Abstract
A method for the [2+3] pyrroline annulation to the six-membered non-aromatic enols using 3-aryl-2H-azirines as annulation agents is developed in the current study. The reaction proceeds as a formal (3+2) cycloaddition via the N1-C2 azirine bond cleavage and is catalyzed by both Cu(II) and Cu(I) compounds. The new annulation method can be applied to prepare pyrrolo[3,2-c]quinoline, chromeno[3,4-b]pyrrole, and naphtho[1,8-ef]indole derivatives in good to excellent yields from enols of the quinolin-2-one, 2H-chromen-2-one, and 1H-phenalen-1-one series.
1. Introduction
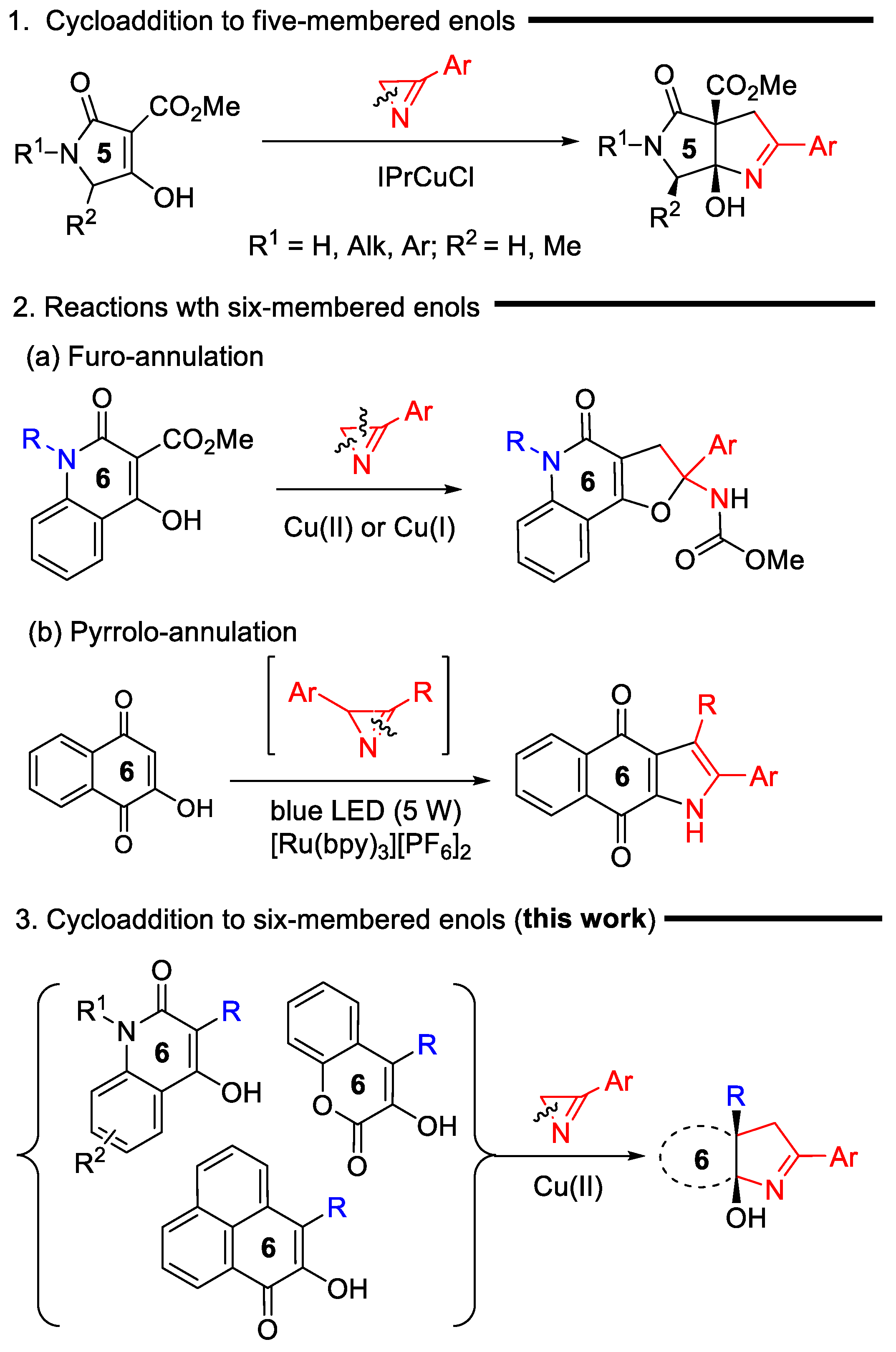
2H-Azirines are widely used for the preparation of various 4–7-membered N-, N,N-, and N,O-heterocycles of varying degrees of unsaturation and different heteroatom arrangements [1,2]. The ability of azirines to open at any of the three bonds of the ring under the action of electrophilic and nucleophilic reagents, as well as transition metal compounds, underlies a powerful strategy for the synthesis of azete [3], pyrrole [4,5,6], oxazole [7,8,9], imidazole [10,11,12], 1,2,3-triazole [13], pyridine [14] derivatives, and other heterocycles, which are hardly accessible with conventional methods. Some of the azirine-ring opening reactions can be applied for the synthesis of ortho-fused, spiro-fused, and bridged heterocycles. These heteropolycycles can be the result of both intramolecular and intermolecular reactions of azirines [1]. Among them, intermolecular cycloaddition reactions are of particular interest, since they satisfy the requirements of green chemistry being atom-economical processes. In contrast to the (2+3)- and (2+4)-cycloaddition reactions of azirines with 1,3-dipoles and 1,3-dienes (or their aza-analogs) [15,16,17,18,19], in which azirines, without the ring opening, provide the incorporation of a diatomic N–C fragment in the resulting heterocycle, the reaction sequence “azirine-ring opening/cycloaddition” provides the incorporation of all atoms of the azirine ring into a new heterocyclic system. This annulation strategy includes transition-metal-catalyzed reactions of azirines with cyclic diazo compounds [20,21,22], Y(OTf)3-catalyzed [3+6] cycloaddition of azirines to fulvenes [23] leading to 3,4-dihydro-2H-cyclopenta[c]pyridine derivatives, photoinduced (3+2) cycloaddition of nitrile ylides, generated via the azirine-ring opening (C2–C3 azirine bond cleavage), to quinones [24] or N-benzylmaleimide [25], synthesis of cycloalkane-fused pyrroles by the Fe(III)-catalyzed decarboxylative (3+2) cycloaddition of the 2H-azirines to cyclic β-ketoacids (N1–C3 azirine bond cleavage) [26], and synthesis of pyrrolo[3,4-b]pyrrole derivatives via Cu(I)-catalyzed (3+2) cycloaddition of azirines to the enol carbon–carbon double bond of 3-methoxycarbonyl-substituted tetramic acids (Scheme 1, reaction 1) [27]. The last of these methods can be effectively applied to the pyrroline annulation of tetronic and thiotetronic acids as well [28]. However, attempts to extend this method to six-membered enols of the quinoline-3-carboxylate series unexpectedly encountered a serious problem associated with the involvement of the ester substituent in the transformation (Scheme 1, reaction 2a). This reaction also proceeds through azirine N1-C2 bond cleavage under both Cu(I) and Cu(II) catalysis, but exclusively produces the furo-annulation product [29]. A visible-light-promoted (3+2) cycloaddition of azirines, derived in situ from vinyl azides, to α-hydroxybenzoquinones is the only successful example of azirine cycloaddition to a multiple bond of a six-membered cyclic enol to date (Scheme 1, reaction 2b) [30]. However, this reaction cannot serve as an alternative to the method of copper-catalyzed annulation of enols with azirines, since it proceeds via the cleavage of not a single N-C2 azirine bond, but a multiple N-C3 bond, and provides a pyrrole ring with another substitution pattern. Thus, the search for the effective conditions for (3+2) cycloadditions of azirines to unsaturated cyclic systems, including cyclic enols, the elucidation of mechanisms of these reactions, assessing their scope and limitations still remains an outstanding challenge.
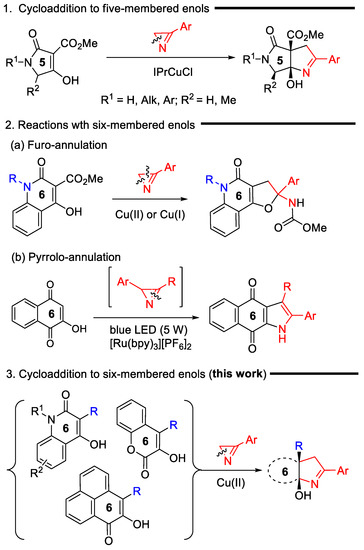
Scheme 1.
Reactions of cyclic enols with 2H-azirines.
In this study, we describe a method for the pyrroline annulation of six-membered non-aromatic enols of the quinolin-2-one, 2H-chromen-2-one, and 1H-phenalen-1-one series. Additionally, a reaction mechanism is presented that allows us to define the scope of this method.
2. Results and Discussion
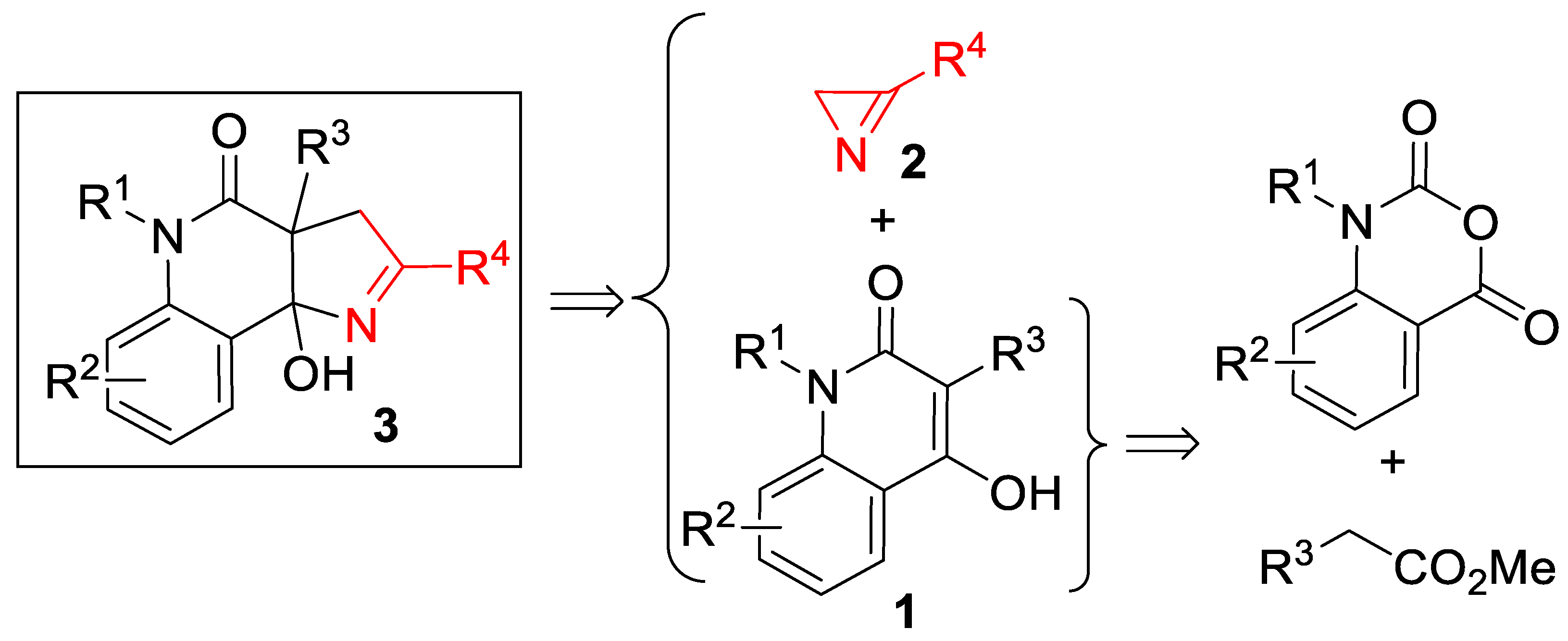
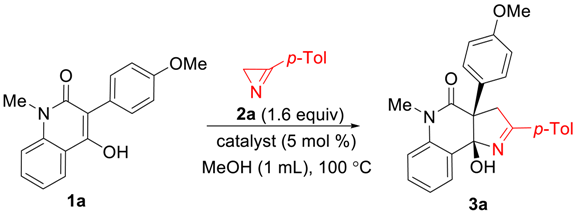
Our initial interest in (3+2)-cycloaddition reactions of azirines 2 was related to their possible use for the rapid assembly of 1H-pyrrolo[3,2-c]quinoline framework 3 from 4-hydroxyquinolone derivatives 1 (Scheme 2). The structural motif of 1H-pyrrolo[3,2-c]quinoline has always attracted the attention of synthetic chemists [31,32], as it is included in many bioactive, natural products [33,34,35,36] and synthetic compounds that possess enzyme modulator [37], antitumor [38], and 5-hydroxytryptamine(6) receptor antagonist activities [39]. The attractiveness of the mentioned approach to these compounds lies in the easy availability of quinolones 1, which can be prepared from isatoic anhydrides. It should also be noted that the synthesis of pyrroline-fused systems bearing a bridgehead hydroxy group is a challenge, since conventional annulation methods either produce unsatisfactory results [40] or require the use of an aggressive medium, such as liquid ammonia [41].
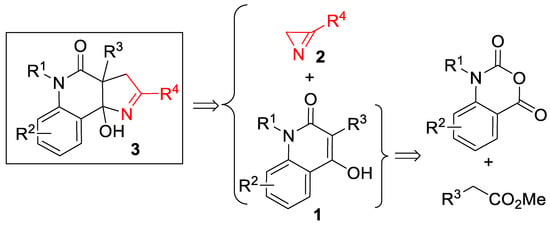
Scheme 2.
Retrosynthetic scheme for 1H-pyrrolo[3,2-c]quinolones 3.
In our initial experiments, we synthesized 3-(4-methoxyphenyl)-substituted quinolin-2-one 1a from N-methylisatoic anhydride and methyl 4-methoxyphenylacetate according to the procedure described in the literature [42]. Enol 1a turned out to be inactive toward azirine 2a when heated at 100 °C in methanol, toluene, or 1,2-dichloroethane (DCE). At higher temperatures, the formation of azirine decomposition products without enol involvement was observed. The reaction between 1a and 2a commenced at 100 °C in methanol when catalytic amounts of the Cu(I)-NHC complex IPrCuCl (5 mol%) were added, and resulted in the formation of the desired annulation product, pyrroloquinolone 3a, in 98% yield in 20 min (Table 1, entry 2). It was notable that a close to quantitative yield of 3a was also achieved with all tested copper(II) catalysts (entries 3–5). The optimal ratio 1a/2a was observed to be 1:1.6, whereas the reaction between equimolar amounts of the reagents provided only 65% yield (entry 6). Replacing anhydrous methanol with 96% aqueous ethanol led to a decrease in the yield (entry 7). A high yield of 3a in the CuCl2-catalyzed reaction was obtained by increasing the amount of the azirine to 2 equiv. To our surprise, cobalt(II) acetate as well as nickel(II) and iron(III) acetylacetonates also catalyzed the reaction, albeit with less efficiency (entries 9–11). As a result, we used the 1:1.6 mixture of enol 1 and azirine 2 in the presence of copper(II) acetate monohydrate (5 mol%) as a catalyst at 100 °C in methanol as optimal conditions for the further experiments.

Table 1.
Optimization of synthesis of 3a.
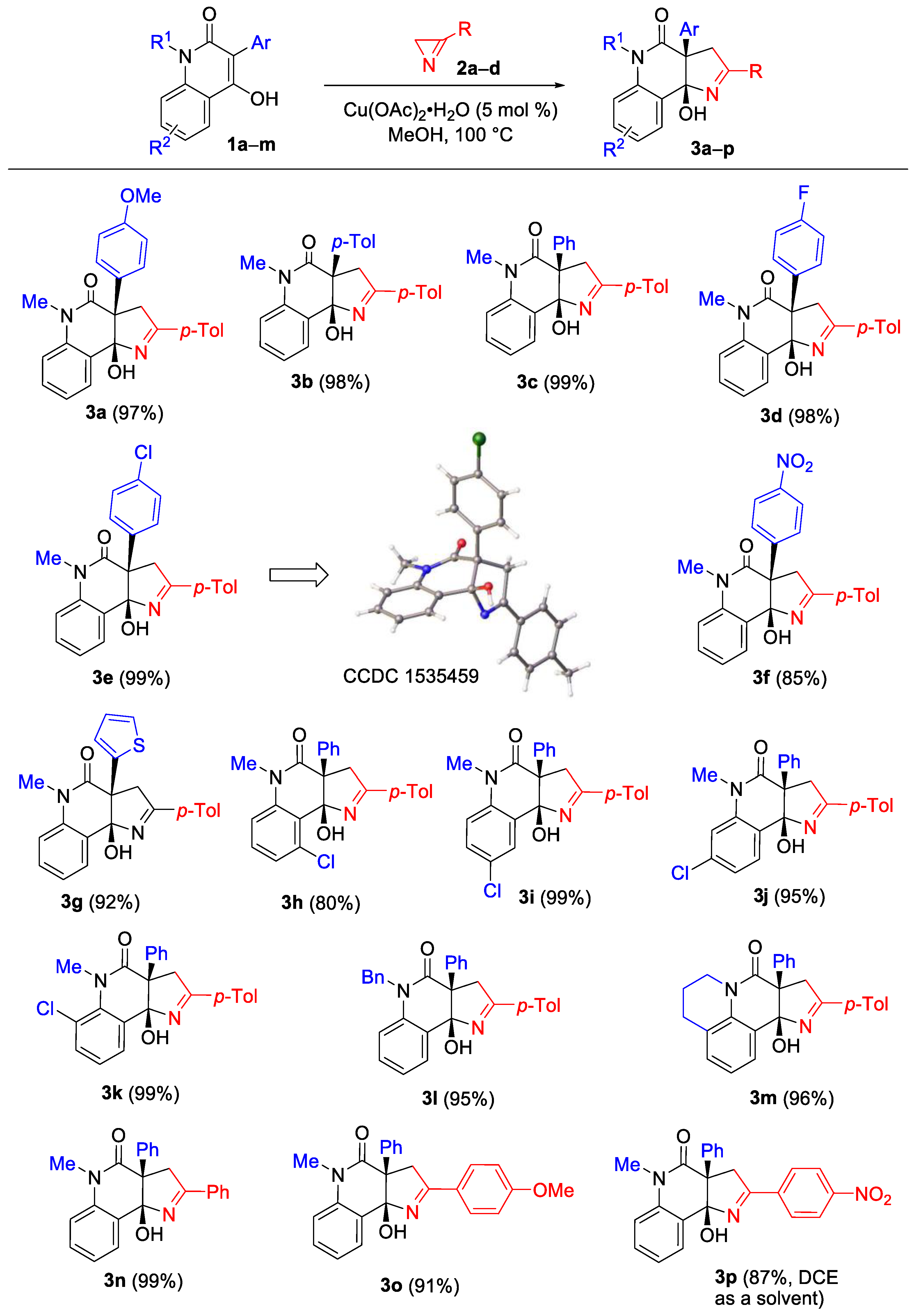
The scope of hydroxyquinolones 1 was then evaluated under the optimized conditions using 3-(p-tolyl)-2H-azirine (2a) as the reaction partner (Scheme 3). The reaction displayed a low sensitivity to the electronic effects of the aryl substituent at C3 of the enol (compounds 3a–f). In addition, the annulation product 3g was synthesized from a quinolone with a 2-thienyl substituent at the C3 in 92% yield. The reaction was observed to be tolerant to the presence of a substituent at any position of the benzene ring of the quinolone moiety and provided high product yields (compounds 3h–k). Additional ortho- and peri-fusion (as in quinolone 1m) also did not influence the product yield (compound 3m).
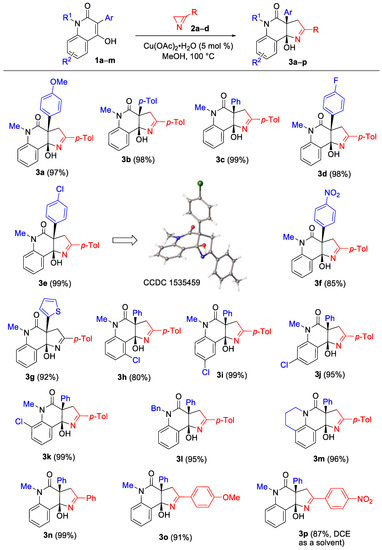
Scheme 3.
Synthesis of pyrroloquinolinones 3.
The structures of compounds 3a–p were established using NMR spectroscopy and HRMS methods. The structure of compound 3e was additionally verified by X-ray diffraction analysis.
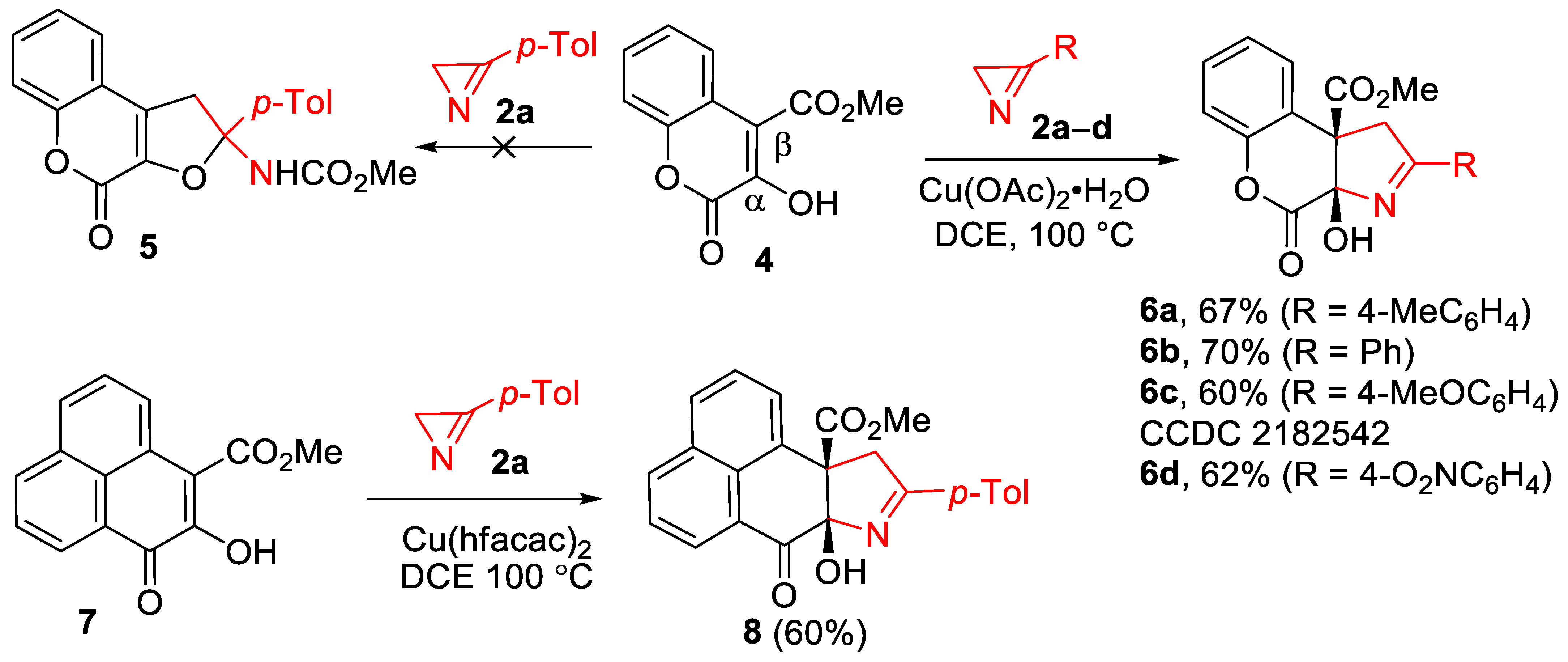
The comparison of the obtained results with the data of our previous work [29] (Scheme 1, reaction 2a) revealed a dramatic dependence of the reaction outcome on the nature of the C3 substituent in quinolones 1: 3-alkoxycarbonyl-substituted derivatives produced the products of furo-annulation, while 3-aryl-substituted derivatives exclusively produced pyrroline-fused products 3. In order to find out how general this pattern was for six-membered enols, we examined compounds 4 and 7, having carbonyl substituents both at the α- and β-carbon atoms of the enol moiety (Scheme 4). The Cu(OAc)2- and IPrCuCl-catalyzed reactions of chromenone 4 with azirine 2a, conducted in methanol at 100 °C, resulted in a complex, inseparable mixture of products. IPrCuCl did not catalyzed the reaction in DCE at all, but, to our delight, the target chromenopyrrole 6a was obtained in 67% yield in DCE using Cu(OAc)2 × H2O (5 mol%) as a catalyst. According to the 1H NMR spectrum of the reaction mixture, no traces of the furo-annulated product, compound 5, were detected. The reaction of chromenone 4 with azirines 2b–d occurred with almost the same efficiency, producing chromenopyrroles 6b–d in 60–70% yields. The structure of compound 6c was confirmed by X-ray diffraction analysis.
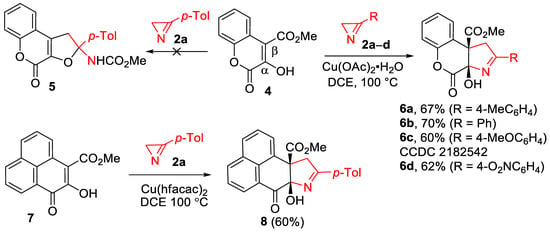
Scheme 4.
Synthesis of chromenopyrrolones 6 and adduct 8.
Phenalenone 7 with a similarly substituted enol moiety included in the ortho- and peri-fused system also reacted well with azirine 2a, producing the tetracyclic annulation adduct 8 in a 60% yield. In this reaction, copper(II) hexafluoroacetylacetonate (10 mol%) was used as a catalyst since it provided a slightly higher product yield than copper(II) acetate.
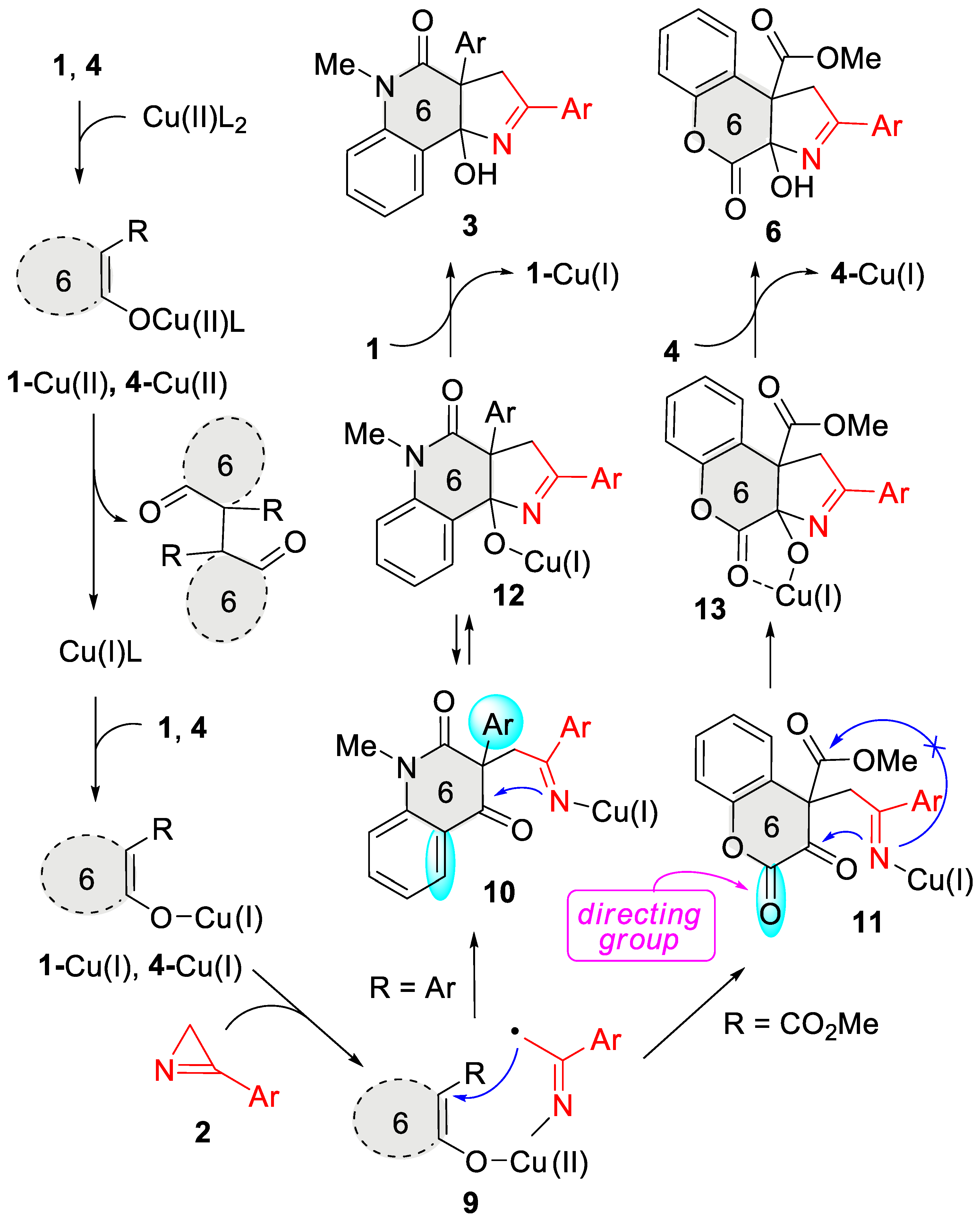
Thus, the substitution pattern of the enol moiety of six-membered cyclic enols controlled the outcome of their catalytic annulation with azirines directing the reaction toward either pyrroline- or furane-fused products. In the presence of an aryl substituent at the β-position of the enol moiety of quinolones 1, its (2+3) cycloaddition to azirines 2 smoothly proceeded to afford pyrroline-annulated products 3. In contrast, the CO2Me group in the same position directs the process toward the formation of furo-annulated products as follows from the results of the previous studies [29]. However, this switching does not occur if the α-carbon of the enol moiety is adjacent to the endocyclic carbonyl carbon atom. We proposed the reaction mechanism (Scheme 5) to address the observed reactivity of non-aromatic six-membered cyclic enols toward azirines under copper catalysis. The key step of the reaction was the azirine-ring opening across the N1-C2 bond to form radical 9 under the action of copper(I) enolate 1-Cu(I)/4-Cu(I). The latter resulted from the oxidative homocoupling of the enol with the copper(II) catalyst. Such an oxidation with copper(II) acetylacetonate was previously reported for tetramic acids [27]. Indeed, enol 1a reacts with Cu(OAc)2 in boiling MeOH, but, unfortunately, our attempts to isolate the oxidative coupling product failed because of the low selectivity of the reaction, which yielded an inseparable mixture of products. Intramolecular radical attack in intermediate 9 afforded copper(I) iminide 10,11. 3-Aryl-substituted iminide 10 underwent the cyclization at the keto group followed by a copper–hydrogen exchange to produce cycloadduct 3. The expected intramolecular nucleophilic attack of the iminide nitrogen on the ester carbonyl in intermediate 11 to form furo-annulation product 5 did not occur because of two reasons: (1) the additional activation of the electrophilic keto group by the lactone carbonyl group, and (2) stabilization of alcoholate 13 by the chelation of the copper by the lactone carbonyl group. As a result, the cyclization in the keto group proceeded rapidly and irreversibly. The copper–hydrogen exchange between alcoholate 13 and enol 4 afforded the final cycloadduct 6 and regenerated enolate 4-Cu(I).
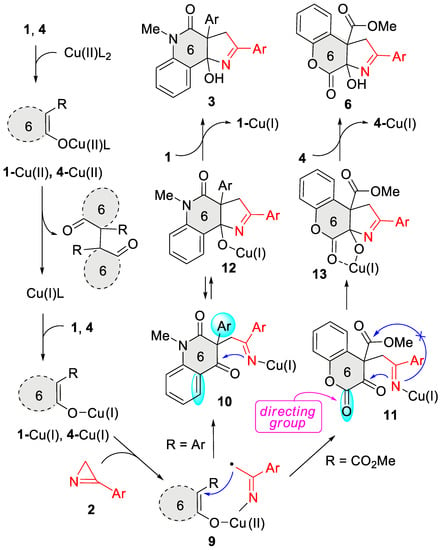
Scheme 5.
Plausible mechanism.
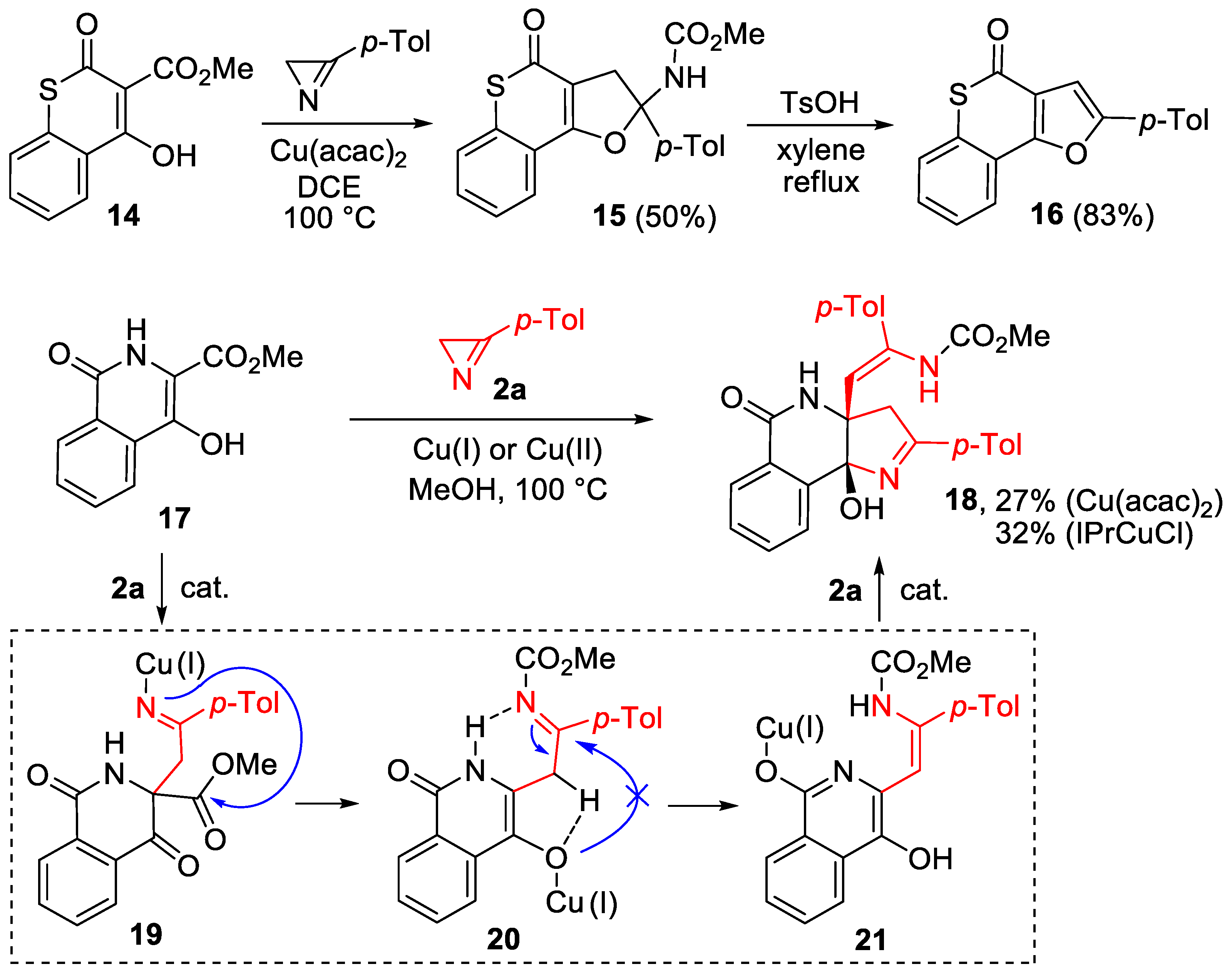
Thus, the lactone carbonyl in intermediate 11 acted as a directing group, enabling the annulation of the pyrroline ring even though the enol system bears an ester substituent at the β-C enol atom. The reaction of methoxycarbonyl-substituted 6-membered cyclic enols, having no directing carbonyl group, should proceed through a furo-annulation involving the CO2Me group. This important conclusion was supported by the reaction between thiochromene-based enol 14 and azirine 2a (Scheme 6). This reaction afforded carbamate 15 as a sole product in the presence of copper(II) acetylacetonate under the standard conditions. Compound 15 can be transformed under acidic conditions into thiochromenofuran 16 in good yields.

Scheme 6.
Synthesis of compounds 15, 16, and 18.
An unexpected result was obtained in the reaction of 4-hydroxyisoquinolinone 17 with azirine 2a catalyzed with Cu(acac)2 (5 mol%) (Scheme 6). The formation of a furo-fused product (similar to 15) did not occur from compound 17, despite the presence of the methoxycarbonyl group at the β-C atom and the absence of the directing carbonyl group at the α-C atom of the enol moiety. Instead, pyrrolino-annulated 1:2 adduct 18 was isolated in 27% yield. Optimization experiments showed that the use of other copper(II) catalysts (Cu(OAc)2, Cu(hfacac)2) did not enhance the efficiency of the reaction, whereas NHC complex IPrCuCl (5 mol%) allowed a slight increase in the yield of 18 (32%). This product resulted from the addition of two molecules of azirine 2a to isoquinolone 17, one of which modified the methoxycarbonyl group of 17 and another one formed the pyrroline ring. The abnormal reaction course can be rationalized in terms of rapid copper–hydrogen exchange in intermediate 20, which is more rapid than the furan ring closure. The (2+3) cycloaddition of aminovinyl-substituted enol 21 to azirine 2a afforded compound 18. The Z configuration of the C=C bond in compound 18 was established based on 2D 1H-1H-NOESY spectrum data (see the Supplementary Materials).
3. Materials and Methods
3.1. General Instrumentation
Melting points were determined on a melting-point apparatus and were uncorrected. 1H (400 MHz) and 13C (100 MHz) NMR spectra were recorded on a Bruker Avance 400 spectrometer in solvent indicated below. 1H NMR spectra were calibrated according to the residual peaks of CDCl3 (δ = 7.26 ppm), DMSO-d6 (δ = 2.50 ppm). 13C{1H} NMR spectra were calibrated according to the carbon atom peaks of CDCl3 (δ = 77.0 ppm), DMSO-d6 (δ = 40.0 ppm). High-resolution mass spectra were recorded with a Bruker maXis HRMS-QTOF, electrospray ionization. X-ray diffraction analysis was performed with an Agilent Technologies Xcalibur Eos (for 3e) and Agilent Technologies Supernova (for 6c) diffractometers. Crystallographic data for the structures 3e (CCDC 1535459) and 6c (CCDC 2182542) were deposited at the Cambridge Crystallographic Data Centre. Thin-layer chromatography (TLC) was conducted on aluminum sheets precoated with SiO2 ALUGRAM SIL G/UV254. Column chromatography was performed on silica gel 60 M (0.04–0.063 mm). Methanol was refluxed for 2 h with magnesium turnings and then distilled. 1,2-Dichloroethane was washed with concentrated H2SO4, water, then distilled from P2O5 and stored over anhydrous K2CO3.
Quinolones 1a–l were prepared from isatoic anhydrides and corresponding methyl acetates according to the procedure described in the literature [42]. Quinolones 1a,l,m [43], and 1c [42] are known compounds. Compounds 2a–c [44], 2d [45], 4 [46], 7 [47], 14 [48], and 17 [49] are known compounds, which were prepared by using the reported procedures.
3.2. Synthesis and Characterization of Quinolones
4-Hydroxy-1-methyl-3-(4-methylphenyl)quinolin-2(1H)-one (1b) [42]. Compound 1b (1.09 g, 82%) was prepared from N-methylisatoic anhydride (0.89 g, 5 mmol), methyl 4-methylphenylacetate (0.82 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 204–205 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.35 (s, 3H), 3.60 (s, 3H), 7.20–7.29 (m, 5H), 7.50 (d, J = 8.4 Hz, 1H), 7.61–7.65 (m, 1H), 8.02–8.04 (m, 1H), 9.92 (s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.4, 29.7, 112.6, 114.8, 116.8, 121.7, 124.0, 128.9, 131.1, 131.3, 131.5, 136.5, 139.3, 156.5, 162.5. HRMS-ESI: [M+H]+ calcd for C17H16NO2+ 266.1176, found 216.1187.
3-(4-Fluorophenyl)-4-hydroxy-1-methylquinolin-2(1H)-one (1d) [42]. Compound 1d (0.89 g, 66%) was prepared from N-methylisatoic anhydride (0.89 g, 5 mmol), methyl 4-fluorophenylacetate (0.84 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 288–290 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.61 (s, 3H), 7.20–7.24 (m, 2H), 7.28 (t, J = 7.5 Hz, 1H), 7.36–7.39 (m, 2H), 7.52 (d, J = 8.4 Hz, 1H), 7.65 (d, J = 7.5 Hz, 1H), 8.05 (d, J = 7.7 Hz, 1H), 10.11 (s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 29.7, 111.7, 114.9, 115.1 (d, J = 21.2 Hz), 116.7, 121.8, 124.0, 130.4 (d, J = 3.2 Hz), 131.5, 133.6 (d, J = 8.1 Hz), 139.4, 156.8, 161.8 (d, J = 243.0 Hz), 162.4. HRMS-ESI: [M+H]+ calcd for C16H13FNO2+ 270.0925, found 270.0931.
3-(4-Chlorophenyl)-4-hydroxy-1-methylquinolin-2(1H)-one (1e) [42]. Compound 1e (1.24 g, 87%) was prepared from N-methylisatoic anhydride (0.89 g, 5 mmol), methyl 4-chlorophenylacetate (0.92 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 279–281 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.60 (s, 3H), 7.28 (t, J = 7.5 Hz, 1H), 7.37 (d, J = 8.4 Hz, 2H), 7.45 (d, J = 8.4 Hz, 2H), 7.51 (d, J = 8.4 Hz, 1H), 7.65 (t, J = 7.3 Hz, 1H), 8.05 (d, J = 7.5 Hz, 1H), 10.21 (br. S, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 29.7, 111.5, 114.9, 116.7, 121.9, 124.1, 128.2, 131.6, 132.1, 133.1, 133.5, 139.5, 156.9, 162.2. HRMS-ESI: [M+H]+ calcd for C16H1335ClNO2+ 286.0629, found 286.0639.
4-Hydroxy-1-methyl-3-(4-nitrophenyl)quinolin-2(1H)-one (1f) [42]. Compound 1f (0.77 g, 52%) was prepared from N-methylisatoic anhydride (0.89 g, 5 mmol), methyl 4-nitrophenylacetate (0.98 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 339–340 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.62 (s, 3H), 7.32 (t, J = 7.5 Hz, 1H), 7.56 (d, J = 8.5 Hz, 1H), 7.67–7.71 (m, 3H), 8.10 (d, J = 7.9 Hz, 1H), 8.26 (d, J = 8.5 Hz, 2H), 10.61 (br. s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 29.3, 110.9, 114.5, 116.4, 121.5, 122.6, 123.9, 131.6, 132.6, 139.5, 141.8, 146.5, 157.3, 161.5. HRMS-ESI: [M+H]+ calcd for C16H13N2O4+ 297.0870, found 297.0885.
4-Hydroxy-1-methyl-3-(thiophen-2-yl)quinolin-2(1H)-one (1g) [42]. Compound 1g (0.71 g, 55%) was prepared from N-methylisatoic anhydride (0.89 g, 5 mmol), methyl (thiophen-2-yl)acetate (0.78 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 175–176 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.68 (s, 3H), 7.13 (dd, J = 5.1 and 3.8 Hz, 1H), 7.33 (t, J = 7.2 Hz, 1H), 7.52 (dd, J = 5.2 and 1.0 Hz, 1H), 7.56 (d, J = 8.3 Hz, 1H), 7.64–7.68 (m, 1H), 8.01 (dd, J = 3.7 and 1.0 Hz, 1H), 8.17 (dd, J = 8.1 and 1.0 Hz, 1H), 11.08 (br. s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 30.1, 107.0, 115.1, 116.4, 122.1, 123.9, 126.2, 126.6, 128.7, 131.5, 135.1, 138.4, 156.4, 161.6. HRMS-ESI: [M+H]+ calcd for C14H12NO2S+ 258.0583, found 258.0595.
5-Chloro-4-hydroxy-1-methyl-3-phenylquinolin-2(1H)-one (1h) [42]. Compound 1h (1.05 g, 74%) was prepared from N-methyl-5-chloroisatoic anhydride (1.06 g, 5 mmol), methyl phenylacetate (0.75 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 208–209 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.61 (s, 3H), 7.31–7.57 (m, 8H), 9.78 (s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 30.7, 114.3, 114.6, 114.8, 125.7, 127.8, 128.5, 131.1 (2C), 131.7, 133.6, 141.8, 157.0, 161.6. HRMS-ESI: [M+H]+ calcd for C16H1335ClNO2+ 286.0629, found 286.0636.
6-Chloro-4-hydroxy-1-methyl-3-phenylquinolin-2(1H)-one (1i) [42]. Compound 1i (0.97 g, 68%) was prepared from N-methyl-6-chloroisatoic anhydride (1.06 g, 5 mmol), methyl phenylacetate (0.75 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 265–267 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.59 (s, 3H), 7.31–7.35 (m, 3H), 7.39–7.43 (m, 2H), 7.53 (d, J = 9.1 Hz, 1H), 7.65 (dd, J = 9.0 and 2.4 Hz, 1H), 8.02 (d, J = 2.4 Hz, 1H), 10.28 (br. s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 29.9, 113.8, 117.0, 118.2, 123.1, 126.2, 127.6, 128.2, 131.0, 131.5, 133.8, 138.2, 155.6, 162.2. HRMS-ESI: [M+H]+ calcd for C16H1335ClNO2+ 286.0629, found 286.0640.
7-Chloro-4-hydroxy-1-methyl-3-phenylquinolin-2(1H)-one (1j) [42]. Compound 1j (1.0 g, 70%) was prepared from N-methyl-7-chloroisatoic anhydride (1.06 g, 5 mmol), methyl phenylacetate (0.75 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 230–231 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.58 (s, 3H), 7.29–7.35 (m, 4H), 7.39–7.43 (m, 2H), 7.57 (d, J = 1.7 Hz, 1H), 8.02 (d, J = 8.6 Hz, 1H), 10.22 (s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 29.9, 113.0, 114.5, 115.7, 121.8, 125.9, 127.6, 128.3, 131.6, 133.8, 136.1, 140.4, 156.2, 162.4. HRMS-ESI: [M+H]+ calcd for C16H1335ClNO2+ 286.0629, found 286.0638.
8-Chloro-4-hydroxy-1-methyl-3-phenylquinolin-2(1H)-one (1k) [42]. Compound 1k (0.87 g, 61%) was prepared from N-methyl-8-chloroisatoic anhydride (1.06 g, 5 mmol), methyl phenylacetate (0.75 g, 5 mmol), and KHDMS (1.99 g, 10 mmol) as a colorless solid. Mp: 238–239 °C (MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.77 (s, 3H), 7.25 (t, J = 7.9 Hz, 1H), 7.31–7.43 (m, 5H), 7.69 (d, J = 7.7 Hz, 1H), 8.02 (d, J = 7.8 Hz, 1H), 10.32 (br. S, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 37.3, 113.0, 119.8, 121.0, 123.1, 123.6, 127.6, 128.3, 131.5, 133.7, 134.4, 137.8, 156.5, 164.2. HRMS-ESI: [M+Na]+ calcd for C16H1235ClNNaO2+ 308.0449, found 308.0461.
3.3. Synthesis of Pyrrolo[3,2-c]quinolin-4-ones 3
General procedure. Quinolone 1a–m (0.2 mmol), Cu(Oac)2·H2O (2 mg, 0.01 mmol), azirine 2 (0.32 mmol), and MeOH (3 mL) (or DCE for compound 3p) were placed consequently into a screw-cap glass tube and heated at 100 °C for 15–20 min under stirring until full consumption of the quinolinone 1 was detected (control by TLC). The solvent was removed in vacuo and the residue was purified by column chromatography on silica gel (eluent hexane/EtOAc from 2:1 to 1:2) followed by recrystallization from hexane/CHCl3 to produce adduct 3 as pure crystals (compounds 3c and 3l) or as solvates with chloroform (compounds 3a,b,d–k,m–p).
rac-(3aR,9bR)-9b-Hydroxy-3a-(4-methoxyphenyl)-5-methyl-2-(4-methylphenyl)-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3a). Colorless solid (3a × 0.48CHCl3, 91 mg, 97%). Mp: 144–145 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.36 (s, 3H), 3.25 (s, 3H), 3.67 (s, 3H), 3.78 (d, J = 16.1 Hz, 1H), 4.27 (d, J = Hz, 1H), 6.17 (s, 1H), 6.78 (d, J = 8.8 Hz, 2H), 7.12 (d, J = 8.8 Hz, 2H), 7.20 (d, J = 8.1 Hz, 1H), 7.25–7.30 (m, 3H), 7.43–7.48 (m, 1H), 7.83 (d, J = 8.1 Hz, 2H), 7.96 (dd, J = 7.6 and 1.3 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.5, 44.2, 55.4, 60.9, 97.5, 113.8, 115.0, 123.7, 127.2, 128.4 (2C), 129.4, 129.7, 129.9, 130.0, 131.3, 138.3, 142.0, 158.7, 171.5, 175.6. HRMS-ESI: [M+H]+ calcd for C26H25N2O3+ 413.1860, found 413.1858.
rac-(3aR,9bR)-9b-Hydroxy-5-methyl-2,3a-di(4-methylphenyl)-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3b). Colorless solid (3b × 1.47CHCl3, 112 mg, 98%). Mp: 178–179 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.21 (s, 3H), 2.36 (s, 3H), 3.25 (s, 3H), 3.82 (d, J = 16.1 Hz, 1H), 4.29 (d, J = 16.1 Hz, 1H), 6.19 (s, 1H), 7.02 (d, J = 7.6 Hz, 2H), 7.10 (d, J = 7.6 Hz, 2H), 7.19 (d, J = 8.0 Hz, 1H), 7.28–7.30 (m, 3H), 7.45 (t, J = 7.2 Hz, 1H), 7.85 (d, J = 7.6 Hz, 2H), 7.98 (d, J = 7.2 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.0, 21.5, 30.5, 44.1, 61.3, 97.5, 115.0, 123.6, 127.2, 128.2, 128.4 (2C), 128.9, 129.7, 129.9, 131.3, 135.0, 136.6, 138.3, 142.0, 171.4, 175.6. HRMS-ESI: [M+H]+ calcd for C26H25N2O2+ 397.1911, found 397.1914.
rac-(3aR,9bR)-9b-Hydroxy-5-methyl-2-(4-methylphenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3c). Pale-yellow solid (76 mg, 99%). Mp: 165–167 °C (CHCl3/Et2O). 1H NMR (400 MHz, CDCl3), δ, ppm: 2.44 (s, 3H), 3.33 (br. S, 3H), 3.53–3.57 (m, 1H), 4.11 (br. S, 1H), 4.27–4.29 (m, 1H), 7.10–7.49 (m, 10H), 7.44–7.53 (m, 1H), 7.67–7.80 (m, 1H), 8.06–8.09 (m, 1H). 13C NMR (100 MHz, CDCl3), δ, ppm: 21.5, 30.4, 44.1, 61.4, 97.7, 114.4, 123.8, 127.0, 127.6 (2C), 128.4 (3C), 129.1, 129.9, 130.1, 136.6, 138.0, 142.5, 170.9, 177.6. HRMS-ESI: [M+H]+ calcd for C25H23N2O2+ 383.1754, found 383.1748.
rac-(3aR,9bR)-3a-(4-Fluorophenyl)-9b-hydroxy-5-methyl-2-(4-methylphenyl)-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3d). Colorless solid (3d × 0.56CHCl3, 91 mg, 98%). Mp: 148–149 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.36 (s, 3H), 3.26 (s, 3H), 3.82 (d, J = 16.2 Hz, 1H), 4.30 (d, J = 16.2 Hz, 1H), 6.29 (s, 1H), 7.06 (t, J = 8.9 Hz, 2H), 7.21–7.30 (m, 6H), 7.44–7.49 (m, 1H), 7.84 (d, J = 8.1 Hz, 2H), 7.97 (dd, J = 7.6 and 1.4 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.6, 44.5, 61.0, 97.5, 115.1 (d, J = 21.1 Hz), 115.2, 123.8, 127.2, 128.2, 128.4, 129.7, 130.0, 130.3 (d, J = 8.0 Hz), 131.1, 134.3 (d, J = 3.1 Hz), 138.1, 142.1, 161.7 (d, J = 243.7 Hz), 171.1, 175.5. HRMS-ESI: [M+H]+ calcd for C25H22FN2O2+ 401.1660, found 401.1655.
rac-(3aR,9bR)-3a-(4-Chlorophenyl)-9b-hydroxy-5-methyl-2-(4-methylphenyl)-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3e). Colorless solid (3e × 0.45CHCl3, 93 mg, 99%). Mp: 202–203 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.35 (s, 3H), 3.27 (s, 3H), 3.84 (d, J = 16.2 Hz, 1H), 4.29 (d, J = 16.2 Hz, 1H), 6.33 (s, 1H), 7.21–7.31 (m, 8H), 7.44–7.48 (m, 1H), 7.84 (d, J = 8.1 Hz, 2H), 7.97 (dd, J = 7.6 and 1.4 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.7, 44.4, 61.2, 97.5, 115.2, 123.9, 127.2, 128.1, 128.4 (2C), 129.7, 130.0, 130.2, 131.1, 132.3, 137.0, 138.1, 142.2, 170.9, 175.4. HRMS-ESI: [M+H]+ calcd for C25H2235ClN2O2+ 417.1364, found 417.1369.
rac-(3aR,9bR)-9b-Hydroxy-5-methyl-2-(4-methylphenyl)-3a-(4-nitrophenyl)-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3f). Colorless solid (3f × 0.39CHCl3, 80 mg, 85%). Mp: 186–187 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.36 (s, 3H), 3.29 (s, 3H), 3.93 (d, J = 16.3 Hz, 1H), 4.33 (d, J = 16.3 Hz, 1H), 6.48 (s, 1H), 7.25–7.32 (m, 4H), 7.46–7.51 (m, 3H), 7.84 (d, J = 8.1 Hz, 2H), 7.96 (dd, J = 7.7 and 1.4 Hz, 1H), 8.11 (d, J = 8.9 Hz, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.8, 44.6, 61.9, 97.7, 115.4, 123.5, 124.1, 127.3, 127.8, 128.5, 129.7, 129.8, 130.2, 130.9, 137.9, 142.3, 145.5, 147.0, 170.2, 175.2. HRMS-ESI: [M+H]+ calcd for C25H22N3O4+ 428.1605, found 428.1608.
rac-(3aR,9bR)-9b-Hydroxy-5-methyl-2-(4-methylphenyl)-3a-(thiophen-2-yl)-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3g). Colorless solid (3g × 0.49CHCl3, 82 mg, 92%). Mp: 112–114 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.36 (s, 3H), 3.28 (s, 3H), 3.77 (d, J = 15.9 Hz, 1H), 4.28 (d, J = 15.9 Hz, 1H), 6.35 (s, 1H), 6.82 (d, J = 3.1 Hz, 1H), 6.87–6.89 (m, 1H), 7.21–7.32 (m, 5H), 7.46 (t, J = 7.2 Hz, 1H), 7.81 (d, J = 8.0 Hz, 2H), 7.93 (d, J = 7.5 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.7, 45.0, 59.2, 97.6, 115.1, 123.8, 125.7, 126.3, 126.6, 127.3, 127.8, 128.4, 129.7, 130.1, 131.0, 138.2, 139.7, 142.2, 170.0, 176.1. HRMS-ESI: [M+H]+ calcd for C23H21N2O2S+ 389.1318, found 389.1333.
rac-(3aR,9bS)-9-Chloro-9b-hydroxy-5-methyl-2-(4-methylphenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3h). Colorless solid (3h × 0.60CHCl3, 78 mg, 80%). Mp: 146–148 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.37 (s, 3H), 3.25 (s, 3H), 3.73 (d, J = 16.4 Hz, 1H), 4.29 (d, J = 16.4 Hz, 1H), 5.86 (s, 1H), 7.21–7.33 (m, 9H), 7.44 (t, J = 8.2 Hz, 1H), 7.87 (d, J = 8.1 Hz, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.6, 31.6, 43.2, 62.5, 98.4, 114.8, 125.5, 126.9, 127.4, 128.4, 128.5 (2C), 129.7, 130.5, 131.0, 134.3, 138.7, 141.0, 142.3, 170.8, 176.2. HRMS-ESI: [M+H]+ calcd for C25H2235ClN2O2+ 417.1364, found 417.1372.
rac-(3aR,9bR)-8-Chloro-9b-hydroxy-5-methyl-2-(4-methylphenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3i). Colorless solid (3i × 0.67CHCl3, 98 mg, 99%). Mp: 124–126 °C (CHCl3/hexane). Compound 3i can be obtained as a solvate with DMSO-d6 by evaporation solution in DMSO-d6 with identical NMR spectra. 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.36 (s, 3H), 3.25 (s, 3H), 3.86 (d, J = 16.3 Hz, 1H), 4.27 (d, J = 16.3 Hz, 1H), 6.41 (s, 1H), 7.17–7.31 (m, 8H), 7.52 (dd, J = 8.7 and 2.6 Hz, 1H), 7.84 (d, J = 8.1 Hz, 2H), 7.91 (d, J = 2.6 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.7, 44.2, 61.5, 97.1, 117.2, 126.8, 127.6, 128.0, 128.3, 128.5 (2C), 129.6, 129.7, 130.5, 131.0, 137.2, 137.6, 142.3, 171.1, 176.1. HRMS-ESI: [M+H]+ calcd for C25H2235ClN2O2+ 417.1364, found 417.1376.
rac-(3aR,9bR)-7-Chloro-9b-hydroxy-5-methyl-2-(4-methylphenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3j). Colorless solid (3j × 0.47CHCl3, 90 mg, 95%). Mp: 120–121 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.36 (s, 3H), 3.27 (s, 3H), 3.88 (d, J = 16.2 Hz, 1H), 4.29 (d, J = 16.2 Hz, 1H), 6.36 (s, 1H), 7.22–7.36 (m, 9H), 7.85 (d, J = 7.7 Hz, 2H), 7.96 (d, J = 8.2 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.5, 30.7, 44.2, 61.6, 97.2, 115.1, 123.5, 127.3, 127.6, 128.3, 128.4, 128.5, 128.9, 129.7, 131.0, 134.4, 137.7, 139.6, 142.2, 171.5, 175.8. HRMS-ESI: [M+H]+ calcd for C25H2235ClN2O2+ 417.1364, found 417.1374.
rac-(3aR,9bR)-6-Chloro-9b-hydroxy-5-methyl-2-(4-methylphenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3k). Colorless solid (3k × 0.68CHCl3, 98 mg, 99%). Mp: 170–171 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.37 (s, 3H), 3.29 (s, 3H), 3.79 (d, J = 15.9 Hz, 1H), 4.31 (d, J = 15.9 Hz, 1H), 6.50 (s, 1H), 7.13–7.34 (m, 8H), 7.55 (d, J = 7.4 Hz, 1H), 7.85–7.90 (m, 3H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.6, 38.1, 44.7, 62.3, 97.2, 122.1, 126.1, 126.2, 127.6, 128.2, 128.4, 128.5, 129.7, 131.2, 132.3, 134.1, 136.2, 137.2, 142.3, 173.3, 177.4. HRMS-ESI: [M+H]+ calcd for C25H2235ClN2O2+ 417.1364, found 417.1372.
rac-(3aR,9bR)-5-Benzyl-9b-hydroxy-2-(4-methylphenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3l). Colorless solid (87 mg, 95%). Mp: 173–175 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.38 (s, 3H), 3.92 (d, J = 16.0 Hz, 1H), 4.34 (d, J = 16.0 Hz, 1H), 5.17 (s, 2H), 6.33 (s, 1H), 6.91–6.93 (m, 2H), 7.07–7.12 (m, 4H), 7.21–7.34 (m, 9H), 7.88 (d, J = 8.1 Hz, 2H), 7.98 (dd, J = 7.6 and 1.5 Hz, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.6, 44.2, 45.3, 61.9, 97.4, 115.7, 123.9, 126.5, 127.2 (2C), 127.6, 128.4 (2C), 128.5, 128.8, 129.0, 129.7, 129.8, 131.4, 137.1, 137.2, 137.8, 142.1, 171.8, 176.1. HRMS-ESI: [M+Na]+ calcd for C31H26N2NaO2+ 481.1886, found 481.1896.
rac-(8aR,11aR)-11a-Hydroxy-10-(4-methylphenyl)-8a-phenyl-5,6,9,11a-tetrahydro-4H-pyrido[3,2,1-ij]pyrrolo[3,2-c]quinolin-8(8aH)-one (3m). Colorless solid (3m × 1.20CHCl3, 106 mg, 96%). Mp: 187–188 °C (CHCl3/MeOH). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 1.79 (s, 2H), 2.36 (s, 3H), 2.81 (br. S, 2H), 3.43–3.45 (m, 1H), 3.85 (d, J = 16.1 Hz, 1H), 3.99–4.02 (m, 1H), 4.29 (d, J = 16.1 Hz, 1H), 6.15 (s, 1H), 7.15–7.30 (m, 9H), 7.78–7.86 (m, 3H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.1, 21.6, 27.5, 42.3, 44.0, 61.3, 97.6, 123.2, 125.0, 125.3, 127.4, 128.1, 128.3, 128.4 (2C), 129.7, 130.2, 131.3, 134.0, 138.2, 142.0, 170.6, 175.4. HRMS-ESI: [M+H]+ calcd for C27H25N2O2+ 409.1911, found 409.1916.
rac-(3aR,9bR)-9b-Hydroxy-5-methyl-2,3a-diphenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3n). Colorless solid (3n × 0.26CHCl3, 79 mg, 99%). Mp: 174–175 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.27 (s, 3H), 3.89 (d, J = 16.2 Hz, 1H), 4.32 (d, J = 16.2 Hz, 1H), 6.26 (s, 1H), 7.22–7.31 (m, 7H), 7.45–7.55 (m, 4H), 7.95–7.98 (m, 3H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 30.6, 44.2, 61.6, 97.6, 115.1, 123.8, 127.1, 127.5, 128.3, 128.4 (3C), 129.1, 130.0, 132.1, 133.8, 138.0, 138.2, 171.2, 175.7. HRMS-ESI: [M+H]+ calcd for C24H21N2O2+ 369.1598, found 369.1592.
rac-(3aR,9bR)-9b-Hydroxy-2-(4-methoxyphenyl)-5-methyl-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3o). Colorless solid (3o × 0.51CHCl3, 83 mg, 91%). Mp: 99–100 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.26 (s, 3H), 3.78–3.82 (m, 4H), 4.26 (d, J = 16.1 Hz, 1H), 6.16 (s, 1H), 7.02 (d, J = 8.8 Hz, 2H), 7.19–7.29 (m, 7H), 7.44–7.48 (m, 1H), 7.88–7.94 (m, 3H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 30.6, 44.0, 55.9, 61.7, 97.4, 114.5, 115.1, 123.7, 126.5, 127.1, 127.4, 128.4 (2C), 128.5, 129.9, 130.2, 138.1, 138.2, 162.4, 171.3, 175.0. HRMS-ESI: [M+H]+ calcd for C25H23N2O3+ 399.1703, found 399.1693.
rac-(3aR,9bR)-9b-Hydroxy-5-methyl-2-(4-nitrophenyl)-3a-phenyl-3,3a,5,9b-tetrahydro-4H-pyrrolo[3,2-c]quinolin-4-one (3p). Compound 3p was prepared according to the general procedure using DCE as a solvent. Colorless solid (3p × 0.45CHCl3, 81 mg, 87%). Mp: 101–102 °C (CHCl3/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.27 (s, 3H), 3.96 (d, J = 16.5 Hz, 1H), 4.34 (d, J = 16.5 Hz, 1H), 6.40 (s, 1H), 7.21–7.31 (m, 7H), 7.47–7.51 (m, 1H), 7.95–7.98 (m, 1H), 8.20 (d, J = 8.8 Hz, 2H), 8.31–8.33 (m, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 30.7, 44.5, 61.7, 97.9, 115.2, 123.9, 124.3, 127.2, 127.6, 127.7, 128.3, 128.5, 129.7, 130.2, 137.6, 138.3, 139.3, 149.6, 171.0, 174.6. HRMS-ESI: [M+H]+ calcd for C24H20N3O4+ 414.1448, found 414.1440.
3.4. Synthesis of Chromenopyrroles 6
General procedure. Hydroxycoumarin 4 (0.2 mmol), Cu(Oac)2 × H2O (2 mg, 0.01 mmol), azirine 2 (0.32 mmol), and DCE (3.0 mL) were consequently placed into a screw-cap glass tube and heated at 100 °C for 20–30 min until the full consumption of 3-hydroxycoumarin 4 was detected (control by TLC). The solvent was removed in vacuo and the residue was purified by column chromatography on silica gel (eluent hexane/EtOAc from 2:1 to 1:2), followed by recrystallization from a hexane–Et2O mixture to produce compound 6.
Methyl rac-(3aR,9bR)-3a-hydroxy-2-(4-methylphenyl)-4-oxo-3a,4-dihydrochromeno[3,4-b]pyrrole-9b(1H)-carboxylate (6a). Colorless solid (47 mg, 67%). Mp: 203–204 °C (Et2O/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.32 (s, 3H), 3.59 (s, 3H), 4.02 and 3.88 (AB-q, J = 17.2 Hz, 2H), 7.11 (d, J = 8.1 Hz, 1H), 7.18 (t, J = 7.4 Hz, 1H), 7.26 (d, J = 7.9 Hz, 2H), 7.34 (t, J = 7.4 Hz, 1H), 7.46 (d, J = 7.5 Hz, 1H), 7.59 (s, 1H), 7.78 (d, J = 8.0 Hz, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.6, 44.6, 53.2, 60.8, 95.5, 117.4, 122.7, 125.8, 128.3, 128.7, 129.8, 130.0, 130.7, 143.3, 150.3, 166.2, 169.7, 179.8. HRMS-ESI: [M-H]− calcd for C20H16NO5− 350.1034, found 350.1039.
Methyl rac-(3aR,9bR)-3a-hydroxy-4-oxo-2-phenyl-3a,4-dihydrochromeno[3,4-b]pyrrole-9b(1H)-carboxylate (6b). Colorless solid (47 mg, 70%). Mp: 171–172 °C (Et2O/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.59 (s, 3H), 3.92 and 4.07 (AB-q, J = 17.3 Hz, 2H), 7.12 (dd, J = 8.2 and 1.0 Hz, 1H), 7.19 (td, J = 7.6 and 1.1 Hz, 1H), 7.32–7.36 (m, 1H), 7.43–7.48 (m, 3H), 7.54 (t, J = 7.4 Hz, 1H), 7.66 (s, 1H), 7.88–7.90 (m, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 44.7, 53.3, 60.9, 95.5, 117.4, 122.6, 125.8, 128.3, 128.7, 129.3, 130.7, 132.6, 133.0, 150.3, 166.1, 169.7, 180.1. HRMS-ESI: [M+H]+ calcd for C19H16NO5+ 338.1023, found 338.1039.
Methyl rac-(3aR,9bR)-3a-hydroxy-2-(4-methoxyphenyl)-4-oxo-3a,4-dihydrochromeno[3,4-b]pyrrole-9b(1H)-carboxylate (6c). Colorless solid (44 mg, 60%). Mp: 199–200 °C (Et2O/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.58 (s, 3H), 3.79 (s, 3H), 3.87 and 4.00 (AB-q, J = 17.1 Hz, 2H), 6.98 (d, J = 8.6 Hz, 2H), 7.11 (d, J = 8.1 Hz, 1H), 7.18 (t, J = 7.4 Hz, 1H), 7.34 (t, J = 7.5 Hz, 1H), 7.45 (d, J = 7.6 Hz, 1H), 7.52 (s, 1H), 7.84 (d, J = 8.6 Hz, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 44.5, 53.2, 55.9, 60.9, 95.4, 114.6, 117.4, 122.8, 125.3, 125.7, 128.3, 130.6, 130.7, 150.3, 163.1, 166.3, 169.8, 179.2. HRMS-ESI: [M+H]+ calcd for C20H18NO6+ 368.1129, found 368.1138.
Methyl rac-(3aR,9bR)-3a-hydroxy-2-(4-nitrophenyl)-4-oxo-3a,4-dihydrochromeno[3,4-b]pyrrole-9b(1H)-carboxylate (6d). Colorless solid (47 mg, 62%). Mp: 190–191 °C (Et2O/hexane). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 3.61 (s, 3H), 3.99 and 4.16 (AB-q, J = 17.5 Hz, 2H), 7.13 (dd, J = 8.2 and 1.0 Hz, 1H), 7.20 (td, J = 7.7 and 1.1 Hz, 1H), 7.34–7.38 (m, 1H), 7.47 (dd, J = 7.8 and 1.3 Hz, 1H), 7.86 (s, 1H), 8.13 (d, J = 8.9 Hz, 2H), 8.27 (d, J = 8.9 Hz, 2H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 45.1, 53.4, 61.0, 95.6, 117.5, 122.2, 124.3, 125.9, 128.4, 130.1, 130.9, 138.0, 150.1, 150.2, 165.8, 169.5, 178.9. HRMS-ESI: [M-H]− calcd for C19H13N2O7− 381.0728, found 381.0720.
3.5. Synthesis of Methyl Rac-(7aR,10aR)-7a-hydroxy-9-(4-methylphenyl)-7oxo-7a,10-dihydronaphtho[1,8-ef]indole-10a(7H)-carboxylate (8)
Enol 7 (51 mg, 0.2 mmol), Cu(hfacac)2 (10 mg, 0.02 mmol), azirine 2a (42 mg, 0.32 mmol), and DCE (3.0 mL) were consequently placed into a screw-cap glass tube and heated at 100 °C for 2.5 h until the full consumption of enol 7 was detected (control by TLC). The solvent was removed in vacuo and the residue was purified by column chromatography on silica gel (eluent hexane/EtOAc from 2:1 to 1:2). Yellow solid (46 mg, 60%). Mp: 208–209 °C (Et2O/hexane). 1H NMR (400 MHz, CDCl3–DMSO-d6 mixture), δ, ppm: 2.26 (s, 3H), 3.49 (s, 3H), 3.83 (d, J = 16.9 Hz, 1H), 4.32 (d, J = 16.9 Hz, 1H), 5.82 (s, 1H), 7.07 (d, J = 7.9 Hz, 2H), 7.48–7.52 (m, 1H), 7.60–7.64 (m, 4H), 7.80–7.82 (m, 1H), 8.08–8.10 (m, 1H), 8.32–8.34 (m, 1H). 13C NMR (100 MHz, CDCl3–DMSO-d6 mixture), δ, ppm 20.7, 46.0, 51.9, 63.0, 97.0, 125.4, 125.8, 125.9, 126.3, 127.1, 127.3, 127.6, 128.4, 129.5, 130.1, 132.3, 132.6, 134.4, 141.8, 169.9, 177.8, 193.3. HRMS-ESI: [M+H]+ calcd for C24H20NO4+ 386.1387, found 386.1393.
3.6. Synthesis of Methyl (2-(4-Methylphenyl)-4-oxo-2,3-dihydro-4H-thiochromeno[4,3-b]furan-2-yl)carbamate (15)
Enol 14 (60 mg, 0.25 mmol), Cu(acac)2 (3 mg, 0.01 mmol), azirine 2a (54 mg, 0.41 mmol), and DCE (3.0 mL) were consequently placed into a screw-cap glass tube and heated at 100 °C for 20 min until the full consumption of enol 14 was detected (control by TLC). The solvent was removed in vacuo and the residue was purified by column chromatography on silica gel (eluent hexane/EtOAc 4:1). Colorless solid (46 mg, 50%). Mp: 183–184 °C (Et2O/hexane). 1H NMR (400 MHz, CDCl3), δ, ppm: 2.38 (s, 3H), 3.46 (d, J = 16.1 Hz, 1H), 3.68 (s, 3H), 4.05 (d, J = 16.1 Hz, 1H), 6.04 (s, 1H), 7.23 (d, J = 7.8 Hz, 2H), 7.42–7.48 (m, 3H), 7.50–7.56 (m, 2H), 8.13 (d, J = 7.8 Hz, 1H). 13C NMR (100 MHz, CDCl3), δ, 21.0, 41.2, 52.3, 98.9, 111.7, 119.9, 124.3, 125.5, 125.9, 126.1, 129.6, 130.4, 138.9, 139.1, 139.5, 154.5, 164.8, 180.1. HRMS-ESI: [M+Na]+ calcd for C20H17NNaO4S+ 390.0770, found 390.0770.
3.7. Synthesis of 2-(4-Methylphenyl)-4H-thiochromeno[4,3-b]furan-4-one (16)
A solution of carbamate 15 (38 mg, 0.1 mmol) and anhydrous p-toluenesulfonic acid (2 mg, 0.01 mmol) in anhydrous o-xylene (2.0 mL) was refluxed for 15 min. The reaction mixture was diluted with EtOAc (10 mL), washed with 0.1M NaOH, and dried over Na2SO4. Following filtration and concentration under vacuum, the residue was purified by flash column chromatography on silica gel (eluent hexane/EtOAc 5:1) to produce compound 16. Colorless solid (25 mg, 83%). Mp: 172–173 °C (Et2O/hexane). 1H NMR (400 MHz, CDCl3), δ, ppm: 2.43 (s, 3H), 7.13 (s, 1H), 7.27–7.29 (m, 2H), 7.45–7.56 (m, 3H), 7.71 (d, J = 8.0 Hz, 2H), 8.22–8.25 (m, 1H). 13C NMR (100 MHz, CDCl3), δ, 21.4, 100.6, 120.6, 121.3, 123.3, 124.5, 125.6, 126.3, 126.6, 128.7, 129.7, 135.2, 139.3, 155.7, 157.5, 180.4. HRMS-ESI: [M+Na]+ calcd for C18H13NaO2S+ 293.0631, found 293.0633.
3.8. Synthesis of Methyl (2-(rac-(3aR,9bR)-9b-Hydroxy-2-(4-methylphenyl)-5-oxo-3,4,5,9b-tetrahydro-3aH-pyrrolo[3,2-c]isoquinolin-3a-yl)-1-(4-methylphenyl)vinyl)carbamate (18)
Enol 17 (44 mg, 0.2 mmol), IPrCuCl (5 mg, 0.01 mmol), azirine 2a (42 mg, 0.32 mmol), and MeOH (3.0 mL) were consequently placed into a screw-cap glass tube and heated at 100 °C for 30 min until the full consumption of enol 17 was detected (control by TLC). The solvent was removed in vacuo and the residue was purified by column chromatography on silica gel (eluent hexane/EtOAc from 2:1 to 1:2). Light-yellow oil (31 mg, 32%). 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.27 (s, 3H), 2.37 (s, 3H), 3.35 (d, J = 17.0 Hz, 1H), 3.42 (d, J = 17.0 Hz, 1H), 3.54 (s, 3H), 5.48 (s, 1H), 6.29 (s, 1H), 7.10 (d, J = 8.0 Hz, 2H), 7.27 (d, J = 8.1 Hz, 2H), 7.32 (d, J = 8.0 Hz, 2H), 7.44–7.48 (m, 1H), 7.64–7.67 (s, 1H), 7.78–7.80 (m, 3H), 7.92–7.95 (m, 1H), 8.74 (s, 1H), 9.16 (br. s, 1H). 13C NMR (100 MHz, DMSO-d6), δ, ppm: 21.2, 21.6, 51.4, 52.1, 79.0, 90.2, 115.9, 126.0, 126.6, 126.8, 127.5, 128.2, 128.3, 128.9, 129.7, 131.1, 132.8, 136.1, 137.6, 138.5, 141.9, 142.2, 154.8, 161.8, 175.6. HRMS-ESI: [M+Na]+ calcd for C29H27N3NaO4+ 504.1894, found 504.1897.
4. Conclusions
In conclusion, we described an effective one-step procedure for the [2+3] pyrroline annulation to six-membered non-aromatic enols with 3-aryl-2H azirines as annulation agents. The reaction could be catalyzed by both Cu(II) and Cu(I) compounds. It proceeded as a formal (2+3) cycloaddition via the N1–C2 azirine bond cleavage with high-atom economy and efficiency. The method can be applied to cyclic enols of the quinolin-2-one, 2H-chromen-2-one, and 1H-phenalen-1-one series. The reaction outcome can be rationalized from the reactivity of the aminide intermediate, which was obtained following the copper(I) enolate-induced azirine-ring opening.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules27175681/s1: X-ray diffraction experiments; NMR spectra of compounds 1, 3, 6, 8, 18 [50,51,52,53].
Author Contributions
Conceptualization, P.A.S. and M.S.N.; methodology, P.A.S., N.V.R. and A.F.K.; investigation, P.A.S.; writing—original draft preparation, P.A.S. and M.S.N.; writing—review and editing, N.V.R. and A.F.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Russian Science Foundation, grant number 20-13-00044.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
This research was conducted using resources from the Centre for Magnetic Resonance, the Research Centre for X-ray Diffraction Studies, and the Centre for Chemical Analysis and Materials of the Science Park of Saint Petersburg State University.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of compounds 3, 6, 8, 15–18 are available from the authors.
References
- Khlebnikov, A.F.; Novikov, M.S.; Rostovskii, N.V. Advances in 2H-azirine chemistry: A seven-year update. Tetrahedron 2019, 75, 2555–2624. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Doyle, A.G. The Chemistry of Transition Metals with Three-Membered Ring Heterocycles. Chem. Rev. 2014, 114, 8153–8198. [Google Scholar] [CrossRef] [PubMed]
- Smetanin, I.A.; Novikov, M.S.; Agafonova, A.V.; Rostovskii, N.V.; Khlebnikov, A.F.; Kudryavtsev, I.V.; Terpilowski, M.A.; Serebriakova, M.K.; Trulioff, A.S.; Goncharov, N.V. A novel strategy for the synthesis of thermally stable and apoptosis-inducing 2,3-dihydroazetes. Org. Biomol. Chem. 2016, 14, 4479–4487. [Google Scholar] [CrossRef] [PubMed]
- Pokhriyal, A.; Karki, B.S.; Kant, R.; Rastogi, N. Redox-Neutral 1,3-Dipolar Cycloaddition of 2H-Azirines with 2,4,6-Triarylpyrylium Salts under Visible Light Irradiation. J. Org. Chem. 2021, 86, 4661–4670. [Google Scholar] [CrossRef]
- Zhao, M.-N.; Ren, Z.-H.; Yang, D.-S.; Guan, Z.-H. Iron-Catalyzed Radical Cycloaddition of 2H-Azirines and Enamides for the Synthesis of Pyrroles. Org. Lett. 2018, 20, 1287–1290. [Google Scholar] [CrossRef]
- Zhao, M.-N.; Ning, G.-W.; Yang, D.-S.; Gao, P.; Fan, M.-J.; Zhao, L.-F. Nickel-catalyzed formal [3+2]-cycloaddition of 2H-azirines with 1,3-dicarbonyl compounds for the synthesis of pyrroles. Tetrahedron Lett. 2020, 61, 152319. [Google Scholar] [CrossRef]
- Zeng, T.-T.; Xuan, J.; Ding, W.; Wang, K.; Lu, L.-Q.; Xiao, W.-J. [3+2] Cycloaddition/Oxidative Aromatization Sequence via Photoredox Catalysis: One-Pot Synthesis of Oxazoles from 2H-Azirines and Aldehydes. Org. Lett. 2015, 17, 4070–4073. [Google Scholar] [CrossRef]
- Duan, X.; Yang, K.; Lu, J.; Kong, X.; Liu, N.; Ma, J. Base-Mediated Cascade Substitution–Cyclization of 2H-Azirines: Access to Highly Substituted Oxazoles. Org. Lett. 2017, 19, 3370–3373. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.; Otani, Y.; Ohwada, T. Base-Induced Transformation of 2-Acyl-3-alkyl-2H-azirines to Oxazoles: Involvement of Deprotonation-Initiated Pathways. J. Org. Chem. 2017, 82, 6313–6326. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Huang, J.; Yuan, C.; Wang, G.; Guo, D.; Wang, J. Switchable assembly of substituted pyrimidines and 2H-imidazoles via Cu(I)-catalysed ring expansion of 2-methoxyl-2H-azirines. Org. Chem. Front. 2022, 9, 3006–3011. [Google Scholar] [CrossRef]
- Angyal, A.; Demjén, A.; Wölfling, J.; Puskás, L.G.; Kanizsai, I. Acid-Catalyzed 1,3-Dipolar Cycloaddition of 2H-Azirines with Nitrones: An Unexpected Access to 1,2,4,5-Tetrasubstituted Imidazoles. J. Org. Chem. 2020, 85, 3587–3595. [Google Scholar] [CrossRef] [PubMed]
- Rossa, T.A.; Fantinel, M.; Bortoluzzi, A.J.; Sá, M.M. Multicomponent Synthesis of Structurally Diverse Imidazoles Featuring Azirines, Amines and Aldehydes. Eur. J. Org. Chem. 2018, 2018, 4171–4177. [Google Scholar] [CrossRef]
- Feng, F.-F.; Li, J.-K.; Liu, X.-Y.; Zhang, F.-G.; Cheung, C.W.; Ma, J.-A. General Synthesis of Tri-Carbo-Substituted N2-Aryl-1,2,3-triazoles via Cu-Catalyzed Annulation of Azirines with Aryldiazonium Salts. J. Org. Chem. 2020, 85, 10872–10883. [Google Scholar] [CrossRef]
- Sujatha, C.; Bhatt, C.S.; Ravva, M.K.; Suresh, A.K.; Namitharan, K. Copper-Catalyzed Ring-Expansion Cascade of Azirines with Alkynes: Synthesis of Multisubstituted Pyridines at Room Temperature. Org. Lett. 2018, 20, 3241–3244. [Google Scholar] [CrossRef]
- Khlebnikov, A.F.; Novikov, M.S.; Petrovskii, P.P.; Stoeckli-Evans, H. An Aza Cyclopropylcarbinyl-Homoallyl Radical Rearrangement–Radical Cyclization Cascade. Synthesis of Dibenzoimidazoazepine and Oxazepine Derivatives. J. Org. Chem. 2011, 76, 5384–5391. [Google Scholar] [CrossRef]
- Borra, S.; Chandrasekhar, D.; Adhikary, S.; Rasala, S.; Gokulnath, S.; Maurya, R.A. Visible-Light Driven Photocascade Catalysis: Union of N,N-Dimethylanilines and α-Azidochalcones in Flow Microreactors. J. Org. Chem. 2017, 82, 2249–2256. [Google Scholar] [CrossRef]
- Angyal, A.; Demjén, A.; Harmat, V.; Wölfling, J.; Puskás, L.G.; Kanizsai, I. 1,3-Dipolar Cycloaddition of Isatin-Derived Azomethine Ylides with 2H-Azirines: Stereoselective Synthesis of 1,3-Diazaspiro[bicyclo[3.1.0]hexane]oxindoles. J. Org. Chem. 2019, 84, 4273–4281. [Google Scholar] [CrossRef]
- Colin, A.R.; Risberg, E.; Somfai, P. Diastereoselective Lewis acid-catalysed [4+2] cycloadditions of 3-alkyl-, 3-aryl- and 3-carboxyl-2H-azirines: A route to aziridine containing azabicyclo[4.1.0]heptanes and azatricyclo[2.2.1.0]nonanes. Tetrahedron 2002, 58, 5983–5987. [Google Scholar] [CrossRef]
- Alves, M.J.; Lemos, A.; Rodriguez-Borges, J.E.; García-Mera, X.; Fortes, A.G. Ethyl 2-(diisopropoxyphosphoryl)-2H-azirine-3-carboxylate: Reactions with nucleophilic 1,3-dienes. Synthesis 2009, 2009, 3263–3266. [Google Scholar] [CrossRef]
- Ding, H.; Wang, Z.; Bai, S.; Lu, P.; Wang, Y. Rh-Catalyzed Conversion of 3-Diazoindolin-2-imines to 5H-Pyrazino[2,3-b]indoles with Photoluminescent Properties. Org. Lett. 2017, 19, 6514–6517. [Google Scholar] [CrossRef]
- Baek, Y.; Maeng, C.; Kim, H.; Lee, P.H. Regioselective Synthesis of Indolopyrazines through a Sequential Rhodium-Catalyzed Formal [3+3] Cycloaddition and Aromatization Reaction of Diazoindolinimines with Azirines. J. Org. Chem. 2018, 83, 2349–2360. [Google Scholar] [CrossRef] [PubMed]
- Ruvinskaya, J.O.; Rostovskii, N.V.; Filippov, I.P.; Khlebnikov, A.F.; Novikov, M.S. A novel approach to 5H-pyrazino[2,3-b]indoles via annulation of 3-diazoindolin-2-imines with 2H-azirines or 5-alkoxyisoxazoles under Rh(II) catalysis. Org. Biomol. Chem. 2018, 16, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Wu, M.-F.; Liao, J.-H.; Hong, B.-C. Formal [6+3] cycloaddition of fulvenes with 2H-azirine: A facile approach to the [2]pyrindines system. Tetrahedron Lett. 2004, 45, 1663–1666. [Google Scholar] [CrossRef]
- Wang, L.; Liu, C.; Li, L.; Wang, X.; Sun, R.; Zhou, M.-D.; Wang, H. Visible-Light-Promoted [3 + 2] Cycloaddition of 2H-Azirines with Quinones: Access to Substituted Benzo[f]isoindole-4,9-diones. Chin. J. Chem. 2022, 40, 719–724. [Google Scholar] [CrossRef]
- Cludius-Brandt, S.; Kupracz, L.; Kirschning, A. [3+2]-Cycloadditions of nitrile ylides after photoactivation of vinyl azides under flow conditions. Beilstein J. Org. Chem. 2013, 9, 1745–1750. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Wang, C.; Lai, H.; Wang, S.; Ni, H.; Yu, W.; Cao, P. The Fe(III)-catalyzed decarboxylative cycloaddition of β-ketoacids and 2H-azirines for the synthesis of pyrrole derivatives. Org. Chem. Front. 2020, 7, 3686–3691. [Google Scholar] [CrossRef]
- Rostovskii, N.V.; Sakharov, P.A.; Novikov, M.S.; Khlebnikov, A.F.; Starova, G.L. Cu(I)–NHC-Catalyzed (2+3)-Annulation of Tetramic Acids with 2H-Azirines: Stereoselective Synthesis of Functionalized Hexahydropyrrolo[3,4-b]pyrroles. Org. Lett. 2015, 17, 4148–4151. [Google Scholar] [CrossRef]
- Sakharov, P.A.; Rostovskii, N.V.; Khlebnikov, A.F.; Novikov, M.S. Annulation of five-membered cyclic enols with 3-aryl-2H-azirines: Catalytic versus non-catalytic cycloaddition. Tetrahedron 2017, 73, 4663–4670. [Google Scholar] [CrossRef]
- Sakharov, P.A.; Rostovskii, N.V.; Khlebnikov, A.F.; Panikorovskii, T.L.; Novikov, M.S. 2H-Azirines as C–C Annulation Reagents in Cu-Catalyzed Synthesis of Furo[3,2-c]quinolone Derivatives. Org. Lett. 2019, 21, 3615–3619. [Google Scholar] [CrossRef]
- Borra, S.; Chandrasekhar, D.; Newar, U.D.; Maurya, R.A. Access to 2,3-Fused Pyrroles via Visible Light Driven Coupling of α-Azidochalcones with 1/2-Naphthols, or 2-Hydroxy-1,4-Naphthoquinone. J. Org. Chem. 2019, 84, 1042–1052. [Google Scholar] [CrossRef]
- Mahto, P.; Shukla, K.; Das, A.; Singh, V.K. Organocatalytic asymmetric synthesis of pyrrolo[3,2-c]quinolines via a formal [3+2] cycloaddition-lactamization cascade reaction using a bifunctional squaramide catalyst. Tetrahedron 2021, 87, 132115. [Google Scholar] [CrossRef]
- Nieman, J.A.; Ennis, M.D. Enantioselective Synthesis of the Pyrroloquinoline Core of the Martinellines. Org. Lett. 2000, 2, 1395–1397. [Google Scholar] [CrossRef] [PubMed]
- Yugandar, S.; Misra, N.C.; Parameshwarappa, G.; Panda, K.; Ila, H. Reaction of Cyclic α-Oxoketene Dithioacetals with Methylene Isocyanides: A Novel Pyrrole Annulation–Ring-Expansion Domino Process. Org. Lett. 2013, 15, 5250–5253. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Liu, J.; Ding, K.; Liu, J.; Cai, Q. Copper-Catalyzed Tandem Reaction of Isocyanides with N-(2-Haloaryl)propiolamides for the Synthesis of Pyrrolo[3,2-c]quinolin-4-ones. J. Org. Chem. 2011, 76, 5346–5353. [Google Scholar] [CrossRef]
- Ma, D.; Xia, C.; Jiang, J.; Zhang, J. First Total Synthesis of Martinellic Acid, a Naturally Occurring Bradykinin Receptor Antagonist. Org. Lett. 2001, 3, 2189–2191. [Google Scholar] [CrossRef]
- Witherup, K.M.; Ransom, R.W.; Graham, A.C.; Bernard, A.M.; Salvatore, M.J.; Lumma, W.C.; Anderson, P.S.; Pitzenberger, S.M.; Varga, S.L. Martinelline and Martinellic Acid, Novel G-Protein Linked Receptor Antagonists from the Tropical Plant Martinella iquitosensis (Bignoniaceae). J. Am. Chem. Soc. 1995, 117, 6682–6685. [Google Scholar] [CrossRef]
- Heidempergher, F.; Pevarello, P.; Pillan, A.; Pinciroli, V.; Della Torre, A.; Speciale, C.; Marconi, M.; Cini, M.; Toma, S.; Greco, F.; et al. Pyrrolo[3,2-c]quinoline derivatives: A new class of kynurenine-3-hydroxylase inhibitors. Farmaco 1999, 54, 152–160. [Google Scholar] [CrossRef]
- Ohashi, T.; Oguro, Y.; Tanaka, T.; Shiokawa, Z.; Shibata, S.; Sato, Y.; Yamakawa, H.; Hattori, H.; Yamamoto, Y.; Kondo, S.; et al. Discovery of pyrrolo[3,2-c]quinoline-4-one derivatives as novel hedgehog signaling inhibitors. Bioorg. Med. Chem. 2012, 20, 5496–5506. [Google Scholar] [CrossRef]
- Grychowska, K.; Satała, G.; Kos, T.; Partyka, A.; Colacino, E.; Chaumont-Dubel, S.; Bantreil, X.; Wesołowska, A.; Pawłowski, M.; Martinez, J.; et al. Novel 1H-Pyrrolo[3,2-c]quinoline Based 5-HT6 Receptor Antagonists with Potential Application for the Treatment of Cognitive Disorders Associated with Alzheimer’s Disease. ACS Chem. Neurosci. 2016, 7, 972–983. [Google Scholar] [CrossRef]
- Ohki, S.; Yoshino, M. Synthesis of nitrogen-containing heterocyclic compounds through nitrilium salt. I. Reaction of nitriles with 2-(α-hydroxy-methyl, -ethyl, and isopropyl)cyclohexanol in the presence of acid. Chem. Pharm. Bull. 1969, 17, 2142–2150. [Google Scholar] [CrossRef] [Green Version]
- Maini, P.N.; Sammes, M.P. The synthesis and chemistry of azolenines. Part 8. The Paal-Knorr reaction with cyclic 2-(acylmethyl)-2-alkyl-l,3-diketones: Isolation of 1-acyl-1H-pyrroles. J. Chem. Soc. Perkin Trans. 1 1988, 161–168. [Google Scholar] [CrossRef]
- Coppola, G.M. An Efficient Synthesis of Arboricine. Synth. Commun. 1985, 15, 135–139. [Google Scholar] [CrossRef]
- Choppakatla, S.; Dachepally, A.K.; Bollikolla, H.B. Palladium-Catalyzed Double C–H Functionalization of 2-Aryl-1,3-Dicarbonyl Compounds: A Facile Access to Alkenylated Benzopyrans. Tetrahedron Lett. 2016, 57, 2488–2491. [Google Scholar] [CrossRef]
- Hortmann, A.G.; Robertson, D.A.; Gillard, B.K. A Convenient Procedure for the Preparation of 2-Arylazirines. J. Org. Chem. 1972, 37, 322–324. [Google Scholar] [CrossRef]
- Wang, Y.; Lei, X.; Tang, Y. Rh(II)-catalyzed cycloadditions of 1-tosyl 1,2,3-triazoles with 2H-azirines: Switchable reactivity of Rh-azavinylcarbene as [2C]- or aza-[3C]-synthon. Chem. Commun. 2015, 51, 4507–4510. [Google Scholar] [CrossRef]
- Eistert, B.; Selzer, H. Umsetzungen Einiger Diazoalkane Mit Isatin, N-Methyl-Isatin, Cumarandion Und Thionaphthenchinon. Chem. Ber. 1963, 96, 1234–1255. [Google Scholar] [CrossRef]
- Eistert, B.; Selzer, H. Ringerweiterung von Acenaphthenchinon Mit Diazoalkanen Unter Abfangen Der Produkte Als Enolate Bzw. Zinkchelate. Chem. Ber. 1963, 96, 314–319. [Google Scholar] [CrossRef]
- Kikionis, S.; McKee, V.; Markopoulos, J.; Igglessi-Markopoulou, O. A prominent C-acylation–cyclisation synthetic sequence and X-ray structure elucidation of benzothiopyranone derivatives. Tetrahedron 2008, 64, 5454–5458. [Google Scholar] [CrossRef]
- Gabriel, S.; Colman, J. Ueber Die Einwirkung von Natriumalkylaten Auf Phtalylglycinester Und Dessen Homologe. Ber. Dtsch. Chem. Ges. 1900, 33, 980–995. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated Space-Group and Crystal-Structure Determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).