders, J.W.M.; Siegert, G.; Bornstein, S.R.; Friberg, P.; Milosevic, D.; Mannelli, M.; Linehan, W.M.; Adams, K.; Timmers, H.J.; et al. Plasma methoxytyramine: A novel biomarker of metastatic pheochromocytoma and paraganglioma in relation to established risk factors of tumour size, location and SDHB mutation status. Eur. J. Cancer 2012, 48, 1739–1749. [Google Scholar] [CrossRef] [Green Version]
- Lefeuvre, S.; Bois-Maublanc, J.; Mongeois, E.; Policarpo, V.; Formaux, L.; Francia, T.; Billaud, E.; Got, L. Quantitation using HRMS: A new tool for rapid, specific and sensitive determination of catecholamines and deconjugated methanephrines metanephrines in urine. J. Chromatogr. B 2021, 1166, 122391. [Google Scholar] [CrossRef]
- Richard, V.; Zahedi, R.; Eintracht, S.; Borchers, C. An LC-MRM assay for the quantification of metanephrines from dried blood spots for the diagnosis of pheochromocytomas and paragangliomas. Anal. Chim. Acta 2020, 1128, 140–148. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, X.; Zhen, Q.; Zou, W.; Chen, H.; Luo, C.; Ding, M. Detection of spot urinary free metanephrines and 3-methoxytyramine with internal reference correction for the diagnosis of pheochromocytomas and paragangliomas. J. Chromatogr. B 2020, 1156, 122306. [Google Scholar] [CrossRef] [PubMed]
- Zuber, S.; Kantorovich, V.; Pacak, K. Hypertension in pheochromocytoma: Characteristics and treatment. Endocrinol. Metab. Clin. 2011, 40, 295–311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrák, O.; Haluzíková, D.; Kaválková, P.; Štrauch, B.; Rosa, J.; Holaj, R.; Brabcová Vránková, A.; Michalsky, D.; Haluzík, M.; Zelinka, T.; et al. Changes in energy metabolism in pheochromocytoma. J. Clin. Endocrinol. Metab. 2013, 98, 1651–1658. [Google Scholar] [CrossRef] [Green Version]
- Grouzmann, E.; Fathi, M.; Gillet, M.; de Torrenté, A.; Cavadas, C.; Brunner, H.; Buclin, T. Disappearance rate of catecholamines, total metanephrines, and neuropeptide Y from the plasma of patients after resection of pheochromocytoma. Clin. Chem. 2001, 47, 1075–1082. [Google Scholar] [CrossRef]
- Zuo, M.; Zhen, Q.; Zhang, X.; Zou, W.; Yang, X.; Tian, G.; Shi, Z.; Li, Q.; Ding, M. High specificity of spot urinary free metanephrines in diagnosis and prognosis of pheochromocytomas and paragangliomas by HPLC with electrochemical detection. Clin. Chim. Acta 2018, 478, 82–89. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.; Graham, K.; van der Molen, J.; Links, T.; Morris, M.; Ross, H.; de Vries, E.; Kema, I. Plasma free metanephrine measurement using automated online solid-phase extraction HPLC tandem mass spectrometry. Clin. Chem. 2007, 53, 1684–1693. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Xu, G.; Wei, F.; Yang, J.; Hu, Q. Preparation of a stir bar coated with molecularly imprinted polymer and its application in analysis of dopamine in urine. J. Pharm. Biomed. Anal. 2014, 94, 118–124. [Google Scholar] [CrossRef]
- Dramou, P.; Zuo, P.; He, H.; Pham-Huy, L.A.; Zou, W.; Xiao, D.; Pham-Huy, C. Development of novel amphiphilic magnetic molecularly imprinted polymers compatible with biological fluids for solid phase extraction and physicochemical behavior study. J. Chromatogr. A 2013, 1317, 110–120. [Google Scholar] [CrossRef]
- Vasapollo, G.; Sole, R.D.; Mergola, L.; Lazzoi, M.R.; Scardino, A.; Scorrano, S.; Mele, G. Molecularly imprinted polymers: Present and future prospective. Int. J. Mol. Sci. 2011, 12, 5908–5945. [Google Scholar] [CrossRef] [Green Version]
- Płotka-Wasylka, J.; Szczepańska, N.; de La Guardia, M.; Namieśnik, J. Modern trends in solid phase extraction: New sorbent media. TrAC Trends Anal. Chem. 2016, 77, 23–43. [Google Scholar] [CrossRef]
- Speltini, A.; Scalabrini, A.; Maraschi, F.; Sturini, M.; Profumo, A. Newest applications of molecularly imprinted polymers for extraction of contaminants from environmental and food matrices: A review. Anal. Chim. Acta 2017, 974, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Beyazit, S.; Bui, B.T.S.; Haupt, K.; Gonzato, C. Molecularly imprinted polymer nanomaterials and nanocomposites by controlled/living radical polymerization. Prog. Polym. Sci. 2016, 62, 1–21. [Google Scholar] [CrossRef]
- Xu, T.; Tang, H.; Chen, D.; Dong, H.; Li, L. Simultaneous determination of 24 polycyclic aromatic hydrocarbons in edible oil by tandem solid-phase extraction and gas chromatography coupled/tandem mass spectrometry. J. AOAC Int. 2015, 98, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Yaripour, S.; Mohammadi, A.; Nojavan, S. Electromembrane extraction of tartrazine from food samples: Effects of nano-sorbents on membrane performance. J. Sep. Sci. 2016, 39, 2642–2651. [Google Scholar] [CrossRef] [PubMed]
- Kopperi, M.; Riekkola, M.-L. Non-targeted evaluation of selectivity of water-compatible class selective adsorbents for the analysis of steroids in wastewater. Anal. Chim. Acta 2016, 920, 47–53. [Google Scholar] [CrossRef]
- Pesavento, M.; D’Agostino, G.; Biesuz, R.; Alberti, G.; Profumo, A. Ion selective electrode for dopamine based on a molecularly imprinted polymer. Electroanalysis 2012, 24, 813–824. [Google Scholar] [CrossRef]
- Chen, L.; Xu, S.; Li, J. Recent advances in molecular imprinting technology: Current status, challenges and highlighted applications. Chem. Soc. Rev. 2011, 40, 2922–2942. [Google Scholar] [CrossRef]
- Sturini, M.; Speltini, A.; Maraschi, F.; Pretali, L.; Profumo, A.; Fasani, E.; Albini, A. Environmental photochemistry of fluoroquinolones in soil and in aqueous soil suspensions under solar light. Environ. Sci. Pollut. Res. 2014, 21, 13215–13221. [Google Scholar] [CrossRef]
- Wen, Y.; Chen, L.; Li, J.; Liu, D.; Chen, L. Recent advances in solid-phase sorbents for sample preparation prior to chromatographic analysis. TrAC Trends Anal. Chem. 2014, 59, 26–41. [Google Scholar] [CrossRef]
- Volkert, A.A.; Haes, A.J. Advancements in nanosensors using plastic antibodies. Analyst 2014, 139, 21–31. [Google Scholar] [CrossRef]
- Pichon, V.; Chapuis-Hugon, F. Role of molecularly imprinted polymers for selective determination of environmental pollutants—A review. Anal. Chim. Acta 2008, 622, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Kubo, T.; Otsuka, K. Recent progress for the selective pharmaceutical analyses using molecularly imprinted adsorbents and their related techniques: A review. J. Pharm. Biomed. Anal. 2016, 130, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Marzi Khosrowshahi, E.; Ghalkhani, M.; Afshar Mogaddam, M.; Farajzadeh, M.; Sohouli, E.; Nemati, M. Evaluation of MXene as an adsorbent in dispersive solid phase extraction of several pesticides from fresh fruit juices prior to their determination by HPLC-MS/MS. Food Chem. 2022, 386, 132773. [Google Scholar] [CrossRef] [PubMed]
- González-Gómez, L.; Gañán, J.; Morante-Zarcero, S.; Pérez-Quintanilla, D.; Sierra, I. Mesostructured Silicas as Cation-Exchange Sorbents in Packed or Dispersive Solid Phase Extraction for the Determination of Tropane Alkaloids in Culinary Aromatics Herbs by HPLC-MS/MS. Toxins 2022, 14, 218. [Google Scholar] [CrossRef] [PubMed]
- Rattanakunsong, N.; Jullakan, S.; Płotka-Wasylka, J.; Bunkoed, O. A hierarchical porous composite magnetic sorbent of reduced graphene oxide embedded in polyvinyl alcohol cryogel for solvent assisted-solid phase extraction of polycyclic aromatic hydrocarbons. J. Sep. Sci. 2022, 45, 1774–1783. [Google Scholar] [CrossRef]
- Lv, S.; Gao, Y.; Zhao, M.; Jiang, X.; Li, X.; Yang, J.; Chen, S.; Cui, S. Biomass-derived porous material synthesized by one-step calcination method for the magnetic solid phase extraction of polychlorinated biphenyls in water. J. Sep. Sci. 2022, 45, 1693–1701. [Google Scholar] [CrossRef]
- Chen, R.; Qiao, X.; Liu, F. Ionic liquid-based magnetic nanoparticles for magnetic dispersive solid-phase extraction: A review. Anal. Chim. Acta 2022, 1201, 339632. [Google Scholar] [CrossRef]
- Liu, M.; Zhu, C.; Li, X.; Wang, F. 3DG Functionalized Magnetic Solid Phase Extraction Materials for the Efficient Enrichment of Hexamethylenetetramine in Vermicelli. Molecules 2022, 27, 1548. [Google Scholar] [CrossRef]
- Nemati, M.; Tuzen, M.; Farazajdeh, M.; Kaya, S.; Afshar Mogaddam, M. Development of dispersive solid-liquid extraction method based on organic polymers followed by deep eutectic solvents elution; application in extraction of some pesticides from milk samples prior to their determination by HPLC-MS/MS. Anal. Chim. Acta 2022, 1199, 339570. [Google Scholar] [CrossRef]
- Bouri, M.; Lerma-García, M.J.; Salghi, R.; Zougagh, M.; Ríos, A. Selective extraction and determination of catecholamines in urine samples by using a dopamine magnetic molecularly imprinted polymer and capillary electrophoresis. Talanta 2012, 99, 897–903. [Google Scholar] [CrossRef]
- Kumar, N.; Narayanan, N.; Gupta, S. Application of magnetic molecularly imprinted polymers for extraction of imidacloprid from eggplant and honey. Food Chem. 2018, 255, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Li, X.; Li, J.; Liu, M.; Lei, F.; Tan, X.; Li, P.; Luo, W. Synthesis and characterization of core–shell magnetic molecularly imprinted polymers for solid-phase extraction and determination of Rhodamine B in food. Food Chem. 2015, 171, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Li, J.; Liu, Y.; Ma, J. Development of magnetic molecularly imprinted polymers with double templates for the rapid and selective determination of amphenicol antibiotics in water, blood, and egg samples. J. Chromatogr. A 2016, 1473, 19–27. [Google Scholar] [CrossRef] [PubMed]


| Template | EA (%) of Standards | ||
|---|---|---|---|
| NMN | MN | 3-MT | |
| DA | 94.5 ± 0.0 | 90.8 ± 0.7 | 94.9 ± 0.1 |
| E | 78.1 ± 1.3 | 73.4 ± 0.6 | 82.0 ± 0.6 |
| NE | 84.6 ± 0.3 | 75.8 ± 0.9 | 87.3 ± 0.6 |
| NMN | 52.5 ± 2.2 | 46.6 ± 0.3 | 61.5 ± 2.3 |
| MN | 57.8 ± 1.1 | 64.3 ± 1.2 | 48.0 ± 0.6 |
| 3-MT | 56.4 ± 0.9 | 32.7 ± 1.4 | 45.7 ± 2.2 |
| DA +MN | 91.3 ± 0.1 | 89.4 ± 0.2 | 94.6 ± 0.0 |
| MNIP | 8.9 ± 1.2 | 9.4 ± 0.5 | 8.5 ± 0.5 |
| Template | Peak Area | ||
|---|---|---|---|
| NMN | MN | 3-MT | |
| DA | 287.5 ± 8.3 | 2.1 ± 0.1 | 44.1 ± 1.6 |
| E | 12.2 ± 0.5 | 12.0 ± 0.4 | 10.1 ± 0.1 |
| NE | 3.8 ± 0.3 | 3.6 ± 0.3 | 82.9 ± 7.3 |
| NMN | 26.3 ± 0.5 | 0.0 | 5.0 ± 0.5 |
| MN | 5.6 ± 0.2 | 42.9 ± 0.3 | 49.0 ± 0.8 |
| 3-MT | 24.6 ± 0.1 | 29.3 ± 0.8 | 51.1 ± 0.7 |
| DA + MN | 25.8 ± 1.0 | 40.3 ± 1.2 | 37.4 ± 1.8 |
| MNIP | 3.2 ± 0.1 | 5.6 ± 0.1 | 3.0 ± 1.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, H.; Zhang, X.; Zhen, Q.; He, Y.; Wang, H.; Zhu, Y.; Sun, Q.; Ding, M. Dual-Template Magnetic Molecularly Imprinted Polymer for Simultaneous Determination of Spot Urine Metanephrines and 3-Methoxytyramine for the Diagnosis of Pheochromocytomas and Paragangliomas. Molecules 2022, 27, 3520. https://doi.org/10.3390/molecules27113520
Zeng H, Zhang X, Zhen Q, He Y, Wang H, Zhu Y, Sun Q, Ding M. Dual-Template Magnetic Molecularly Imprinted Polymer for Simultaneous Determination of Spot Urine Metanephrines and 3-Methoxytyramine for the Diagnosis of Pheochromocytomas and Paragangliomas. Molecules. 2022; 27(11):3520. https://doi.org/10.3390/molecules27113520
Chicago/Turabian StyleZeng, Hongyu, Xiaoqing Zhang, Qianna Zhen, Yifan He, Haoran Wang, Yang Zhu, Qi Sun, and Min Ding. 2022. "Dual-Template Magnetic Molecularly Imprinted Polymer for Simultaneous Determination of Spot Urine Metanephrines and 3-Methoxytyramine for the Diagnosis of Pheochromocytomas and Paragangliomas" Molecules 27, no. 11: 3520. https://doi.org/10.3390/molecules27113520





