The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: ‘Talented’ Taxa from the Amaryllidaceae
Abstract
1. Introduction
2. The Genus Tulbaghia
2.1. Botanical Description
2.2. Geographical Distribution and Traditional Uses of Tulbaghia Species
2.3. Phytochemistry of Tulbaghia
2.4. Pharmacological Studies of Tulbaghia Species
2.4.1. Antimicrobial and Antiparasitic Activity
2.4.2. Anticancer Activity
2.4.3. Antioxidant Activity
2.4.4. Antidiabetic, Anticardiovascular and Antithrombogenic Activity
2.4.5. Miscellaneous Pharmacological Activity
3. The Genus Allium
3.1. Botanical Description
3.2. Traditional Uses of Genus Allium
3.3. Phytochemistry of Allium
3.4. Pharmacological Effects of Allium
3.4.1. Antimicrobial Activities
3.4.2. Antioxidant Properties
3.4.3. Anti-Inflammatory Properties
3.4.4. Anticancer Activity
3.4.5. Other Pharmacological Effects of Allium Species
4. The Genus Crinum
4.1. Geographical Distribution of Crinum
4.2. Traditional Uses of Crinum
4.3. Phytochemistry of Crinum
4.4. Pharmacological Activities of Crinum
4.4.1. Anti-Inflammatory and Analgesic Effects
4.4.2. Anticancer and Cytotoxicity Effects
4.4.3. Antimicrobial Properties
4.4.4. Antioxidant Properties
4.4.5. Other Pharmacological Properties
5. The Genus Cyrtanthus
5.1. Botanical Description
5.2. Geographical Distribution
5.3. Traditional Uses
5.4. Phytochemistry of Cyrtanthus
5.5. Pharmacological Activities
5.5.1. Antioxidant Activity
5.5.2. Anti-Inflammatory Activity
5.5.3. Inhibition of Acetylcholinesterase
5.5.4. Antimicrobial Activity
5.5.5. Cytotoxic Activity
5.5.6. Miscellaneous Pharmacological Activities
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Barile, E.; Capasso, R.; Izzo, A.A.; Lanzotti, V.; Sajjadi, S.E.; Zolfaghari, B. Structure-activity relationships for saponins from Allium hirtifolium and Allium elburzense and their antispasmodic activity. Planta Med. 2005, 71, 1010–1018. [Google Scholar] [CrossRef] [PubMed]
- Chase, M.W.; Reveal, J.L.; Fay, M.F. A subfamilial classification for the expanded asparagalean families Amaryllidaceae, Asparagaceae and Xanthorrhoeaceae. Bot. J. Linn. Soc. 2009, 161, 132–136. [Google Scholar] [CrossRef]
- Takos, A.M.; Rook, F. Towards a Molecular Understanding of the Biosynthesis of Amaryllidaceae Alkaloids in Support of Their Expanding Medical Use. Int. J. Mol. Sci. 2013, 14, 11713–11741. [Google Scholar] [CrossRef] [PubMed]
- Vosa, C.G.; Siebert, S.J.; Van Wyk, A.E.B. Micromorphology and cytology of Prototulbaghia siebertii, with notes on its taxonomic significance. Upsp. Inst. Repos. 2011, 41, 311–314. [Google Scholar]
- Elgorashi, E.E.; van Staden, J. Bioactivity and Bioactive Compounds of African Amaryllidaceae; ACS Publications: Washington, DC, USA, 2009; ISBN 1947-5918. [Google Scholar]
- Fenwick, G.R.; Hanley, A.B.; Whitaker, J.R. The genus allium—Part 1. Crit. Rev. Food Sci. Nutr. 1985, 22, 199–271. [Google Scholar] [CrossRef]
- Fenwick, G.R.; Hanley, A.B. The genus allium—Part 2. Crit. Rev. Food Sci. Nutr. 1985, 22, 273–377. [Google Scholar] [CrossRef]
- Aremu, A.O.; Van Staden, J. The genus Tulbaghia (Alliaceae)—A review of its ethnobotany, pharmacology, phytochemistry and conservation needs. J. Ethnopharmacol. 2013, 149, 387–400. [Google Scholar] [CrossRef]
- Styger, G.; Aboyade, O.M.; Gibson, D.; Hughes, G. Tulbaghia—A Southern African Phytomedicine. J. Altern. Complement. Med. 2016, 22, 255–261. [Google Scholar] [CrossRef]
- Jagtap, U.B.; Lekhak, M.M.; Fulzele, D.P.; Yadav, S.R.; Bapat, V.A. Analysis of selected Crinum species for galanthamine alkaloid: An anti-Alzheimer drug. Curr. Sci. 2014, 107, 2008–2010. [Google Scholar]
- Nair, J.J.; Aremu, A.O.; Van Staden, J. Isolation of narciprimine from Cyrtanthus contractus (Amaryllidaceae) and evaluation of its acetylcholinesterase inhibitory activity. J. Ethnopharmacol. 2011, 137, 1102–1106. [Google Scholar] [CrossRef]
- Torras-Claveria, L.; Berkov, S.; Codina, C.; Viladomat, F.; Bastida, J. Daffodils as potential crops of galanthamine. Assessment of more than 100 ornamental varieties for their alkaloid content and acetylcholinesterase inhibitory activity. Ind. Crops Prod. 2013, 43, 237–244. [Google Scholar] [CrossRef]
- Shang, A.; Cao, S.-Y.; Xu, X.-Y.; Gan, R.-Y.; Tang, G.-Y.; Corke, H.; Mavumengwana, V.; Li, H.-B. Bioactive Compounds and Biological Functions of Garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Rani, R.; Chaturvedi, M.; Rohilla, P.; Yadav, J.P. In silico and in vitro approach of Allium cepa and isolated quercetin against MDR bacterial strains and Mycobacterium smegmatis. S. Afr. J. Bot. 2019, 124, 29–35. [Google Scholar] [CrossRef]
- Stoica, F.; Aprodu, I.; Enachi, E.; Stănciuc, N.; Condurache, N.N.; Duță, D.E.; Bahrim, G.E.; Râpeanu, G. Bioactive’s Characterization, Biological Activities, and In Silico Studies of Red Onion (Allium cepa L.) Skin Extracts. Plants 2021, 10, 2330. [Google Scholar] [CrossRef] [PubMed]
- Albishi, T.; John, J.A.; Al-Khalifa, A.S.; Shahidi, F. Antioxidant, anti-inflammatory and DNA scission inhibitory activities of phenolic compounds in selected onion and potato varieties. J. Funct. Foods 2013, 5, 930–939. [Google Scholar] [CrossRef]
- Benmalek, Y.; Yahia, O.A.; Belkebir, A.; Fardeau, M.-L. Anti-microbial and anti-oxidant activities of Illicium verum, Crataegus oxyacantha ssp monogyna and Allium cepa red and white varieties. Bioengeineered 2013, 4, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Elberry, A.A.; Mufti, S.; Al-Maghrabi, J.; Abdel Sattar, E.; Ghareib, S.A.; Mosli, H.A.; Gabr, S.A. Immunomodulatory effect of red onion (Allium cepa Linn) scale extract on experimentally induced atypical prostatic hyperplasia in Wistar rats. Mediat. Inflamm. 2014, 2014, 640746. [Google Scholar] [CrossRef]
- Lanzotti, V. The analysis of onion and garlic. J. Chromatogr. 2006, 1112, 3–22. [Google Scholar] [CrossRef]
- Rouf, R.; Uddin, S.J.; Sarker, D.K.; Islam, M.T.; Ali, E.S.; Shilpi, J.A.; Nahar, L.; Tiralongo, E.; Sarker, S.D. Antiviral potential of garlic (Allium sativum) and its organosulfur compounds: A systematic update of pre-clinical and clinical data. Trends Food Sci. Technol. 2020, 104, 219–234. [Google Scholar] [CrossRef]
- Harazem, R.; El Rahman, S.A.; El-Kenawy, A. Evaluation of Antiviral Activity of Allium cepa and Allium sativum Extracts Against Newcastle Disease Virus. Alex. J. Vet. Sci. 2019, 61, 108–118. [Google Scholar] [CrossRef]
- Elmi, T.; Hajialiani, F.; Asadi, M.R.; Sadeghi, S.; Namazi, M.J.; Tabatabaie, F.; Zamani, Z. Antimalarial effects of the hydroalcoholic extract of Allium paradoxum in vitro and in vivo. J. Parasit. Dis. 2021, 45, 1055–1064. [Google Scholar] [CrossRef]
- Ruslan, M.S.; Baba, M.S. In vivo antimalarial assessment and toxicity evaluation of garlic (Allium sativum) in plasmodium berghei NK65-induced mice. Malays. Appl. Biol. 2018, 47, 17–24. [Google Scholar]
- Syaban, M.F.R.; Rachman, H.A.; Arrahman, A.D.; Hudayana, N.; Khamid, J.P.; Pratama, F.A. Allium sativum as antimalaria agent via falciapin protease-2 inhibitor mechanism: Molecular docking perspective. Clin. Res. J. Intern. Med. 2021, 2, 130–135. [Google Scholar] [CrossRef]
- Upadhyay, R.K. Nutritional and therapeutic potential of Allium vegetables. J. Nutr. Ther. 2017, 6, 18–37. [Google Scholar] [CrossRef]
- Thomson, M.; Ali, M. Garlic [Allium sativum]: A review of its potential use as an anti-cancer agent. Curr. Cancer Drug Targets 2003, 3, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Corea, G.; Fattorusso, E.; Lanzotti, V.; Capasso, R.; Izzo, A.A. Antispasmodic saponins from bulbs of red onion, Allium cepa L. var. Tropea. J. Agric. Food Chem. 2005, 53, 935–940. [Google Scholar] [CrossRef]
- Galmarini, C.R.; Goldman, I.L.; Havey, M.J. Genomics Genetic analyses of correlated solids, flavor, and health-enhancing traits in onion (Allium cepa L.). Mol. Genet. Genom. 2001, 265, 543–551. [Google Scholar] [CrossRef]
- Takahashi, M.; Shibamoto, T. Chemical compositions and antioxidant/anti-inflammatory activities of steam distillate from freeze-dried onion (Allium cepa L.) sprout. J. Agric. Food Chem. 2008, 56, 10462–10467. [Google Scholar] [CrossRef]
- Nishimura, H.; Wijaya, C.H.; Mizutani, J. Volatile flavor components and antithrombotic agents: Vinyldithiins from Allium victorialis L. J. Agric. Food Chem. 1988, 36, 563–566. [Google Scholar] [CrossRef]
- Brace, L.D. Cardiovascular benefits of garlic (Allium sativum L.). J. Cardiovasc. Nurs. 2002, 16, 33–49. [Google Scholar] [CrossRef]
- Ali, M.; Thomson, M.; Afzal, M. Garlic and onions: Their effect on eicosanoid metabolism and its clinical relevance. Prostaglandins Leukot. Essent. Fat. Acids 2000, 62, 55–73. [Google Scholar] [CrossRef] [PubMed]
- Sabiu, S.; Madende, M.; Ajao, A.A.; Aladodo, R.A.; Nurain, I.O.; Ahmad, J.B. The Genus Allium (Amaryllidaceae: Alloideae): Features, Phytoconstituents, and Mechanisms of Antidiabetic Potential of Allium cepa and Allium sativum, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128138229. [Google Scholar]
- Muñoz-Torrero López-Ibarra, D. Recent Advances in Pharmaceutical Sciences I; Transworld Research Network: Trivandrum, India, 2011; ISBN 8178955288. [Google Scholar]
- Akash, M.S.H.; Rehman, K.; Chen, S. Spice plant Allium cepa: Dietary supplement for treatment of type 2 diabetes mellitus. Nutrition 2014, 30, 1128–1137. [Google Scholar] [CrossRef] [PubMed]
- Corzo-Martínez, M.; Corzo, N.; Villamiel, M. Biological properties of onions and garlic. Trends Food Sci. Technol. 2007, 18, 609–625. [Google Scholar] [CrossRef]
- Kumar, K.P.S.; Debjit, B.; Pankaj, T. Allium cepa: A traditional medicinal herb and its health benefits. J. Chem. Pharm. Res. 2010, 2, 283–291. [Google Scholar]
- Shri, R.; Bora, K.S. Neuroprotective effect of methanolic extracts of Allium cepa on ischemia and reperfusion-induced cerebral injury. Fitoterapia 2008, 79, 86–96. [Google Scholar] [CrossRef]
- Kongkwamcharoen, C.; Itharat, A.; Pipatrattanaseree, W.; Ooraikul, B. Effects of Various Preextraction Treatments of Crinum asiaticum Leaf on Its Anti-Inflammatory Activity and Chemical Properties. Evid. Based. Complement. Alternat. Med. 2021, 2021, 8850744. [Google Scholar] [CrossRef] [PubMed]
- Fennell, C.W.; Van Staden, J. Crinum species in traditional and modern medicine. J. Ethnopharmacol. 2001, 78, 15–26. [Google Scholar] [CrossRef]
- Maroyi, A. Ethnobotanical, phytochemical and pharmacological properties of Crinum bulbispermum (Burm f) Milne-Redh and Schweick (Amaryllidaceae). Trop. J. Pharm. Res. 2016, 15, 2497–2506. [Google Scholar] [CrossRef]
- Takaidza, S.; Pillay, M.; Mtunzi, F.M. Biological activities of species in the genus Tulbaghia: A review. Afr. J. Biotechnol. 2015, 14, 3037–3043. [Google Scholar]
- Herrera, M.R.; Machocho, A.K.; Nair, J.J.; Campbell, W.E.; Brun, R.; Viladomat, F.; Codina, C.; Bastida, J. Alkaloids from Cyrtanthus elatus. Fitoterapia 2001, 72, 444–448. [Google Scholar] [CrossRef]
- Nair, J.J.; van Staden, J. Chemical and biological studies of the South African Amaryllidaceae genera Crinum, Ammocharis, Amaryllis, Cyrtanthus and Brunsvigia. S. Afr. J. Bot. 2021, 142, 467–476. [Google Scholar] [CrossRef]
- Heinrich, M.; Teoh, H.L. Galanthamine from snowdrop—the development of a modern drug against Alzheimer’s disease from local Caucasian knowledge. J. Ethnopharmacol. 2004, 92, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Nair, J.J.; van Staden, J. Pharmacological and toxicological insights to the South African Amaryllidaceae. Food Chem. Toxicol. 2013, 62, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Govaerts, R. World Checklist of Selected Plant Species. Facilitated by the Royal Botanic Gardens, Kew. 2015. Available online: https://wcsp.science.kew.org/about.do (accessed on 25 March 2022).
- Kubec, R.; Velíšek, J.; Musah, R.A. The amino acid precursors and odor formation in society garlic (Tulbaghia violacea Harv.). Phytochemistry 2002, 60, 21–25. [Google Scholar] [CrossRef]
- Dillon, H.; Nelson, E.C. Tulbaghia leucantha: Alliaceae. Kew Mag. 1991, 8, 12–15. [Google Scholar] [CrossRef]
- Makunga, N.P. Medicinal Plants of South Africa; Briza Publications: Pretoria, South Africa, 2010; Volume 105. [Google Scholar]
- Van Wyk, B.E. The potential of South African plants in the development of new food and beverage products. S. Afr. J. Bot. 2011, 77, 857–868. [Google Scholar] [CrossRef]
- Raji, I.A.; Obikeze, K.; Mugabo, P.E. Potential beneficial effects of tulbaghia violacea william henry harvey (Alliaceae) on cardiovascular system—A Review. Trop. J. Pharm. Res. 2015, 14, 1111–1117. [Google Scholar] [CrossRef]
- Pooley, E. A Field Guide to the Wild Flowers of KwaZulu-Natal and the Eastern Region. Natal Flora Publ. Trust. Pg 2005, 93, 630. [Google Scholar]
- Sander, T.; Freyss, J.; Von Korff, M.; Rufener, C. DataWarrior: An open-source program for chemistry aware data visualization and analysis. J. Chem. Inf. Model. 2015, 55, 460–473. [Google Scholar] [CrossRef]
- Pino, J.A.; Quijano-Celís, C.E.; Fuentes, V. Volatile compounds of tulbaghia violacea harv. J. Essent. Oil-Bear. Plants 2008, 11, 203–207. [Google Scholar] [CrossRef]
- Ranglová, K.; Krejčová, P.; Kubec, R. The effect of storage and processing on antimicrobial activity of Tulbaghia violacea. S. Afr. J. Bot. 2015, 97, 159–164. [Google Scholar] [CrossRef]
- Smith, S.; Stansbie, J. Flora of Tropical East Africa. Crown Agents for Oversea Governments and Administration; CRC Press: Boca Raton, FL, USA, 2003; p. 230. [Google Scholar]
- Takaidza, S.; Mtunzi, F.; Pillay, M. Analysis of the phytochemical contents and antioxidant activities of crude extracts from Tulbaghia species. J. Tradit. Chin. Med. 2018, 38, 272–279. [Google Scholar] [CrossRef]
- Staffa, P.; Nyangiwe, N.; Msalya, G.; Nagagi, Y.P.; Nchu, F. The effect of Beauveria bassiana inoculation on plant growth, volatile constituents, and tick (Rhipicephalus appendiculatus) repellency of acetone extracts of Tulbaghia violacea. Vet. World 2020, 13, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Devi, K.P.; Malar, D.S.; Nabavi, S.F.; Sureda, A.; Xiao, J.; Nabavi, S.M.; Daglia, M. Kaempferol and inflammation: From chemistry to medicine. Pharmacol. Res. 2015, 99, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Teffo, L.S.; Aderogba, M.A.; Eloff, J.N. Antibacterial and antioxidant activities of four kaempferol methyl ethers isolated from Dodonaea viscosa Jacq. var. angustifolia leaf extracts. S. Afr. J. Bot. 2010, 76, 25–29. [Google Scholar] [CrossRef]
- Yang, C.; Yang, W.; He, Z.; Guo, J.; Yang, X.; Wang, R.; Li, H. Kaempferol Alleviates Oxidative Stress and Apoptosis Through Mitochondria-dependent Pathway During Lung Ischemia-Reperfusion Injury. Front. Pharmacol. 2021, 12, 11. [Google Scholar] [CrossRef]
- Murray, C.J. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 388, 629–655. [Google Scholar] [CrossRef]
- Anand, U.; Jacobo-Herrera, N.; Altemimi, A.; Lakhssassi, N. A comprehensive review on medicinal plants as antimicrobial therapeutics: Potential avenues of biocompatible drug discovery. Metabolites 2019, 9, 258. [Google Scholar] [CrossRef]
- Kokoska, L.; Kloucek, P.; Leuner, O.; Novy, P. Plant-Derived Products as Antibacterial and Antifungal Agents in Human Health Care. Curr. Med. Chem. 2018, 26, 5501–5541. [Google Scholar] [CrossRef]
- Salam, A.M.; Quave, C.L. Opportunities for plant natural products in infection control. Curr. Opin. Microbiol. 2018, 45, 189–194. [Google Scholar] [CrossRef]
- Krstin, S.; Sobeh, M.; Braun, M.S.; Wink, M. Tulbaghia violacea and Allium ursinum extracts exhibit anti-parasitic and antimicrobial activities. Molecules 2018, 23, 313. [Google Scholar] [CrossRef] [PubMed]
- Eid, H.H.; Metwally, G.F. Phytochemical and biological study of callus cultures of Tulbaghia violacea Harv. Cultivated in Egypt. Nat. Prod. Res. 2017, 31, 1717–1724. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front. Microbiol. 2017, 7, 2170. [Google Scholar] [CrossRef] [PubMed]
- Belewa, V.; Baijnath, H.; Frost, C.; Somai, B.M. Tulbaghia violacea Harv. plant extract affects cell wall synthesis in Aspergillus flavus. J. Appl. Microbiol. 2017, 122, 921–931. [Google Scholar] [CrossRef]
- Somai, B.M.; Belewa, V.; Frost, C. Tulbaghia violacea (Harv) Exerts its Antifungal Activity by Reducing Ergosterol Production in Aspergillus flavus. Curr. Microbiol. 2021, 78, 2989–2997. [Google Scholar] [CrossRef]
- Pretorius, J.C. Extracts and Compounds from Tulbaghia Violacea and Their Use as Biological Plant Protecting Agents 2014. Google Patents US8697149B2, 15 April 2014. [Google Scholar]
- Ncise, W.; Daniels, C.W.; Etsassala, N.G.E.R.; Nchu, F. Interactive effects of light intensity and ph on growth parameters of a bulbous species (Tulbaghia violacea l.) in hydroponic cultivation and its antifungal activities. Med. Plants 2021, 13, 442–451. [Google Scholar] [CrossRef]
- Ncise, W.; Daniels, C.W.; Nchu, F. Effects of light intensities and varying watering intervals on growth, tissue nutrient content and antifungal activity of hydroponic cultivated Tulbaghia violacea L. under greenhouse conditions. Heliyon 2020, 6, 3906. [Google Scholar] [CrossRef] [PubMed]
- Malungane, M.M.F.; Florah, M.M. Effect of Crude Extracts of Tulbaghia violacea (Wild Garlic) on Growth of Tomato and Supression of Meloidogyne Species; University of Limpopo: Mankweng, South Africa, 2014. [Google Scholar]
- Kaushik, I.; Ramachandran, S.; Prasad, S.; Srivastava, S.K. Drug rechanneling: A novel paradigm for cancer treatment. In Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2021; Volume 68, pp. 279–290. [Google Scholar]
- Cragg, G.M.; Grothaus, P.G.; Newman, D.J. Impact of natural products on developing new anti-cancer agents. Chem. Rev. 2009, 109, 3012–3043. [Google Scholar] [CrossRef]
- Mthembu, N.N.; Motadi, L.R. Apoptotic potential role of Agave palmeri and Tulbaghia violacea extracts in cervical cancer cells. Mol. Biol. Rep. 2014, 41, 6143–6155. [Google Scholar] [CrossRef]
- Motadi, L.R.; Choene, M.S.; Mthembu, N.N. Anticancer properties of Tulbaghia violacea regulate the expression of p53-dependent mechanisms in cancer cell lines. Sci. Rep. 2020, 10, 12924. [Google Scholar] [CrossRef]
- Rivas-García, L.; Romero-Márquez, J.M.; Navarro-Hortal, M.D.; Esteban-Muñoz, A.; Giampieri, F.; Sumalla-Cano, S.; Battino, M.; Quiles, J.L.; Llopis, J.; Sánchez-González, C. Unravelling potential biomedical applications of the edible flower Tulbaghia violacea. Food Chem. 2022, 381, 132096. [Google Scholar] [CrossRef]
- Bianchini, G.; Balko, J.M.; Mayer, I.A.; Sanders, M.E.; Gianni, L. Triple-negative breast cancer: Challenges and opportunities of a heterogeneous disease. Nat. Rev. Clin. Oncol. 2016, 13, 674–690. [Google Scholar] [CrossRef] [PubMed]
- Dlamini, Z.; Alouna, M.; Hull, R.; Penny, C. Abstract 2843: The effects of extracts of the indigenous South African plant, Tulbaghia violacea, on triple negative breast cancer cells. In Proceedings of the NCRI Cancer Conference, London, UK, 8–12 November 2021. [Google Scholar]
- Deepak, K.G.K.; Vempati, R.; Nagaraju, G.P.; Dasari, V.R.; Nagini, S.; Rao, D.N.; Malla, R.R. Tumor microenvironment: Challenges and opportunities in targeting metastasis of triple negative breast cancer. Pharmacol. Res. 2020, 153, 104683. [Google Scholar] [CrossRef] [PubMed]
- Takaidza, S.; Kumar, A.M.; Ssemakalu, C.C.; Natesh, N.S.; Karanam, G.; Pillay, M. Anticancer activity of crude acetone and water extracts of Tulbaghia violacea on human oral cancer cells. Asian Pac. J. Trop. Biomed. 2018, 8, 456–462. [Google Scholar] [CrossRef]
- Lyantagaye, S.L. Two new pro-apoptotic glucopyranosides from Tulbaghia violacea. J. Med. Plants Res. 2013, 7, 2214–2220. [Google Scholar] [CrossRef]
- Lyantagaye, S. Characterization of the Biochemical Pathway of Apoptosis Induced by D-glucopyranoside Derivatives from Tulbaghia violacea. Annu. Res. Rev. Biol. 2014, 4, 962–977. [Google Scholar] [CrossRef]
- Lushchak, V.I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chem. Biol. Interact. 2014, 224, 164–175. [Google Scholar] [CrossRef]
- Thorpe, G.W.; Fong, C.S.; Alic, N.; Higgins, V.J.; Dawes, I.W. Cells have distinct mechanisms to maintain protection against different reactive oxygen species: Oxidative-stress-response genes. Proc. Natl. Acad. Sci. USA 2004, 101, 6564–6569. [Google Scholar] [CrossRef]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of natural plant origins: From sources to food industry applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef]
- Madike, L.N.; Pillay, M.; Popat, K.C. Antithrombogenic properties of Tulbaghia violacea–loaded polycaprolactone nanofibers. J. Bioact. Compat. Polym. 2020, 35, 102–116. [Google Scholar] [CrossRef]
- Arhin, I.; Depika, D.; Ajay, B.; Delon, N.; Irene, M. Biochemical, phytochemical profile and angiotensin-1 converting enzyme inhibitory activity of the hydro-methanolic extracts of Tulbaghia acutiloba harv. J. Nat. Remedies 2019, 19, 221–235. [Google Scholar] [CrossRef]
- Madike, L.N.; Pillay, M.; Popat, K.C. Antithrombogenic properties of: Tulbaghia violacea aqueous leaf extracts: Assessment of platelet activation and whole blood clotting kinetics. RSC Adv. 2021, 11, 30455–30464. [Google Scholar] [CrossRef] [PubMed]
- Glovaci, D.; Fan, W.; Wong, N.D. Epidemiology of Diabetes Mellitus and Cardiovascular Disease. Curr. Cardiol. Rep. 2019, 21, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global Prevalence of Diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef]
- Matheus, A.S.D.M.; Tannus, L.R.M.; Cobas, R.A.; Palma, C.C.S.; Negrato, C.A.; Gomes, M.D.B. Impact of diabetes on cardiovascular disease: An update. Int. J. Hypertens. 2013, 2013, 653789. [Google Scholar] [CrossRef] [PubMed]
- Moodley, K.; Joseph, K.; Naidoo, Y.; Islam, S.; Mackraj, I. Antioxidant, antidiabetic and hypolipidemic effects of Tulbaghia violacea Harv. (wild garlic) rhizome methanolic extract in a diabetic rat model. BMC Complement. Altern. Med. 2015, 15, 408. [Google Scholar] [CrossRef] [PubMed]
- Moodley, K.; Mackraj, I. Metabolic effects of tulbaghia violacea harv. In a diabetic model. Afr. J. Tradit. Complement. Altern. Med. 2016, 13, 113–122. [Google Scholar] [CrossRef]
- Raji, I.; Obikeze, K.; Mugabo, P. Comparison of the acute effects of Tulbaghia violacea William Henry Harvey (Alliaceae) on blood pressure and heart rate of ageing male normotensive Wistar kyoto rats and adult male spontaneously hypertensive rats. Trop. J. Pharm. Res. 2016, 15, 2429–2434. [Google Scholar] [CrossRef][Green Version]
- Moodley, K.; Naidoo, Y.; Mackraj, I. Effects of Tulbaghia violacea Harv. (Alliaceae) rhizome methanolic extract on kidney function and morphology in Dahl salt-sensitive rats. J. Ethnopharmacol. 2014, 155, 1194–1203. [Google Scholar] [CrossRef]
- Navar, L.G. The role of the kidneys in hypertension. J. Clin. Hypertens. 2005, 7, 542–549. [Google Scholar] [CrossRef]
- Davì, G.; Patrono, C. Platelet Activation and Atherothrombosis. N. Engl. J. Med. 2007, 357, 2482–2494. [Google Scholar] [CrossRef] [PubMed]
- Masoud, K.A.A.; Okobi, E.; Ekpo, G.J. Amabeoku Investigation of Some Possible Mechanisms Involved in the Anticonvulsant Activity of Tulbaghia violacea Harv. J. Pharm. Pharmacol. 2017, 5. [Google Scholar] [CrossRef][Green Version]
- Madike, L.N.; Takaidza, S.; Ssemakalu, C.; Pillay, M. Genotoxicity of aqueous extracts of Tulbaghia violacea as determined through an Allium cepa assay. S. Afr. J. Sci. 2019, 115, 1–6. [Google Scholar] [CrossRef]
- Madike, L.N.; Takaidza, S.; Ssemakalu, C.C.; Pillay, M. The effect of extracts of Tulbaghia violacea on the proliferation of a murine macrophage cell line. S. Afr. J. Bot. 2020, 130, 185–197. [Google Scholar] [CrossRef]
- Stavělíková, H. Morphological characteristics of garlic (Allium sativum L.) genetic resources collection—Information. Hortic. Sci. 2008, 35, 130–135. [Google Scholar] [CrossRef]
- Wheeler, E.J.; Mashayekhi, S.; Mcneal, D.W.; Travis Columbus, J.; Chris Pires, J. Molecular systematics of Allium subgenus Amerallium (Amaryllidaceae) in North America. Am. J. Bot. 2013, 100, 701–711. [Google Scholar] [CrossRef]
- Fernandes, S.; Gois, A.; Mendes, F.; Perestrelo, R.; Medina, S.; Câmara, J.S. Typicality Assessment of Onions (Allium cepa) from Different Geographical Regions Based on the Volatile Signature and Chemometric Tools. Foods 2020, 9, 375. [Google Scholar] [CrossRef]
- Jo, J.; Purushotham, P.M.; Han, K.; Lee, H.-R.; Nah, G.; Kang, B.-C. Development of a Genetic Map for Onion (Allium cepa L.) Using Reference-Free Genotyping-by-Sequencing and SNP Assays. Front. Plant Sci. 2017, 8, 1342. [Google Scholar] [CrossRef]
- Najeebullah, S.; Shinwari, Z.K.; Jan, S.A.; Khan, I.; Ali, M. Ethno medicinal and phytochemical properties of genus Allium: A review of recent advances. Pak. J. Bot. 2021, 53, 135–144. [Google Scholar] [CrossRef]
- Lawande, K.E. Onion. In Handbook of Herbs and Spices; Elsevier: Amsterdam, The Netherlands, 2012; pp. 417–429. [Google Scholar]
- Debin, W.; Jiande, G.; Guangshu, L. General situation of Allium crops in China. In Proceedings of the IV International Symposium on Edible Alliaceae 688, Beijing, China, 21–26 April 2004; pp. 327–332. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/faostat/en/#data (accessed on 26 March 2022).
- Bartolucci, F.; Iocchi, M.; De Castro, O.; Conti, F. Allium ducissae (A. subgen. Polyprason, Amaryllidaceae) a New Species from the Central Apennines (Italy). Plants 2022, 11, 426. [Google Scholar] [CrossRef]
- Friesen, N. The genus Allium L. in the flora of Mongolia. Feddes Repert. 1995, 106, 59–81. [Google Scholar] [CrossRef]
- Sinitsyna, T.A. Genus Allium L.(Alliaceae) in Siberia. Vavilovia 2020, 2, 3–22. [Google Scholar] [CrossRef]
- Temperate Plants Database, Ken Fern. Available online: https://temperate.theferns.info/ (accessed on 26 March 2022).
- Kawano, S.; Nagai, Y. Life-history monographs of Japanese plants. 4: Allium victorialis L. ssp. platyphyllum (Makino) Hultén (Alliaceae) Syn. Allium victorialis L. var. platyphyllum Makino; A. latissimum Prokh. Plant Species Biol. 2005, 20, 219–225. [Google Scholar] [CrossRef]
- Kitamura, S.; Murata, G.; Hori, M. Coloured Illustrations of Herbaceous Plants of Japan; Hoikusha Publishing Co.: Osaka, Japan, 1958. [Google Scholar]
- Arifin, N.S.; Okudo, H. Geographical distribution of allozyme patterns in shallot (Allium cepa var. ascalonicum Backer) and wakegi onion (A. × wakegi Araki). Euphytica 1996, 91, 305–313. [Google Scholar] [CrossRef]
- Bah, A.A.; Wang, F.; Huang, Z.; Shamsi, I.H.; Zhang, Q.; Jilani, G.; Hussain, S.; Hussain, N.; Ali, E. Biology Phyto-characteristics, Cultivation and Medicinal Prospects of Chinese Jiaotou (Allium chinense). Int. J. Agric. Biol. 2012, 14, 650–657. [Google Scholar]
- Blattner, F.R.; Friesen, N. Relationship between Chinese Chive (Allium tuberosum) and Its Putative Progenitor A. Ramosum as Assessed by Random Amplified Polymorphic DNA (RAPD); California University Press: Los Angeles, CA, USA, 2006; pp. 134–142. [Google Scholar]
- Pandey, A.; Pradheep, K.; Gaikwad, A.B.; Gupta, R.; Malav, P.K.; Rai, M. Systematics study on a morphotype of Allium tuberosum Rottler ex Spreng. (Alliaceae) from Ladakh, India. Indian J. Plant Genet. Resour. 2019, 32, 223–231. [Google Scholar] [CrossRef]
- Sajad, M.A.; Khan, M.S.; Bahadur, S.; Naeem, A.; Ali, H.; Batool, F.; Shuaib, M.; Khan, M.A.S.; Batool, S. Evaluation of chromium phytoremediation potential of some plant species of Dir Lower, Khyber Pakhtunkhwa, Pakistan. Acta Ecol. Sin. 2020, 40, 158–165. [Google Scholar] [CrossRef]
- Keusgen, M.; Fritsch, R.M.; Hisoriev, H.; Kurbonova, P.A.; Khassanov, F.O. Wild Allium species (Alliaceae) used in folk medicine of Tajikistan and Uzbekistan. J. Ethnobiol. Ethnomed. 2006, 2, 18. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ijaz, F.; Iqbal, Z.; Rahman, I.U.; Alam, J.; Khan, S.M.; Shah, G.M.; Khan, K.; Afzal, A. Investigation of traditional medicinal floral knowledge of Sarban Hills, Abbottabad, KP, Pakistan. J. Ethnopharmacol. 2016, 179, 208–233. [Google Scholar] [CrossRef]
- Ajaib, M.; Ishtiaq, M.; Bhatti, K.H.; Hussain, I.; Maqbool, M.; Hussain, T.; Mushtaq, W.; Ghani, A.; Azeem, M.; Khan, S.M.R.; et al. Inventorization of traditional ethnobotanical uses of wild plants of Dawarian and Ratti Gali areas of District Neelum, Azad Jammu and Kashmir Pakistan. PLoS ONE 2021, 16, e0255010. [Google Scholar] [CrossRef]
- Islam, M.; Inamullah, A.I.; Akhtar, N.; Alam, J.; Razzaq, A.; Mohammad, K.; Mahmood, T.; Khan, F.U.; Muhammad Khan, W.; Ishtiaq, A.; et al. Medicinal plants resources of Western Himalayan Palas Valley, Indus Kohistan, Pakistan: Their uses and degrees of risk of extinction. Saudi J. Biol. Sci. 2021, 28, 3076–3093. [Google Scholar] [CrossRef] [PubMed]
- Amjad, M.S.; Qaeem, M.F.; Ahmad, I.; Khan, S.U.; Chaudhari, S.K.; Malik, N.Z.; Shaheen, H.; Khan, A.M. Descriptive study of plant resources in the context of the ethnomedicinal relevance of indigenous flora: A case study from Toli Peer National Park, Azad Jammu and Kashmir, Pakistan. PLoS ONE 2017, 12, e0171896. [Google Scholar] [CrossRef] [PubMed]
- Tavares, L.; Santos, L.; Zapata Noreña, C.P. Bioactive compounds of garlic: A comprehensive review of encapsulation technologies, characterization of the encapsulated garlic compounds and their industrial applicability. Trends Food Sci. Technol. 2021, 114, 232–244. [Google Scholar] [CrossRef]
- Zhao, X.X.; Lin, F.J.; Li, H.; Li, H.B.; Wu, D.T.; Geng, F.; Ma, W.; Wang, Y.; Miao, B.H.; Gan, R.Y. Recent Advances in Bioactive Compounds, Health Functions, and Safety Concerns of Onion (Allium cepa L.). Front. Nutr. 2021, 8, 463. [Google Scholar] [CrossRef] [PubMed]
- Beretta, H.V.; Bannoud, F.; Insani, M.; Berli, F.; Hirschegger, P.; Galmarini, C.R.; Cavagnaro, P.F. Relationships Between Bioactive Compound Content and the Antiplatelet and Antioxidant Activities of Six Allium Vegetable Species. Food Technol. Biotechnol. 2017, 55, 266–275. [Google Scholar] [CrossRef]
- Majewski , M. Allium sativum: Facts and myths regarding human health. Rocz Panstw Zakl Hig. 2014, 65, 1–8. [Google Scholar] [PubMed]
- Marrelli, M.; Amodeo, V.; Statti, G.; Conforti, F. Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities. Molecules 2019, 24, 119. [Google Scholar] [CrossRef]
- Teshika, J.D.; Zakariyyah, A.M.; Zaynab, T.; Zengin, G.; Rengasamy, K.R.; Pandian, S.K.; Fawzi, M.M. Traditional and modern uses of onion bulb (Allium cepa L.): A systematic review. Crit. Rev. Food Sci. Nutr. 2019, 59, S39–S70. [Google Scholar] [CrossRef]
- Turati, F.; Pelucchi, C.; Guercio, V.; La Vecchia, C.; Galeone, C. Allium vegetable intake and gastric cancer: A case-control study and meta-analysis. Mol. Nutr. Food Res. 2015, 59, 171–179. [Google Scholar] [CrossRef]
- Zhou, X.F.; Ding, Z.S.; Liu, N.B. Allium vegetables and risk of prostate cancer: Evidence from 132,192 subjects. Asian Pac. J. Cancer Prev. 2013, 14, 4131–4134. [Google Scholar] [CrossRef]
- Turati, F.; Guercio, V.; Pelucchi, C.; La Vecchia, C.; Galeone, C. Colorectal cancer and adenomatous polyps in relation to allium vegetables intake: A meta-analysis of observational studies. Mol. Nutr. Food Res. 2014, 58, 1907–1914. [Google Scholar] [CrossRef] [PubMed]
- Guercio, V.; Turati, F.; La Vecchia, C.; Galeone, C.; Tavani, A. Allium vegetables and upper aerodigestive tract cancers: A meta-analysis of observational studies. Mol. Nutr. Food Res. 2016, 60, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Kuete, V. Moringa oleifera, in Medicinal Spices and Vegetables from Africa. Med. Spices Veg. Afr. 2017, 43, 605–610. [Google Scholar]
- Peltola, R. Allium victoralis. Available online: https://portal.mtt.fi/portal/page/portal/mtt/hankkeet/BARENTSPEC (accessed on 20 March 2022).
- Shafakatullah, N.; Chandra, M. Isolation of lactic acid bacteria from Allium cepa var. aggregatum and study of their probiotic properties. Int. J. Pharma Sci. Res. 2015, 6, 749–752. [Google Scholar]
- Lawless, J.W.; Latham, M.C.; Stephenson, L.S.; Kinoti, S.N.; Pertet, A.M. Iron supplementation improves appetite and growth in anemic Kenyan primary school children. J. Nutr. 1994, 124, 645–654. [Google Scholar] [CrossRef]
- Lee, Y.M.; Lim, D.Y.; Choi, H.J.; Jung, J.I.; Chung, W.Y.; Park, J.H.Y. Induction of cell cycle arrest in prostate cancer cells by the dietary compound isoliquiritigenin. J. Med. Food 2009, 12, 8–14. [Google Scholar] [CrossRef]
- Kaiser, P.; Youssouf, M.S.; Tasduq, S.A.; Singh, S.; Sharma, S.C.; Singh, G.D.; Gupta, V.K.; Gupta, B.D.; Johri, R.K. Anti-allergic effects of herbal product from Allium cepa (bulb). J. Med. Food 2009, 12, 374–382. [Google Scholar] [CrossRef]
- Mohammadi-Motlagh, H.R.; Mostafaie, A.; Mansouri, K. Anticancer and anti-inflammatory activities of shallot (Allium ascalonicum) extract. Arch. Med. Sci. 2011, 7, 38–44. [Google Scholar] [CrossRef]
- Handbook of Chinese Herbs and Formulas; Yeung, H., Ed.; Institute of Chinese Medicine: London, UK, 1985; Volume 1. [Google Scholar]
- Jannat, K.; Rahman, T.; Rahmatullah, M. Traditional uses, phytochemicals and pharmacological properties of Allium tuberosum Rottler ex spreng. J. Med. Plants Stud. 2019, 7, 214–220. [Google Scholar]
- Sabha, D.; Hiyasat, B.; Grtzinger, K.; Hennig, L.; Schlegel, F.; Mohr, F.W.; Rauwald, H.W.; Dhein, S. Allium ursinum L.: Bioassay-guided isolation and identification of a galactolipid and a phytosterol exerting antiaggregatory effects. Pharmacology 2012, 89, 260–269. [Google Scholar] [CrossRef]
- Carotenuto, A.; De Feo, V.; Fattorusso, E.; Lanzotti, V.; Magno, S.; Cicala, C. The flavonoids of Allium ursinum. Phytochemistry 1996, 41, 531–536. [Google Scholar] [CrossRef]
- Sobolewska, D.; Podolak, I.; Makowska-Wąs, J. Allium ursinum: Botanical, phytochemical and pharmacological overview. Phytochem. Rev. 2015, 14, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, A.; Mikhova, B.; Najdenski, H.; Tsvetkova, I.; Kostova, I. Chemical composition and antimicrobial activity of wild garlic Allium ursinum of Bulgarian origin. Nat. Prod. Commun. 2009, 4, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Ruan, J.; Ding, Z.; Zhao, W.; Hao, M.; Zhang, Y.; Jiang, H.; Zhang, Y.; Wang, T. molecules Phytochemistry and Comprehensive Chemical Profiling Study of Flavonoids and Phenolic Acids in the Aerial Parts of Allium Mongolicum Regel and Their Intestinal Motility Evaluation. Molecules 2020, 25, 577. [Google Scholar] [CrossRef]
- Marefati, N.; Ghorani, V.; Shakeri, F.; Boskabady, M.; Kianian, F.; Rezaee, R.; Boskabady, M.H. A review of anti-inflammatory, antioxidant, and immunomodulatory effects of Allium cepa and its main constituents. Pharm. Biol. 2021, 59, 287–302. [Google Scholar] [CrossRef]
- Fossen, T.; Slimestad, R.; Andersen, Ø.M. Anthocyanins with 4′-glucosidation from red onion, Allium cepa. Phytochemistry 2003, 64, 1367–1374. [Google Scholar] [CrossRef]
- Xiao, H.; Parkin, K.L. Isolation and identification of potential cancer chemopreventive agents from methanolic extracts of green onion (Allium cepa). Phytochemistry 2007, 68, 1059–1067. [Google Scholar] [CrossRef]
- Fossen, T.; Andersen, Ø.M. Anthocyanins from red onion, Allium cepa, with novel aglycone. Phytochemistry 2003, 62, 1217–1220. [Google Scholar] [CrossRef]
- Lanzotti, V.; Romano, A.; Lanzuise, S.; Bonanomi, G.; Scala, F. Antifungal saponins from bulbs of white onion, Allium cepa L. Phytochemistry 2012, 74, 133–139. [Google Scholar] [CrossRef]
- Nile, A.; Nile, S.H.; Cespedes-Acuña, C.L.; Oh, J.W. Spiraeoside extracted from red onion skin ameliorates apoptosis and exerts potent antitumor, antioxidant and enzyme inhibitory effects. Food Chem. Toxicol. 2021, 154, 112327. [Google Scholar] [CrossRef]
- Nohara, T.; Fujiwara, Y.; El-Aasr, M.; Ikeda, T.; Ono, M.; Nakano, D.; Kinjo, J. Thiolane-type sulfides from garlic, onion, and Welsh onion. J. Nat. Med. 2021, 75, 741–751. [Google Scholar] [CrossRef] [PubMed]
- El-Aasr, M.; Fujiwara, Y.; Takeya, M.; Ikeda, T.; Tsukamoto, S.; Ono, M.; Nakano, D.; Okawa, M.; Kinjo, J.; Yoshimitsu, H.; et al. Onionin A from Allium cepa inhibits macrophage activation. J. Nat. Prod. 2010, 73, 1306–1308. [Google Scholar] [CrossRef] [PubMed]
- Terahara, N.; Yamaguchi, M.; Honda, T. Malonylated anthocyanins from bulbs of red onion, Allium cepa L. Biosci. Biotechnol. Biochem. 1994, 58, 1324–1325. [Google Scholar] [CrossRef]
- Pontin, M.; Bottini, R.; Burba, J.L.; Piccoli, P. Allium sativum produces terpenes with fungistatic properties in response to infection with Sclerotium cepivorum. Phytochemistry 2015, 115, 152–160. [Google Scholar] [CrossRef]
- Lanzotti, V.; Barile, E.; Antignani, V.; Bonanomi, G.; Scala, F. Antifungal saponins from bulbs of garlic, Allium sativum L. var. Voghiera. Phytochemistry 2012, 78, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, T.; Fujita, K.; Shibata, S.; Hoson, M.; Kawada, T.; Masaki, M.; Yamate, N. Effects of Chinese drugs “xiebai” and “dasuan” on human platelet aggregation (Allium bakeri, A. sativum). Planta Med. 1989, 55, 242–244. [Google Scholar] [CrossRef]
- Timité, G.; Mitaine-Offer, A.C.; Miyamoto, T.; Tanaka, C.; Mirjolet, J.F.; Duchamp, O.; Lacaille-Dubois, M.A. Structure and cytotoxicity of steroidal glycosides from Allium schoenoprasum. Phytochemistry 2013, 88, 61–66. [Google Scholar] [CrossRef]
- Fossen, T.; Slimestad, R.; Øvstedal, D.O.; Andersen, Ø.M. Covalent anthocyanin-flavonol complexes from flowers of chive, Allium schoenoprasum. Phytochemistry 2000, 54, 317–323. [Google Scholar] [CrossRef]
- Barile, E.; Bonanomi, G.; Antignani, V.; Zolfaghari, B.; Sajjadi, S.E.; Scala, F.; Lanzotti, V. Saponins from Allium minutiflorum with antifungal activity. Phytochemistry 2007, 68, 596–603. [Google Scholar] [CrossRef]
- Carotenuto, A.; Fattorusso, E.; Lanzotti, V.; Magno, S.; De Feo, V.; Cicala, C. The flavonoids of Allium neapolitanum. Phytochemistry 1997, 44, 949–957. [Google Scholar] [CrossRef]
- Chehri, Z.; Zolfaghari, B.; Sadeghi Dinani, M. Isolation of Cinnamic Acid Derivatives from the Bulbs of Allium tripedale. Adv. Biomed. Res. 2018, 7, 60. [Google Scholar] [CrossRef]
- Jan, K.; Michael, K. Cysteine sulfoxides and volatile sulfur compounds from Allium tripedale. J. Agric. Food Chem. 2010, 58, 1129–1137. [Google Scholar] [CrossRef]
- Fattorusso, E.; Lanzotti, V.; Taglialatela-Scafati, O.; Cicala, C. The flavonoids of leek, Allium porrum. Phytochemistry 2001, 57, 565–569. [Google Scholar] [CrossRef]
- Carotenuto, A.; Fattorusso, E.; Lanzotti, V.; Magno, S. Spirostanol saponins of Allium porrum L. Phytochemistry 1999, 51, 1077–1082. [Google Scholar] [CrossRef]
- Peng, J.P.; Yao, X.S.; Tezuka, Y.; Kikuchi, T. Furostanol glycosides from bulbs of Allium chinense. Phytochemistry 1996, 41, 283–285. [Google Scholar] [CrossRef]
- Kuroda, M.; Mimaki, Y.; Kameyama, A.; Sashida, Y.; Nikaido, T. Steroidal saponins from Allium chinense and their inhibitory activities on cyclic AMP phosphodiesterase and Na+/K+ ATPase. Phytochemistry 1995, 40, 1071–1076. [Google Scholar] [CrossRef]
- Baba, M.; Ohmura, M.; Kishi, N.; Okada, Y.; Shibata, S.; Peng, J.; Yao, S.S.; Nishino, H.; Okuyama, T. Saponins isolated from Allium chinense G. Don and antitumor-promoting activities of isoliquiritigenin and laxogenin from the same drug. Biol. Pharm. Bull. 2000, 23, 660–662. [Google Scholar] [CrossRef]
- Peng, J.; Yao, X.; Kobayashi, H.; Ma, C. Novel furostanol glycosides from Allium macrostemon. Planta Med. 1995, 61, 58–61. [Google Scholar] [CrossRef]
- Peng, J.P.; Wu, Y.; Yao, X.S.; Okuyama, T.; Narui, T. Two new steroidal saponins from Allium macrostemon. Yao Xue Xue Bao 1992, 27, 918–922. [Google Scholar] [PubMed]
- Peng, J.P.; Wang, X.; Yao, X.S. Studies on two new furostanol glycosides from Allium macrostemon Bunge. Yao Xue Xue Bao 1993, 28, 526–531. [Google Scholar]
- Usui, A.; Matsuo, Y.; Tanaka, T.; Ohshima, K.; Fukuda, S.; Mine, T.; Yakashiro, I.; Ishimaru, K. Ferulic acid esters of glucosylglucose from Allium macrostemon Bunge. J. Asian Nat. Prod. Res. 2017, 19, 215–221. [Google Scholar] [CrossRef]
- Kawashima, K.; Mimaki, Y.; Sashida, Y. Steroidal saponins from the bulbs of Allium schubertii. Phytochemistry 1993, 32, 1267–1272. [Google Scholar] [CrossRef]
- Zou, Z.M.; Yu, D.Q.; Cong, P.Z. A steroidal saponin from the seeds of Allium tuberosum. Phytochemistry 2001, 57, 1219–1222. [Google Scholar] [CrossRef]
- Sang, S.; Zou, M.; Xia, Z.; Lao, A.; Chen, Z.; Ho, C.T. New spirostanol saponins from Chinese chives (Allium tuberosum). J. Agric. Food Chem. 2001, 49, 4780–4783. [Google Scholar] [CrossRef] [PubMed]
- Sang, S.M.; Zou, M.L.; Zhang, X.W.; Lao, A.N.; Chen, Z.L. Tuberoside M, a new cytotoxic spirostanol saponin from the seeds of Allium tuberosum. J. Asian Nat. Prod. Res. 2002, 4, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Mimaki, Y.; Kawashima, K.; Kanmoto, T.; Sashida, Y. Steroidal glycosides from Allium albopilosum and A. ostrowskianum. Phytochemistry 1993, 34, 799–805. [Google Scholar] [CrossRef]
- Zolfaghari, B.; Yazdiniapour, Z.; Sadeghi, M.; Akbari, M.; Troiano, R.; Lanzotti, V. Cinnamic acid derivatives from welsh onion (Allium fistulosum) and their antibacterial and cytotoxic activities. Phytochem. Anal. 2021, 32, 84–90. [Google Scholar] [CrossRef]
- Sang, S.; Lao, A.; Wang, Y.; Chin, C.K.; Rosen, R.T.; Ho, C.T. Antifungal constituents from the seeds of Allium fistulosum L. J. Agric. Food Chem. 2002, 50, 6318–6321. [Google Scholar] [CrossRef]
- Tsuruoka, T.; Ishikawa, K.; Hosoe, T.; Davaajab, D.; Duvjir, S.; Surenjav, U. A new cinnamoylphenethylamine derivative from a Mongolian Allium species, Allium carolinianum. J. Nat. Med. 2018, 72, 332–334. [Google Scholar] [CrossRef]
- Zamri, N.; Hamid, H.A. Comparative Study of Onion (Allium cepa) and Leek (Allium ampeloprasum): Identification of Organosulphur Compounds by UPLC-QTOF/MS and Anticancer Effect on MCF-7 Cells. Plant Foods Hum. Nutr. 2019, 74, 525–530. [Google Scholar] [CrossRef]
- Kang, L.-P.; Liu, Z.-J.; Zhang, L.; Tan, D.-W.; Zhao, Y.; Zhao, Y.; Chen, H.-B.; Ma, B.-P. New furostanol saponins from Allium ascalonicum L. Magn. Reson. Chem. Magn. Reson. Chem 2007, 45, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Kubec, R.; Cody, R.B.; Dane, A.J.; Musah, R.A.; Schraml, J.; Vattekkatte, A.; Block, E. Applications of direct analysis in real time-mass spectrometry (DART-MS) in Allium chemistry. (Z)-butanethial S-oxide and 1-butenyl thiosulfinates and their S-(E)-1-butenylcysteine S-oxide precursor from Allium siculum. J. Agric. Food Chem. 2010, 58, 1121–1128. [Google Scholar] [CrossRef]
- Hu, X.-P.; Zhou, H.; Du, Y.-M.; Ou, S.-Y.; Yan, R.; Wang, Y. Two new flavonoids from the bark of Allium chrysanthum. J. Asian Nat. Prod. Res. 2017, 19, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Kusterer, J.; Vogt, A.; Keusgen, M. Isolation and identification of a new cysteine sulfoxide and volatile sulfur compounds from Allium subgenus Melanocrommyum. J. Agric. Food Chem. 2010, 58, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Morita, T.; Ushiroguchi, T.; Hayashi, N.; Matsuura, H.; Itakura, Y.; Fuwa, T. Steroidal saponins from elephant garlic, bulbs of Allium ampeloprasum L. Chem. Pharm. Bull. 1988, 36, 3480–3486. [Google Scholar] [CrossRef]
- Lee, K.T.; Choi, J.H.; Kim, D.H.; Son, K.H.; Kim, W.B.; Kwon, S.H.; Park, H.J. Constituents and the antitumor principle of Allium victorialis var. platyphyllum. Arch. Pharm. Res. 2001, 24, 44–50. [Google Scholar] [CrossRef]
- Akhov, L.S.; Musienko, M.M.; Piacente, S.; Pizza, C.; Oleszek, W. Structure of steroidal saponins from underground parts of Allium nutans L. J. Agric. Food Chem. 1999, 47, 3193–3196. [Google Scholar] [CrossRef]
- Mimaki, Y.; Matsumoto, K.; Sashida, Y.; Nikaido, T.; Ohmoto, T. New steroidal saponins from the bulbs of Allium giganteum exhibiting potent inhibition of cAMP phosphodiesterase activity. Chem. Pharm. Bull. 1994, 42, 710–714. [Google Scholar] [CrossRef]
- Ren, L.; Yaun-Fei, W.; Qian, S.; Hua-Bin, H. Chemical composition and antimicrobial activity of the essential oil from Allium hookeri consumed in Xishuangbanna, Southwest China. Nat. Prod. Commun. 2014, 9, 863–864. [Google Scholar]
- El-Saber Batiha, G.; Magdy Beshbishy, A.; Wasef, L.G.; Elewa, Y.H.A.; Al-Sagan, A.; Abd El-Hack, M.E.; Taha, A.E.; Abd-Elhakim, M.Y.; Prasad Devkota, H. Chemical Constituents and Pharmacological Activities of Garlic (Allium sativum L.): A Review. Nutrients 2020, 12, 872. [Google Scholar] [CrossRef]
- Kuda, T.; Iwai, A.; Yano, T. Effect of red pepper Capsicum annuum var. conoides and garlic Allium sativum on plasma lipid levels and cecal microflora in mice fed beef tallow. Food Chem. Toxicol. 2004, 42, 1695–1700. [Google Scholar] [CrossRef] [PubMed]
- Wallock-Richards, D.; Doherty, C.J.; Doherty, L.; Clarke, D.J.; Place, M.; Govan, J.R.W.; Campopiano, D.J. Garlic revisited: Antimicrobial activity of allicin-containing garlic extracts against Burkholderia cepacia complex. PLoS ONE 2014, 9, e112726. [Google Scholar] [CrossRef] [PubMed]
- Ross, Z.M.; O’Gara, E.A.; Hill, D.J.; Sleightholme, H.V.; Maslin, D.J. Antimicrobial properties of garlic oil against human enteric bacteria: Evaluation of methodologies and comparisons with garlic oil sulfides and garlic powder. Appl. Environ. Microbiol. 2001, 67, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Pârvu, M.; Moţ, C.A.; Pârvu, A.E.; Mircea, C.; Stoeber, L.; Roşca-Casian, O.; Ţigu, A.B. Allium sativum Extract Chemical Composition, Antioxidant Activity and Antifungal Effect against Meyerozyma guilliermondii and Rhodotorula mucilaginosa Causing Onychomycosis. Molecules 2019, 24, 3958. [Google Scholar] [CrossRef]
- Fufa, B.K. Anti-bacterial and Anti-fungal Properties of Garlic Extract (Allium sativum): A Review. Microbiol. Res. J. Int. 2019, 28, 1–5. [Google Scholar] [CrossRef]
- Zhen, H.; Fang, F.; Ye, D.; Shu, S.; Zhou, Y.; Dong, Y.; Nie, X.; Li, G. Experimental study on the action of allitridin against human cytomegalovirus in vitro: Inhibitory effects on immediate-early genes. Antivir. Res. 2006, 72, 68–74. [Google Scholar] [CrossRef]
- Danquah, C.A.; Tetteh, M.; Amponsah, I.K.; Mensah, A.Y.; Buabeng, K.O.; Gibbons, S.; Bhakta, S. Investigating ghanaian Allium species for anti-infective and resistance-reversal natural product leads to mitigate multidrug-resistance in tuberculosis. Antibiotics 2021, 10, 902. [Google Scholar] [CrossRef]
- Satvati, S.A.R.; Shooriabi, M.; Amin, M.; Shiezadeh, F. Evaluation of the Antimicrobial Activity of Tribulus terrestris, Allium sativum, Salvia officinalis, and Allium hirtifolium Boiss Against Enterococcus faecalis. Int. J. Enteric. Pathog. 2017, 5, 63–67. [Google Scholar] [CrossRef][Green Version]
- Abdel-Hafeez, E.H.; Ahmad, A.K.; Kamal, A.M.; Abdellatif, M.Z.M.; Abdelgelil, N.H. In vivo antiprotozoan effects of garlic (Allium sativum) and ginger (Zingiber officinale) extracts on experimentally infected mice with Blastocystis spp. Parasitol. Res. 2015, 114, 3439–3444. [Google Scholar] [CrossRef]
- Gruhlke, M.C.H.; Nicco, C.; Batteux, F.; Slusarenko, A.J. The Effects of Allicin—A Reactive Sulfur Species from Garlic, on a Selection of Mammalian Cell Lines. Antioxidants 2016, 6, 1. [Google Scholar] [CrossRef]
- Danquah, C.A.; Kakagianni, E.; Khondkar, P.; Maitra, A.; Rahman, M.; Evangelopoulos, D.; McHugh, T.D.; Stapleton, P.; Malkinson, J.; Bhakta, S.; et al. Analogues of Disulfides from Allium stipitatum Demonstrate Potent Anti-tubercular Activities through Drug Efflux Pump and Biofilm Inhibition. Sci. Rep. 2018, 8, 1150. [Google Scholar] [CrossRef] [PubMed]
- Asdaq, S.M.B.; Inamdar, M.N. Pharmacodynamic and Pharmacokinetic Interactions of Propranolol with Garlic (Allium sativum) in Rats. Evid. Based. Complement. Alternat. Med. 2011, 2011, 824042. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.-J.; Lee, H.-J.; Yoon, D.-K.; Ji, D.-S.; Kim, J.-H.; Lee, C.-H. Antioxidant and antimicrobial activities of fresh garlic and aged garlic by-products extracted with different solvents. Food Sci. Biotechnol. 2018, 27, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Daim, M.M.; Shaheen, H.M.; Abushouk, A.I.; Toraih, E.A.; Fawzy, M.S.; Alansari, W.S.; Aleya, L.; Bungau, S. Thymoquinone and diallyl sulfide protect against fipronil-induced oxidative injury in rats. Environ. Sci. Pollut. Res. Int. 2018, 25, 23909–23916. [Google Scholar] [CrossRef]
- Putnoky, S.; Caunii, A.; Butnariu, M. Study on the stability and antioxidant effect of the Allium ursinumwatery extract. Chem. Cent. J. 2013, 7, 21. [Google Scholar] [CrossRef]
- Asgarpanah, J.; Ghanizadeh, B. Pharmacologic and medicinal properties of Allium hirtifolium Boiss. Afr. J. Pharm. Pharmacol. 2012, 6, 1809–1814. [Google Scholar] [CrossRef]
- Ahmad, T.A.; El-Sayed, B.A.; El-Sayed, L.H. Development of immunization trials against Eimeria spp. Trials Vaccinol. 2016, 5, 38–47. [Google Scholar] [CrossRef]
- Hobauer, R.; Frass, M.; Gmeiner, B.; Kaye, A.D.; Frost, E.A. Garlic extract (Allium sativum) reduces migration of neutrophils through endothelial cell monolayers. Middle East J. Anaesthesiol. 2000, 15, 649–658. [Google Scholar]
- Gu, X.; Wu, H.; Fu, P. Allicin attenuates inflammation and suppresses HLA-B27 protein expression in ankylosing spondylitis mice. Biomed. Res. Int. 2013, 2013, 171573. [Google Scholar] [CrossRef]
- Jeong, Y.Y.; Ryu, J.H.; Shin, J.-H.; Kang, M.J.; Kang, J.R.; Han, J.; Kang, D. Comparison of Anti-Oxidant and Anti-Inflammatory Effects between Fresh and Aged Black Garlic Extracts. Molecules 2016, 21, 430. [Google Scholar] [CrossRef]
- Jin, P.; Kim, J.-A.; Choi, D.-Y.; Lee, Y.-J.; Jung, H.S.; Hong, J.T. Anti-inflammatory and anti-amyloidogeniceffects of a small molecule, 2,4-bis(p-hydroxyphenyl)-2-butenal in Tg2576 Alzheimer’sdisease mice model. J. Neuroinflammation 2013, 10, 767. [Google Scholar] [CrossRef] [PubMed]
- Krejčová, P.; Kučerová, P.; Stafford, G.I.; Jäger, A.K.; Kubec, R. Antiinflammatory and neurological activity of pyrithione and related sulfur-containing pyridine N-oxides from Persian shallot (Allium stipitatum). J. Ethnopharmacol. 2014, 154, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Karunanidhi, A.; Ghaznavi-Rad, E.; Jeevajothi Nathan, J.; Abba, Y.; van Belkum, A.; Neela, V. Allium stipitatum Extract Exhibits In Vivo Antibacterial Activity against Methicillin-Resistant Staphylococcus aureus and Accelerates Burn Wound Healing in a Full-Thickness Murine Burn Model. Evid. Based Complement. Altern. Med. 2017, 2017, 1914732. [Google Scholar] [CrossRef]
- Kim, J.E.; Park, K.M.; Lee, S.Y.; Seo, J.H.; Yoon, I.S.; Bae, C.S.; Yoo, J.C.; Bang, M.A.; Cho, S.S.; Park, D.H. Anti-inflammatory effect of Allium hookeri on carrageenan-induced air pouch mouse model. PLoS ONE 2017, 12, e0190305. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Le, W.; Cui, Z. A novel therapeutic anticancer property of raw garlic extract via injection but not ingestion. Cell Death Discov. 2018, 4, 108. [Google Scholar] [CrossRef] [PubMed]
- Chhabria, S.V.; Akbarsha, M.A.; Li, A.P.; Kharkar, P.S.; Desai, K.B. In situ allicin generation using targeted alliinase delivery for inhibition of MIA PaCa-2 cells via epigenetic changes, oxidative stress and cyclin-dependent kinase inhibitor (CDKI) expression. Apoptosis 2015, 20, 1388–1409. [Google Scholar] [CrossRef]
- Fleischauer, A.T.; Arab, L. Garlic and cancer: A critical review of the epidemiologic literature. J. Nutr. 2001, 131, 1032S–1040S. [Google Scholar] [CrossRef]
- Piscitelli, S.C.; Burstein, A.H.; Welden, N.; Gallicano, K.D.; Falloon, J. The effect of garlic supplements on the pharmacokinetics of saquinavir. Clin. Infect. Dis. 2002, 34, 234–238. [Google Scholar] [CrossRef]
- Dall’Acqua, S.; Maggi, F.; Minesso, P.; Salvagno, M.; Papa, F.; Vittori, S.; Innocenti, G. Identification of non-alkaloid acetylcholinesterase inhibitors from Ferulago campestris (Besser) Grecescu (Apiaceae). Fitoterapia 2010, 81, 1208–1212. [Google Scholar] [CrossRef]
- Szychowski, K.A.; Rybczyńska-Tkaczyk, K.; Gaweł-Bęben, K.; Świeca, M.; Karaś, M.; Jakubczyk, A.; Matysiak, M.; Binduga, U.E.; Gmiński, J. Characterization of Active Compounds of Different Garlic (Allium sativum L.) Cultivars. Pol. J. Food Nutr. Sci. 2018, 68, 73–81. [Google Scholar] [CrossRef]
- Lu, S.-H.; Wu, J.W.; Liu, H.-L.; Zhao, J.-H.; Liu, K.-T.; Chuang, C.-K.; Lin, H.-Y.; Tsai, W.-B.; Ho, Y. The discovery of potential acetylcholinesterase inhibitors: A combination of pharmacophore modeling, virtual screening, and molecular docking studies. J. Biomed. Sci. 2011, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- Mathew, B.; Biju, R. Neuroprotective effects of garlic a review. Libyan J. Med. 2008, 3, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Qidwai, W.; Ashfaq, T. Role of garlic usage in cardiovascular disease prevention: An evidence-based approach. Evid. Based. Complement. Alternat. Med. 2013, 2013, 125649. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, R.; Aamir, K.; Shaikh, A.R.; Ahmed, T. Effects of garlic on dyslipidemia in patients with type 2 diabetes mellitus. J. Ayub Med. Coll. Abbottabad 2005, 17, 60–64. [Google Scholar]
- Zhai, B.; Zhang, C.; Sheng, Y.; Zhao, C.; He, X.; Xu, W.; Huang, K.; Luo, Y. Hypoglycemic and hypolipidemic effect of S-allyl-cysteine sulfoxide (alliin) in DIO mice. Sci. Rep. 2018, 8, 3527. [Google Scholar] [CrossRef]
- Lee, M.-S.; Kim, I.-H.; Kim, C.-T.; Kim, Y. Reduction of body weight by dietary garlic is associated with an increase in uncoupling protein mRNA expression and activation of AMP-activated protein kinase in diet-induced obese mice. J. Nutr. 2011, 141, 1947–1953. [Google Scholar] [CrossRef]
- Sobenin, I.; Andrianova, I.; Ionova, V.; Karagodin, V.; Orekhov, A. Anti-aggregatory and fibrinolytic effects of time-released garlic powder tablets. Med. Heal. Sci. J. 2012, 10, 47–51. [Google Scholar] [CrossRef]
- Embuscado, M.E. Bioactives from culinary spices and herbs: A review. J. Food Bioact. 2019, 6. [Google Scholar] [CrossRef]
- Mohammadi-rika, A.; Beigi-boroujeni, M.; Rajabzadeh, A.; Zarei, L. Effect of Extract of Allium stipitatum on Excisional Wound Healing in Rats. Iran. J. Vet. Surg. 2021, 16, 5–11. [Google Scholar]
- Velten, R.; Erdelen, C.; Gehling, M.; Ghrt, A.; Gondol, D.; Lenz, J.; Loekhoff, O.; Wachendorff, U.; Wendisch, D.; Cripowellin, A. Cripowellin A and B, Novel Type of Amaryllidaceae Alkaloid from Crinum powellii. Tetrahedron Lett. 1998, 39, 1737–1740. [Google Scholar] [CrossRef]
- Zvetkova, E.; Wirleitner, B.; Tram, N.T.; Schennach, H.; Fuchs, D. Aqueous extracts of Crinum latifolium and Camellia sinensis show immunomodulatory properties in human peripheral blood mononuclear cells. Sci. Pharm. 2001, 79, 2143–2150. [Google Scholar] [CrossRef]
- Ulrich, M.R.; Davies, F.T., Jr.; Koh, Y.C.; Duray, S.A.; Egilla, J.N. Micropropagation of Crinum `Ellen Bosanquet’ by tri-scales. Sci. Hortic. 1999, 82, 95–102. [Google Scholar] [CrossRef]
- Thi Ngoc Tram, N.; Titorenkova, T.V.; Bankova, V.S.; Handjieva, N.V.; Popov, S.S. Crinum L. (Amaryllidaceae). Fitoterapia 2002, 73, 183–208. [Google Scholar] [CrossRef]
- Singh, K.A.; Nayak, M.K.; Jagannadham, M.V.; Dash, D. Thrombolytic along with anti-platelet activity of crinumin, a protein constituent of Crinum asiaticum. Blood Cells. Mol. Dis. 2011, 47, 129–132. [Google Scholar] [CrossRef]
- Ghosal, S.; Rao, P.H.; Jaiswal, D.K.; Kumar, Y.; Frahm, A.W. Alkaloids of Crinum pratense. Phytochemistry 2007, 20, 2003–2007. [Google Scholar] [CrossRef]
- Yoshisuke, T.; Noriaki, K.; Vijaya, K. The Alkaloidal Constituents of Goda-Manel (Crinum zeylanicum L.), a Sri Lankan Folk Medicine. Chem. Pharm. Bull. 1984, 32, 3023–3027. [Google Scholar]
- Nordal, I.; Wahlstrom, R. Studies in the Crinum zeylanicum complex in East Africa. Nord. J. Bot. 1982, 2, 465–473. [Google Scholar] [CrossRef]
- Bastida, J.; Peeters, P.; Rubiralta, M.; Naturals, D.D.P.; De Farmficia, F.; Barcelona, U. De Alkaloids from crinum kirkii. Phytochemistry 1995, 40, 1291–1293. [Google Scholar] [CrossRef]
- Nair, J.J.; Machocho, A.K.; Campbell, W.E.; Brun, R.; Viladomat, F.; Codina, C.; Bastida, J. Alkaloids from Crinum macowanii. Phytochemistry 2000, 54, 5. [Google Scholar] [CrossRef]
- Elgorashi, E.E.; Drewes, S.E.; Staden, J. Van Alkaloids from Crinum bulbispermum. Phytochemistry 1999, 52, 533–536. [Google Scholar] [CrossRef]
- Tallini, L.R.; Carrasco, A.; Acosta León, K.; Vinueza, D.; Bastida, J.; Oleas, N.H. Alkaloid Profiling and Cholinesterase Inhibitory Potential of Crinum × amabile Donn. (Amaryllidaceae) Collected in Ecuador. Plants 2021, 10, 2686. [Google Scholar] [CrossRef] [PubMed]
- Tallini, L.R.; Torras-Claveria, L.; de Borges, W.S.; Kaiser, M.; Viladomat, F.; Zuanazzi, J.A.S.; Bastida, J. N-oxide alkaloids from Crinum amabile (Amaryllidaceae). Molecules 2018, 23, 1277. [Google Scholar] [CrossRef] [PubMed]
- Panthong, K.; Ingkaninan, K. Amabiloid A from Crinum × amabile Donn ex Ker Gawl. Nat. Prod. Res. 2021, 35, 3220–3225. [Google Scholar] [CrossRef] [PubMed]
- Bordoloi, M.; Kotoky, R.; Mahanta, J.J.; Sarma, T.C.; Kanjilal, P.B. Anti-genotoxic hydrazide from Crinum defixum. Eur. J. Med. Chem. 2009, 44, 2754–2757. [Google Scholar] [CrossRef]
- Fennell, C.W.; Elgorashi, E.E.; Van Staden, J. Alkaloid production in Crinum moorei cultures. J. Nat. Prod. 2003, 66, 1524–1526. [Google Scholar] [CrossRef]
- Masi, M.; Koirala, M.; Delicato, A.; Di Lecce, R.; Merindol, N.; Ka, S.; Seck, M.; Tuzi, A.; Desgagne-Penix, I.; Calabrò, V.; et al. Isolation and Biological Characterization of Homoisoflavanoids and the Alkylamide N-p-Coumaroyltyramine from Crinum biflorum Rottb., an Amaryllidaceae Species Collected in Senegal. Biomolecules 2021, 11, 1298. [Google Scholar] [CrossRef]
- Elgorashi, E.; Drewes, S.E.; Van Staden, J. Alkaloids from Crinum moorei. Phytochemistry 2001, 56, 637–640. [Google Scholar] [CrossRef]
- Yu, M.; Wang, B.; Qi, Z.; Xin, G.; Li, W. Response surface method was used to optimize the ultrasonic assisted extraction of flavonoids from Crinum asiaticum. Saudi J. Biol. Sci. 2019, 26, 2079–2084. [Google Scholar] [CrossRef]
- Khumkhrong, P.; Piboonprai, K.; Chaichompoo, W.; Pimtong, W.; Khongkow, M.; Namdee, K.; Jantimaporn, A.; Japrung, D.; Asawapirom, U.; Suksamrarn, A.; et al. Crinamine Induces Apoptosis and Inhibits Proliferation, Migration, and Angiogenesis in Cervical Cancer SiHa Cells. Biomolecules 2019, 9, 494. [Google Scholar] [CrossRef]
- Sun, Q.; Shen, Y.H.; Tian, J.M.; Tang, J.; Su, J.; Liu, R.H.; Li, H.L.; Xu, X.K.; Zhang, W.D. Chemical constituents of Crinum asiaticum L. var. sinicum Baker and their cytotoxic activities. Chem. Biodivers. 2009, 6, 1751–1757. [Google Scholar] [CrossRef]
- Kim, S.C.; Kang, J.; Kim, M.K.; Hyun, J.H.; Boo, H.J.; Park, D.B.; Lee, Y.J.; Yoo, E.S.; Kim, Y.H.; Kim, Y.H.; et al. Promotion effect of norgalanthamine, a component of Crinum asiaticum, on hair growth. Eur. J. Dermatol. 2010, 20, 42–48. [Google Scholar] [CrossRef]
- Kogure, N.; Katsuta, N.; Kitajima, M.; Takayama, H. Two new alkaloids from Crinum asiaticum var. sinicum. Chem. Pharm. Bull. 2011, 59, 1545–1548. [Google Scholar] [CrossRef] [PubMed]
- Do, K.M.; Shin, M.K.; Kodama, T.; Win, N.N.; Prema, P.; Nguyen, H.M.; Hayakawa, Y.; Morita, H. Flavanols and Flavanes from Crinum asiaticum and their Effects on LPS Signaling Pathway through the Inhibition of NF-κB Activation. Planta Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Chen, Y.; Liu, Y.; Xu, Y.; Wang, B. Efficient polysaccharides from Crinum asiaticum L.’s structural characterization and anti-tumor effect. Saudi J. Biol. Sci. 2019, 26, 2085–2090. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Sugiura, Y.; Funasaki, M.; Kagechika, H.; Ishibashi, M.; Ohsaki, A. Two new alkaloids from Crinum asiaticum var. japonicum. J. Nat. Med. 2019, 73, 648–652. [Google Scholar] [CrossRef]
- Min, B.S.; Gao, J.J.; Nakamura, N.; Kim, Y.H.; Hattori, M. Cytotoxic alkaloids and a flavan from the bulbs of Crinum asiaticum var. japonicum. Chem. Pharm. Bull. 2001, 49, 1217–1219. [Google Scholar] [CrossRef]
- Kim, Y.H.; Park, E.J.; Park, M.H.; Badarch, U.; Woldemichael, G.M.; Beutler, J.A. Crinamine from Crinum asiaticum var. japonicum inhibits hypoxia inducible factor-1 activity but not activity of hypoxia inducible factor-2. Biol. Pharm. Bull. 2006, 29, 2140–2142. [Google Scholar] [CrossRef][Green Version]
- Machocho, A.K.; Bastida, J.; Codina, C.; Viladomat, F.; Brun, R.; Chhabra, S.C. Augustamine type alkaloids from Crinum kirkii. Phytochemistry 2004, 65, 3143–3149. [Google Scholar] [CrossRef]
- Presley, C.C.; Du, Y.; Dalal, S.; Merino, E.F.; Butler, J.H.; Rakotonandrasana, S.; Rasamison, V.E.; Cassera, M.B.; Kingston, D.G.I. Isolation, structure elucidation, and synthesis of antiplasmodial quinolones from Crinum firmifolium. Bioorganic Med. Chem. 2017, 25, 4203–4211. [Google Scholar] [CrossRef]
- Chen, M.X.; Huo, J.M.; Hu, J.; Xu, Z.P.; Zhang, X. Amaryllidaceae alkaloids from Crinum latifolium with cytotoxic, antimicrobial, antioxidant, and anti-inflammatory activities. Fitoterapia 2018, 130, 48–53. [Google Scholar] [CrossRef]
- Tian, H.; Liu, Q.J.; Wang, J.T.; Zhang, L. Antimicrobial crinane-type alkaloids from the bulbs of Crinum latifolium. J. Asian Nat. Prod. Res. 2021, 23, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Nam, N.H.; Kim, Y.; You, Y.J.; Hong, D.H.; Kim, H.M.; Ahn, B.Z. New constituents from Crinum latifolium with inhibitory effects against tube-like formation of human umbilical venous endothelial cells. Nat. Prod. Res. 2004, 18, 485–491. [Google Scholar] [CrossRef]
- N’Tamon, A.D.; Okpekon, A.T.; Bony, N.F.; Bernadat, G.; Gallard, J.F.; Kouamé, T.; Séon-Méniel, B.; Leblanc, K.; Rharrabti, S.; Mouray, E.; et al. Streamlined targeting of Amaryllidaceae alkaloids from the bulbs of Crinum scillifolium using spectrometric and taxonomically-informed scoring metabolite annotations. Phytochemistry 2020, 179, 112485. [Google Scholar] [CrossRef] [PubMed]
- Berkov, S.; Romani, S.; Herrera, M.; Viladomat, F.; Codina, C.; Momekov, G.; Ionkova, I.; Bastida, J. Antiproliferative alkaloids from Crinum zeylanicum. Phytother. Res. 2011, 25, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Ka, S.; Masi, M.; Merindol, N.; Di Lecce, R.; Plourde, M.B.; Seck, M.; Górecki, M.; Pescitelli, G.; Desgagne-Penix, I.; Evidente, A. Gigantelline, gigantellinine and gigancrinine, cherylline- and crinine-type alkaloids isolated from Crinum jagus with anti-acetylcholinesterase activity. Phytochemistry 2020, 175, 112390. [Google Scholar] [CrossRef]
- Cortes, N.; Sierra, K.; Alzate, F.; Osorio, E.H.; Osorio, E. Alkaloids of Amaryllidaceae as Inhibitors of Cholinesterases (AChEs and BChEs): An Integrated Bioguided Study. Phytochem. Anal. 2017, 29, 217–227. [Google Scholar] [CrossRef]
- Abebe, B.; Tadesse, S.; Hymete, A.; Bisrat, D. Antiproliferative Effects of Alkaloids from the Bulbs of Crinum abyscinicum Hochst. ExA. Rich. Evid. Based. Complement. Alternat. Med. 2020, 2020, 2529730. [Google Scholar] [CrossRef]
- Presley, C.C.; Krai, P.; Dalal, S.; Su, Q.; Cassera, M.; Goetz, M.; Kingston, D.G.I. New potently bioactive alkaloids from Crinum erubescens. Bioorganic Med. Chem. 2016, 24, 5418–5422. [Google Scholar] [CrossRef]
- Abdel-Halim, O.B.; Marzouk, A.M.; Mothana, R.; Awadh, N. A new tyrosinase inhibitor from Crinum yemense as potential treatment for hyperpigmentation. Pharmazie 2008, 63, 405–407. [Google Scholar] [PubMed]
- Abdel-Halim, O.B.; Morikawa, T.; Ando, S.; Matsuda, H.; Yoshikawa, M. New crinine-type alkaloids with inhibitory effect on induction of inducible nitric oxide synthase from Crinum yemense. J. Nat. Prod. 2004, 67, 1119–1124. [Google Scholar] [CrossRef]
- Aboul-Ela, M.A.; El-Lakany, A.M.; Hammoda, H.M. Alkaloids from the bulbs of Crinum bulbispermum. Pharmazie 2004, 59, 894–896. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, M.A.; Kamel, M.S.; Ohtani, K.; Kasai, R.; Yamasaki, K. Minor phenolics from Crinum bulbispermum bulbs. Phytochemistry 2000, 54, 891–896. [Google Scholar] [CrossRef]
- Ali, A.; Ramadan, M.; Frahm, A. Alkaloidal Constituents of Crinum bulbispermum III: Bulbispermine—A New Alkaloid of Crinum bulbispermum. Planta Med. 1984, 50, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Kissling, J.; Ioset, J.R.; Marston, A.; Hostettmann, K. Bio-guided isolation of cholinesterase inhibitors from the bulbs of Crinum x powellii. Phytother. Res. 2005, 19, 984–987. [Google Scholar] [CrossRef]
- Niño, J.; Hincapié, G.M.; Correa, Y.M.; Mosquera, O.M. Alkaloids of Crinum x powellii “Album” (Amaryllidaceae) and their topoisomerase inhibitory activity. Z. Naturforsch. C 2007, 62, 223–226. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Houghton, P.J.; Agbedahunsi, J.M.; Adegbulugbe, A. Choline esterase inhibitory properties of alkaloids from two Nigerian Crinum species. Phytochemistry 2004, 65, 2893–2896. [Google Scholar] [CrossRef]
- Nkanwen, E.R.S.; Gatsing, D.; Ngamga, D.; Fodouop, S.P.C.; Tane, P. Antibacterial agents from the leaves of Crinum purpurascens herb (Amaryllidaceae). Afr. Health Sci. 2009, 9, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Refaat, J.; Kamel, M.S.; Ramadan, M.A.; Ali, A.A. Crinum; An endless source of bioactive principles: A Review. Part V. Biological Profile. Int. J. Pharm. Sci. Res. 2013, 4, 1239–1252. [Google Scholar]
- Mahomoodally, M.F.; Sadeer, N.B.; Suroowan, S.; Jugreet, S.; Lobine, D.; Rengasamy, K.R.R. Ethnomedicinal, phytochemistry, toxicity and pharmacological benefits of poison bulb—Crinum asiaticum L. S. Afr. J. Bot. 2021, 136, 16–29. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, K.H.; Han, C.S.; Park, S.H.; Yang, H.C.; Lee, B.Y.; Eom, S.-Y.; Kim, Y.-S.; Kim, J.-H.; Lee, N.H. Anti-inflammatory activity of Crinum asiaticum Linne var. japonicum extract and its application as a cosmeceutical ingredient. J. Cosmet. Sci. 2008, 59, 419–430. [Google Scholar]
- Samud, A.M.; Asmawi, M.Z.; Sharma, J.N.; Yusof, A.P.M. Anti-inflammatory activity of Crinum asiaticum plant and its effect on bradykinin-induced contractions on isolated uterus. Immunopharmacology 1999, 43, 311–316. [Google Scholar] [CrossRef]
- Rahman, A.S.M.; Azad, H.; Nazim, U.A. Analgesic and anti-inflammatory effects of Crinum asiaticum leaf alcoholic extract in animal models. Afr. J. Biotechnol. 2013, 12, 212–218. [Google Scholar] [CrossRef][Green Version]
- Minkah, P.A.B.; Danquah, C.A. Anti-infective, anti-inflammatory and antipyretic activities of the bulb extracts of Crinum jagus (J. Thomps.) Dandy (Amaryllidaceae). Sci. Afr. 2021, 12, e00723. [Google Scholar] [CrossRef]
- Ratnasooriya, W.D.; Deraniyagala, S.A.; Bathige, S.D.N.K.; Hettiarachchi, H.D.I. Leaf extract of Crinum bulbispermum has antinociceptive activity in rats. J. Ethnopharmacol. 2005, 97, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, B. Chemical composition and antifungal, phytotoxic, brine shrimp cytotoxicity, insecticidal and antibacterial activities of the essential oils of Acacia modesta. J. Med. Plants Res. 2012, 6, 4653–4659. [Google Scholar] [CrossRef]
- Alawode, T.T.; Lajide, L.; Owolabi, B.J.; Olaleye, M.T. Evaluation of Extracts of Leaves of Crinum jagus for Antimicrobial Properties. J. Appl. Sci. Environ. Manag. 2020, 24, 1197–1201. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Nguyen, N.Y.T.; Chau, N.T.N.; Nguyen, A.B.T.; Tran, V.K.T.; Hoang, V.; Le, T.M.; Wang, H.C.; Yen, C.H. Bioassay-guided discovery of potential partial extracts with cytotoxic effects on liver cancer cells from vietnamese medicinal herbs. Processes 2021, 9, 1956. [Google Scholar] [CrossRef]
- Mannan, A.; Kawser, M.J.; Ahmed, A.M.A.; Islam, N.N.; Alam, S.M.M.; Emon, M.A.E.K.; Gupta, S. Das Assessment of antibacterial, thrombolytic and cytotoxic potential of cassia alata seed oil. J. Appl. Pharm. Sci. 2011, 1, 56–59. [Google Scholar]
- Yui, S.; Mikami, M.; Kitahara, M.; Yamazaki, M. The inhibitory effect of lycorine on tumor cell apoptosis induced by polymorphonuclear leukocyte-derived calprotectin. Immunopharmacology 1998, 40, 151–162. [Google Scholar] [CrossRef]
- Patel, D. Crinum asiaticum Linn: A Medicinal Herb as Well as Ornamental Plant in Central India. Int. J. Environ. Sci. Nat. Resour. 2017, 6, 1–7. [Google Scholar] [CrossRef]
- Hyun, J.H.; Kang, J.; Kim, S.C.; Kim, E.; Kang, J.H.; Kwon, J.M.; Park, D.B.; Lee, Y.J.; Yoo, E.S.; Kang, H.K. The effects of crinum asiaticum on the apoptosis induction and the reversal of multidrug resistance in hl-60/mx2. Toxicol. Res. 2008, 24, 29–36. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yusoff, S.M. Anti-angiogenesis as a possible mechanism of action for anti-tumor (potential anti-cancer) activity of Crinum asiaticum leaf methanol extract. J. Angiother. 2017, 1, E012–E017. [Google Scholar] [CrossRef]
- Tan, W.-N.; Shahbudin, F.N.; Mohamed Kamal, N.N.S.N.; Tong, W.-Y.; Leong, C.-R.; Lim, J.-W. Volatile Constituents of the Leaf Essential Oil of Crinum asiaticum and their Antimicrobial and Cytotoxic Activities. J. Essent. Oil Bear. Plants 2019, 22, 947–954. [Google Scholar] [CrossRef]
- Lim, H.S.; Kim, Y.; Kim, Y.J.; Sohn, E.; Kim, J.H.; Jeong, S.J. The Effects of Crinum asiaticum var. japonicum Baker Seeds on Neuroprotection and Antineuroinflammation in Neuronal Cell Lines. Nat. Prod. Commun. 2020, 15, 10. [Google Scholar] [CrossRef]
- Seoposengwe, K.; van Tonder, J.J.; Steenkamp, V. In vitro neuroprotective potential of four medicinal plants against rotenone-induced toxicity in SH-SY5Y neuroblastoma cells. BMC Complement. Altern. Med. 2013, 13, 353. [Google Scholar] [CrossRef] [PubMed]
- Ofori, M.; Danquah, C.A.; Ativui, S.; Doe, P.; Asamoah, W.A. In-Vitro Anti-Tuberculosis, Anti-Efflux Pumps and Anti-Biofilm Effects of Crinum Asiaticum Bulbs. Biomed. Pharmacol. J. 2021, 14, 1905–1915. [Google Scholar] [CrossRef]
- Goswami, S.; Das, R.; Ghosh, P.; Chakraborty, T.; Barman, A.; Ray, S. Comparative antioxidant and antimicrobial potentials of leaf successive extract fractions of poison bulb, Crinum asiaticum L. Ind. Crops Prod. 2020, 154, 112667. [Google Scholar] [CrossRef]
- Fu, L.; Zheng, Y.; Zhang, P.; Zhang, H.; Xu, Y.; Zhou, J.; Zhang, H.; Karimi-Maleh, H.; Lai, G.; Zhao, S.; et al. Development of an electrochemical biosensor for phylogenetic analysis of Amaryllidaceae based on the enhanced electrochemical fingerprint recorded from plant tissue. Biosens. Bioelectron. 2020, 159, 112212. [Google Scholar] [CrossRef]
- Min, B.S.; Kim, Y.H.; Tomiyama, M.; Nakamura, N.; Miyashiro, H.; Otake, T.; Hattori, M. Inhibitory effects of Korean plants on HIV-1 activities. Phytother. Res. 2001, 15, 481–486. [Google Scholar] [CrossRef]
- Naira, J.J.; Van Staden, J.; Bonnet, S.L.; Wilhelm, A. Antibacterial properties of the family amaryllidaceae: Evaluation of plant extracts in vitro. Nat. Prod. Commun. 2017, 12, 1145–1151. [Google Scholar] [CrossRef]
- Surain, P.; Aneja, K.R. Anticandidal potential of Crinum asiaticum leaves extract against selected oral and vaginal Candida pathogens. J. Innov. Biol. 2014, 6473, 27–30. [Google Scholar]
- Noubissi, P.A.; Fokam Tagne, M.A.; Fankem, G.O.; Ngakou Mukam, J.; Wambe, H.; Kamgang, R. Effects of Crinum jagus Water/Ethanol Extract on Shigella flexneri-Induced Diarrhea in Rats. Evid. -Based Complement. Altern. Med. 2019, 2019, 9537603. [Google Scholar] [CrossRef] [PubMed]
- Udegbunam, S.O.; Udegbunam, R.I.; Nnaji, T.O.; Anyanwu, M.U.; Kene, R.O.C.; Anika, S.M. Antimicrobial and antioxidant effect of methanolic Crinum jagus bulb extract in wound healing. J. Intercult. Ethnopharmacol. 2015, 4, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Azikiwe, C.; Amazu, L. The potential organo-toxicity safety of Morpholine and Crinum jagus in rats. Discovery 2015, 10, 113–120. [Google Scholar]
- Akintola, A.O.; Kehinde, A.O.; Adebiyi, O.E.; Ademowo, O.G. Anti-tuberculosis activities of the crude methanolic extract and purified fractions of the bulb of Crinum jagus. Niger. J. Physiol. Sci. 2013, 28, 135–140. [Google Scholar]
- Ka, S.; Merindol, N.; Sow, A.A.; Singh, A.; Landelouci, K.; Plourde, M.B.; Pépin, G.; Masi, M.; Di Lecce, R.; Evidente, A.; et al. Amaryllidaceae Alkaloid Cherylline Inhibits the Replication of Dengue and Zika Viruses. Antimicrob. Agents Chemother. 2021, 65, e0039821. [Google Scholar] [CrossRef]
- Maroyi, A. A review of ethnoboatany, therapeutic value, phytochemistry and pharmacology of Crinum macowanii Baker: A highly traded bulbous plant in Southern Africa. J. Ethnopharmacol. 2016, 194, 595–608. [Google Scholar] [CrossRef]
- Ilavenil, S.; Kaleeswaran, B.; Sumitha, P.; Tamilvendan, D.; Ravikumar, S. Protection of human erythrocyte using Crinum asiaticum extract and lycorine from oxidative damage induced by 2-amidinopropane. Saudi J. Biol. Sci. 2011, 18, 181–187. [Google Scholar] [CrossRef]
- Uddin, Z.; Bin, T.; Kumar, A.; Jenny, A.; Dutta, M.; Morshed, M.; Kawsar, H. Anti-Inflammatory and Antioxidant Activity of Leaf extract of Crinum asiaticum. J. Pharm. Res. 2015, 5, 5553–5556. [Google Scholar]
- Indradevi, S.; Ilavenil, S.; Kaleeswaran, B.; Srigopalram, S.; Ravikumar, S. Ethanolic extract of Crinum asiaticum attenuates hyperglycemia-mediated oxidative stress and protects hepatocytes in alloxan induced experimental diabetic rats. J. King Saud. Univ. Sci. 2015, 24, 171–177. [Google Scholar] [CrossRef]
- Ghane, S.G.; Attar, U.A.; Yadav, P.B.; Lekhak, M.M. Antioxidant, anti-diabetic, acetylcholinesterase inhibitory potential and estimation of alkaloids (lycorine and galanthamine) from Crinum species: An important source of anticancer and anti-Alzheimer drug. Ind. Crops Prod. 2018, 125, 168–177. [Google Scholar] [CrossRef]
- Alawode, T.T.; Lajide, L.; Owolabi, B.J.; Olaleye, M.T. Studies on In vitro Antioxidant and Anti-Inflammatory Activities of Crinum jagus Leaves and Bulb Extracts. Int. J. Biochem. Res. Rev. 2019, 28, 1–9. [Google Scholar] [CrossRef]
- Adewusi, E.A.; Steenkamp, V. In vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from southern Africa. Asian Pac. J. Trop. Med. 2011, 4, 829–835. [Google Scholar] [CrossRef]
- Chahal, S.; Lekhak, M.M.; Kaur, H.; Shekhawat, M. Unraveling the medicinal potential and conservation of Indian Crinum Unraveling the medicinal potential and conservation of Indian Crinum (Amaryllidaceae) species. S. Afr. J. Bot. 2020, 136, 7–15. [Google Scholar] [CrossRef]
- Kang, J.; Choi, J.H.; Lee, J.G.; Yoo, E. The Mechanism of Crinum asiaticum var. japonicum on the Activation of Anagen. Korean J. Pharmacogn. 2017, 48, 148–154. [Google Scholar]
- Jeong, Y.J.; Sohn, E.H.; Jung, Y.H.; Yoon, W.J.; Cho, Y.M.; Kim, I.; Lee, S.R.; Kang, S.C. Anti-obesity effect of Crinum asiaticum var. japonicum Baker extract in high-fat diet-induced and monogenic obese mice. Biomed. Pharmacother. 2016, 82, 35–43. [Google Scholar] [CrossRef]
- Taiwe, G.S.; Tchoya, T.B.; Menanga, J.R.; Dabole, B.; De Waard, M. Anticonvulsant activity of an active fraction extracted from Crinum jagus L. (Amaryllidaceae), and its possible effects on fully kindled seizures, depression-like behaviour and oxidative stress in experimental rodent models. J. Ethnopharmacol. 2016, 194, 421–433. [Google Scholar] [CrossRef]
- Heinrich, M. Galanthamine from Galanthus and other Amaryllidaceae--chemistry and biology based on traditional use. Alkaloids. Chem. Biol. 2010, 68, 157–165. [Google Scholar] [CrossRef]
- Jilani, M.S.; Tagwireyi, D.; Gadaga, L.L.; Maponga, C.C.; Mutsimhu, C. Cognitive-Enhancing Effect of a Hydroethanolic Extract of Crinum macowanii against Memory Impairment Induced by Aluminum Chloride in BALB/c Mice. Behav. Neurol. 2018, 2018, 2057219. [Google Scholar] [CrossRef]
- Snijman, D.A.; Meerow, A.W. Floral and macroecological evolution within Cyrtanthus (Amaryllidaceae): Inferences from combined analyses of plastid ndhF and nrDNA ITS sequences. S. Afr. J. Bot. 2010, 76, 217–238. [Google Scholar] [CrossRef]
- Galley, C.; Bytebier, B.; Bellstedt, D.U.; Linder, H.P. The Cape element in the Afrotemperate flora: From Cape to Cairo? Proc. R. Soc. B Biol. Sci. 2007, 274, 535–543. [Google Scholar] [CrossRef]
- Mucina, L.; Rutherford, M.C. The Vegetation of South Africa, Lesotho and Swaziland; South African National Biodiversity Institute: Cape Town, South Africa, 2006. [Google Scholar]
- Born, J.; Linder, H.P.; Desmet, P. The Greater Cape Floristic Region. J. Biogeogr. 2007, 34, 147–162. [Google Scholar] [CrossRef]
- Cowling, R.M.; Procheş, Ş.; Vlok, J.H.J. On the origin of southern African subtropical thicket vegetation. S. Afr. J. Bot. 2005, 71, 1–23. [Google Scholar] [CrossRef]
- Bhat, R.B.; Jacobs, T.V. Traditional herbal medicine in Transkei. J. Ethnopharmacol. 1995, 48, 7–12. [Google Scholar] [CrossRef]
- Nwude, N.; Ebong, O.O. Some plants used in the treatment of leprosy in Africa. Lepr. Rev. 1980, 51, 11–18. [Google Scholar] [CrossRef]
- Rárová, L.; Ncube, B.; Van Staden, J.; Fürst, R.; Strnad, M.; Gruz, J. Identification of Narciclasine as an in Vitro Anti-Inflammatory Component of Cyrtanthus contractus by Correlation-Based Metabolomics. J. Nat. Prod. 2019, 82, 1372–1376. [Google Scholar] [CrossRef]
- Hutchings, A.; Scott, A.H.; University of Zululand; National Botanical Institute (South Africa). Zulu Medicinal Plants: An Inventory; University of Natal Press: Scottsville, South Africa, 1996; ISBN 0869808931. [Google Scholar]
- Nair, J.J.; Bastida, J.; Codina, C.; Viladomat, F.; Van Staden, J. Alkaloids of the South African amaryllidaceae: A review. Nat. Prod. Commun. 2013, 8, 1335–1350. [Google Scholar] [CrossRef]
- Mahlangeni, N.T.; Moodley, R.; Jonnalagadda, S.B. Phytochemical analysis of Cyrtanthus obliquus bulbs from the informal street market of Kwazulu-Natal, South Africa. Afr. J. Tradit. Complement. Altern. Med. 2015, 12, 28–34. [Google Scholar] [CrossRef][Green Version]
- Brine, N.D.; Campbell, W.E.; Bastida, J.; Herrera, M.R.; Viladomat, F.; Codina, C.; Smith, P.J. A dinitrogenous alkaloid from Cyrtanthus obliquus. Phytochemistry 2002, 61, 443–447. [Google Scholar] [CrossRef]
- Masi, M.; Mubaiwa, B.; Mabank, T.; Karakoyun; Cimmino, A.; Van Otterlo, W.A.L.; Green, I.R.; Evidente, A. Alkaloids isolated from indigenous South African Amaryllidaceae: Crinum buphanoides (Welw. ex Baker), Crinum graminicola (I. Verd.), Cyrtanthus mackenii (Hook. f) and Brunsvigia grandiflora (Lindl). S. Afr. J. Bot. 2018, 118, 188–191. [Google Scholar] [CrossRef]
- Elgorashi, E.E.; Van Staden, J. Pharmacological screening of six Amaryllidaceae species. J. Ethnopharmacol. 2004, 90, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Elgorashi, E.E.; Van Staden, J. Alkaloids from Cyrtanthus falcatus. S. Afr. J. Bot. 2003, 69, 593–594. [Google Scholar] [CrossRef][Green Version]
- Weniger, B.; Italiano, L.; Beck, J.P.; Bastida, J.; Bergonon, S.; Codina, C.; Lobstein, A.; Anton, R. Cytotoxic activity of Amaryllidaceae alkaloids. Planta Med. 1995, 61, 77–79. [Google Scholar] [CrossRef] [PubMed]
- Elgorashi, E.E.; Stafford, G.I.; Mulholland, D.; Van Staden, J. Isolation of captan from Cyrtanthus suaveolens: The effect of pesticides on the quality and safety of traditional medicine. S. Afr. J. Bot. 2004, 70, 512–514. [Google Scholar] [CrossRef][Green Version]
- Kim, H.W.; Wang, M.; Leber, C.A.; Nothias, L.F.; Reher, R.; Kang, K.B.; van der Hooft, J.J.J.; Dorrestein, P.C.; Gerwick, W.H.; Cottrell, G.W. NPClassifier: A Deep Neural Network-Based Structural Classification Tool for Natural Products. J. Nat. Prod. 2021, 84, 2795–2807. [Google Scholar] [CrossRef] [PubMed]
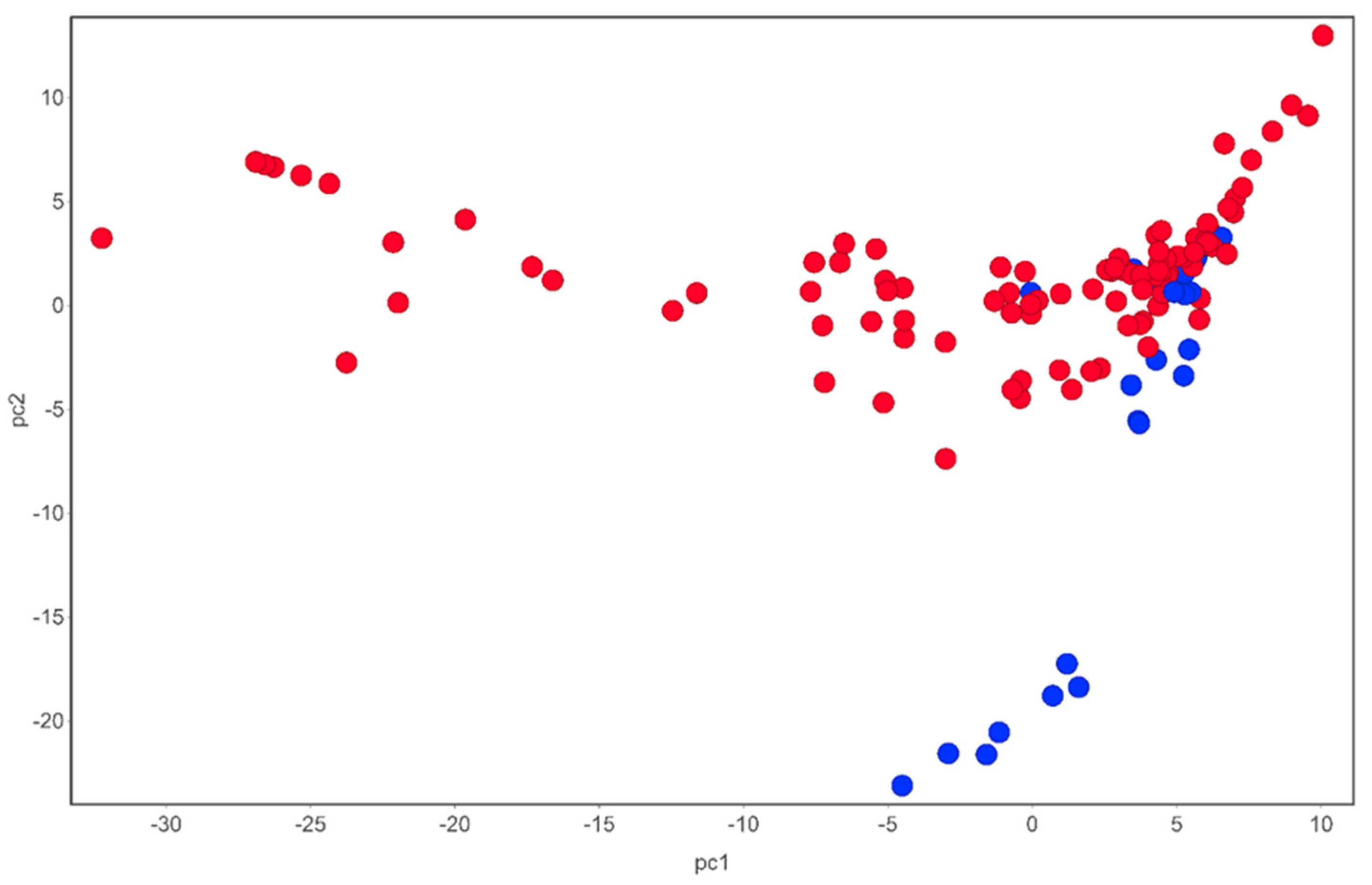


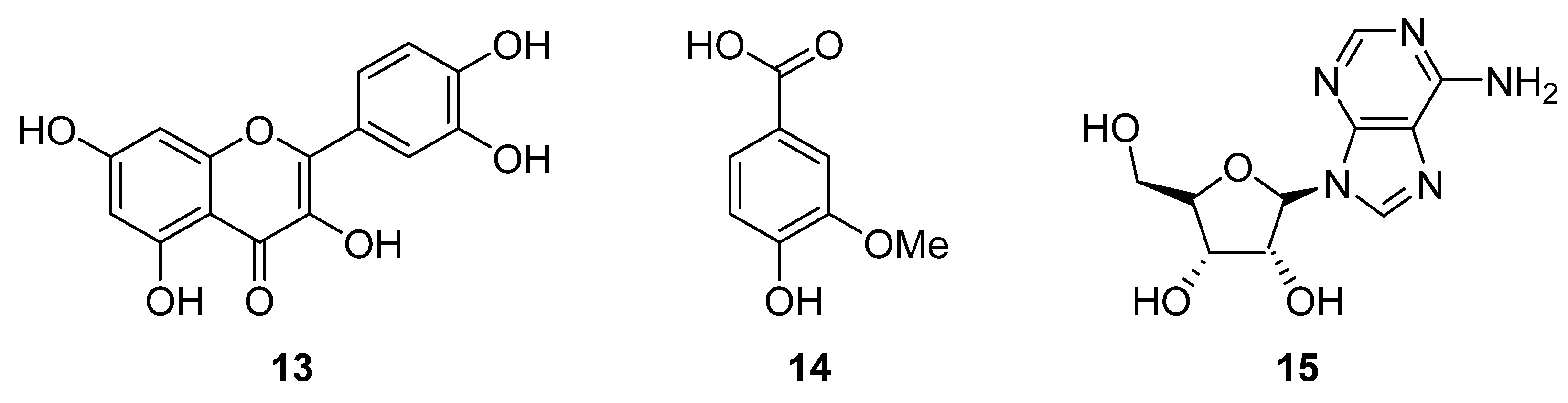


| Plant Species | Geographical Distribution | Traditional Uses | References |
|---|---|---|---|
| T. violacea | Indigenous to the Eastern Cape, KwaZulu-Natal, Gauteng, Free State and Mpumalanga Provinces of South Africa. | The leaves and bulbs are used in the management of fever and colds, tuberculosis, asthma, and stomach problems. The leaves are eaten as vegetables and for the management of oesophageal cancer. It is also used as a snake repellent. | [8,50] |
| T. alliacea | Native to South Africa and grows mostly in the Eastern Cape and southern KwaZulu-Natal Provinces of South Africa. | Its bruised rhizome is used locally in bathwater to relieve fever, rheumatism, and paralysis, and in small doses as a laxative. T. alliacea is used for the management of stomach problems, asthma, and pulmonary tuberculosis. Its rhizome infusion is administered as an enema. | [8,51] |
| T. simmleri | Native to the South African Drakensberg mountains growing as isolated plants on rocky ledges. | Bulbs and leaves are used as a remedy for gastrointestinal ailments, enemas, high blood pressure, heart problems, chest complaints, high cholesterol, constipation, rheumatism, asthma, fever, pulmonary tuberculosis, earache, human immunodeficiency virus (HIV), paralysis, and cardiovascular diseases. | [50,52] |
| T. acutiloba | Found in the rainfall regions of southern Africa, occurring in the Eastern Cape, KwaZulu-Natal, Limpopo, Free State, Gauteng, North West, and Mpumalanga Provinces of South Africa, as well as in Lesotho, Swaziland and Botswana. | T. acutiloba leaves are used as a culinary herb and snake repellent. It is used to treat barrenness, flu, bad breath, and as an aphrodisiac. It is also cultivated to keep snakes away from the homestead. | [8] |
| T. natalensis | Although native to South Africa, but is now grown worldwide. | It is used as a culinary herb and snake repellent. | [53] |
| T. cernua | Commonly found in the Eastern Cape, Free State, Gauteng, KwaZulu-Natal, Limpopo, Mpumalanga, North West and Western Cape Provinces of South Africa. | It is used for ornamental purposes. | [8] |
| T. leucantha | Widely distributed in southern Africa including Botswana, Lesotho, South Africa, Swaziland, Zambia, and Zimbabwe. | Its rhizome is scraped clean and boiled in stews or roasted as a vegetable. Its leaves and stems are used as a culinary herb and protective charm. | [53] |
| T. ludwigiana | Commonly found in the Eastern Cape, KwaZulu-Natal, Northern Provinces of South Africa and in Swaziland. | It is traditionally used as a love charm. | [53] |
| Country | No. of Documents * |
|---|---|
| South Africa | 99 |
| United Kingdom | 15 |
| United States | 12 |
| Czech Republic | 8 |
| Italy | 7 |
| India | 6 |
| Germany | 5 |
| Australia | 3 |
| China | 3 |
| Belgium | 2 |
| Disease | No. of Published Studies # |
|---|---|
| Antimicrobial | 26 |
| Cancer | 11 |
| Antioxidant | 13 |
| Diabetes | 2 |
| Cardiovascular | 12 |
| Antithrombogenic | 2 |
| Miscellaneous | 17 |
| Plant Species | Extraction Solvent | Plant Part Used | Biological Activity | References |
|---|---|---|---|---|
| T. violacea | Dichloromethane | Bulbs | MIC ranging from 20 to 300 µg/mL against Bacillus subtilis, methicillin-resistant S. aureus, S. epidermidis, E. coli, K. pneumoniae, P. aeruginosa, C. albicans and C. parapsilosis. | [67] |
| T. violacea | Hexane and ethanol | Flowers and callus cultures | Moderate to strong broad-antimicrobial (E. coli, P. aeruginosa, S. aureus, Aspergillus niger and C. albicans) activity observed by zone of inhibition in the agar well disc diffusion method. | [68] |
| T. violacea | Water | Bulbs | Significant reduction in A. flavus β-glucan and chitin synthesis corresponding to a dose-dependent inhibition of the enzymes β-glucan and chitin synthase, respectively.This results in inhibition of ergosterol production in the fungus. | [70,71] |
| T. violacea | Acetone | Bulbs | Varied light intensities, pH and watering frequencies substantially impacted both growth and potency of plant extracts against the fungi F. oxysporum. | [73,74] |
| T. violacea | Water | Roots, bulbs, leaves and flowers | Significantly compromised population densities of the nematode M. incognita race 2 on tomato roots and in the soil. | [75] |
| T. violacea | Dichloromethane | Bulbs | Antiparasitic activity against T. brucei (IC50 = 2.83 µg/mL) and L. tarentolae (IC50 = 6.29 µg/mL). | [67] |
| Plant Species | Extraction Solvent | Plant Part Used | Biological Activity | References |
|---|---|---|---|---|
| T. violacea | Methanol | Leaves and roots | Marked time- and dose-dependent cytotoxic effect on cancer cell lines. Induced apoptosis using p53-independent pathway. | [78] |
| T. violacea | Methanol, butanol, and hexane | Leaves | Methanol extract was prolific against multiple cell lines. Hela and ME-180 cell lines treated with methanol and hexane extracts showed an increase in caspase 3/7 activity. Both methanol and hexane extracts induced a 10-fold increase in expression of p53 gene in Hela cells. | [79] |
| T. violacea | Methanol:water:formic acid (80:20:0.1, v/v/v) | Flowers | Demonstrated activity against ovarian tumor cells. | [80] |
| T. violacea | Water and methanol | Leaves | Water-soluble extract emerged as the most cytotoxic (IC50 = 314 µg/mL), compared to the methanol extract (IC50 = 780 µg/mL), against the MDA-MB-231 triple-negative breast cancer cell line. Water-soluble extract prevented cell migration completely for 13 h at 300 µg/mL. | [82] |
| T. violacea | Hexane and ethanol | Flowers and callus cultures | Extracts showed marked cytotoxicity (60–74% growth inhibition at 250 µg/mL) against three different cell lines (Hep G2, PC-3 and MCF-7). | [68] |
| T. violacea | Acetone and water | Leaves | Anticancer activity against oral cancer with an IC50 (acetone extract) of 0.2 mg/mL; IC50 (water extract) of 1 mg/mL. | [84] |
| T. violacea | Methanol:water (1:1) | Whole plants | Two pro-apoptotic glucopyranosides d-fructofuranosyl-β (2→6)-methyl-α-d-glucopyranoside and β-d-fructofuranosyl-(2→6)-α-d-glucopyranoside isolated and identified as active anticancer agents in the plant. | [85] |
| T. violacea | Water | Whole plants | MoA of the three compounds, namely methyl-α-d-glucopyranoside, d-fructofuranosyl-β (2→6)-methyl-α-d-glucopyranoside and β-d-fructofuranosyl-(2→6)-α-d-glucopyranoside isolated from the water extract, deciphered to be through induction of apoptosis by targeting the mitochondrial (intrinsic) pathway | [86] |
| Plant Species | Extraction Solvent | Plant Part Used | Biological Activity | References |
|---|---|---|---|---|
| T. violacea | Water | Leaves | Dose-dependent antioxidant activity measured using the DPPH (Log IC50 = 0.49 mg/mL) and ABTS (Log IC50 = 0.24 mg/mL) assays | [92] |
| T. violacea | Methanol/water/formic acid (80:20:0.1, v/v/v) | Flowers | Marked antioxidant activity was observed using 3 different types of assays, namely DPPH, FRAP and TREC | [80] |
| T. acutiloba | Hydro-methanolic extracts | Roots, rhizomes, leaves and flowers | Dose-dependent antioxidant activity observed with the rhizome extract emerging as the most active plant part (IC50 DPPH = 0.202 mg/mL and peak scavenging activity of 95) | [91] |
| T. violacea | Hexane and ethanol | Flowers and callus cultures | Dose-dependent antioxidant activity with IC50 ranging from 1.933 to 7.350 mg/mL in the DPPH assay | [68] |
| T. violacea | Acetone | Leaves | IC50 DPPH = 0.08 mg/mL; IC50 ABTS = 0.03 mg/mL | [84] |
| T. acutiloba | Acetone | Leaves | IC50 DPPH = 0.16 mg/mL; IC50 ABTS = 0.07 mg/mL | [84] |
| T. alliacea | Acetone | Leaves | IC50 DPPH = 0.06 mg/mL; IC50 ABTS = 0.06 mg/mL | [84] |
| T. cernua | Acetone | Leaves | IC50 DPPH = 0.21 mg/mL; IC50 ABTS = 2.34 mg/mL | [84] |
| T. leucantha | Acetone | Leaves | IC50 DPPH = 0.39 mg/mL; IC50 ABTS = 0.03 mg/mL | [84] |
| T. ludwigiana | Acetone | Leaves | IC50 DPPH = 0.26 mg/mL; IC50 ABTS = 0.09 mg/mL | [84] |
| T. natalensis | Acetone | Leaves | IC50 DPPH = 2.70 mg/mL; IC50 ABTS = 0.04 mg/mL | [84] |
| Plant Species | Extraction Solvent | Plant Part Used | Biological Activity | References |
|---|---|---|---|---|
| Diabetes | ||||
| T. violacea | Methanol | Rhizome | Attenuated diabetes associated physiological complications in streptozotocin-induced diabetic rats. | [96] |
| T. violacea | Methanol | Rhizome | Noticeably reduced blood glucose and serum lipid (TG, TC, and VLDL) levels while raising plasma insulin in a streptozotocin-induced diabetic rat model. | [97] |
| Cardiovascular | ||||
| T. violacea | Methanol | Leaves | Markedly reduced systolic BP, diastolic BP, mean arterial pressure and the heart rate in both age-induced and spontaneous hypertensive rats. | [98] |
| T. violacea | Methanol | Rhizome | 50 mg/kg significantly improved kidney function in vivo. | [99] |
| T. acutiloba | Hydro-methanolic extracts | Roots, rhizomes, leaves and flowers | All extracts inhibited the Angiotensin-1-Converting Enzyme in vitro (> 50 % inhibition at a concentration range of 125–1000 μg/mL). Extracts of leaves demonstrated activity comparable to that of the control drug ramipril. | [91] |
| Antithrombogenic | ||||
| T. violacea | Water | Leaves | Noticeable inhibition of platelet adhesion by a novel scaffold consisting of polycaprolactone incorporated with 10 % (w/w) plant extracts. | [90] |
| T. violacea | Water | Leaves | Marked inhibition of platelet adhesion (70% inhibition at 0.1 mg/mL within 15 min post-exposure). | [92] |
| Plant Species | Extraction Solvent | Plant Part Used | Biological Activity | References |
|---|---|---|---|---|
| T. violacea | Methanol/water/formic acid (80:20:0.1, v/v/v) | Flowers | Reduced 1-42 β-amyloid peptide formation and arrested oxidative stress in vivo. | [80] |
| T. violacea | Methanol | Leaves | Demonstrated in vivo anticonvulsant activity by attenuating tonic convulsions induced by either pentylenetetrazole, bicuculline, picrotoxin, strychnine or NMDLA. | [102] |
| T. violacea | Acetone | Mixture of leaves and bulbs | Marked tick repellence properties of fungus-exposed plants at low treatment concentrations (5 % w/v and 10 % w/v). | [59] |
| T. violacea | Water | Leaves, stems, and roots | Induced conspicuous genotoxicity effects at high test concentrations (250, 500 and 1000 µg/mL) in the A. cepa assay. | [103] |
| T. violacea | Water and ethanol | Leaves, stems, and roots | Broad murine macrophage antiproliferative and cytotoxicity activity influenced by both extract test concentrations, type of solvent and plant part used. | [104] |
| Plant Species | Mode of Preparation | Traditional Medicinal Uses | Reference |
|---|---|---|---|
| A. cepa | Raw, juice of bulb or rhizome, paste, decoctions, cataplasm, maceration, infusion | Alopecia, antilithic (stone disease), anti-obesity, blood purifying, bronchitis, constipation, cardiovascular disease, cough, diabetes, eye diseases, erectile dysfunction, fever, hearing loss, headaches, hemorrhoids, epilepsy, oligomenorrhea, jaundice, lower gastrointestinal bleeding, prostate cancer, rheumatism, rubefacient, sinusitis, stomach pains, snake bites, skin diseases, teeth disorders, reduce flatulence, wound healing | [133,134,135] |
| A. sativum | Extracts of leaves or bulb | Antiseptic, anthelmintic, antithrombotic, antilipidemic, aphrodisiac, anti-greying of hair, bronchitis, carminative, cough, colic, cancers (gastric, prostate, colorectal adenomatous polyps, squamous cell carcinoma), diabetes, diaphoretic, dysentery, eczema, facial paralysis, fever, flatulence, galactagogue, high blood pressure, intestinal worms, liver disorders, rheumatism, scabies, tetanus, stomach pains, tuberculosis | [135,136,137,138,139] |
| A. umbilicatum | Raw or cooked bulb, leaves, flowers | Non-specific reduction in blood cholesterol levels, tonify digestive and circulatory systems | [116] |
| A. victoralis | Fresh, pickled, boiled and salted flowers, leaves and roots | Appetizer, amenorrhea, pediatric otitis, bronchitis, diarrhea, dropsy, expectorant, hypofunction of stomach, inflammatory eye diseases, meteorism, gastroenteritis, heart diseases (atherosclerosis), rheumatism | [140] |
| A. ascalonicum/A. cepa var aggregatum | Bulb and leaves | Allergies, appetizer, cold, cancers, fever, obesity, rheumatoid arthritis, soothes nerves, diabetes, post-menopausal syndrome | [141,142,143,144,145] |
| A. chinense | Flower, leaves, roots, seedpods | Angina pectoris, astringent, bronchitis, carminative, chest pains, diarrhea, expectorant, pleurisy, tenesmus in cases of dysentery, reducing cholesterol, tonic to the digestive and circulatory systems | [146] |
| A. tuberosum | Raw or cooked leaves, roots, oils from seed | Asthma, abdominal pain, carminative, cuts and wounds, diabetes, diarrhea, kidney and bladder weakness, nocturnal emission, urinary incontinence, spermatorrhea, stomachic | [147] |
| A. griffithianum | Leaves and bulb | Carminative, colic indigestion, dyspepsia, diabetes control | |
| A. oreoprasum | Leaves and bulb | Cough and cold, diabetes control, diarrhea, dysentery, fever, gastritis, oedema, headache, jaundice, stomachache, rheumatism, numbness of limbs | [124] |
| Plant Species | Plant Part | Country | Isolated Compounds | Bioactivity | References |
|---|---|---|---|---|---|
| A. ursinum L. | Leaves, underground parts, fresh flowers | Poland Bulgaria | 1,2-di-O-α-linolenoyl-3-O-β-d-galactopyranosyl-sn-glycerol; β-sitosterol3-O-β-d-glucopyranoside; kaempferol 3-O-β-glucopyranoside and kaempferol 3-O-β-neohesperidoside. (-S-)-spirost-5-en-3β-ol tetrasaccharide, (25R)-spirost-5,25(27)-dien-3 β-ol tetrasaccharide, 3-hydroxypregna-5,16-dien-20-one glycoside. Thymidine, adenosine, astragalin (kaempferol-3-O-β-d-glucopyranoside, kaempferol-3-O-β-d-glucopyranosyl-7-O-β-d-glucopyranoside, kaempferol-3-O-β-d-neohesperoside, and kaempferol-3-O-β-d-neohesperoside-7-O-β-d glucopyranoside. | Anti-ADP-aggregation activity in human blood platelets. Inhibition of human platelet aggregation. Cytotoxic activity against murine melanoma B16 and sarcoma XC. | [148,149,150,151] |
| A. mongolicum | Aerial parts | China | Mongoflavonoids A1, A2, A3, A4, B1, B2 and monogophenosides A1, A2, A3, B. | Increase in the height of mouse small intestine. | [152] |
| A. cepa. A. cepa L. | Pigmented scales of red onion, bulbs, red onion skin waste | Naples | Quercetin. 3-O-(3″-O-β-glucopyranosyl-6″-O-malonyl-β-glucopyranoside)-4-O-β-glucopyranoside, cyanidin 3,4′-di-O-β-glucopyranoside, cyanidin-4′-O-β-glucoside, peonidin 3-O-(6 ″-O-malonyl-β-glucopyranoside). 5-hydroxy-3-methyl-4-propylsulfanyl-5H-furan-2-one, (hydroxymethyl) furfural, acetovanillone, methyl 4-hydroxyl cinnamate and ferulic acid methyl ester. 3-O-β-glucopyranoside and 3-O-(6″-O-malonyl-β-glucopyranoside) of 5-carboxypyranocyanidin. Ceposide A, ceposide B and ceposide C. Spiraeoside (4′-O-glucoside of quercetin). Onionin A1, onionin A2, onionin A3, onionin B1 and B2. Onionin A1 (3,4-dimethyl-5-(1E-propenyl)-tetrahydrothiophen-2-sulfoxide-S-oxide). Cyanidin 3-glucoside (Cy 3-Glc), 3-malonylglucoside (Cy3-MaGlc), cyanidin 3-laminaribioside (Cy 3-Lam) and 3-malonyllaminaribioside (Cy 3-MaLam). | Anti-inflammatory and immunomodulatory effect. Induction of quinone reductase. Antifungal activity. Radical scavenging, anti-inflammatory, inhibition of the expression of B-cell lymphoma 2. Suppression of tumor progression in mouse ovarian cancer (Onionin A1). Suppression of tumor-cell proliferation through the inhibition of polarization of M2 activated macrophages. | [153,154,155,156,157,158,159,160,161] |
| A. sativum. A. sativum L. var. voghiera. A. sativum L. | Root, protobulb, leaf sheath and blade, bulbs, tuber | Italy | Nerolidol, α-pinene, terpinolene. Voghieroside A1/A2, voghieroside B1/B2, voghieroside C1/C2, voghieroside D1/D2 and voghieroside E1/E2. Adenosine and guanosine. | Antifungal activity against Sclerotium cepivorum. Antimicrobial activity. Strong inhibitory effect on human platelet aggregation generated by 2 μM ADP in both primary and secondary waves (adenosine). | [162,163,164] |
| A. schoenoprasum | Whole plant, pale-purple flowers | (20S, 25S)-spirost-5-en-3β, 12β,21-triol 3-O-α-L-rhamnopyranosyl-(1→2)-β-d-glucopyranoside, (20S, 25S)-spirost-5-en-3β, 11α,21-triol 3-O-α-L-rhamnopyranosyl-(1→2)-β-d-glucopyranoside, laxogenin 3-O-α-L-rhamnopyranosyl--(1→2)-[β-d-glucopyranosyl-(1→4)]-[β-d-glucopyranoside, (25R)-5α-spirostan-3β, 11α-diol 3-O-β-d-glucopyranosyl-(1→4)]-β-d-galactopyranoside. (cyanidin 3-O-β-glucosideAII) (kaempferol 3-O-(2-O-β-glucosylFIII-β-glucosideFII)-7-O-β-glucosiduronic acid FIV) malonate AIII (AII-6→AIII-1, FIV-2→AIII-3), 1, (cyanidin 3-O-(3-O-acetyl-β-glucosideAII) (kaempferol 3-O-(2-O-β-glucosylFIII-β-glucosideFII)-7-O-β-glucosiduronic acid FIV) malonate AIII (AII-6→AIII-1, FIV-2→AIII-3), 2, and 7-O-(methyl-O-β-glucosiduronateFIV). | Cytotoxicity against HCT 116 and HT-29 human colon cancer lines. | [165,166] | |
| A. minutiflorum Regel | Bulbs | Minutoside A, minutoside B, Minutoside C, alliogenin, neoagigenin | Antifungal activity. | [167] | |
| A. neapolitanum | Extracts | 3-O-{[2-O-α-1-rhamnopyrnosyl-4-O-β-d-glucopyranosyl]-β-d-glucopyranoside}, isorhamnetin; 3-O-{[2-O-α-1-rhamnopyrnosyl-6-O-β-d-glucopyranosyl]-β-d-glucopyranoside}, isorhamnetin; 3-O-{[2-O-α-1-rhamnopyranosyl-4-O-β-d-glucopyranosyl]-β-d-glucopyranoside}-7-O-β-d-glucopyranoside and isorhamnetin; 3-O-{[2-O-α-1-rhamnopyranosyl-6-O-β-d-gentiobiosyl]-β-d-glucopyranoside}. | Antiplatelet aggregation activity. | [168] | |
| A. tripedale | Bulbs, leaves | Iran | 6,7-dimethoxy-N-trans-caffeoyltyramine; N-trans-feruloyltyramine. (+)-S-(1-butenyl)-L-cysteine sulfoxide (homoisoalliin), S-(1-butenyl)-L-cysteine (desoxyhomoisoalliin). | NR. | [167,168] |
| A. porrum L. | Bulbs | Kaempferol 3-O-[2-O-(trans-3-methoxy-4-hydroxycinnamoyl)-β-d-galactopyranosyl]-(1→4)-O-β-d-glucopyranoside; Kaempferol 3-O-[2-O-(trans-3-methoxy-4-hydroxycinnamoyl)-β-d-glucopyranosyl]-(1→6)-O-β-d-glucopyranoside. (25R)-5 α-spirostan-3 β, 6 β-diol 3-O-[O-β-d-glucopyranosyl-(1→2)-O-[β-d-xylopyranosyl-(1→3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside}; (25R)-5 α-spirostan-3 β, 6 β-diol 3-O-{O-β-d-glucopyranosyl-(1→3)-O-β-d-glucopyranosyl-(1→2)-O-[β-d-xylopyranosyl-(1→3)]-O-β-d-galactopyranosyl-(1→4)-β-d-galactopyranoside} | Antiplatelet aggregation activity. Antifungal activity. | [169,170,171] | |
| A. chinense. A. chinense G. Don | Bulbs | Chinenoside II and chinenoside III. (25 R,S)-5 α-Spirostan-3β-ol tetrasaccharide, (25R)-3 β-hydroxy-5 α-spirostan-6-one di- and tri-saccharides. Xiebai-saponin I (laxogenin 3-O-β-xylopyranosyl (1→4)-[α-arabinopyranosyl (1→6)-β-glucopyranoside), laxogenin 3-O-α-arabinopyranosyl (1→6)-β-glucopyranoside, laxogenin, isoliquiritigenin, isoliquiritigenin-4-O-glucoside, and β-sitosterol glucoside. | Inhibition of cAMP phosphodiesterase. Antitumor-promoting activity (laxogenin). | [172,173,174,175] | |
| A. macrostemon. A. macrostemon Bunge | Bulbs, leaves | Japan | Macrostemonoside G (26-O-β-d-glucopyranosyl-22-hydroxy-5-β-furost-25(27)-ene-3 β,12 β,26 triol 3-O-β-d-glucopyranosyl(1-->2)-β-d-galactopyranoside) and I (26-O-β-d-glucopyranosyl-22-hydroxy-5 β-furost-25(27)-ene-12-one-3 β,26-diol 3-O-β-d-glucopyranosyl(1→2)-β-d-galactopyranoside). tigogenin-3-O-β-d-glucopyranosyl(1-->2) [β-d-glucopyranosyl(1→3)1]-β-d-glucopyranosyl(1→4)-β-d-galactopyranoside (1) and tigogenin-3-O-β-d-glucopyranosyl(1→2)[β-d-glucopyranosyl (1→3)(6-O-acetyl-β-d-glucopyranosyl)] (1→4)-β-d-galactopyranoside (2). Macrostemonoside E-(25R)-26-O-β-d-glucopyranosyl-5 α-furost-20(22)-ene-3 β,26-diol-3-O-β-d-glucopyranosyl (1→2) [β-d-glucopyranosyl (1→3)]-β-d-glucopyranosyl (1→4)-β-d-galactopyranoside; Macrostemonoside F(II)-(25R)-26-O-β-d-glucopyranosyl-5 β-furost-20(22)-ene-3 β,26-diol-3-O-β-d-glucopyranosyl (1→2)-β-d-galactoside. Allimacronoid A (1-O-(E)-feruloyl-β-d-glucopyranosyl (1-2)-[β-d-glucopyranosyl (1-6)]-β-d-glucopyranose), Allimacronoid B (1-O-(E)-feruloyl-{β-d-glucopyranosyl (1-4)-[β-d-glucopyranosyl (1-2)]}-[β-d-glucopyranosyl (1-6)]-β-d-glucopyranose) and Allimacronoid Cn1-O-(E)-feruloyl-{β-d-glucopyranosyl (1-6)-[β-d-glucopyranosyl (1-2)]}-[β-d-glucopyranosyl (1-6)]-β-d-glucopyranose. | In vitro inhibition of ADP-induced human platelet aggregation (macrostemonoside G). Inhibitory activity against rabbit platelet aggregation induced by ADP (1). | [176,177,178,179] |
| A. schubertii | Bulbs | (25R and S)-5 α-spirostan-2 α,3 β,6 β-triol 3-O-β-d-glucopyranosyl-(1→2)-O-[4-O-benzoyl-β-d-xylopyranosyl-(1→3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside, (25R and S)-5 α-spirostan-2α,3β,6 β-triol 3-O-β-d-glucopyranosyl-(1→2)-O-[3-O-benzoyl-β-d-xylopyranosyl-(1→3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside, (25R and S)-5 α-spirostan-2α,3β,6 β-triol 3-O-β-d-glucopyranosyl-(1→2)-O-[4-O-(3S)-3-hydroxy-3-methylglutaroyl-β-d-xylopyranosyl-(1→3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside and 26-O-β-d-glucopyranosyl-(25R and S)-5 α-furostan-2α,3β,6β,22 zeta,26-pentol 3-O-β-d-glucopyranosyl-(1→2)-O-[β-d-xylopyranosyl-(1→3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside. | NR. | [180] | |
| A. tuberosum | Seeds | Shanghai | (2α, 3β, 5α, 25S)-2,3,27-trihydroxyspirostane 3-O-α-L-rhamnopyranoyl-(1→2)-O-[α-L-rhamnopyranosyl-(1→4)]-β-d-glucopyranoside. Tuberoside J-(25R)-5 α-spirostan-2α,3 β,27-triol 3-O-α-L-rhamnopyranosyl-(1-->2)-β-d-glucopyranoside; Tuberoside K-(25R)-5α-spirostan-2α,3β 27-triol 3-O-α-L-rhamnopyranosyl-(1→2)-[α-L-rhamnopyranosyl-(1→4)]-β-d-glucopyranoside; and Tuberoside L-27-O-β-d-glucopyranosyl-(25R)-5α-spirostan-2α,3 β,27-triol 3-O-α-d-rhamnopyranosyl-(1-->2)-[α-L-rhamnopyranosyl-(1→4)]-β-d-glucopyranoside. Tuberoside M-(25S)-5β-spirostane-β,3 β-diol 3-O-α-L-rhamnopyranosyl-(1→4)-β-d-glucopyranoside. Tuber-ceramide (N-(2′,3′-dihydroxy-tetracosenoyl)-2-amino-1,3,4-trihydroxy octadecane), and Cerebroside (N-(2′,3′-dihydrox-tetra-cosenoyl)-2-amino-1,3,4-trihydroxy octadecane). | Tuberoside M inhibits the proliferation of the human promyelocytic leukemia cell line (HL-60) | [181,182,183] |
| A.albopilosum and A. ostrowskianum | Bulbs | (25 R and S)-5 α-spirostane-2α, 3 β,6 β-triol 3-O-(O-β-d-glucopyranosyl-(1→2)-O-[3-O-acetyl-β-d-xylopyranosyl-(1→3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside), (25R)-2-O-[(S)-3-hydroxy-3-methylglutaroyl]-5 α-spirostane-2α, 3β, 6β-triol 3-O-(O-β-d-glucopyranosyl-(1→2)-O-[β-d-xylopyranosyl-(1— >3)]-O-β-d-glucopyranosyl-(1→4)-β-d-galactopyranoside), (22S)-cholest-5-ene-1β, β,16 β,22-tetraol 1-O-α-L-rhamnopyranoside 16-O-(O-α-L-rhamnopyranosyl-(1→3)-β-d-glucopyranoside), 1β, 3β, 16β-trihydroxycholest-5-en-22-one 1-O-aα-L-rhamnopyranoside 16-O-(O-α-L-rhamnopyranosyl-(1→3)-β-d-glucopyranoside), 1β,3β,16 bβ-trihydroxy-5 α-cholestan-22-one 1-O-α-L-rhamnopyranoside 16-O-(O-α-L-rhamnopyranosyl-(1→3)-β-d-glucopyranoside) and (22S)-cholest-5-ene-1β,3β, 16β,22-tetraol 16-O-(O-β-d-glucopyranosyl-(1→3)-β-d-glucopyranoside). | NR. | [184] | |
| A.fistulosum. A.fistulosum L. | Whole plant, leaves, seeds | Iran | Fistulomidate A ((1Z,2E)-Methyl3-(3,4-dimethoxyphenyl)-N-(4-hydroxyphenethyl) acrilimidate) and Fistulomidate B ((1Z,2E)-Methyl3-(3,4-dihydroxyphenyl)-N-(4-hydroxyphenethyl)acrilimidat). Onionin A1, onionin A2, and onionin A3. Glycerol mono-(E)-8,11,12-trihydroxy-9-octadecenoate, tianshic acid, 4-(2-formyl-5-hydroxymethylpyrrol-1-yl) butyric acid, p-hydroxybenzoic acid, vanillic acid, and daucosterol. | Antibacterial and cytotoxic activity. Suppression of tumor progression in mouse ovarian cancer (onionin A1). Inhibition of the growth of Phytophtohora capsici on V8 media (glycerol mono-(E)-8,11,12-trihydroxy-9-octadecenoate and V). | [159,185,186] |
| A.carolinianum DC | Bulb | Mongolia | Cinnamoylphenethylamine derivative | Weak cytotoxic activity | [187] |
| A.ampeloprasum var. porrum (Leek) | Plant parts | A-β- d -glucopyranoside | Anticancer activity against MCF-7 human breast cancer cell. | [187] | |
| A.ascalonicum L. | China | Ascalonicoside C-(25R)-26-O-β-d-glucopyranosyl-22-hydroxy-5α-furost-2-one-3β,5,6β, 26-tetraol-3-O-α-L-rhamnopyranosyl-(1→2)-β-d-glucopyranoside. Ascalonicoside d-(25R)-26-O-β-d-glucopyranosyl-22-methoxy-5α-furost-2-one-3β,5,6β, 26-tetraol-3-O-α-L-rhamnopyranosyl-(1→2)-β-d-glucopyranoside. (25R)-26-O-β-d-glucopyranosyl-22-hydroxy-5-ene-furostan-3β,26-diol-3-O-α-L-rhamnopyranosyl-(1→4)-α-L-rhamnopyranosyl-(1→4)-[ α-L-rhamnopyranosyl-(1→2)]-β-d-glucopyranoside. 25R)-26-O-β-d-glucopyranosyl-22-hydroxy-5-ene-furostan-3β, 26-diol-3-O-α-L-rhamnopyranosyl-(1→2)-[α-L-arabinofuranosyl-(1→4)]-β-d-glucopyranoside. | NR. | [188,189] | |
| A.siculum | Bulbs | Zwanenburg, The Netherlands | (Z)-Butanethial S-oxide, (R(S),R(C),E)-S-(1-butenyl)cysteine S-oxide (homoisoalliin). | NR. | [190] |
| A.chrysanthum | Barks | Guangzhou, China | Chrysanthumones A (6″,6″-dimethyl-4″,5″-dihydropyrano [2″,3″: 8,7]-6″′,6″′-dimethyl-prenyl-4″′,5″′-dihydropyrano [2″′,3″′:2′,3′]apigenin) and B ((E)-5,7-dihydroxy-2-(4-hydroxyphenyl)-8-(3-methylbut-1-enyl)-4H-chromen-4-one). | NR. | [191] |
| A. L. melanocrommyum section Megaloprason. | Bulbs | Central Asia | L-(+)-S-(2-pyridyl)-cysteine sulfoxide. | NR. | [192] |
| A.ampeloprasum L. | Bulbs | United States of America | Ampeloside Bs1 (apigenin 3-O-β-glucopyranosyl (1 → 3)-β-glucopyranosyl (1 → 4)-β-galactopyranoside), ampelosides Bf1 ((25R)-26-O-β-glucopyranosyl-22-hydroxy-5α-furostane-2α,3β,6β,26-tetraol-3-O-β-glucopyranosyl(1 → 3)-β-glucopyranosyl-(1 → 4)-β-galactopyranoside) and Bf2 ((25R)-26-O-β-glucopyranosyl-22-hydroxy-5α-furostane-2α,3β,6β,26-tetraol-3-O-β-glucopyranosyl(1 → 4)-β-galactopyranoside). | Weak antifungal activity by ampeloside Bs1. | [193] |
| A.bakeri Reg. | Tuber | Adenosine, guanosine, and tryptophan, β-sitosterol β-d-glucoside. | Strong inhibitory effect on human platelet aggregation generated by 2 μM ADP in both primary and secondary waves (adenosine). | [162] | |
| A.victorialis var. platyphyllum | Aerial parts, bulbs | Korea | Gitogenin 3-O-lycotetroside, astragalin and kaempferol 3, 4′-di-O-β-d-glucoside. | Cytotoxic activity. | [194] |
| A.nutans L. | Underground plant parts | Deltoside, nolinofuroside D, 25R Δ(5)-spirostan 3β-ol-3-O-α-L-rhamnopyranosyl(1-->2)-[β-d-glucopyranosyl(1→4)]-O-β-d-galactopyranoside and 25R Δ(5)-spirostan 1 β, 3β-diol 1-O-β-d-galactopyranoside. | NR. | [195] | |
| A.giganteum | Bulbs | Japan | 3-O-acetyl-(24S,25S)-5α-spirostane-2α,3β,5α,6β,24-pentol 2-O-β-d-glucopyranoside. | Inhibition of cAMP phosphodiesterase activity. | [196] |
| A.hookeri Thwaites | Rhizomes | China | Di-2-propenyl trisulfide, diallyl disulfide, and dipropyl trisulfide. | Antimicrobial activity against Aspergillus fumigatus and C. albicans. | [197] |
| Plant Species | Plant Part | Country | Isolated Compounds | Bioactivity | References |
|---|---|---|---|---|---|
| C. x amabile C. x amabile Donn ex Ker Gawl | Bulbs Stems, roots | Ecuador Brazil Thailand | Haemanthamine/crimine-type alkaloid. Lycorine-type alkaloid Galanthamine-type alkaloid. Augustine N-oxide, buphanisine N-oxide. Amabiloid A. | Anticholinesterase (anti-AChE) and antibutyrylcholinesterase (anti-BuChE) activity. | [249,250,251] |
| C. defixum Ker-Gawl | Bulbs | India | Hydrazide derivative. (E)-N-[(E)-2-butenoyl]-2-butenoylhydrazide. | Anti-genotoxic activity. | [252] |
| C. moorei | Bulblets | Cherylline, crinamidine, crinine, epibuphanisine, lycorine, powelline, undulatine, 1-epideacetylbowdensine, 3-O-acetylhamayne. 3-[4′-(8′-aminoethyl) phenoxy] bulbispermine, mooreine. | NR. | [253] | |
| C. biflorum | Bulbs | Senegal | 5,6,7-trimethoxy-3-(4 hydroxybenzyl) chroman-4-one, 3-hydroxy-5,6,7-trimethoxy-3-(4-hydroxybenzyl) chroman-4-one, 3-hydroxy-5,6,7-trimethoxy-3-(4-methoxybenzyl) chroman-4-one, 5,6,7-trimethoxy-3-(4-methoxybenzyl) chroman-4-one, (E)-N-(4-hydroxyphenethyl)-3-(4-hydroxyphenyl) acrylamide. | Anticancer, anti-AChE, anti-glucosidase activity. | [254,255] |
| C. asiaticum C. asiaticum var. sinicum C. asiaticum L. C. asiaticum var. japonicum. C. asiaticum L. var. sinicum. | Seeds, rhizome, fruits Bulbs, stems, leaves | Beijing, China, Hainan Province, Japan, Island of Jeju in Korea | Flavonoids Isopowellaminone. (2R,3S)-7-methoxyflavan-3-ol (1:), (2R,3S)-7-hydroxy-flavan-3-ol (2:), (2R,3S)-2 ′-hydroxy-7-methoxy-flavan-3-ol (3:). Norgalanthamine. Crinamine CAL-n. Crijaponine A, crijaponine B, ungeremine, lycorine, 2-O-acetyllycorine, 1,2-O-diacetyllycorine, (-)-crinine, 11-hydroxyvittatine, hamayne,(+)-epibuphanisine, crinamine, yemenine A, epinorgalanthamine. Criasiaticidine A, pratorimine, Lycorine, 4′-hyd’oxy-7-methoxyflavan. Crinamine, lycorine, norgalanthamine, epinorgalanthamine. Asiaticumines A, asiaticumines B. | Inhibitory activity against LPS-induced nitric oxide production. Anticancer activity (against cervical cancer SiHa cells). Inhibition of platelet aggregation. Promotion of hair growth through dermal papilla proliferation. Inhibition of the growth of HepG2 tumor cells. Anti-AChE activity, cytotoxic activity. Cytotoxic against Meth-A (mouse sarcoma) and Lewis lung carcinoma (mouse lung carcinoma). Inhibition of the activity of hypoxia inducible factor-1 (crinamine). Cytotoxicity. | [256,257,258,259,260,261,262,263,264,265,266] |
| C. kirkii Baker | Bulbs | Noraugustamine, 4aN-dedihydronoraugustamine, 3-O-acetylsanguinine, 1,2-diacetyllycorine. | Antiparasitic activity against Trypanosoma brucei (T. brucei) rhodesiense, Trypanosoma cruzi (T. cruzi). | [267,268] | |
| C. macowanii | Bulbs | Macowine, lycorine, cherylline, crinine, krepowine, powelline, buphanidrine, crinamidine, undulatine, 1-epideacetylbowdensine, 4a-dehydroxycrinamabine. | NR. | [249] | |
| C. firmifolium | Leaves | Madagascar | 2-alkylquinolin-4(1H), 2-alkylquinolin-4(1H). | Antiplasmodial activity. | [269] |
| C. latifolium | Bulbs Leaves | China. Hanoi, Vietnam | 4,8-dimethoxy-cripowellin C. 4,8-dimethoxy-cripowellin D, 9-methoxy-cripowellin B, 4-methoxy-8-hydroxy-cripowellin B, cripowellin C. C. latines A, C. latines B and C. latines C. 4-senecioyloxymethyl-3,4-dimethoxycoumarin, 5,6,3 ′-trihydroxy-7,8,4 ′-trimethoxyflavone. 4-methyloxysenecioyl-6,7-dimethoxycoumarin, 5,6,3′-trihydroxy-7,8,4′trimethoxyflavone. | Cytotoxic against tumor cell lines, antimicrobial activity, antioxidant activity. Inhibitory activity against human umbilical venous endothelial cells. | [270,271,272] |
| C. scillifolium | Bulbs | Scillitazettine, scilli-N-desmethylpretazettine. | Mild antiplasmodial activity | [273] | |
| C. zeylanicum (L) | Bulbs, leaves, flowers, fruits | Cuba Sri Lanka | Crinine, Lycorine, 11-O-acetoxyambelline, ambelline, 6-hydroxybuphanidrine, 6-ethoxybuphanidrine, 3-acetylhamayne, 6-hydroxycrinamine, hamayne, 6-methoxycrinamine. | Antiproliferative effect. | [246,274] |
| C. jagus (J. Thomps) Dandy | Bulbs, leaves | Senegal Ghana | Gigantelline, gigantellinine, gigancrinine, sanguinine, cherylline, lycorine, crinine, flexinine, hippadine. Galanthamine, galanthamine N-oxide, powelline. | Anti-AChE activity, inhibitors of TcAchE, hAChE and hBChE | [275,276] |
| C. abyscinicum Hochst. ExA. Rich | Bulbs | Ethiopia | 6-hydroxycrinamine, lycorine. | Antiproliferative activity against A2780 epithelial ovarian cancer and MV4-11 acute myeloid leukemia cell lines. | [277] |
| C. erubescens | Above ground plant parts | Puntarenas, Costa Rica | Cripowellin A, cripowellin B, cripowellin C, cripowellin D, hippadine. | Antiplasmodial activity. | [278] |
| C. yemense | Bulbs | Yemen | 6-hydroxy-2H-pyran-3-carbaldehyde. Yemenines A, B and C, 1, (+)-bulbispermine, (+)-crinamine, (+)-6-hydroxycrinamine, (-)-lycorine. | Tyrosinase inhibitor. Inhibit nitric oxide production, induce nitric oxide synthase. | [277,278,279,280] |
| C. bulbispermum C. bulbispermum III | Bulbs | Egypt | 8-hydroxylycorin-7-one, 2-deoxylycorine, vittatine, 11-hydroxyvittatine, hippamine. 4-hydroxy-2′,4′-dimethoxydihydrochalcone, 4,5-methylenedioxy-4′-hydroxy-2-aldehyde [1,1′-biphenyl], hippacine, 4′-hydroxy-7-methoxyflavan-3-ol, 2(S),3′,4′-dihydroxy-7-methoxy flavan, isolarrien, isoliquiritigenin, liquiritigenin. Bulbispermine. | NR. | [281,282,283] |
| C. powellii | Bulbs | Switzerland Colombia | Linoleic acid ethyl ester, alkaloid hippadine, calleryanin, 4′-hydroxy-7-methoxyflavan. Lycorine, 1-O-acetyllycorine, ismine. | AChE inhibitor (linoleic acid ethyl ester). Inhibition of topoisomerase 1 activity. | [284,285] |
| C. glaucum | Bulbs | Nigeria | Hamayne, lycorine, haemanthamane, crinamine. | Choline esterase inhibitory activity. | [286] |
| C. purpurascens | Leaves | Cameroon | 4,5-ethano-9,10-methlenedioxy-7-phenanthridone, 4,5-ethano-9-hydroxy-10-methoxy-7-phenanthridone, α-d-glucopyranoside. | Antibacterial activity | [287] |
| Lineage | Location | Species | References |
|---|---|---|---|
| Clade A | Southern Africa Grassland Southeastern African temperate grasslands Grassland of the Highveld in the northern parts Subtropical Indian Ocean Coastal Belt East Africa and Angola | C. attenuatus, C. macowanii, C. epiphyticus, C. mackenii subsp. cooperi, C. huttonii, C. macmasteri, C. suaveolens, C. stenanthus var. stenanthus, C. flanaganii C. tuckii var. transvaalensis C. angustifolius, C. fergusoniae C. aureolinus, C. mackenii subsp. Mackenii, C. brachyscyphus C. breviflorus | [328] |
| Clade B | Baviaanskloof Mountains and Eastern Cape (Fynbos and Albany Thicket Biomes) Semi-arid Succulent Karoo Greater Cape Region (“the Cape”) Coastal and inland mountains of the southern Cape Cape Peninsula into the Eastern Cape | C. labiatus, C. montanus C. herrei C. carneus, C. elatus, C. guthrieae, C. labiatus, C. leptosiphon, C. leucanthus, C. montanus, C. odorus C. collinus C. ventricosus | [328] |
| Clade C | Albany Thicket Biome Savanna Biome Northwards from the Albany region through South Africa, Zimbabwe, Western Mozambique and East Africa into Sudan Albany Thicket and Savanna Biomes Extends beyond the Savanna Biome into the Sub-Escarpment and Highveld grasslands Fynbos Biome Southern parts of the Nama Karoo | C. flammosus, C. spiralis C. eucallus, C. galpinii C. sanguineus C. helictus C. contractus C. wellandii C. smithiae | [328] |
| Plant Species | Traditional Uses | Compounds | Pharmacological Activities | References |
|---|---|---|---|---|
| C. obliquus | Chronic cough, headache and scrofula | 5,7-dihydroxy-6-methoxy-3-(4′-methoxybenzyl)chroman-4-one, 5,7-dihydroxy-6-methoxy-3-(4′-hydroxybenzyl)chroman-4-one | Antioxidant activity | [338] |
| C. contractus | Mental illness, protective charm against evil spirits | NarciclasineNarciprimine | Anti-inflammatory activity (via inhibition of E-selectin, blockade of the expression of endothelial adhesion molecule ICAM-1)Acetylcholinesterase inhibitor | [337] |
| C. breviflorus | Emesis, worm infestations, protective charm against evil spirits | haemanthamine, lycorine, crinamine hydrochloride and tazettine | Antihelminthic | [337,342] |
| C. elatus | Cough, headache, labor induction | Haemanthamine, zephyranthine, galanthamine and 1,2-O-diacetylzephyranthine | Antiprotozoan activity, selective cytotoxic activity | [43,44,337] |
| C. falcatus | Not known to be used by the traditional South African people | Papyramine, epipapyramine, maritidine, O-methylmaritidine and tazettine | Antibacterial activity against B. subtilis S. aureus and E. coli, mutagenicity, cytotoxic activity | [342,343,344] |
| C. suaveolens | No traditional use has been reported | Captan | Mutagenicity, anti-inflammatory activity via inhibition of COX-2, fungicide | [342,344] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Danquah, C.A.; Minkah, P.A.B.; Agana, T.A.; Moyo, P.; Ofori, M.; Doe, P.; Rali, S.; Osei Duah Junior, I.; Amankwah, K.B.; Somuah, S.O.; et al. The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: ‘Talented’ Taxa from the Amaryllidaceae. Molecules 2022, 27, 4475. https://doi.org/10.3390/molecules27144475
Danquah CA, Minkah PAB, Agana TA, Moyo P, Ofori M, Doe P, Rali S, Osei Duah Junior I, Amankwah KB, Somuah SO, et al. The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: ‘Talented’ Taxa from the Amaryllidaceae. Molecules. 2022; 27(14):4475. https://doi.org/10.3390/molecules27144475
Chicago/Turabian StyleDanquah, Cynthia Amaning, Prince Amankwah Baffour Minkah, Theresa A. Agana, Phanankosi Moyo, Michael Ofori, Peace Doe, Sibusiso Rali, Isaiah Osei Duah Junior, Kofi Bonsu Amankwah, Samuel Owusu Somuah, and et al. 2022. "The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: ‘Talented’ Taxa from the Amaryllidaceae" Molecules 27, no. 14: 4475. https://doi.org/10.3390/molecules27144475
APA StyleDanquah, C. A., Minkah, P. A. B., Agana, T. A., Moyo, P., Ofori, M., Doe, P., Rali, S., Osei Duah Junior, I., Amankwah, K. B., Somuah, S. O., Nugbemado, I. N., Maharaj, V. J., Bhakta, S., & Gibbons, S. (2022). The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: ‘Talented’ Taxa from the Amaryllidaceae. Molecules, 27(14), 4475. https://doi.org/10.3390/molecules27144475










