1. Introduction
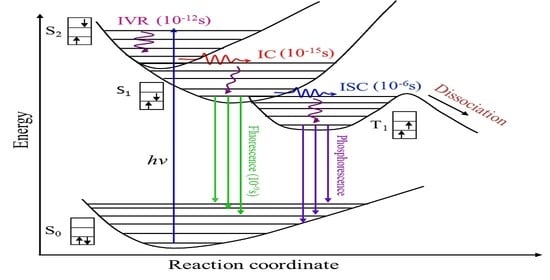
Molecules are excited to electronic excited states after absorbing ultraviolet light, and the excited state can rapidly initiate ultrafast non-adiabatic process with an energy flow, which can lead to some excited-state photochemical processes that are harmful to living organisms [
1,
2]. Intersystem crossing and internal conversion are the primary excited-state relaxation pathways essential for many biological processes, such as photosynthesis and DNA self-repair. Hence, it is important to study electron relaxation dynamics in these non-adiabatic processes [
3,
4]. Because molecules can be ionized from arbitrary excited states with various multiplicities, there is a need to observe the levels of dark states in real time via intersystem crossing (ISC) [
5]. Quantum-yield and fluorescence measurements can reveal non-adiabatic dynamical information. However, there is an urgent need for a comprehensive method to elucidate all of the dynamics.
Pyrazine aromatic hydrocarbon compounds are stable, highly toxic, carcinogenic, teratogenic, and widely distributed in nature [
6]. They are important in biological research, pharmaceutical and pesticide production, chemical synthesis, and environmental protection. They can be electronically excited with ultraviolet light, and the excited state can rapidly initiate ultrafast non-adiabatic process with an energy flow that is important in natural photophysical and photochemical processes. Related theoretical and experimental studies have focused on aromatic hydrocarbon compounds such as benzene and its derivatives. With the development of laser technology to explore chemical reactions on the femtosecond timescale, a new, extremely fast reaction channel has been observed in benzene. Radloff [
7] et al. studied the internal conversion of its excited S
2 state and observed that it decayed to the S
1 state in 40 ± 10 fs. The vibrationally excited S
1 state was then internally converted to the S
0 state in 6.7 ± 0.3 ps. Fielding [
8] et al. studied the channel-three effect of the S
1 state of benzene and found that when it was excited to a level 3000 cm
−1 above the S
1 state, approximately 20% of the excited-state population was transferred to the T
1 state. At the same time, approximately 6% of the population oscillated back and forth between the S
1 and T
2 states. They estimated that these processes all occurred within a few-hundred femtoseconds. After being excited to the S
2 state, benzene is rapidly converted to the S
1 state. The non-adiabatic transitions of S
1 to the S
0 or T
1 states generally occurs in nanoseconds, and the internal conversion and intersystem crossing compete with each other [
9]. Many chemical reactions occur in the femtosecond timescale [
10,
11,
12,
13,
14,
15]. The corresponding lifetimes of the S
2 and S
1 states are usually within 100 fs and between 4.3 and 8.8 ps, respectively, and there is a “channel-three” effect that plays a key role. (Because there is no standard kinetic process associated with it, Callomon [
16] et al. referred to this unknown process as “channel three”.) This effect refers to the region 2000 cm
−1 above the excited state. The lifetime in this region is much shorter than that of the S
1 state band. After molecules are excited to this region, the lifetime suddenly becomes much shorter because of intra-transitions or inter-system crossover; hence, the channel-three effect [
17].
The excited-state dynamics of nitrogen-containing aromatic hydrocarbon compounds, especially pyrazine, have attracted great attention [
18,
19,
20] with regard to their ultrafast and radiation-free dynamics [
21]. However, research on the dynamics of electronically excited 2-methylpyrazine has been less frequent. Here, because the dynamics in the channel-three region in 2-methylpyrazine is still controversial [
22,
23,
24], we performed time-resolved photoelectron imaging. The non-degeneracy of 2-methylpyrazine should lead to less symmetry prohibition and a higher vibrational level density. Therefore, we expected to observe non-adiabatic kinetics, and we investigated the channel-three effect of the S
1 state using femtosecond pump–probe photoelectron imaging. The Rydberg states were identified via photoelectron spectroscopy and angular distributions.
3. Results and Discussion
Since 2-methylpyrazine only reported the experimental value of the energy of the S
1 state, and no experimental and theoretical values were found for other excited states, density functional theory (DFT) was used with a B3PW91/6-311++G (d, p) basis set. Transition characteristics, vertical excitation energies (Eexc), and corresponding oscillator strengths (f) of various electronically excited states were calculated, as summarized in
Table 1. It is found that the oscillator strength of the S
2 state is very small, so it is believed that our pump light excites the high vibrational state of the S
1 state.
Experimentally, double-light and single-light mass spectrometries were performed. The pump and probe pulse energies were controlled so that there was almost no signal for a single light. At zero-time, the double-light mass spectrum had two peaks, and the stronger signal corresponded to the parent (C5H6N2+) ion. There were also weaker signals corresponding to ionized fragments of the parent ions. The calculated signal intensity of the parent ion was several factors of ten higher than that of the fragment ions, indicating that photoelectrons from the fragment ions were negligible. In addition, the intensities of the parent and fragment ion signals changed similarly with time. Therefore, most of the photoelectron signals were directly ionized from the parent ions.
Figure 1 is a parent (C
5H
6N
2+) ion signal as a function of time. It was fitted via a convolution of an exponentially rising and exponentially decaying functions and a Gaussian function. The convolution was necessary because in the femtosecond time-resolved experiment, both the pump and probe light pulses had Gaussian pulse widths, while the photoelectric conversion devices had certain response times. Thus, the recorded ion signal intensity was a convolution of the real signal of the excited-state layout number over time with the correlation function of the pump and probe light pulses. For example, a molecule could be excited to state A. However, because A is unstable, it will decay over time. That is, the A state will decay to another excited state, state B, and state B may also decay to the state C over time. The rate constant of the A-state decay can be denoted as g
1, and that of the B state to the C state can be denoted as g
2. Hence, an equation will have the form:
In the above equations, the variation in the number of layouts of excited states with time is a single-exponential decay and a single-exponential rise function. Because the femtosecond pump and probe pulses were Gaussian, the correlation function of the two Gaussian pulses was still Gaussian. In the experiment, the correlation function of the pump and the probe pulses was measured first, and then the mass-spectral signals under the measured pump and probe time delays were fitted by the convolution of the exponential and Gaussian functions to obtain the precise lifetime of the molecule in the excited state:
In Equation (6), is the amplitude, is the lifetime, is the full-width at the half-maximum of the correlation function, and is the zero-point time that represents the convolution operation.
As shown in
Figure 1, the time-resolved ion signal could be fitted by a convolution of an exponential rise, an exponential decay, and a Gaussian function to obtain an exponential rise and decay time of 23 ps. The 260 nm pump light excited the high vibrational dynamics of the S
1 state of 2-methylpyrazine. In pyrazine, the S
2 state is short-lived at less than 20 fs. We acquired the absorption spectrum of 2-methylpyrazine, which showed broadband absorption near the S
2 state. Therefore, we deduced that the S
2 state of 2-methylpyrazine was also short-lived. However, we did not observe this transient state in experiments. The pump–probe cross-correlation function was approximately 200 fs, which may have limited the time resolution of the short S
2 state lifetime. We considered the lifetime of the S
1 state after the transition from the excited S
2 state. The ion signal could be fitted by the convolution noted above with a decay of 23 ps, which was attributed to the decay of the S
1 state. A 23-ps rising signal was also observed with the decay of the S
1 state, which probably reflected the layout of the T
1 state. To better understand the decay process following the 260 nm pump pulse, we collected photoelectron images for different pump–probe time delays.
Figure 2 shows photoelectron images obtained for various pump–probe time delays. The upper row shows original images, which are projections of the 3D photoelectron distributions on the two-dimensional detector. The lower row shows the corresponding reconstructed 3D images after BASEX transformations. These images consisted of four rings, where the outer three rings disappeared at longer pump–probe time delays, while the innermost ring remained. To examine the decay processes more clearly, photoelectron spectra at different time delays were obtained from the photoelectron images.
We obtained photoelectron spectra at different time delays from the photoelectron images, as shown in
Figure 3. There were four peaks: the first three peaks decayed over time, while the fourth peak near 0 remained over time. The photoelectron images and corresponding photoelectron spectra at 44.5 ps and 279.5 ps are shown in
Figure 4. From the spectra, there were three photoelectron peaks at 44.5 ps. All three decayed over time, while the fourth peak appeared at 279.5 ps. Because the S
1 state had already decayed after 279.5 ps, the fourth peak was attributed to the ionization of the T
1 state. The S
1 state decayed at the same time as the T
1 layout. When pumping at 260 nm, intersystem crossing was still the main channel for S
1 decay. The T
1 lifetime in 2-methylpyrazine was likely to be similar to that in pyrazine measured in the time-resolved photoelectron imaging experiments of Wang et al. [
27]. We observed that the S
1 state decays to the T
1 state through the intersystem crossing process after being laid out. After S
1 is laid out, it is also possible to decay to the high vibrational state of the S
0 state through the internal conversion process. However, the high vibrational state of the S
0 state has a vibrational energy of 4.76 eV, and the 400 nm probe light cannot ionize such a high vibrational state of the ground state. Therefore, in our experiment, there is no way to observe the channel that converts S
1 to S
0, but we believe that the internal conversion from S
1 to S
0 after the S
1 layout is also an important channel.
In the photoelectron kinetic energy distributions in
Figure 3, all four peaks were sharp. Hence, the excited state of 2-methylpyrazine had an accidental resonance with a Rydberg state during ionization. The 2-methylpyrazine molecule absorbs probe photons to resonate with the Rydberg state. For example, the presence of the 3s and 3p Rydberg states observed in pyrazine and pyridazine enhanced (1 + 2’) resonant multiphoton ionization, and the angular distribution anisotropy of the photoelectrons generated from the 3s Rydberg state was stronger than that of the 3p Rydberg state. Therefore, we expected to observe similar behavior here. Ionization from the Rydberg state to the cationic state involved Δv = 0, and the photoelectron kinetic energy (PKE) could be expressed as [
28]:
where
IP is the ionization potential,
hω1 and
hω2, are the pump and probe photon energies, respectively,
TR is the Rydberg state vibrational energy,
n is the principal quantum number,
δ is the quantum defect, and
R is the Rydberg state constant (13.606 eV).
The four peaks in
Figure 4 are located at 0.95 eV, 0.72 eV, 0.22 eV, and 0.14 eV. From Equation (7), the quantum defects of the four Rydberg states are 0.48, 0.61, 0.83, and 0.87, respectively. Because the value of the quantum defect δ that generally corresponds to the s orbital of the Rydberg state is approximately 0.9–1.2, that for the p orbital is between 0.3 and 0.5, and that for the d orbital is 0. The value of the quantum defect also indicated that the fourth peak was likely to arise from an accidental resonance with the Rydberg state.
Table 2 lists the Rydberg state assignments and δ values of pyrazine and 2-methylpyrazine. By comparing these Rydberg state assignments for pyrazine and 2-methylpyrazine, the first and second peaks came from the 3p Rydberg state, the third peak was from the singlet (S
1) 3s Rydberg state, and the fourth peak was from the triplet (T
1) 3s Rydberg state.
Various photoelectron angular distributions reflect different molecular-orbital characteristics.
Figure 5 shows that the angular distributions of the different rings come from different intermediate states. In the atomic images, pure p-waves are generated from s-state ionization, and p-state ionization generates s- and d-waves. The interference of s- and d-waves will weaken the angular distribution anisotropy of the photoelectrons. Hence, in general, the photoelectron angular distribution anisotropy from the s state was stronger than that of the p state. The same holds true for Rydberg states in molecules with similar electronic configurations. Therefore, the photoelectron angular distributions were also a good means to understand the Rydberg states.
When exciting the high vibrational dynamics of the S1 state, we still observed intersystem crossover. After the S1 state was laid out, its attenuation to the triplet T1 state was still a very important channel. To study the competition between S1-to-T1 intersystem crossing and internal conversion from S1 to S0, we used 244 nm pump light to excite higher vibrational states in S1.
Figure 6 shows a signal from the 244 nm pump and 400 nm probe pulses fitted with a convolution of an exponential decay function and a Gaussian function, resulting in a decay time of 11 ps. When the pump light is 244 nm, we did not observe intersystem crossing because the decay of the S
1 state should have been an internal conversion process directly to the S
0 state.
As the wavelength gets shorter, the intersystem crossing process probably compete with the internal conversion process. When the pump light was 260 nm, we observed intersystem crossing from S
1 to T
1. Although we did not observe internal conversion from S
1 to S
0, it most likely occurred. The proportion of the intersystem crossing process appears to be smaller for the 260 nm pump than for the 323 nm pump [
31]. However, when the pump light is 244 nm, we did not observe intersystem crossing because the decay of the S
1 state should have been an internal conversion process directly to the S
0 state. Therefore, the high vibrational dynamics of the S
1 state had different decay processes for 260 nm and 244 nm pumping.
The kinetic process of the S
1 state of the 2-methylpyrazine molecule is summarized in
Figure 7. The high vibrational state of the S
1 state of the 2-methylpyrazine molecule is excited by the 260 nm pump light. When pumping at 260 nm, the first, second, and third peaks come from the ionization of the S
1 state, and the fourth peak comes from the ionization of the T
1 state. During the ionization process, the S
1 and T
1 states accidentally resonate with the Rydberg state. After the S
1 state is laid out, the attenuation channel of the S
1 state is the S
1→T
1 intersystem crossing and the S
1→S
0 internal conversion process.