A Novel Polysaccharide from AuriculariaAuricula Alleviates Thrombosis Induced by Carrageenan in Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Purification of AAP-b2
2.2. Monosaccharide Composition of AAP-b2
2.3. Methylation Analysis of AAP-b2
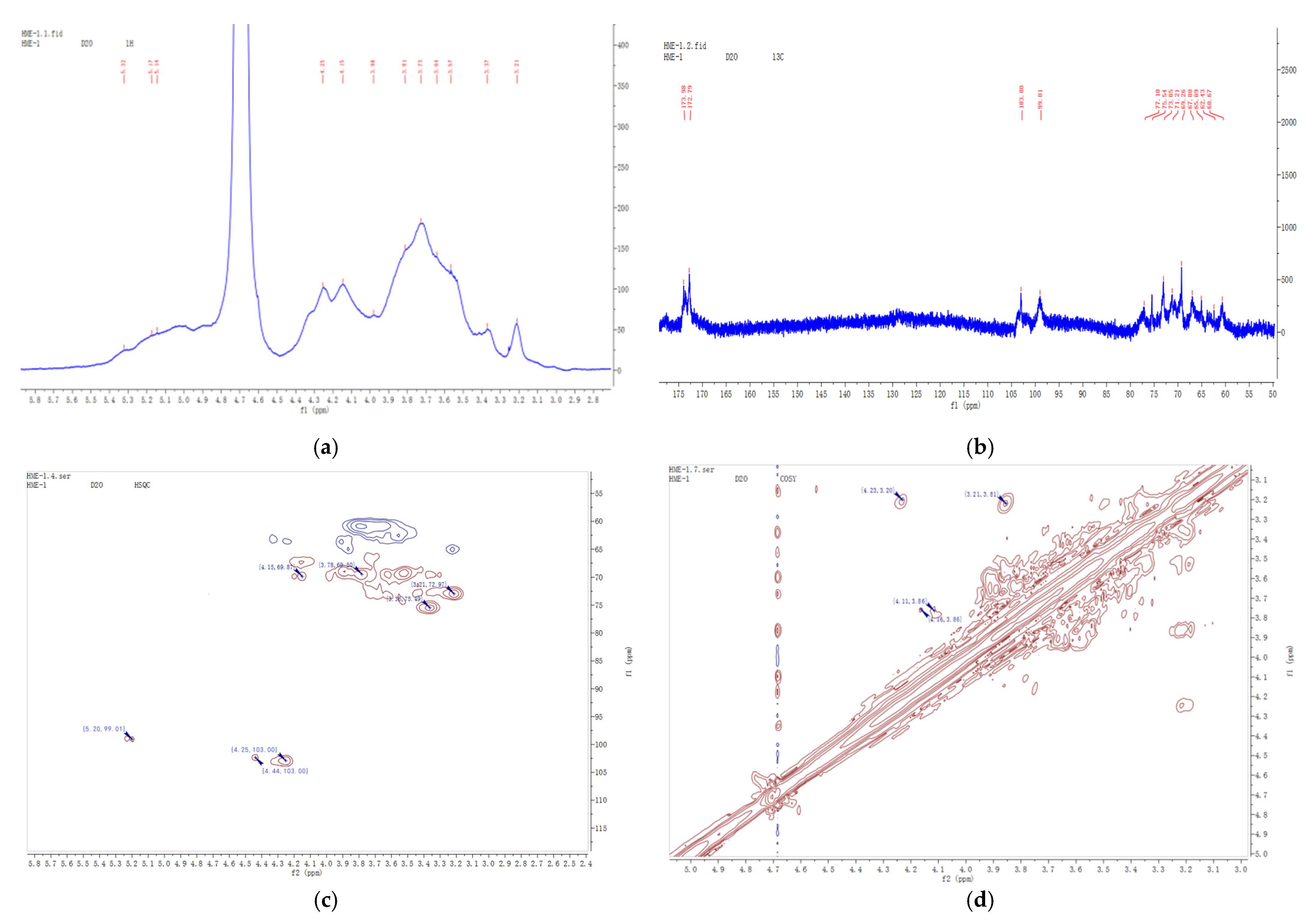
2.4. NMR Analysis of AAP-b2
2.5. Effects of AAP-b2 on Platelet Aggregation In Vitro
2.6. Effects of AAP-b2 on Thrombus Length
2.7. Effects of AAP-b2 on Coagulation Parameter
2.8. Histological Examination
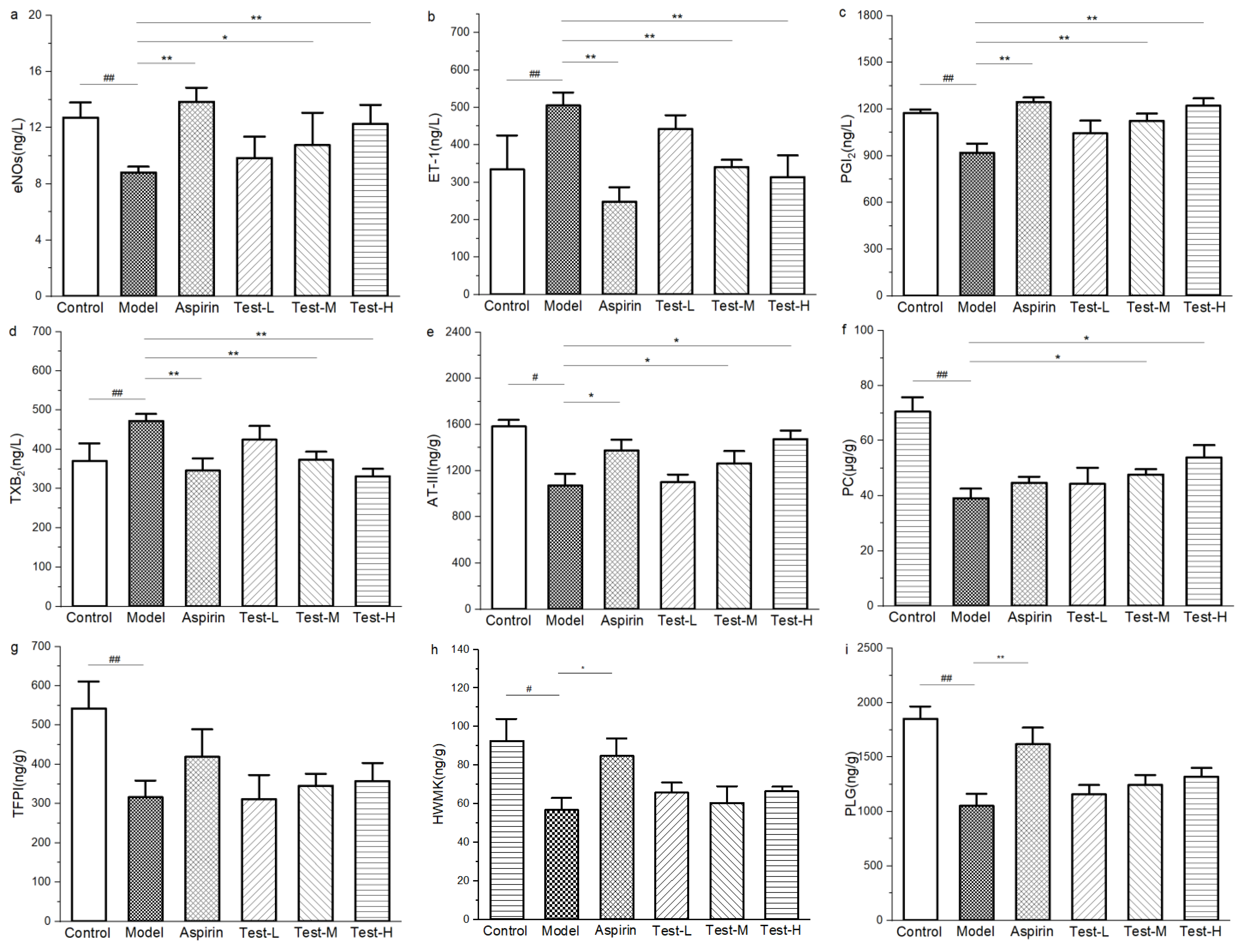
2.9. Antithrombotic Pathway Analysis of AAP-b2
2.9.1. Effect of AAP-b2 on Platelet Activation in Mice
2.9.2. Effect of AAP-b2 on Anticoagulant Activity
2.9.3. Effect of AAP-b2 on Fibrinolytic Capacity
3. Materials and Methods
3.1. Materials and Reagents
3.2. Extraction and Purification of Polysaccharides
3.3. Structure Elucidation
3.3.1. Molecular Weight Determination
3.3.2. Monosaccharide Composition Determination
3.3.3. Methylation Analysis
3.3.4. NMR Spectroscopy Analysis
3.4. Animal Experiments
3.4.1. Animals and Treatments
3.4.2. Platelet aggregation
3.4.3. Determination of Thrombus Length
3.4.4. Measurement of APTT, PT, TT and FIB
3.4.5. Histological Examination
3.4.6. Enzyme-linked Immunosorbent Assay
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability Statement
References
- Zeng, S.; Yi, R.; Tan, F.; Sun, P.; Cheng, Q.; Zhao, X. Lactobacillus plantarum HFY05 attenuates carrageenan-induced thrombosis in mice by regulating NF-kappa B pathway-associated inflammatory responses. Front. Nutr. 2022, 9, 813899. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Majida, A.; Niu, L.; Zhou, F.; Zhang, Y.; Wang, M.; Ren, J.; Fan, J.; Zhang, B.; Wang, L. Degradation enhances the anticoagulant and antiplatelet activities of polysaccharides from Lycium barbarum L. leaves. Int. J. Biol. Macromol. 2019, 133, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Zhang, Y.; Wang, X.; Wei, J.; Kang, W. Antithrombotic effect and mechanism of Rubus spp. Blackberry. Food Func. 2017, 8, 2000–2012. [Google Scholar] [CrossRef] [PubMed]
- Goto, S.; Tomita, A. Antithrombotic therapy for prevention of various thrombotic diseases. Drug Develop. Res. 2013, 74, 568–574. [Google Scholar] [CrossRef]
- Qiu, M.; Huang, S.; Luo, C.; Wu, Z.; Liang, B.; Huang, H.; Ci, Z.; Zhang, D.; Han, L.; Lin, J. Pharmacological and clinical application of heparin progress: An essential drug for modern medicine. Biomed. Pharmacother. 2021, 139, 111561. [Google Scholar] [CrossRef]
- Dedhia, N.; Marathe, S.; Singhal, R. Food polysaccharides: A review on emerging microbial sources, bioactivities, nanoformulations and safety considerations. Carbohydr. Polym. 2022, 287, 119355. [Google Scholar] [CrossRef]
- Liu, Z.; Jiao, Y.; Lu, H.; Shu, X.; Chen, Q. Chemical characterization, antioxidant properties and anticancer activity of exopolysaccharides from Floccularia luteovirens. Carbohydr. Polym. 2020, 229, 115432. [Google Scholar] [CrossRef]
- Qu, H.; Gao, X.; Zhao, H.; Wang, Z.; Yi, J. Structural characterization and in vitro hepatoprotective activity of polysaccharide from pine nut (Pinus koraiensis Sieb. et Zucc.). Carbohydr. Polym. 2019, 223, 115056. [Google Scholar] [CrossRef]
- Souza, R.; Assreuy, A.; Madeira, J.; Chagas, F.; Parreiras, L.; Santos, G.; Mourao, P.; Pereira, M. Purified polysaccharides of Geoffroea spinosa barks have anticoagulant and antithrombotic activities devoid of hemorrhagic risks. Carbohydr. Polym. 2015, 124, 208–215. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Lian, P.; Yu, Q.; Wei, J.; Kang, W. Antithrombotic mechanism of polysaccharides in blackberry (Rubus spp.) seeds. Food Nutr. Res. 2017, 61, 1379862. [Google Scholar] [CrossRef] [Green Version]
- Chagas, F.; Lima, G.; dos Santos, V.; Costa, L.; de Sousa, W.; Sombra, V.; de Araujo, D.; Barros, F.; Marinho, E.; Feitosa, J.; et al. Sulfated polysaccharide from the red algae Gelidiella acerosa: Anticoagulant, antiplatelet and antithrombotic effects. Int. J. Biol. Macromol. 2020, 159, 415–421. [Google Scholar] [CrossRef]
- Su, Y.; Li, L. Structural characterization and antioxidant activity of polysaccharide from four auriculariales. Carbohydr. Polym. 2020, 229, 115407. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Zhang, H.; Wang, Z.; Liu, D.; Liu, S.; Han, W.; Regenstein, J.; Geng, L. The antitumor effect of folic acid conjugated-Auricularia auricular polysaccharide-cisplatin complex on cervical carcinoma cells in nude mice. Int. J. Biol. Macromol. 2018, 107, 2180–2189. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, J.; Fan, Z.; Qiu, J.; Rumbani, M.; Yang, X.; Zhang, H.; Wang, Z. Effects of polysaccharide from the fruiting bodies of Auricularia auricular on glucose metabolism in Co-60-gamma-radiated mice. Int. J. Biol. Macromol. 2019, 135, 887–897. [Google Scholar] [CrossRef] [PubMed]
- Bian, C.; Wang, Z.; Shi, J. Extraction optimization, structural characterization, and anticoagulant activity of acidic polysaccharides from Auricularia auricula-judae. Molecules 2020, 25, 710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, Y.; Yu, L.; Liang, J.; Yang, B.; Kuang, H. Chromatography and mass spectrometry-based approaches for perception of polysaccharides in wild and cultured fruit bodies of Auricularia auricular-judae. Int. J. Biol. Macromol. 2019, 137, 1232–1244. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.; Bilan, M.; Dmitrenok, A.; Silchenko, A.; Grebnev, B.; Stonik, V.; Nifantiev, N.; Usov, A. Fucosylated chondroitin sulfates from the sea cucumbers Paracaudina chilensis and Holothuria hilla: Structures and anticoagulant activity. Mar. Drugs 2020, 18, 540. [Google Scholar] [CrossRef]
- Cui, K.; Tai, W.; Shan, X.; Hao, J.; Li, G.; Yu, G. Structural characterization and anti-thrombotic properties of fucoidan from Nemacystus decipiens. Int. J. Biol. Macromol. 2018, 120, 1817–1822. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, J.; Li, D.; Song, S.; Song, L.; Fu, Y.; Zhang, L. Structural investigation of a uronic acid-containing polysaccharide from abalone by graded acid hydrolysis followed by PMP-HPLC–MSn and NMR analysis. Carbohyd. Res. 2015, 402, 95–101. [Google Scholar] [CrossRef]
- Jin, W.; He, X.; Wu, W.; Bao, Y.; Wang, S.; Cai, M.; Zhang, W.; Wang, C.; Zhang, F.; Linhardt, R.; et al. Structural analysis of a glucoglucuronan derived from laminarin and the mechanisms of its anti-lung cancer activity. Int. J. Biol. Macromol. 2020, 163, 776–787. [Google Scholar] [CrossRef]
- Qu, H.; Gao, X.; Cheng, C.; Zhao, H.; Wang, Z.; Yi, J. Hepatoprotection mechanism against alcohol-induced liver injury in vivo and structural characterization of Pinus koraiensis pine nut polysaccharide. Int. J. Biol. Macromol. 2020, 154, 1007–1021. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, L.; Wang, Z. Structural characterization and antioxidant activity of polysaccharide from Hohenbuehelia serotina. Int. J. Biol. Macromol. 2017, 98, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ruan, D.; Jin, Y.; Shashkov, A.; Senchenkova, S.; Kilcoyne, M.; Savage, A.; Na, N. Chemical structure of aeromonas gum-extracellular polysaccharide from Aeromonas nichidenii 5797. Carbohyd. Res. 2004, 339, 1631–1636. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Song, H.; Wang, L.; Peng, H.; Sun, Y.; Ai, C.; Wen, C.; Zhu, B.; Song, S. Structural characterization and SARS-CoV-2 inhibitory activity of a sulfated polysaccharide from Caulerpa lentillifera. Carbohydr. Polym. 2022, 280, 119006. [Google Scholar] [CrossRef]
- Gong, P.; Tian, Y.; Guo, Y.; Gu, L.; Li, J.; Qi, J.; Yu, B. Comparisons of antithrombosis, hematopoietic effects and chemical profiles of dried and rice wine-processed Rehmanniae Radix extracts. J. Ethnopharmacol. 2019, 231, 394–402. [Google Scholar] [CrossRef]
- Nyansah, W.; Koffuor, G.; Ben, I.; Gyanfosu, L.; Ehigiator, B. Antithrombotic property of an aqueous extract from Pseudocedrela kotschyi and Adenia cissampeloides. Res. Pharm. Sci. 2021, 16, 436–446. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xi, M.; Choi, Y.; Lee, B. Antithrombotic effect of fermented Ophiopogon japonicus in thrombosis-induced rat models. J. Med. Food 2017, 20, 637–645. [Google Scholar] [CrossRef]
- He, N.; Wang, P.; Niu, Y.; Chen, J.; Li, C.; Kang, W. Evaluation antithrombotic activity and action mechanism of myricitrin. Ind. Crop. Prod. 2019, 129, 536–541. [Google Scholar] [CrossRef]
- Wei, X.; Zhang, B.; Wei, F.; Ding, M.; Luo, Z.; Han, X.; Tan, X. Gegen Qinlian pills alleviate carrageenan-induced thrombosis in mice model by regulating the HMGB1/NF-κB/NLRP3 signaling. Phytomedicine 2022, 100, 154083. [Google Scholar] [CrossRef] [PubMed]
- Lazze, M.; Pizzala, R.; Perucca, P.; Cazzalini, O.; Savio, M.; Forti, L.; Vannini, V.; Bianchi, L. Anthocyanidins decrease endothelin-1 production and increase endothelial nitric oxide synthase in human endothelial cells. Mol. Nutr. Food Res. 2006, 50, 44–51. [Google Scholar] [CrossRef]
- Chirkov, Y.; Nguyen, T.; Horowitz, J. Impairment of anti-aggregatory responses to nitric oxide and prostacyclin: Mechanisms and clinical implications in cardiovascular disease. Int. J. Mol. Sci. 2022, 23, 1042. [Google Scholar] [CrossRef]
- Ren, D.; Liu, Y.; Zhao, Y.; Yang, X. Hepatotoxicity and endothelial dysfunction induced by high choline diet and the protective effects of phloretin in mice. Food Chem.Toxicol. 2016, 94, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Badimon, L.; Vilahur, G.; Rocca, B.; Patrono, C. The key contribution of platelet and vascular arachidonic acid metabolism to the pathophysiology of atherothrombosis. Cardiovasc. Res. 2021, 117, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhai, X.; Ren, D.; Luo, Y.; Hu, Y.; Yang, X. Chemical characteristics of an Ilex Kuding tea polysaccharide and its protective effects against high fructose-induced liver injury and vascular endothelial dysfunction in mice. Food Funct. 2017, 8, 2536. [Google Scholar] [CrossRef] [PubMed]
- Aburima, A.; Berger, M.; Spurgeon, B.; Webb, B.; Wraith, K.; Febbraio, M.; Poole, A.; Naseem, K. Thrombospondin-1 promotes hemostasis through modulation of cAMP signaling in blood platelets. Blood 2021, 137, 678–689. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Han, R.; Zhu, Z.; Zhang, Y.; Wang, X.; Wang, L.; Shen, Z. Hemostatic variation during perioperative period of orthotopic liver transplantation without venovenous bypass. Thromb. Res. 2008, 122, 161–166. [Google Scholar] [CrossRef]
- Lu, G.; Lin, J.; Curnutte, J.; Conley, P. Andexanet alfa, a universal antidote Under development for Factor Xa inhibitors, reverses rivaroxaban-induced inhibition of thrombin generation initiated by the intrinsic coagulation pathway independent of TFPI. Blood 2016, 128, 3831. [Google Scholar] [CrossRef]
- Roser, L.; Monica, S.; Pura, M. Plasmin generation dependent on alpha-enolase-type plasminogen receptor is required for myogenesis. Thromb. Haemost. 2003, 90, 724–733. [Google Scholar]
- Needs, P.; Selvendran, R. Avoiding oxidative degradation during sodium hydroxide/methyl iodide-mediated carbohydrate methylation in dimethyl sulfoxide. Carbohyd. Res. 1993, 245, 1–10. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, R.; Wang, L.; Li, Y.; Han, J.; Yang, Y.; Zheng, H.; Lu, M.; Shen, Y.; Yang, H. Purification and characterization of a novel thermostable anticoagulant protein from medicinal leech Whitmania pigra Whitman. J. Ethnopharmacol. 2022, 288, 114990. [Google Scholar] [CrossRef]
- Zhou, J.; Zhai, J.; Zheng, W.; Han, N.; Liu, Z.; Lv, G.; Zheng, X.; Chang, S.; Yin, J. The antithrombotic activity of the active fractions from the fruits of Celastrus orbiculatus Thunb through the anti-coagulation, anti-platelet activation and anti-fibrinolysis pathways. J. Ethnopharmacol. 2019, 241, 111974. [Google Scholar] [CrossRef] [PubMed]







| Groups | APTT (s) | PT (s) | TT (s) | FIB (mg/mL) |
|---|---|---|---|---|
| Control | 35.86 ± 0.05 | 17.60 ± 0.29 | 21.07 ± 0.48 | 1.86 ± 0.03 |
| Model | 30.67 ± 0.37 ## | 13.37 ± 0.34 # | 16.90 ± 0.29 ## | 3.50 ± 0.06 ## |
| Aspirin | 34.63 ± 0.09 ** | 16.27 ± 0.37 ** | 19.77 ± 0.57 ** | 2.49 ± 0.02 ** |
| Test-L | 30.87 ± 0.12 | 12.70 ± 0.22 | 16.80 ± 0.29 | 3.50 ± 0.07 |
| Test-M | 31.30 ± 0.37 | 12.77 ± 0.25 | 18.03 ± 0.59 | 3.24 ± 0.02 ** |
| Test-H | 32.57 ± 0.29 ** | 14.00 ± 0.22 * | 18.73 ± 0.37 ** | 2.70 ± 0.02 ** |
| Groups | Experimental Treatments | |||
|---|---|---|---|---|
| Saline | AAP-b2 | Aspirin | Carrageenan (0.5%) | |
| Control | a | - | - | - |
| Model | a | - | - | 10 mL/kg b.w. |
| Aspirin | - | - | 20 mg/kg b.w. | 10 mL/kg b.w. |
| Test-L | - | 50 mg/kg b.w. | - | 10 mL/kg b.w. |
| Test-M | - | 100 mg/kg b.w. | - | 10 mL/kg b.w. |
| Test-H | - | 200 mg/kg b.w. | - | 10 mL/kg b.w. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bian, C.; Ji, L.; Qu, H.; Wang, Z. A Novel Polysaccharide from AuriculariaAuricula Alleviates Thrombosis Induced by Carrageenan in Mice. Molecules 2022, 27, 4831. https://doi.org/10.3390/molecules27154831
Bian C, Ji L, Qu H, Wang Z. A Novel Polysaccharide from AuriculariaAuricula Alleviates Thrombosis Induced by Carrageenan in Mice. Molecules. 2022; 27(15):4831. https://doi.org/10.3390/molecules27154831
Chicago/Turabian StyleBian, Chun, Lanyang Ji, Hang Qu, and Zhenyu Wang. 2022. "A Novel Polysaccharide from AuriculariaAuricula Alleviates Thrombosis Induced by Carrageenan in Mice" Molecules 27, no. 15: 4831. https://doi.org/10.3390/molecules27154831





