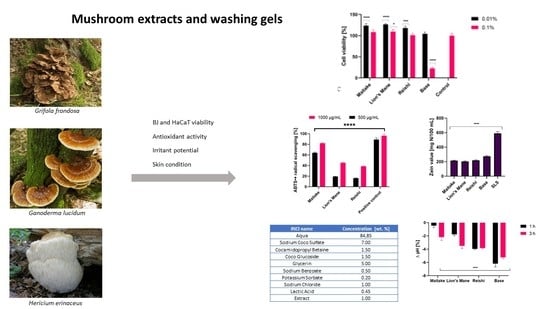
2.2. Determination of Antioxidant Properties
In order to determine the antioxidant properties of the tested extracts, a DPPH radical scavenging assay was performed. The obtained results, for the samples in a concentration of 1000 µg/mL, are presented in
Figure 2. The most effective scavenging of the DPPH radical was noted for the Maitake extract, which reached the highest value after 20 min of incubation (39.84%). The Reishiextract obtained the least efficient scavenging of the DPPH radical and its value did not change during the 20 min of the measurement. At the beginning of the measurement, it was 6.47%, while after 20 min it was only 9.29%. The situation wassimilar in the case of the Lion’s Mane extract. At the beginning of the measurement, it was 7.11%, and after 20 min it was 12.29%.
The second test used to determine the antioxidant properties of the examined extracts was ABTS
• scavengingassay. The obtained results, for the samples in concentrations of 500 and 1000 µg/mL, are presented on the
Figure 3. For all extracts, the highest values of ABTS
• radical scavenging wereobtained in the concentration of 1000 µg/mL. The most effective scavenging was noted for Maitake (82.12%), and the least for Reishi (38.50%). The situation wassimilar in the case of the 500 µg/mLconcentration. The highest value was obtained for Maitake (64.19%) and the lowest for Reishi (16.22%). Due to the fact that Fe
2+ ions are involved in the formation of reactive oxygen species, the next test evaluating the antioxidant properties was the test assessing the ability to chelate these ions (
Figure 4). Results were obtained for extract samples also at concentrations of 500 and 1000 µg/mL. As in previous tests, the highest chelating capacity for Fe
2+ ions was recorded for the Maitake extract (31.67%) in the concentration of 1000 µg/mL. The reducing activity of the tested Maitake, Lion’s Mane and Reishi extracts was also measured by the FRAP method (
Table 5). The results of the experiments were expressed as μmol of Trolox equivalent/g of dry weight of individual mushrooms. As for the other tests assessing antioxidant activity, Maitake extract also showed the most favorable properties obtaining 21.1 ± 3.2 μmol Trolox/g dry weight.
Both for the extracts and for the prepared cosmetics containing the analyzed mushrooms, a test was carried out to detect the ability of the compounds to generate intracellular production of reactive oxygen species. The test was performed using fluorogenic H2DCFDA dye, which is oxidized inside cells into highly fluorescent 20,70-dichlorofluorescein in the presence of ROS.
As shown in
Figure 5, all tested extracts did not increase the amount of ROS in fibroblasts. Reishi showed values similar to the control (cells not treated with the extracts), while Maitake and Lion’s Mane achieved values lower than the control, which means that the presence of these extracts lowers ROS levels in the cells. In this respect, the Maitake extract wasthe most effective, compared with the Reishi and Lion’s Mane extracts. The situation wassimilar with keratinocytes; only Reishi extract obtained a higher fluorescencevalue than the control. The rest of the extracts did not increase the intracellular production of ROS.
A test was also carried out on washing gels containing extracts from the studied mushrooms, the results of which are presented in
Figure 6. In the case of fibroblasts, all washing gels containing mushrooms extracts achieved higher fluorescence values than the control, but lower than cells treated with only base. In the case of keratinocytes, the situation wassimilar. The Maitake, Reishi and Lion’s Mane washing gels obtained a fluorescence value higher than the control, but lower than the base, which may suggest that the presence of the mushroom extract reduces the harmful effects of the compounds present in the cosmetic base, both on fibroblasts and keratinocytes.
When analyzing the obtained results, it can be concluded that the tested mushrooms showed antioxidant activity. This is caused by the presence of compounds such as malic acid, fumaric acid, citric acid, hericenone, erinacine, ganoderic acid and lucidenic acid, which have the ability to protect cells against the harmful effects of free radicals [
47,
48,
49,
50,
51,
52,
53,
54,
55,
56]. Maitake extract showed the best antioxidant properties.
Wang and Xu investigated the antioxidant properties of various mushroom species, including Lion’s Mane and Maitake. They compared, among other, total phenolic content in acetone, ethanol, water, and hot water extracts. For both Lion’s Mane and Maitake, the highest phenolics were found in the aqueous extracts, namely, 3.08 mg GAE/g (Lion’s Mane) and 3.78 mg GAE/g (Maitake). Moreover, DPPH free radical scavenging capacities were determined, which proved to be better for Lion’s Mane (2.85 µmole TE/g) than Maitake (1.75 µmole TE/g) [
57]. Yeh et al. tested two Maitake strains, for which the phenol content in the water extracts was 39.78 mg/g (T1 strain) and 38.96 m/g (T2 strain). The researchers also determined the content of flavonoids, which was lower: 1.09 mg/g for the T1 strain, and 0.52 mg/g for the T2 strain. The presence of ascorbic acid, which also exhibits antioxidant properties, was also confirmed in these extracts. The ability of the extracts to scavenge DPPH radicals was also tested; at a concentration of 20 mg/mL for cold water extracts of Maitake T1 and T2, it was 50.62% and 59.58%, respectively. Much higher results were recorded for ethanolic extracts: 99.19% (T1) and 84.36% (T2) at 20 mg/mL [
58]. On the other hand, in the work of Rahman et al., the antioxidant properties of two Reishi strains were compared. Total polyphenol content for Ganoderma lucidum-5 was 33.30 mg/100g, and for Ganoderma lucidum-7 was 43.49 mg/100g. Total flavonoid content was 34.09 mg/100g (Ganoderma lucidum-5) and 38.08 mg/100g (Ganoderma lucidum-7). The ability to scavenge the DPPH radicals by strains Ganoderma lucidum-5 and Ganoderma lucidum-7, was 24.27% and 23.66%, respectively [
59].
2.3. Cytotoxicity Assessment
In assessing the potential use of extracts from various natural raw materials, including fungi, it is very important to assess their cytotoxicity to skin cells. Hence, as part of this study, the impact of the three prepared extracts, and gels containing these extracts, on the viability of fibroblasts and keratinocytes was assessed. The first of the tests to assess the metabolic activity of the studied cells was alamarBlueassay (AB). As shown in
Figure 6, Lion’s Mane and Reishi extracts at all tested concentrations showed no toxic effects on keratinocytes (HaCaT) and fibroblasts (BJ cells). Maitake extract at the concentration of 100 µg/mL was not cytotoxic for BJ and HaCaT cells, but with the increase in concentration the cytotoxicity increased, and at the highest concentration (1000 µg/mL) the viability of these cells decreased below 60%. The developed gels in a concentration of 0.01%, containing extracts from the tested fungi, did not show any cytotoxicity. However, in the case of their higher concentration (0.1%), they showed cytotoxicity towards keratinocytes, depending on the type of fungus used. It should be noted that compared with the gels base, the addition of the extract significantly reduced their cytotoxicity, which indicates an increase in their safety of application to skin. The positive effect was observed to the greatest extent in the case of Lion’s Mane, for which at a concentration of 0.1%, an increase in keratinocyte viability by 70% compared with the gel base was observed. The cytotoxic effect of the gel base itself is probably related to the activity of surfactants present in the cosmetic base, the cytotoxic effect of which has been proven in numerous studies [
60,
61]. Based on the results presented in
Figure 7C, it can be concluded that the presence of fungal extracts in the developed washing preparations minimizes the unfavorable effect of surfactants, and thus increases the proliferation and viability of cells.
The evaluation of cytotoxicity was also carried out using the Neutral Red uptake assay (NR), and the results are shown in
Figure 8. The obtained results indicate that Lion’s Mane and Reishi extracts at all tested concentrations showed no cytotoxicity to both BJ and HaCaT cells. Moreover, treating keratinocytes with Reishi extracts increased the viability of these cells. On the other hand, Maitake extract showed cytotoxicity to BJ cells at all tested concentrations, and this effect was stronger with increasing concentration. In the case of HaCaT cells, this extract showed no cytotoxicity at the concentrations of 100 µg/mL and 500 µg/mL, but it inhibited the viability of these cells at the concentration of 1000 µg/mL. In the case of the developed gels containing extracts of the tested mushrooms, none showed cytotoxicity. At a concentration of 0.01%, all showed the ability to increase the proliferation of HaCaT cells by about 20% compared with the control (cells not treated with gels). Similar to the AB test, the studies showed that the gel base at a concentration of 0.01% did not show cytotoxicity, while when 0.1% concentration wasused, the cytotoxicity wassignificant and reduced the keratinocyte viability to just over 20%. On the other hand, the addition of extracts from all three tested fungi to the gel formula eliminated the cytotoxicity of the tested washing preparations, which indicates the legitimacy of including these extracts in the formulathedeveloped gels.
Research on skin cell lines is an important element that allows assessment of the safety of the obtained extracts, and to predict their possible effects in the next stages of research, such as in vivo tests or clinical trials. The results obtained in this study, indicating the lack of cytotoxicity of the tested extracts used in a given concentration range and the possibility of reducing the negative effects of various cosmetic ingredients, allow the estimate that these raw materials can be more and more willingly included in the formulations of a wide range of cosmetic preparations. In addition to the lack of a negative impact on the viability and proliferation of keratinocytes and fibroblasts, scientific works indicate that these extracts also exhibit multidirectional activity in in vitro conditions.
The available literature data do not contain any reports on the cytotoxicity of Maitake extracts to skin cells. There are reports mainly describing antitumor, immunomodulating and antioxidant properties of this fungus, but the influence of this type of extract on the viability of keratinocytes and fibroblasts has not been assessed [
62,
63,
64]. Contrary to the extracts of Lion’s Mane and Reishi, the analyses carried out in this study showed that these extracts, in the higher concentrations used, exert a cytotoxic effect on skin cells in vitro, mainly fibroblasts. This may be the result of the action of compounds contained in these extracts, including alpha-hydroxy acids, which, despite their antioxidant activity, may exert an antiproliferative effect on HaCaT cells [
65]. Scientific research shows that malic acid inhibits the proliferation of keratinocytes by inhibiting the progression of the cell cycle in the G0/G1 phase. Additionally, this acid can induce the expression of endoplasmic reticulum stress-related proteins such as GRP78, GADD153 and ATF6α [
65]. Other studies carried out on human dermal fibroblasts (HDF) exposed to ultraviolet A radiation, however, indicate that exopolysaccharide isolated from this fungus has photoprotective potential. This polysaccharide has an inhibitory effect on the expression of human interstitial collagenase (matrix metalloproteinase, MMP-1), which may reduce skin photoaging by reducing the matrix degradation system associated with MMP-1 [
66]. Thus, in order to use the potential of this fungus, it is necessary to select theconcentration of the extract that will show the desired biological activity with the simultaneous lack of cytotoxicity. This is important because, as shown in this work, the addition of Maitake extract may reduce the negative effect of cosmetic cleansing preparations on skin cells.
The effect of Lion’s Mane extract on keratinocytes and fibroblasts in vitro has not been described to date. Thus, in this study, for the first time, we demonstrated the lack of cytotoxic effect of the studied extract on these skin cells, and the possibility of stimulating their viability and proliferation. This may be the result of reducing the level of free radicals in these cells, which was also shown in this work. The inhibitory effect on the production of ROS was also indicated by Chang et al., who demonstrated that the extracts of this fungus trigger the expression of the antioxidant genes of heme oxidase-1 (HO-1), γ-glutamylcysteine synthetase (γ-GCLC), and affect the level of glutathione. The antioxidant activity demonstrated by these authors on human endothelial cells (EA.hy926) is associated with increased nuclear translocation and transcriptional activation of NF-E2 associated factor 2 (Nrf2) [
67].
Due to the fact that Reishi is considered to be one of the strongest adaptogens found in nature and exhibits not only antioxidant but also anti-inflammatory, immunomodulating and anti-cancer properties, interest in it in the context of skin care and treatment of skin diseases is growing [
68]. Abate et al. showed that Reishiextract inducesthe proliferation of keratinocytes and increases the expression of cyclic protein kinases, such as CDK2 and CDK6. Additionally, these authors indicated that keratinocytes treated with Reishiextract show an increased migration rate and an increase in activation of tissue remodeling factors such as matrix metalloproteinases 2 (MMP2) and 9 (and MMP-9). Moreover, this extract, through its antioxidant activity, protects keratinocytes against H
2O
2-induced cytotoxicity. These studies indicate the legitimacy of the cosmetic use of this fungus due to the possibility of accelerating wound healing processes, protecting cells against oxidative stress and intensified re-epithelialization [
69]. Kim et al. indicated the inhibitory effect of Reishi extract on the activity of tyrosinase and melanin biosynthesis in B16F10 melanoma cells. They also demonstrated the possibility of inhibiting the expression of tyrosinase-related protein 1 (TRP-1), TRP-2, as well as microphthalmia-related transcription factor (MITF), thus reducing the production of melanin. This extract also influences the mitogen-activated kinase (MAPK) cascade and cyclic adenosine monophosphate (cAMP)-dependent signaling pathway, which has a significant effect on the melanogenesis of B16F10 melanoma cells [
20]. Hu et al., on the other hand, indicated that the polysaccharides contained in Reishi increase the viability of fibroblasts and the ability to migrate these cells. Moreover, these polysaccharides increase the expression of β-catenin, CICP and TGF-β1 in fibroblasts in vitro. Additionally, in vivo studies in mice indicated that these compounds significantly improved healing rates and wound healing time. This is likely the result of activation of the Wnt/β-catenin signaling pathway and elevated levels of TGF-β1 [
1].
To sum up, the lack of cytotoxic effect of Lion’s Mane and Reishi extracts on skin cells, and a reduction in the cytotoxicity of the base of cleansing preparations by extracts from all three tested mushrooms, indicate that they can be perceived as valuable cosmetic raw materials with a broad spectrum of activity.
2.4. Transepidermal Water Loss (TEWL), Skin Hydration and Skin pH Measurements
Due to the content of hydroxyl groups in molecules, bioactive ingredients of the studied extracts have a positive effect on the condition of our skin. They mainly affect the retention of water in the skin, but also reduce the evaporation of water from the upper layer of the epidermis [
70,
71,
72]. In addition, these extracts are characterized by several health-promoting properties and therefore can be successfully used as active ingredients in cosmetic products. The conducted research assessed the effect of preparations containing extracts on basic skin parameters, such as skin hydration, TEWL and pH. The tested samples were products containing 1% mushroom extract (Reishi, Lion’s Mane, Maitake) and the base sample, for conductinga comparison. The analyses were carried out at twotime intervals, 1 h and 3 h, after the application of the product. The analysis of the obtained results showed the positive effect of the contained extract on the condition of the skin. The results are presented in the
Figure 8,
Figure 9 and
Figure 10.
Analysis of skin moisture showed differences between examined model gels with extracts, which will be discussed one by one. For the gel containing Maitake extract, aminimal influence was observed. After 1 h, the effect was similar to the base, for which decreased moisture was recorded, but registered values were around 20% lower when compared with the base. After 3 h it turned into a 20% relative increase with reference to the base level. In the case of Lion’s Mane, the initial effect measured after 1 h was also negative, showing around 10% decrease in moisture compared with the base, but after 3 h the result turned into a significant 200% increase in moisture. The best results were noted for the Reishi extract, where, even after the first 1 h, a slight increase in moisture was observed, bearing in mind that for the base a noticeable decrease was observed. After 3 h, high values were observed, as the moisture was 350% higher when compared with the base. From that perspective, Reishi showed the most preferable properties, with better results, compared with the rest of the examined extracts.
Analysis of the TEWL showed a positive effect of each of the model gels with mushroom extracts, however there were still some differences between them which are worth discussing. First, values observed after 1 h were all preferable, showing values around −7.5% and −8% for Maitake and Reishi, respectively, and even better valuearound −9.5% for Lion’s Mane. Values measured after 3 h showed, in case of Maitake extract, the TEWL value stayed at almost the same level of −8%, but in the case of Lion’s Mane and Reishi extracts even better values were observed, equaling −11% for both. In conclusion, the Lion’s Mane extract exhibited the most preferable properties, slightly ahead of the Reishi extract.
Analysis of the pH showed noticeable differences between model gels with mushroom extracts (
Figure 11). Zero level means no change in the skin pH in relation to the control field, i.e., the physiological pH of the test volunteers, therefore, the most favorable values would beclose to zero. First, values observed for the base werethe most distant from zero level, being the least preferable. Values observed for the Maitake extract appeared to be the best among those compared, showing differences of −0.5 and −2, respectively, after 1 h and 3 h. It is worth emphasizing that the deviation observed after 1h wasvery close to the perfect natural skin pH.NextwastheLion’s Mane extract, for which differences of −2 and −3.5,after 1 h and 3 h, respectively, wereobserved. The worst results were measured for the Reishi extract, where the deviation from the optimal pH was close to −4 after both 1 h and 3 h. From that perspective, the Maitake extract results werethe most preferable.
2.5. Irritant Potential of Model Body Wash Gels
The lack of adverse effects of washing cosmetics on skin is one of the most important requirements for this group of products. The measurement results of the irritating potential of the analyzed washing gels are presented in
Figure 12.
The method used to determine the irritation potential of the model washing gels was the determination of the zein number. Due to the fact that the irritant potential of washing cosmetics is caused by interactions of surfactants with proteins that build the stratum corneum layer, this method closely imitates the strength of the interactions of surfactants used in the formulation of the model washing gels, with skin proteins. The irritating potential generated by surfactants results from the ability of these substances to denature epidermal proteins and then wash them out of the skin. Contact of the analyzed samples with zein, a protein with a structure similar to the main proteins in skin, may indicate their protein-denaturing capacity, which is a measure of irritating potential.
Obtained results showed that the addition of analyzed mushroom extracts to the base model washing gel reduced the irritant potential (zein number). The base sample (without the addition of the extracts) was characterized by a zein number at the level of 270 mg N/100 mL and, according to the accepted classification, was classified as moderately irritant for the skin. Samples containing Maitake and Reishi extracts had lower irritant potential and their zein valueswere about 20% lower in relation to the base sample (about 220 mg N/100 mL). The lowest irritant potential was observed for the sample that contained Lion’s Mane extract. The zein value of this sample was about 25% lower than the base sample. The obtained results indicate that samples with the addition of the analyzed extracts can be classified on the border of non-irritating and slightly irritating.
The anionic surfactant, sodium coco sulfate, was used in the formulation of the analyzed model washing gels as the main washing agent. As mentioned in the introduction, anionic surfactants have the strongest irritant potential due to the fact that they interact with skin proteins through strong ionic bonds, and their denaturing potential is significantly higher than for nonionic and amphoteric surfactants. The lower irritant potential observed for the samples containing mushroom extracts wasmost probably caused by carbohydrates and proteins, that are the main ingredients of mushrooms’ dry weight, and are extracted, in the extraction process, by polar solvents. These substances may be incorporated into surfactant’s micelles, causing anincrease in their stability and size. Formation of mixed micelles that contain both surfactants and high molecular weight carbohydrates and proteins limits the number of free surfactants in the form of monomers that are released to the bulk phase, and leads to a lowering of the irritant potential of the product [
23,
25,
26,
27].