Role of Natural Compounds and Target Enzymes in the Treatment of Alzheimer’s Disease
Abstract
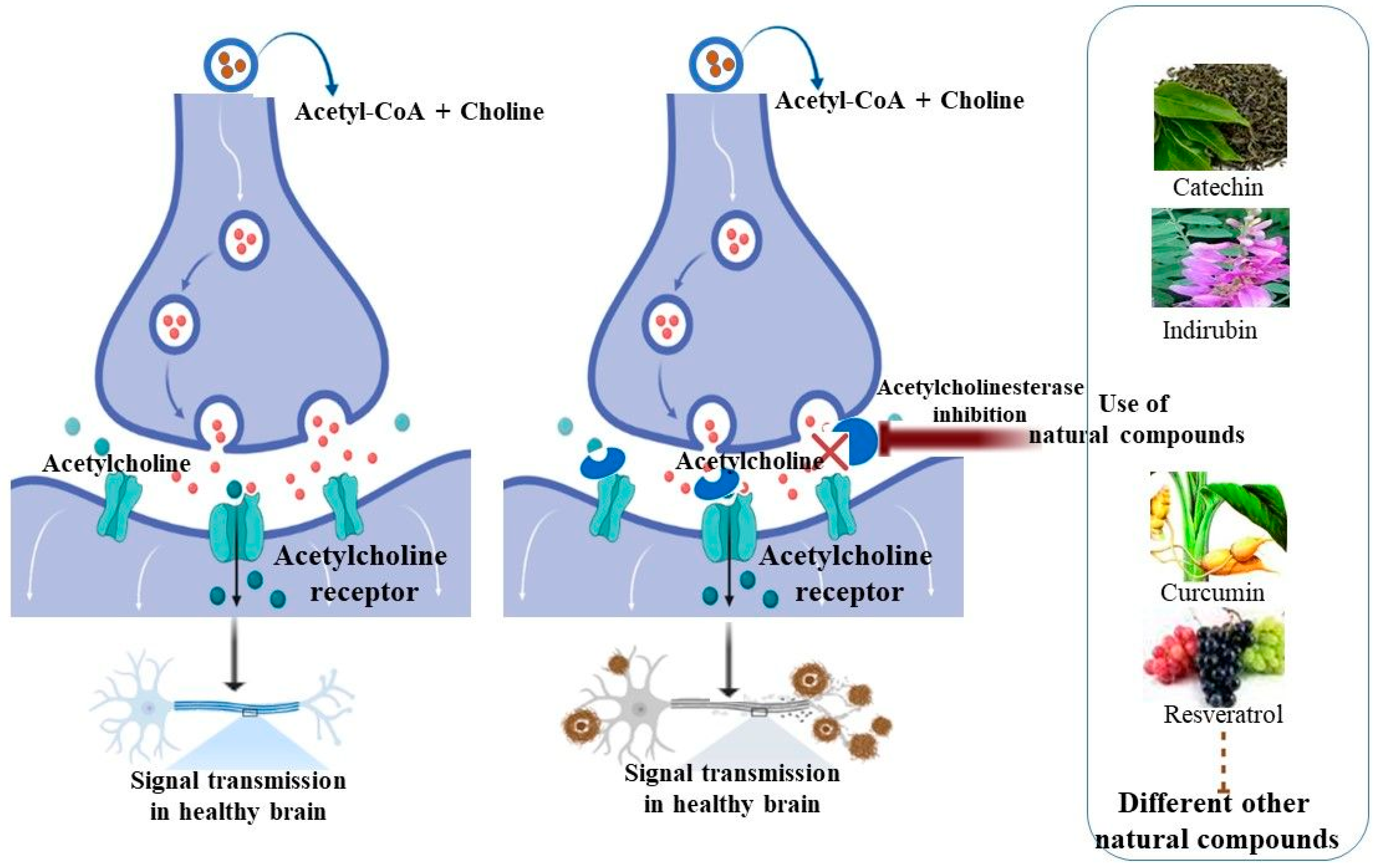
:1. Introduction
2. Natural Compounds and Alzheimer’s Disease
3. Inhibition of Acetylcholinesterase Activity Using Natural Compounds
4. Inhibition of BACE1 Activity Using Natural Compounds
5. Inhibition of Monoamine Oxidase Activity Using Natural Compounds
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| AChE | Acetylcholinesterase |
| BuChE | Butyrylcholinesterase |
| Ach | Acetylcholine |
| CNS | Central nervous system |
| ND | Neurodegenerative disease |
| Aβ | β-amyloid |
| APP | Amyloid precursor protein |
| MAOs | Monoamine oxidases |
References
- Dey, A.; Bhattacharya, R.; Mukherjee, A.; Pandey, D.K. Natural products against Alzheimer’s disease: Pharmaco-therapeutics and biotechnological interventions. Biotechnol. Adv. 2017, 35, 178–216. [Google Scholar] [CrossRef] [PubMed]
- Noetzli, M.; Eap, C.B. Pharmacodynamic, Pharmacokinetic and Pharmacogenetic Aspects of Drugs Used in the Treatment of Alzheimer’s Disease. Clin. Pharmacokinet. 2013, 52, 225–241. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, R.A. Review article What causes alzheimer’s disease? Folia Neuropathol. 2013, 3, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Scheltens, P.; Blennow, K.; Breteler, M.M.; de Strooper, B.; Frisoni, G.B.; Salloway, S.; van der Flier, W.M. Alzheimer’s disease. Lancet 2016, 388, 505–517. [Google Scholar] [CrossRef]
- Alzheimer’s Association. 2021 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2021, 17, 327–406. [Google Scholar] [CrossRef]
- Iqubal, A.; Rahman, S.O.; Ahmed, M.; Bansal, P.; Haider, R.; Iqubal, M.K.; Najmi, A.K.; Pottoo, F.H.; Haque, S.E. Current Quest in Natural Bioactive Compounds for Alzheimer’s Disease: Multi-Targeted-Designed-Ligand Based Approach with Preclinical and Clinical Based Evidence. Curr. Drug Targets 2021, 22, 685–720. [Google Scholar] [CrossRef]
- Scheltens, P.; De Strooper, B.; Kivipelto, M.; Holstege, H.; Chételat, G.; Teunissen, C.E.; Cummings, J.; van der Flier, W.M. Alzheimer’s disease. Lancet 2021, 397, 1577–1590. [Google Scholar] [CrossRef]
- Marucci, G.; Buccioni, M.; Ben, D.D.; Lambertucci, C.; Volpini, R.; Amenta, F. Efficacy of acetylcholinesterase inhibitors in Alzheimer’s disease. Neuropharmacology 2021, 190, 108352. [Google Scholar] [CrossRef]
- Andrade, S.; Ramalho, M.J.; Loureiro, J.A.; Pereira, M.D.C. Natural Compounds for Alzheimer’s Disease Therapy: A Systematic Review of Preclinical and Clinical Studies. Int. J. Mol. Sci. 2019, 20, 2313. [Google Scholar] [CrossRef] [Green Version]
- Kaur, D.; Behl, T.; Sehgal, A.; Singh, S.; Sharma, N.; Bungau, S. Multifaceted Alzheimer’s Disease: Building a Roadmap for Advancement of Novel Therapies. Neurochem. Res. 2021, 46, 2832–2851. [Google Scholar] [CrossRef]
- Trottier, G.; Boström, P.J.; Lawrentschuk, N.; Fleshner, N.E. Nutraceuticals and prostate cancer prevention: A current review. Nat. Rev. Urol. 2009, 7, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Regulation of “Nutraceuticals”. Science 1999, 285, 1853–1855. [Google Scholar] [CrossRef] [PubMed]
- Sadhukhan, P.; Saha, S.; Dutta, S.; Mahalanobish, S.; Sil, P.C. Nutraceuticals: An emerging therapeutic approach against the pathogenesis of Alzheimer’s disease. Pharmacol. Res. 2018, 129, 100–114. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.S.; Khalid, M.; Kamal, M.A.; Younis, K. Study of Nutraceuticals and Phytochemicals for the Management of Alzheimer’s Disease: A Review. Curr. Neuropharmacol. 2021, 19, 1. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, T.; Reker, D.; Schneider, P.; Schneider, G. Counting on natural products for drug design. Nat. Chem. 2016, 8, 531–541. [Google Scholar] [CrossRef]
- Woo, J.; Yang, H.; Yoon, M.; Gadhe, C.G.; Pae, A.N.; Cho, S.; Lee, S.C.A.C.J. 3-Carene, a Phytoncide from Pine Tree Has a Sleep-enhancing Effect by Targeting the GABAA-benzodiazepine Receptors. Exp. Neurobiol. 2019, 28, 593–601. [Google Scholar] [CrossRef] [Green Version]
- Vallianou, I.; Hadzopoulou-Cladaras, M. Camphene, a Plant Derived Monoterpene, Exerts Its Hypolipidemic Action by Affecting SREBP-1 and MTP Expression. PLoS ONE 2016, 11, e0147117. [Google Scholar] [CrossRef] [Green Version]
- Girola, N.; Figueiredo, C.R.; Farias, C.F.; Azevedo, R.A.; Ferreira, A.K.; Teixeira, S.F.; Capello, T.M.; Martins, E.G.; Matsuo, A.L.; Travassos, L.R.; et al. Camphene isolated from essential oil of Piper cernuum (Piperaceae) induces intrinsic apoptosis in melanoma cells and displays antitumor activity in vivo. Biochem. Biophys. Res. Commun. 2015, 467, 928–934. [Google Scholar] [CrossRef]
- Lenis-Rojas, O.A.; Robalo, M.P.; Tomaz, A.I.; Carvalho, A.; Fernandes, A.R.; Marques, F.; Folgueira, M.; Yáñez, J.; Vázquez-García, D.; Torres, M.L.; et al. RuII(p-cymene) Compounds as Effective and Selective Anticancer Candidates with No Toxicity In Vivo. Inorg. Chem. 2018, 57, 13150–13166. [Google Scholar] [CrossRef]
- Hou, J.; Zhang, Y.; Zhu, Y.; Zhou, B.; Ren, C.; Liang, S.; Guo, Y. α-Pinene Induces Apoptotic Cell Death via Caspase Activation in Human Ovarian Cancer Cells. Med. Sci. Monit. 2019, 25, 6631–6638. [Google Scholar] [CrossRef]
- Aydin, E.; Türkez, H.; Taşdemir, Ş. Anticancer and Antioxidant Properties of Terpinolene in Rat Brain Cells. Arch. Ind. Hyg. Toxicol. 2013, 64, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Liu, Q.F.; Choi, B.; Shin, C.; Lee, B.; Yuan, C.; Song, Y.J.; Yun, H.S.; Lee, I.-S.; Koo, B.-S.; et al. Neuroprotective Effects of Limonene (+) against Aβ42-Induced Neurotoxicity in a Drosophila Model of Alzheimer’s Disease. Biol. Pharm. Bull. 2020, 43, 409–417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabogal-Guáqueta, A.M.; Hobbie, F.; Keerthi, A.; Oun, A.; Kortholt, A.; Boddeke, E.; Dolga, A. Linalool attenuates oxidative stress and mitochondrial dysfunction mediated by glutamate and NMDA toxicity. Biomed. Pharmacother. 2019, 118, 109295. [Google Scholar] [CrossRef] [PubMed]
- Islam, A.; Hellman, B.; Nyberg, F.; Amir, N.; Jayaraj, R.; Petroainu, G.; Adem, A. Myrcene Attenuates Renal Inflammation and Oxidative Stress in the Adrenalectomized Rat Model. Molecules 2020, 25, 4492. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Luan, J.; Jiang, R.P.; Liu, J.; Ma, Y. Myrcene exerts anti-asthmatic activity in neonatal rats via modulating the matrix remodeling. Int. J. Immunopathol. Pharmacol. 2020, 34, 2058738420954948. [Google Scholar] [CrossRef]
- Kumar, A.; Agarwal, K.; Singh, M.; Saxena, A.; Yadav, P.; Maurya, A.K.; Yadav, A.; Tandon, S.; Chanda, D.; Bawankule, D.U. Essential oil from waste leaves of Curcuma longa L. alleviates skin inflammation. Inflammopharmacology 2018, 26, 1245–1255. [Google Scholar] [CrossRef]
- Baldissera, M.D.; Grando, T.H.; Souza, C.F.; Gressler, L.T.; Stefani, L.M.; da Silva, A.S.; Monteiro, S.G. In vitro and in vivo action of terpinen-4-ol, γ-terpinene, and α-terpinene against Trypanosoma evansi. Exp. Parasitol. 2016, 162, 43–48. [Google Scholar] [CrossRef]
- Falk, A.; Löf, A.; Hagberg, M.; Hjelm, E.W.; Wang, Z. Human exposure to 3-carene by inhalation: Toxicokinetics, effects on pulmonary function and occurrence of irritative and CNS symptoms. Toxicol. Appl. Pharmacol. 1991, 110, 198–205. [Google Scholar] [CrossRef]
- Schlumpf, M.; Durrer, S.; Faass, O.; Ehnes, C.; Fuetsch, M.; Gaille, C.; Henseler, M.; Hofkamp, L.; Maerkel, K.; Reolon, S.; et al. Developmental toxicity of UV filters and environmental exposure: A review. Int. J. Androl. 2008, 31, 144–151. [Google Scholar] [CrossRef]
- Dörsam, B.; Wu, C.-F.; Efferth, T.; Kaina, B.; Fahrer, J. The eucalyptus oil ingredient 1,8-cineol induces oxidative DNA damage. Arch. Toxicol. 2014, 89, 797–805. [Google Scholar] [CrossRef]
- Ravichandran, C.; Badgujar, P.C.; Gundev, P.; Upadhyay, A. Review of toxicological assessment of d-limonene, a food and cosmetics additive. Food Chem. Toxicol. 2018, 120, 668–680. [Google Scholar] [CrossRef] [PubMed]
- Audrain, H.; Kenward, C.; Lovell, C.; Green, C.; Ormerod, A.; Sansom, J.; Chowdhury, M.; Cooper, S.; Johnston, G.; Wilkinson, S.M.; et al. Allergy to oxidized limonene and linalool is frequent in the U.K. Br. J. Dermatol. 2014, 171, 292–297. [Google Scholar] [CrossRef] [PubMed]
- De Groot, A.C.; Schmidt, E. Tea tree oil: Contact allergy and chemical composition. Contact Dermat. 2016, 75, 129–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuboyama, T.; Tohda, C.; Komatsu, K. Effects of Ashwagandha (Roots of Withania somnifera) on Neurodegenerative Diseases. Biol. Pharm. Bull. 2014, 37, 892–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corona, J.C. Natural Compounds for the Management of Parkinson’s Disease and Attention-Deficit/Hyperactivity Disorder. BioMed Res. Int. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Rehman, M.U.; Wali, A.F.; Ahmad, A.; Shakeel, S.; Rasool, S.; Ali, R.; Rashid, S.M.; Madkhali, H.; Ganaie, M.A.; Khan, R. Neuroprotective Strategies for Neurological Disorders by Natural Products: An update. Curr. Neuropharmacol. 2019, 17, 247–267. [Google Scholar] [CrossRef]
- de Araújo, F.F.; de Paulo Farias, D.; Neri-Numa, I.A.; Pastore, G.M. Polyphenols and their applications: An approach in food chemistry and innovation potential. Food Chem. 2020, 338, 127535. [Google Scholar] [CrossRef]
- Silman, I.; Sussman, J.L. Acetylcholinesterase: How is structure related to function? Chem. Interactions 2008, 175, 3–10. [Google Scholar] [CrossRef]
- Lane, R.M.; Kivipelto, M.; Greig, N.H. Acetylcholinesterase and Its Inhibition in Alzheimer Disease. Clin. Neuropharmacol. 2004, 27, 141–149. [Google Scholar] [CrossRef]
- Lv, M.; Xu, H. Acetylcholinesterase: A Primary Target for Drugs and Insecticides. Mini-Rev. Med. Chem. 2017, 17, 1665–1676. [Google Scholar] [CrossRef]
- Ahmad, S.S.; Younis, K.; Philippe, J.; Aschner, M.; Khan, H. Strategic approaches to target the enzymes using natural compounds for the management of Alzheimer’s disease: A review. CNS Neurol. Disord. Drug Targets 2021, 21, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.A.; Dar, T.A.; Dar, P.A.; Ganie, S.A.; Kamal, M.A. A Current Perspective on the Inhibition of Cholinesterase by Natural and Synthetic Inhibitors. Curr. Drug Metab. 2017, 18, 96–111. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.P.; Kang, M.-G.; Lee, J.Y.; Oh, J.M.; Baek, S.C.; Leem, H.H.; Park, D.; Cho, M.-L.; Kim, H. Potent inhibition of acetylcholinesterase by sargachromanol I from Sargassum siliquastrum and by selected natural compounds. Bioorganic Chem. 2019, 89, 103043. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.; Santos, S.; Pereira, H. In Vitro Screening for Acetylcholinesterase Inhibition and Antioxidant Activity of Quercus suber Cork and Corkback Extracts. Evid. Based Complement. Altern. Med. 2020, 2020, 1–8. [Google Scholar] [CrossRef]
- Cespedes, C.L.; Muñoz, E.; Salazar, J.R.; Yamaguchi, L.; Werner, E.; Alarcon, J.; Kubo, I. Inhibition of cholinesterase activity by extracts, fractions and compounds from Calceolaria talcana and C. integrifolia (Calceolariaceae: Scrophulariaceae). Food Chem. Toxicol. 2013, 62, 919–926. [Google Scholar] [CrossRef]
- Krstić, D.Z.; Čolović, M.; Kralj, M.B.; Franko, M.; Krinulović, K.; Trebše, P.; Vasić, V. Inhibition of AChE by malathion and some structurally similar compounds. J. Enzym. Inhib. Med. Chem. 2008, 23, 562–573. [Google Scholar] [CrossRef] [Green Version]
- Hassan, S.H.A.; Fry, J.R.; Abu Bakar, M.F. Phytochemicals Content, Antioxidant Activity and Acetylcholinesterase Inhibition Properties of IndigenousGarcinia parvifoliaFruit. BioMed Res. Int. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Ahmad, S.S.; Waheed, T.; Rozeen, S.; Mahmood, S.; Kamal, M.A. Therapeutic Study of Phytochemicals Against Cancer and Alzheimer’s Disease Management. Curr. Drug Metab. 2020, 20, 1006–1013. [Google Scholar] [CrossRef]
- Calfio, C.; Gonzalez, A.; Singh, S.K.; Rojo, L.E.; Maccioni, R.B. The Emerging Role of Nutraceuticals and Phytochemicals in the Prevention and Treatment of Alzheimer’s Disease. J. Alzheimer’s Dis. 2020, 77, 33–51. [Google Scholar] [CrossRef]
- Gregory, J.; Vengalasetti, Y.; Bredesen, D.; Rao, R. Neuroprotective Herbs for the Management of Alzheimer’s Disease. Biomolecules 2021, 11, 543. [Google Scholar] [CrossRef]
- Karthika, C.; Appu, A.P.; Akter, R.; Rahman, H.; Tagde, P.; Ashraf, G.M.; Abdel-Daim, M.M.; Hassan, S.S.U.; Abid, A.; Bungau, S. Potential innovation against Alzheimer’s disorder: A tricomponent combination of natural antioxidants (vitamin E, quercetin, and basil oil) and the development of its intranasal delivery. Environ. Sci. Pollut. Res. 2022, 29, 10950–10965. [Google Scholar] [CrossRef] [PubMed]
- Hartman, R.E. Effects and mechanisms of actions of phytochemicals on Alzheimer rsquo s disease neuropathology. Front. Biosci. 2018, 10, 300–333. [Google Scholar] [CrossRef]
- Camandola, S.; Plick, N.; Mattson, M.P. Impact of Coffee and Cacao Purine Metabolites on Neuroplasticity and Neurodegenerative Disease. Neurochem. Res. 2018, 44, 214–227. [Google Scholar] [CrossRef] [PubMed]
- Fernando, W.M.; Somaratne, G.; Goozee, K.G.; Williams, S.; Singh, H.; Martins, R.N. Diabetes and Alzheimer’s Disease: Can Tea Phytochemicals Play a Role in Prevention? J. Alzheimer’s Dis. 2017, 59, 481–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hardy, J.; Allsop, D. Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol. Sci. 1991, 12, 383–388. [Google Scholar] [CrossRef]
- Taylor, H.A.; Przemylska, L.; Clavane, E.M.; Meakin, P.J. BACE1: More than just a beta-secretase. Obes. Rev. 2022, 23, e13430. [Google Scholar] [CrossRef]
- König, G.; Mönning, U.; Czech, C.; Prior, R.; Banati, R.; Schreiter-Gasser, U.; Bauer, J.; Masters, C.; Beyreuther, K. Identification and differential expression of a novel alternative splice isoform of the beta A4 amyloid precursor protein (APP) mRNA in leukocytes and brain microglial cells. J. Biol. Chem. 1992, 267, 10804–10809. [Google Scholar] [CrossRef]
- Norstrom, E. Metabolic processing of the amyloid precursor protein––New pieces of the Alzheimer’s puzzle. Discov. Med. 2017, 23, 269–276. [Google Scholar]
- Sathya, M.; Premkumar, P.; Karthick, C.; Moorthi, P.; Jayachandran, K.; Anusuyadevi, M. BACE1 in Alzheimer’s disease. Clin. Chim. Acta 2012, 414, 171–178. [Google Scholar] [CrossRef]
- He, X.; Zhu, G.; Koelsch, G.; Rodgers, K.K.; Zhang, X.C.; Tang, J. Biochemical and Structural Characterization of the Interaction of Memapsin 2 (β-Secretase) Cytosolic Domain with the VHS Domain of GGA Proteins. Biochemistry 2003, 42, 12174–12180. [Google Scholar] [CrossRef]
- Hrabinova, M.; Pejchal, J.; Kucera, T.; Juna, D.; Schmidt, M.; Soukup, O. Is It the Twilight of BACE1 Inhibitors? Curr. Neuropharmacol. 2020, 19, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, M.E.; Wang, W.; Song, L.; Lee, J.; Zhang, L.; Wong, G.; Wang, L.; Parker, E. Measuring human β-secretase (BACE1) activity using homogeneous time-resolved fluorescence. Anal. Biochem. 2003, 319, 49–55. [Google Scholar] [CrossRef]
- Eketjäll, S.; Janson, J.; Kaspersson, K.; Bogstedt, A.; Jeppsson, F.; Fälting, J.; Haeberlein, S.B.; Kugler, A.R.; Alexander, R.C.; Cebers, G. AZD3293: A Novel, Orally Active BACE1 Inhibitor with High Potency and Permeability and Markedly Slow Off-Rate Kinetics. J. Alzheimer’s Dis. 2016, 50, 1109–1123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neumann, U.; Ufer, M.; Jacobson, L.; Rouzade-Dominguez, M.; Huledal, G.; Kolly, C.; Lüönd, R.M.; Machauer, R.; Veenstra, S.J.; Hurth, K.; et al. The BACE -1 inhibitor CNP 520 for prevention trials in Alzheimer’s disease. EMBO Mol. Med. 2018, 10, e9316. [Google Scholar] [CrossRef]
- Kennedy, M.E.; Stamford, A.W.; Chen, X.; Cox, K.; Cumming, J.N.; Dockendorf, M.F.; Egan, M.; Ereshefsky, L.; Hodgson, R.A.; Hyde, L.A.; et al. The BACE1 inhibitor verubecestat (MK-8931) reduces CNS β-amyloid in animal models and in Alzheimer’s disease patients. Sci. Transl. Med. 2016, 8, 363ra150. [Google Scholar] [CrossRef]
- Sakamoto, K.; Matsuki, S.; Matsuguma, K.; Yoshihara, T.; Uchida, N.; Azuma, F.; Russell, M.; Hughes, G.; Haeberlein, S.B.; Alexander, R.C.; et al. BACE1 Inhibitor Lanabecestat (AZD3293) in a Phase 1 Study of Healthy Japanese Subjects: Pharmacokinetics and Effects on Plasma and Cerebrospinal Fluid Aβ Peptides. J. Clin. Pharmacol. 2017, 57, 1460–1471. [Google Scholar] [CrossRef]
- Timmers, M.; Van Broeck, B.; Ramael, S.; Slemmon, J.; De Waepenaert, K.; Russu, A.; Bogert, J.; Stieltjes, H.; Shaw, L.M.; Engelborghs, S.; et al. Profiling the dynamics of CSF and plasma Aβ reduction after treatment with JNJ-54861911, a potent oral BACE inhibitor. Alzheimer’s Dementia: Transl. Res. Clin. Interv. 2016, 2, 202–212. [Google Scholar] [CrossRef] [Green Version]
- Lopez, C.L.; Tariot, P.N.; Caputo, A.; Langbaum, J.B.; Liu, F.; Riviere, M.; Langlois, C.; Rouzade-Dominguez, M.; Zalesak, M.; Hendrix, S.; et al. The Alzheimer’s Prevention Initiative Generation Program: Study design of two randomized controlled trials for individuals at risk for clinical onset of Alzheimer’s disease. Alzheimer’s Dementia: Transl. Res. Clin. Interv. 2019, 5, 216–227. [Google Scholar] [CrossRef]
- Zhang, C. Natural compounds that modulate BACE1-processing of amyloid-beta precursor protein in Alzheimer’s disease. Discov. Med. 2012, 14, 189–197. [Google Scholar]
- Youn, K.; Park, J.-H.; Lee, S.; Lee, S.; Lee, J.; Yun, E.-Y.; Jeong, W.-S.; Jun, M. BACE1 Inhibition by Genistein: Biological Evaluation, Kinetic Analysis, and Molecular Docking Simulation. J. Med. Food 2018, 21, 416–420. [Google Scholar] [CrossRef]
- Naushad, M.; Durairajan, S.S.K.; Bera, A.K.; Senapati, S.; Li, M. Natural Compounds with Anti-BACE1 Activity as Promising Therapeutic Drugs for Treating Alzheimer’s Disease. Planta Med. 2019, 85, 1316–1325. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Paudel, P.; Seong, S.H.; Kim, J.A.; Jung, H.A.; Choi, J.S. Computational insights into β-site amyloid precursor protein enzyme 1 (BACE1) inhibition by tanshinones and salvianolic acids from Salvia miltiorrhiza via molecular docking simulations. Comput. Biol. Chem. 2018, 74, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Youdim, M.B.H.; Edmondson, D.; Tipton, K.F. The therapeutic potential of monoamine oxidase inhibitors. Nat. Rev. Neurosci. 2006, 7, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Shih, J.C.; Chen, K.; Ridd, M.J. MONOAMINE OXIDASE: From Genes to Behavior. Annu. Rev. Neurosci. 1999, 22, 197–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tipton, K.F.; Boyce, S.; O’Sullivan, J.; Davey, G.P.; Healy, J. Monoamine Oxidases: Certainties and Uncertainties. Curr. Med. Chem. 2004, 11, 1965–1982. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Dwivedi, A.R.; Sarkar, B.; Gupta, S.K.; Krishnamurthy, S.; Mantha, A.K.; Parkash, J.; Kumar, V. 4,6-Diphenylpyrimidine Derivatives as Dual Inhibitors of Monoamine Oxidase and Acetylcholinesterase for the Treatment of Alzheimer’s Disease. ACS Chem. Neurosci. 2018, 10, 252–265. [Google Scholar] [CrossRef]
- Orhan, I.E. Potential of Natural Products of Herbal Origin as Monoamine Oxidase Inhibitors. Curr. Pharm. Des. 2015, 22, 268–276. [Google Scholar] [CrossRef]
- Juvekar, A.R.; Khatri, D. Kinetics of inhibition of monoamine oxidase using curcumin and ellagic acid. Pharmacogn. Mag. 2016, 12, 116–120. [Google Scholar] [CrossRef] [Green Version]
- Baek, S.C.; Ryu, H.W.; Kang, M.-G.; Lee, H.; Park, D.; Cho, M.-L.; Oh, S.-R.; Kim, H. Selective inhibition of monoamine oxidase A by chelerythrine, an isoquinoline alkaloid. Bioorganic Med. Chem. Lett. 2018, 28, 2403–2407. [Google Scholar] [CrossRef]
- Chaurasiya, N.D.; Midiwo, J.; Pandey, P.; Bwire, R.N.; Doerksen, R.J.; Muhammad, I.; Tekwani, B.L. Selective Interactions of O-Methylated Flavonoid Natural Products with Human Monoamine Oxidase-A and -B. Molecules 2020, 25, 5358. [Google Scholar] [CrossRef]
- Santos, G.; Giraldez-Alvarez, L.D.; Avila-Rodríguez, M.; Capani, F.; Galembeck, E.; Neto, A.G.; Barreto, G.E.; Andrade, B. SUR1 Receptor Interaction with Hesperidin and Linarin Predicts Possible Mechanisms of Action of Valeriana officinalis in Parkinson. Front. Aging Neurosci. 2016, 8, 97. [Google Scholar] [CrossRef] [PubMed]
- González-Muñoz, G.C.; Arce, M.P.; Pérez, C.; Romero, A.; Villarroya, M.; Lopez, M.G.; Conde, S.; Rodríguez-Franco, M.I. Dibenzo[1,4,5]thiadiazepine: A hardly-known heterocyclic system with neuroprotective properties of potential usefulness in the treatment of neurodegenerative diseases. Eur. J. Med. Chem. 2014, 81, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, D.; Kaur Dogra, A.; Tahrani, A.; Herrmann, F.; Wink, M. Extracts from Traditional Chinese Medicinal Plants Inhibit Acetylcholinesterase, a Known Alzheimer’s Disease Target. Molecules 2016, 21, 1161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, A.K.; Singh, S.K.; Nandi, M.K.; Mishra, G.; Maurya, A.; Rai, A.; Rai, G.K.; Awasthi, R.; Sharma, B.; Kulkarni, G.T. Berberine: A Plant-derived Alkaloid with Therapeutic Potential to Combat Alzheimer’s disease. Central Nerv. Syst. Agents Med. Chem. 2019, 19, 154–170. [Google Scholar] [CrossRef] [PubMed]
- Howes, M.-J.R.; Simmonds, M.S. The role of phytochemicals as micronutrients in health and disease. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Larit, F.; Elokely, K.M.; Chaurasiya, N.D.; Benyahia, S.; Nael, M.A.; Leon, F.; Abu-Darwish, M.S.; Efferth, T.; Wang, Y.-H.; Belouahem-Abed, D.; et al. Inhibition of human monoamine oxidase A and B by flavonoids isolated from two Algerian medicinal plants. Phytomedicine 2017, 40, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Quispe, C.; Imran, M.; Rauf, A.; Nadeem, M.; Gondal, T.A.; Ahmad, B.; Atif, M.; Mubarak, M.S.; Sytar, O.; et al. Genistein: An Integrative Overview of Its Mode of Action, Pharmacological Properties, and Health Benefits. Oxidative Med. Cell. Longev. 2021, 2021, 1–36. [Google Scholar] [CrossRef]
- Menze, E.T.; Tadros, M.G.; Abdel-Tawab, A.M.; Khalifa, A. Potential neuroprotective effects of hesperidin on 3-nitropropionic acid-induced neurotoxicity in rats. NeuroToxicology 2012, 33, 1265–1275. [Google Scholar] [CrossRef]
- Chakraborty, S.; Bandyopadhyay, J.; Chakraborty, S.; Basu, S. Multi-target screening mines hesperidin as a multi-potent inhibitor: Implication in Alzheimer’s disease therapeutics. Eur. J. Med. Chem. 2016, 121, 810–822. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and Anti-Inflammatory Properties of the Citrus Flavonoids Hesperidin and Hesperetin: An Updated Review of their Molecular Mechanisms and Experimental Models. Phytotherapy Res. 2014, 29, 323–331. [Google Scholar] [CrossRef]
- Carmona, V.; Martín-Aragón, S.; Goldberg, J.; Schubert, D.; Bermejo-Bescós, P. Several targets involved in Alzheimer’s disease amyloidogenesis are affected by morin and isoquercitrin. Nutr. Neurosci. 2018, 23, 575–590. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.A.; Wang, X.-Q.; Yan, H.-C. Morin hydrate: A comprehensive review on novel natural dietary bioactive compound with versatile biological and pharmacological potential. Biomed. Pharmacother. 2021, 138, 111511. [Google Scholar] [CrossRef] [PubMed]
- Shal, B.; Ding, W.; Ali, H.; Kim, Y.S.; Khan, S. Anti-neuroinflammatory Potential of Natural Products in Attenuation of Alzheimer’s Disease. Front. Pharmacol. 2018, 9, 548. [Google Scholar] [CrossRef] [PubMed]
- Dar, N.J.; Hamid, A.; Ahmad, M. Pharmacologic overview of Withania somnifera, the Indian Ginseng. Exp. 2015, 72, 4445–4460. [Google Scholar] [CrossRef]
- Fang, J.; Liu, R.; Tian, Q.; Hong, X.-P.; Wang, S.-H.; Cao, F.-Y.; Pan, X.-P.; Wang, J.-Z. Dehydroevodiamine attenuates calyculin A-induced tau hyperphos-phorylation in rat brain slices. Acta Pharmacol. Sin. 2007, 28, 1717–1723. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.-Y.; Zheng, W.; Wang, T.; Xie, J.-W.; Wang, S.-L.; Zhao, B.-L.; Teng, W.-P.; Wang, Z.-Y. Huperzine A Activates Wnt/β-Catenin Signaling and Enhances the Nonamyloidogenic Pathway in an Alzheimer Transgenic Mouse Model. Neuropsychopharmacology 2011, 36, 1073–1089. [Google Scholar] [CrossRef]
- Yang, Z.-D.; Zhang, X.; Du, J.; Ma, Z.-J.; Guo, F.; Li, S.; Yao, X.-J. An aporphine alkaloid from Nelumbo nucifera as an acetylcholinesterase inhibitor and the primary investigation for structure–activity correlations. Nat. Prod. Res. 2012, 26, 387–392. [Google Scholar] [CrossRef]
- Xian, Y.-F.; Mao, Q.-Q.; Wu, J.C.; Su, Z.-R.; Chen, J.-N.; Lai, X.-P.; Ip, S.-P.; Lin, Z.-X. Isorhynchophylline Treatment Improves the Amyloid-β-Induced Cognitive Impairment in Rats via Inhibition of Neuronal Apoptosis and Tau Protein Hyperphosphorylation. J. Alzheimer’s Dis. 2014, 39, 331–346. [Google Scholar] [CrossRef]
- Haj, E.; Losev, Y.; KrishnaKumar, V.G.; Pichinuk, E.; Engel, H.; Raveh, A.; Gazit, E.; Segal, D. Integrating in vitro and in silico approaches to evaluate the “dual functionality” of palmatine chloride in inhibiting and disassembling Tau-derived VQIVYK peptide fibrils. Biochim. Et Biophys. Acta (BBA) Gen. Subj. 2018, 1862, 1565–1575. [Google Scholar] [CrossRef]
- López, S.; Bastida, J.; Viladomat, F.; Codina, C. Acetylcholinesterase inhibitory activity of some Amaryllidaceae alkaloids and Narcissus extracts. Life Sci. 2002, 71, 2521–2529. [Google Scholar] [CrossRef]
- Rollinger, J.M.; Schuster, D.; Baier, E.; Ellmerer, E.P.; Langer, T.; Stuppner, H. Taspine: Bioactivity-Guided Isolation and Molecular Ligand−Target Insight of a Potent Acetylcholinesterase Inhibitor from Magnolia x soulangiana. J. Nat. Prod. 2006, 69, 1341–1346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, S.; Khan, M.; Ahmad, K.; Lim, J.-H.; Shaikh, S.; Lee, E.-J.; Choi, I. Biocomputational Screening of Natural Compounds against Acetylcholinesterase. Molecules 2021, 26, 2641. [Google Scholar] [CrossRef] [PubMed]
- Qi, T.; Li, H.; Li, S. Indirubin improves antioxidant and anti-inflammatory functions in lipopolysaccharide-challenged mice. Oncotarget 2017, 8, 36658–36663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, S.S.; Sinha, M.; Ahmad, K.; Khalid, M.; Choi, I. Study of Caspase 8 Inhibition for the Management of Alzheimer’s Disease: A Molecular Docking and Dynamics Simulation. Molecules 2020, 25, 2071. [Google Scholar] [CrossRef]
- Ahmad, S.S.; Akhtar, S.; Rizvi, S.M.D.; Kamal, M.A.; Sayeed, U.; Khan, M.K.A.; Siddiqui, M.H.; Arif, J.M. Screening and Elucidation of Selected Natural Compounds for Anti- Alzheimer’s Potential Targeting BACE-1 Enzyme: A Case Computational Study. Curr. Comput. Aided-Drug Des. 2017, 13, 311–318. [Google Scholar] [CrossRef]
- Kou, X.; Chen, N. Resveratrol as a Natural Autophagy Regulator for Prevention and Treatment of Alzheimer’s Disease. Nutrients 2017, 9, 927. [Google Scholar] [CrossRef] [Green Version]
- Tang, M.; Taghibiglou, C. The Mechanisms of Action of Curcumin in Alzheimer’s Disease. J. Alzheimer’s Dis. 2017, 58, 1003–1016. [Google Scholar] [CrossRef]
- Ji, H.-F.; Zhang, H.-Y. Multipotent natural agents to combat Alzheimer’s disease. Functional spectrum and structural features. Acta Pharmacol. Sin. 2008, 29, 143–151. [Google Scholar] [CrossRef] [Green Version]

| S.No. | Compound | Pub Chem ID | Properties | Work Type | Therapeutic Actions/Function | Reference |
|---|---|---|---|---|---|---|
| 1. | Apigenin | 5280443 | Antioxidant and antiinflammatory | in vitro | Decrease Aβ burden | [1,81] |
| in vivo (mouse model) | induced neurogenesis | |||||
| 2. | Dibenzo[1,4,5]thiadiazepine | 71358659 | antioxidant | in vitro (neuroblastoma cells) | neuroprotective and antioxidant properties | [82] |
| 3. | Berberine | 2353 | anti-inflammatory | in vitro (rat model) | inhibition of AChE | [83,84] |
| 4. | Catechin | 9064 | antioxidant | in vivo (rat model of AD) | inhibition of AChE | [85] |
| 5. | Genistein | 5280961 | Antioxidant and anti-inflammatory | in silico and in vitro (model of AD) | inhibition of human monoamine oxidase A and B | [86,87] |
| 6. | Hesperidin | 10621 | antioxidant and anti-inflammatory | in silico and in vivo (rat model of AD) | inhibition of BACE1 and Aβ aggregation | [88,89,90] |
| 7. | Morin | 5281670 | antioxidant, anti-inflammatory and neuroprotective | (MC65 cells) | BACE1, γ-secretase, Aβ fibrillogenesis, amyloid plaque, and tau hyperphosphorylation | [91,92] |
| 8. | Naringenin | 932 | anti-inflammatory | in vitro (rat model) | decrease inflammatory cytokines | [93] |
| 9. | Withanone | 21679027 | neuroprotective | in vivo (rat model of AD) | decrease Aβ fibril formation | [1,94] |
| 10. | Dehydroevodiamine | 9817839 | anti-inflammatory | rat brain slices against AD | inhibition of tau phosphorylation | [95] |
| 11. | Huperzine A | 449069 | neuroprotective | Alzheimer transgenic mouse model | reduces the level of Aβ | [96] |
| 12. | N-methylasimilobine | 197017 | Antioxidant | in vitro | inhibition of AChE | [97] |
| 13. | Isorhynchophylline | 3037048 | neuroprotective | rat model | restore Aβ–induced cognitive impairment | [98] |
| 14. | Palmatine | 19009 | anti-inflammatory and anti-neurodegenerative | in vitro, in vivo | inhibit tau aggregation | [99] |
| 15. | Sanguinarine | 5154 | Antitumor properties | in vitro | inhibition of AChE | [100] |
| 16. | Taspine | 215159 | anti-inflammatory | in vitro | inhibition of AChE | [101] |
| 17. | Indirubin | 10177 | antioxidant and anti-inflammatory | in silico | inhibition of AChE | [102,103] |
| 18. | Rutaecarpine | 65752 | anti-inflammatory | in silico | inhibition of Caspase 8 | [104] |
| 19. | Ajmalicine | 441975 | antihypertensive | in silico | inhibition of BACE1 | [105] |
| 20. | Resveratrol | 445154 | Antioxidant | in vitro and in vivo (AD models) | neuroprotective role in AD | [106] |
| 21. | Curcumin | 969516 | antioxidant, anticarcinogenic, anti-inflammatory, antiangiogenic | in vivo and in vitro | inhibition of AChE | [107] |
| 22. | Resveratrol | 445154 | Antioxidant | in vitro | inhibition of MAOA for AD treatment | [108] |
| 23. | Genistein | 5280961 | Antioxidant and anti-inflammatory | in vitro | anti-AD activities | [70] |
| 24. | Quercetin | 5280343 | Antioxidant | - | Anti-BACE1 Activity | [71] |
| 25. | Ellagic acid | 5281855 | antioxidant, antimutagenic, and anticancer properties | in vitro | MAO inhibitor for ND treatment | [78] |
| 26. | Chelerythrine | 2703 | anti-inflammatory | in vitro | MAO-A inhibitor | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Kong, X.; Chen, Z.; Wang, G.; Zhang, J.; Wang, J. Role of Natural Compounds and Target Enzymes in the Treatment of Alzheimer’s Disease. Molecules 2022, 27, 4175. https://doi.org/10.3390/molecules27134175
Wang S, Kong X, Chen Z, Wang G, Zhang J, Wang J. Role of Natural Compounds and Target Enzymes in the Treatment of Alzheimer’s Disease. Molecules. 2022; 27(13):4175. https://doi.org/10.3390/molecules27134175
Chicago/Turabian StyleWang, Shanshan, Xianbo Kong, Zhangjing Chen, Guopin Wang, Juan Zhang, and Jing Wang. 2022. "Role of Natural Compounds and Target Enzymes in the Treatment of Alzheimer’s Disease" Molecules 27, no. 13: 4175. https://doi.org/10.3390/molecules27134175





