Abstract
‘Seolhyang’ strawberry is harvested before it is fully ripened and treated with CO2 to extend the shelf-life. However, the volatile changes in the ‘Seolhyang’ strawberry after short-term CO2 treatment have not been investigated, although the volatile profile is an important quality attribute. Herein, we investigated the effect of short-term high CO2 treatment on the changes in the composition of volatile compounds in ‘Seolhyang’ strawberries at two ripening stages (i.e., half-red and bright-red) during cold storage using headspace solid-phase microextraction and gas chromatography-mass spectrometry. Furthermore, the effect of CO2 treatment on fruit quality with respect to the aroma was investigated. A total of 30 volatile compounds were identified. Storage increased the volatile compound concentrations, and the total concentration of volatiles in the CO2-treated strawberries was lower than that of the untreated strawberries during storage. The production of some characteristic strawberry volatiles (e.g., 4-methoxy-2,5-dimethyl-3(2H)-furanone) was inhibited in CO2-treated strawberries. However, CO2 treatment helped maintain the concentrations of hexanal and 2-hexenal, which are responsible for the fresh odor in strawberries. Interestingly, CO2 treatment suppressed the production of off-odor volatiles, acetaldehyde, and hexanoic acid during strawberry storage. Thus, short-term CO2 treatment may help maintain the fresh aroma of strawberries during cold storage.
1. Introduction
‘Seolhyang’ (Fragaria × ananassa Duch) strawberry was developed in 2005 by cross-breeding the ‘Akihime’ and ‘Red Pearl’ cultivars in Korea [1]. This cultivar, which has a sweet taste and a characteristic sweet aroma, accounted for 87.5% of the Korean strawberry production in 2017 [2]. The strawberry cultivar is exported from Korea to various Asian countries. In 2020, approximately 1602 and 1208 tons of strawberries were exported from Korea to Hong Kong and Singapore, respectively [3]. However, the ‘Seolhyang’ cultivar, owing to its soft texture, has a short shelf-life compared with other traditional strawberry cultivars, such as ‘Maehyang’ and ‘Keumhyang’ [4]. Thus, harvesting unripened strawberries and postharvest treatments may prolong the shelf-life of ‘Seolhyang’ strawberry to enable shipping over long distances [4].
The characteristic aroma of the strawberry influences consumer acceptance [5]. Volatile compounds are responsible for strawberry aroma, and the volatile composition of strawberries depends on the cultivar and agricultural practice (e.g., harvesting location) [6]. ‘Carezza’ strawberry cultivar contains higher concentrations of hexyl acetate (8.11 µg/kg) than those in the ‘Darselect’ and ‘Marmolada’ cultivars (3.48–4.10 µg/kg), which have higher methyl hexanoate and methyl butanoate concentrations than those in the ‘Carezza’ cultivar, despite being grown in the same region in Switzerland [7]. Furthermore, the γ-decalactone content of 10 strawberry cultivars (i.e., Rubygem, Fortuna, Festival, Calinda, FL 127, Plared, Sahara, Sabrina, Victory, and E-22) ranged from n.d. to 62.76 µg/kg depending on the cultivar [8]. Among the volatile compounds found in strawberries, 2,5-dimethyl-4-hydroxy-3(2H)-furanone, methyl butanoate, ethyl butanoate, ethyl hexanoate, and methyl butanoate are responsible for the characteristic volatile compounds of ‘Elsanta’ strawberry grown in Belgium [9].
Furthermore, the ripening stage also affects the volatile compound composition of strawberries, and the total volatiles usually increase after ripening [7]. Especially, butyl acetate, methyl 2-methyl butanoate, ethyl butanoate, propyl butanoate, isopropyl hexanoate, and 3-phenyl-1-propanol are produced during ripening [7]. Additionally, C6 aldehydes are formed when lipoxygenase and hydroperoxide lyase activity increase during strawberry ripening [10]. Moreover, the activity of other enzymes, such as alcohol acyltransferase in some cultivars, increases during ripening, affecting the volatile composition of strawberries [11]. When the fruits are wounded, the lipoxygenase and hydroperoxide lyase activity increase up to 1.2 and 2.0 fold, respectively, resulting in an increase in 3-hexenal and trans-2-hexanal production [12].
Additionally, the volatile composition of strawberries changes during storage. For example, β-citronellol, hexanoic acid, and ethyl butanoate concentrations increased 5.3, 11.6, and 5.6 folds, respectively, after 7 d of cold storage at 10 °C [13]. Postharvest treatment also affects the volatile composition of strawberries. A previous study reported that while some volatiles, such as octyl acetate, butyl benzene, and 1-methyl-2-propyl benzene were not detected in high O2 (90% O2/10% N2)-exposed strawberries, other volatiles, such as α-terpineol, were newly formed [14]. In addition, hexanal treatment during storage increased the alcohol percentage of strawberry volatiles at the end of their shelf-life [15]. Storage under high CO2 exposure is an effective postharvest treatment that maintains fruit quality [16]. Elevated CO2 was more effective in inhibiting strawberry aroma changes than elevated O2 [17]. ‘Diamante’ strawberries, stored under elevated CO2 (20 kpa), maintained their characteristic volatile profile for 11 d at 5 °C [16]. However, long-term CO2 treatment may produce an unpleasant aroma, color change, and a decrease in the sugar and organic acid concentrations of strawberries [18]. Thus, short-term CO2 treatment may be required to extend the shelf-life of strawberries without quality loss. In our previous study, short-term CO2-treated ‘Seolhyang’ strawberries at two ripening stages showed similar texture, color, and metabolite profiles with lower decay rates after cold storage compared with the control fruit [19]. However, the volatile changes in the ‘Seolhyang’ strawberry after short-term CO2 treatment have not been investigated, although the volatile profile is an important quality attribute of strawberries. To the best of our knowledge, this is the first study investigating changes in the volatile composition of ‘Seolhyang’ strawberries depending on the postharvest treatment (i.e., short-term high CO2 treatment), maturity, and storage conditions.
Short-term CO2 treatment may help maintaining the fresh aroma of strawberries during cold storage. In this study, the effect of short-term high CO2 treatment on the volatile composition of ‘Seolhyang’ strawberries was investigated at two ripening stages (half-red: 80% ripeness; bright-red: 100% ripeness) during 9 d of cold storage. These two ripening stages were chosen because unripened strawberries are harvested to prolong the shelf-life to be shipped over long distance, and ripened strawberries are also harvested to be shipped to short distance. In addition, ‘Seolhyang’ strawberries were chosen because the cultivar accounted for 87.5% of Korean strawberry production [2].
2. Results and Discussion
2.1. Identification of Volatile Compounds in Strawberries
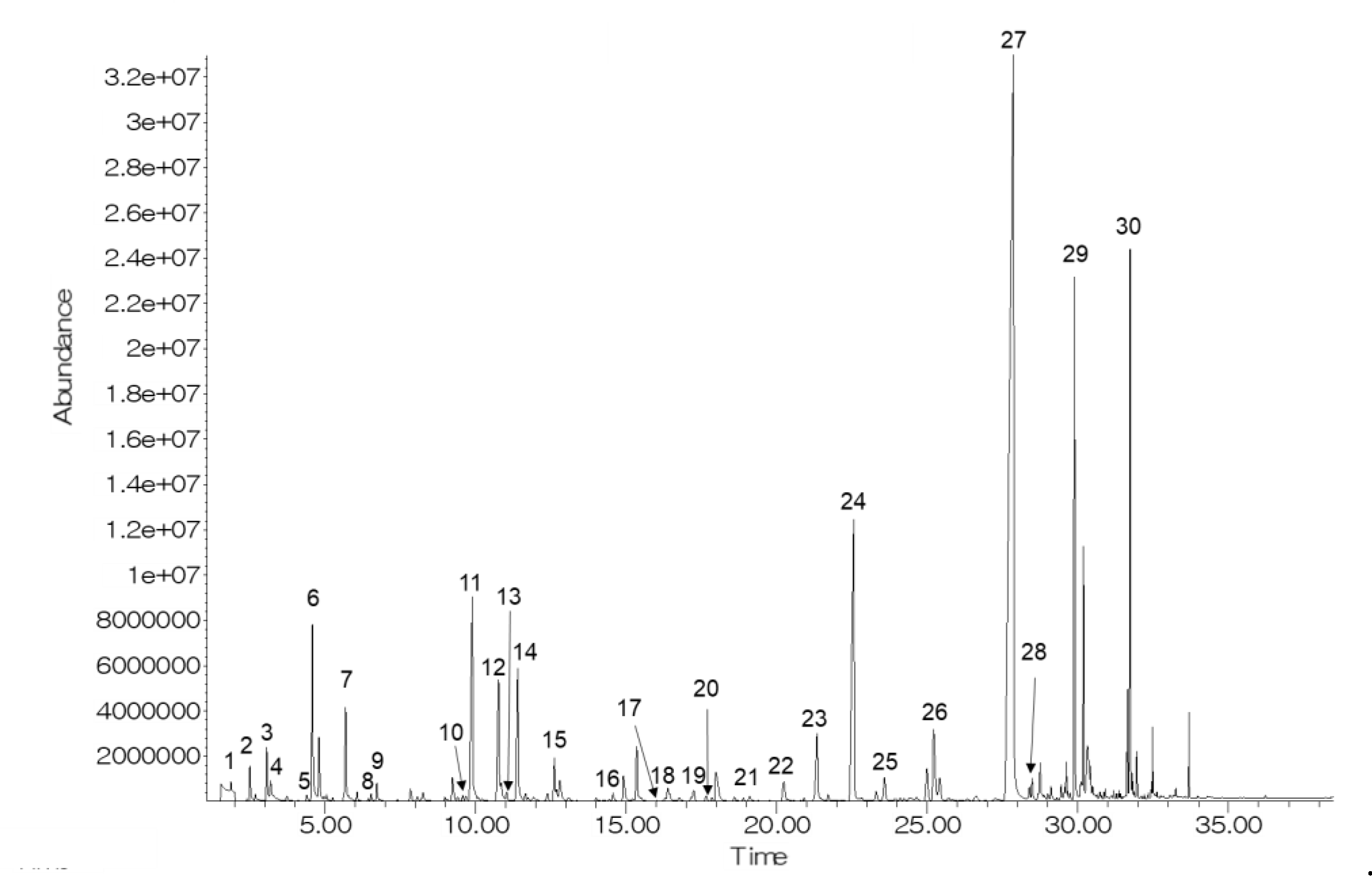
Figure 1 shows a representative GC chromatogram of CO2-treated bright-red strawberries after 9 d of storage. Thirty volatiles (5 aldehydes, 17 esters, 3 ketones, 2 alcohols, 2 acids, and 1 furanone) were identified (Table 1). Among the identified compounds, 11 volatile compounds (hexanal, 2-hexenal, nonanal, benzaldehyde, methyl acetate, methyl hexanoate, ethyl hexanoate, ethyl octanoate, 2-heptanone, butyric acid, and hexanoic acid) were identified by comparing them with the retention time and mass spectra of authentic standards. In previous studies, methyl butanoate, ethyl butanoate, methyl hexanoate, ethyl hexanoate, and 4-methoxy-2,5-dimethyl-3-(2H)-furanone were reported as characteristic volatiles of strawberries [20,21].
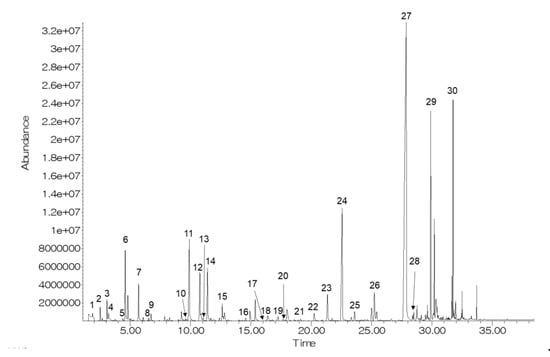
Figure 1.
A representative GC chromatogram of CO2-treated bright-red strawberries after 9 d of storage at 10 °C. Volatile compound codes are provided in Table 1.

Table 1.
Volatile compounds identified using HS-SPME-GC/MS in strawberries.
At day 0, some volatiles occurred or were present in higher concentrations only in the bright-red strawberries. For example, (2E)-hexenyl acetate was not present in half-red strawberries but was present in bright-red strawberries at day 0. (2E)-Hexenyl acetate was also reported in seven Fragaria × ananassa cultivars by Dong et al. [20]; however, it was not present in the ‘Camarosa’ and ‘Chandler’ strawberries harvested in India [22]. Thus, the volatile composition of strawberries may be affected by the cultivar type, maturity, and/or agricultural practices, such as the growing region. [23].
During cold storage, (2E)-hexenyl acetate and ethyl octanoate were newly produced in half-red and bright-red strawberries, respectively. The volatile composition of strawberries during cold storage was affected by the ripening stage when harvested. For example, ethyl octanoate was not found in fresh strawberries (day 0) during the two ripening stages; however, it was produced only in bright-red strawberries after cold storage, regardless of CO2 treatment. Previous studies found that ethyl octanoate was not present in strawberries of the ‘Cigaline’, ‘Chandler’, ‘Albion’, and ‘Festival’ cultivars [24,25,26]. Wang et al. reported that ethyl octanoate is formed after 5 d of storage in ‘Hong Yan’ strawberries [27]. The concentrations of some volatiles decreased or were absent after CO2 treatment. For example, the acetaldehyde content decreased, and 2-butanone was absent in bright-red strawberries after CO2 treatment.
2.2. Changes in the Composition of Volatile Compounds in Strawberries
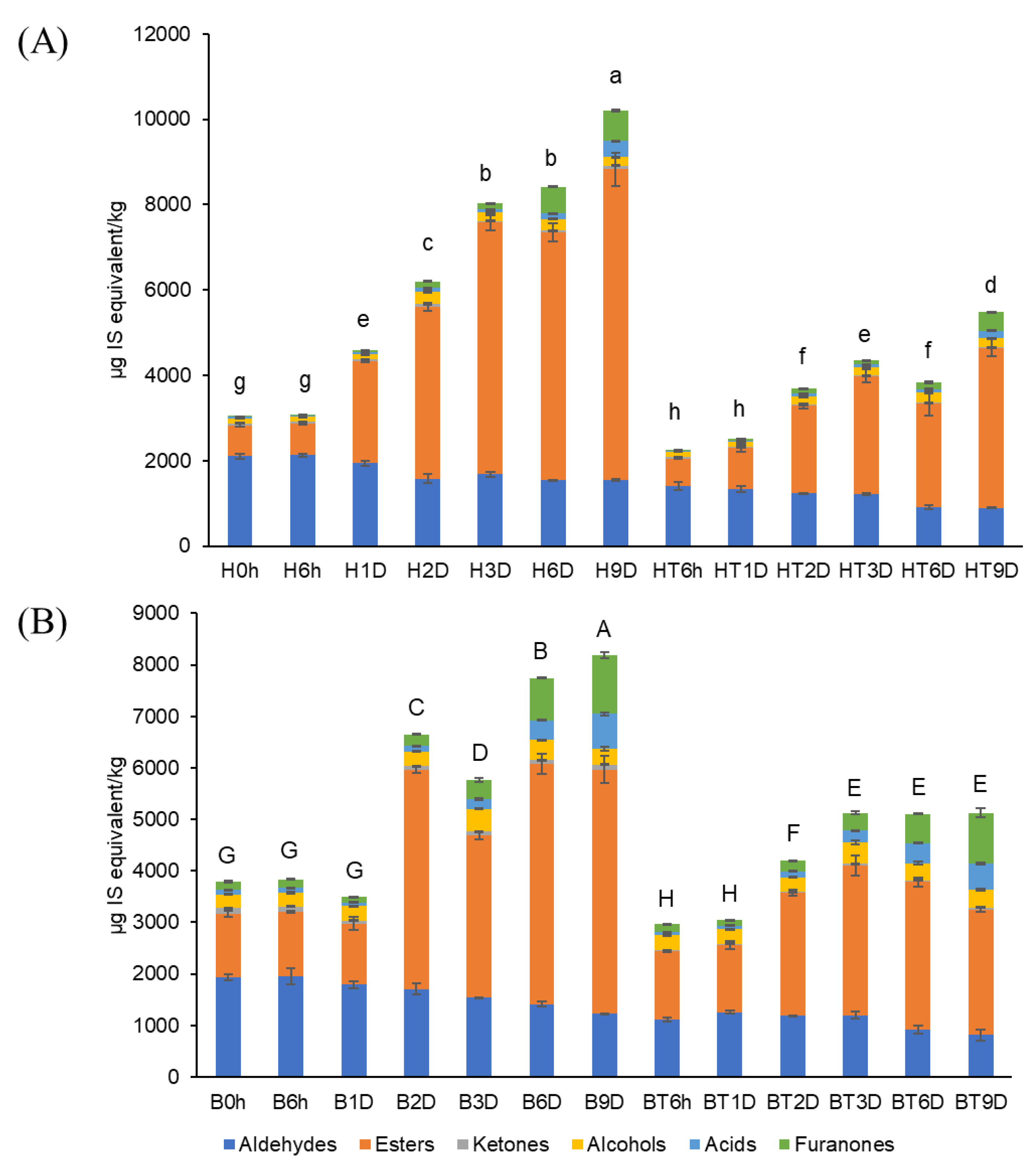
Figure 2 shows the sum of the volatile compounds of the strawberries. Before storage (0 d), the sum of the volatiles in bright-red strawberries (B0h, 3787 µg/kg) was 1.24-fold higher than that of the half-red strawberries (H0h, 3044 µg/kg). These findings agree with those of previous studies, showing that the sum of the volatiles in ‘Cigaline’ strawberries increased during maturity [25]. After 9 d of storage, the volatile concentrations of strawberries used as control increased to a greater extent (B9D, 8,186 µg/kg; H9D, 10,198 µg/kg) than those of CO2-treated strawberries (BT9D, 5130 µg/kg; HT9D, 5480 µg/kg). Thus, short-term CO2 treatment of strawberries at two ripening stages may inhibit the production of volatiles during 9 d of storage. The MANOVA results showed that CO2 treatment and storage time affected the concentrations of the total volatiles (i.e., sum of volatiles) (p < 0.001), as shown in Supplementary Materials, Table S1.
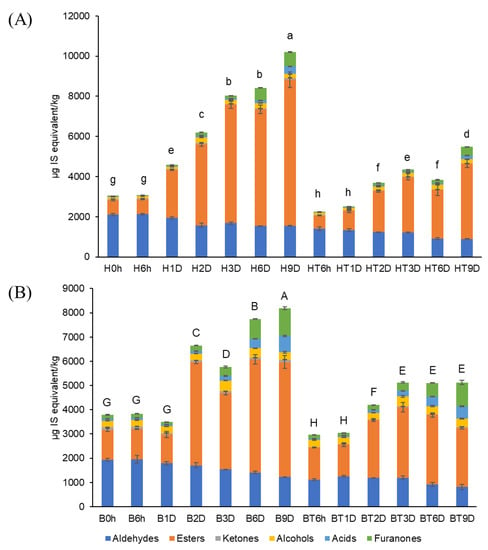
Figure 2.
Sum of volatile compounds in (A) half-red and (B) bright-red strawberries stored at 10 °C for 9 d (H: half-red, B: bright-red, T: CO2-treated, h: hours, D: days). Lowercase and capital letters indicate significant difference for half-red and bright-red strawberries, respectively (p < 0.05).
The volatile composition of strawberries varied with maturity. For example, bright-red strawberries showed a higher ester/aldehyde ratio compared to that of half-red strawberries at day 0. After 9 d of storage, the aldehyde contents of half-red strawberries (H9D) and bright-red strawberries (B9D) decreased to 78% and 71%, respectively, compared to that at day 0. However, the ester contents of H9D and B9D increased 3.0-fold and 1.8-fold, respectively, compared to that at day 0. The ester content of CO2-treated half-red strawberries (HT9D) was 68.5% that of the total volatiles. This value was similar to that of the half-red.
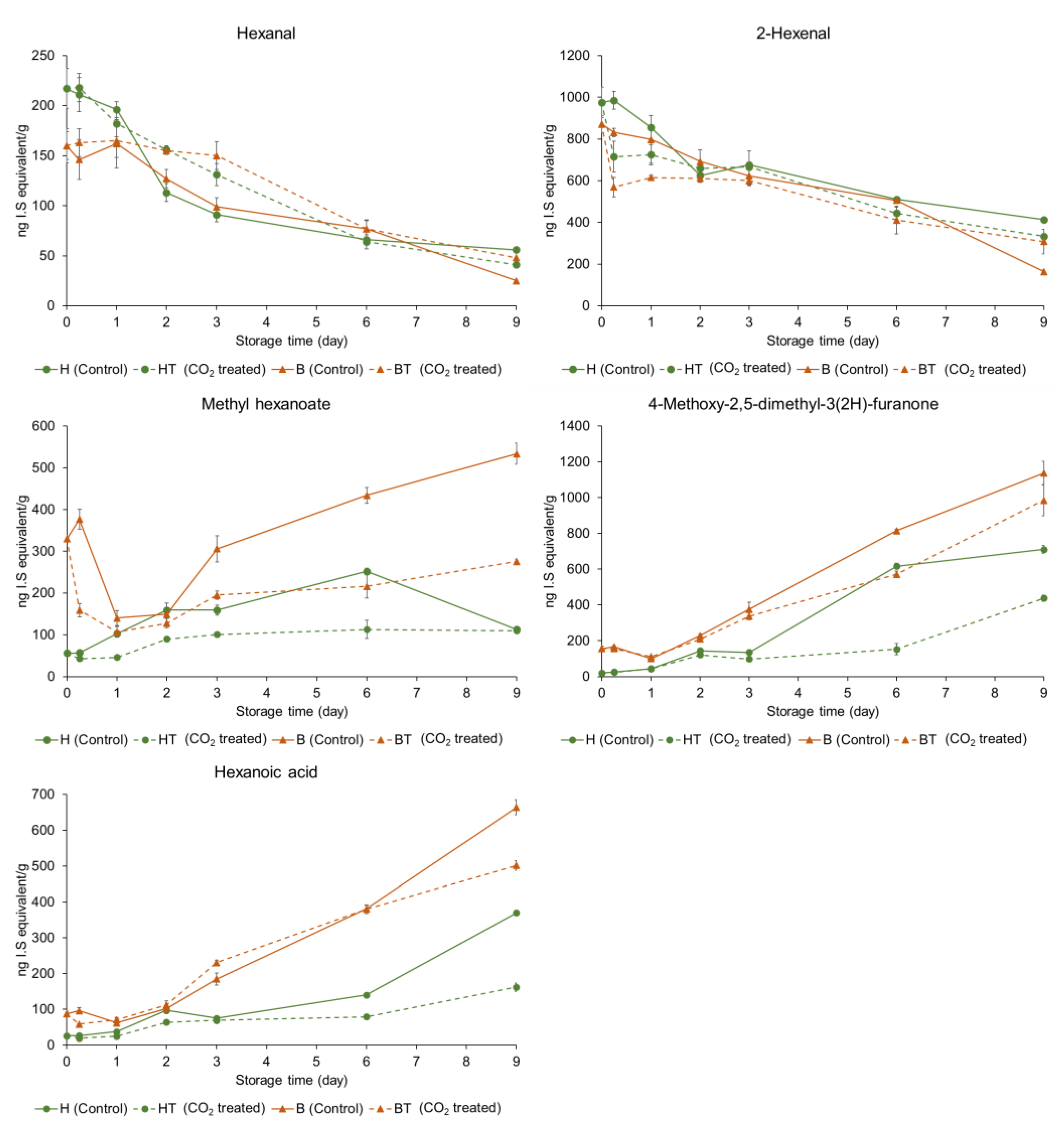
The relative concentrations of individual volatile compounds in the control and CO2-treated strawberries at two ripening stages—(A) half-red and (B) bright-red—during storage are shown in Table 2 and Table 3. The relative concentrations of characteristic volatile compounds of the strawberries are shown in Figure 3. The concentration of each volatile compound was described as the relative concentration of the appropriate internal standard. In half-red strawberries, the hexanal concentration significantly decreased from 217 µg/kg (H0h) to 41 µg/kg (HT9D) and 56 µg/kg (H9D) after 9 d in storage (p < 0.05). Furthermore, the 2-hexenal concentrations significantly decreased from 975 µg/kg (H0h) to 413 µg/kg (H9D) and 333 µg/kg (HT9D). No significant difference was observed in the hexanal and 2-hexenal concentrations between H9D and HT9D (p > 0.05). The hexanal concentrations of stored bright-red strawberries (B9D, 25 µg/kg; BT9D, 48 µg/kg) decreased compared to those in fresh bright-red strawberries (B0h, 160 µg/kg). Furthermore, the 2-hexenal concentrations of stored bright-red strawberries (B9D, 164 µg/kg; BT9D, 308 µg/kg) decreased compared to those of fresh half-red strawberries (B0h, 871 µg/kg). The 2-hexenal concentration of BT9D was significantly higher than that of B9D (p < 0.05). A previous study reported that the concentrations of hexanal and 2-hexenal, the characteristic volatile compounds responsible for the fresh and fruity odor of fresh strawberries, decreased during storage [28]. Hexanal may help delay the ripening process through the inactivation of phospholipase D, membrane lipid-degrading enzymes, and downstream oxidative signaling, and enhance shelf-life, as shown in mango fruits [29]. Moreover, hexanal may help reduce postharvest deterioration by preventing the growth of microorganism and fungi [30]. Interestingly, postharvest hexanal treatment on ‘Rubygem’ strawberries increased the alcohol percentage of the fruit at the end of their shelf-life [15]. However, in this study, short-term CO2 treatment on half-red strawberries did not increase the alcohol content at the end of the storage period. In this study, short-term CO2 treatment did not affect the hexanal and 2-hexenal concentrations in half-red strawberries but maintained their concentrations in bright-red strawberries. Thus, CO2 treatment on bright-red strawberries may help maintain fruit quality during storage.

Table 2.
Changes in the relative concentrations of volatile compounds in untreated (control) and CO2-treated half-red strawberries.

Table 3.
Changes in the relative concentrations of volatile compounds in untreated (control) and CO2-treated bright-red strawberries.
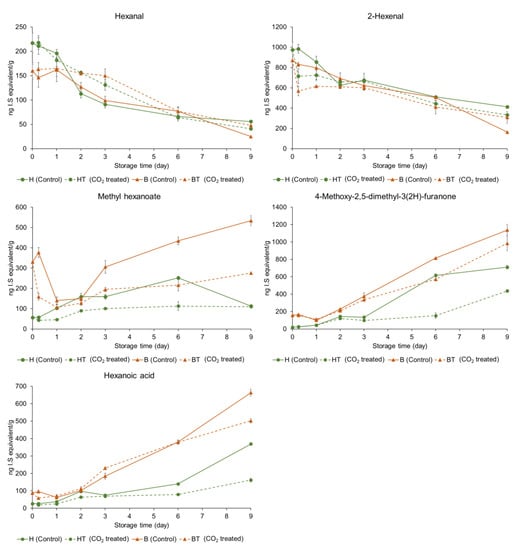
Figure 3.
Comparisons of relative concentrations of characteristic volatile compounds in control and CO2-treated strawberries at varying stages of ripening stored at 10 °C for 9 d (H: half-red, B: bright-red, T: CO2 treated, h: hours, D: days). The concentration of each volatile compound was described as the relative concentration of the appropriate internal standard.
Characteristic strawberry volatile compounds include esters, such as ethyl butanoate, ethyl hexanoate, methyl butanoate, and methyl hexanoate [5]. Among them, methyl hexanoate showed a higher concentration than that of other esters. It has been previously suggested as a maturity marker for other strawberry cultivars (e.g., ‘Albion’, ‘Malling Pearl’, ‘Florin’, ‘Charlotte’, ‘Anabelle’, ‘Florin 2’, ‘Montery’, ‘San Andreas’, and ‘Portola’) [31]. The methyl hexanoate concentration of B0h (330 µg/kg) was 5.9-folds higher than that of H0h (56 µg/kg) before storage (day 0). The methyl hexanoate concentration of control half-red control strawberries increased and then decreased during storage. Ayala-Zavala et al. also showed that the methyl hexanoate concentration of commercially matured ‘Chandler’ strawberries increased 1.8-fold when stored at 5 °C for 5 d, but it decreased to the concentrations lower than the original concentration when harvested [32]. However, the methyl hexanoate concentration continued to increase at a slower rate in CO2-treated half-red (HT) strawberries but not in the in control half-red strawberries. The methyl hexanoate concentrations decreased during early storage (≈2 d) and then increased both in the control and CO2-treated bright-red strawberries. The concentrations decreased to a greater extent in CO2-treated bright-red strawberries until 2 d of storage and increased at a slower rate in CO2-treated bright-red strawberries compared to the control bright-red strawberries stored under the same conditions. Thus, CO2 treatment may induce a decline in the methyl hexanoate concentration during the early storage period and then inhibit methyl hexanoate formation during storage, regardless of the ripening stage. Interestingly, the production of methyl butanoate, ethyl butanoate, and ethyl hexanoate was inhibited in CO2-treated half-red strawberries during storage; however, the effect was not observed or was less frequently observed in bright-red strawberries. The concentration of esters in CO2-treated bright-red strawberries was 81–98% that of the control at 9 d of storage.
The 4-methoxy-2,5-dimethyl-3(2H)-furanone concentration of bright-red strawberries (B0h, 156 µg/kg) was 7.8-fold higher than that of half-red strawberries (H0h, 20 µg/kg). Furanones, such as 2,5-dimethyl-4-hydroxy-3(2H)-furanone and 4-methoxy-2,5-dimethyl-3(2H)-furanone, are known as characteristic volatile compounds of strawberry [5]. However, 2,5-dimethyl-4-hydroxy-3(2H)-furanone was not detected in the ‘Seolhyang’ strawberry. The concentration of 4-methoxy-2,5-dimethyl-3(2H)-furanone of half-red strawberries (H0h, 20 µg/kg) increased 35.6-folds after 9 d of storage (H9D, 711 µg/kg). The concentration of 4-methoxy-2,5-dimethyl-3(2H)-furanone of bright-red strawberries (B0h, 156 µg/kg) increased by 7.3-folds after 9 d storage (B9D, 1,137 µg/kg). For the CO2 treatment groups, the 4-methoxy-2,5-dimethyl-3(2H)-furanone concentrations also increased but were lower compared to the control groups (H9D (711 µg/kg) vs. HT9D (411 µg/kg); B9D (1,137 µg/kg) vs. BT9D (985 µg/kg)). Thus, CO2 treatment may inhibit the production of 4-methoxy-2,5-dimethyl-3(2H)-furanone during storage.
High CO2 treatment of strawberries may induce a decrease in alcohol acetyltransferase activity, which is responsible for ester biosynthesis from acetyl CoA [33]. Thus, short-term CO2 treatment of half-red and bright-red strawberries may inhibit the production of esters during storage. In a previous study, CO2 treatment of strawberries inhibited the production of glucose, which is a carbon source for 4-hydroxy-2,5-dimethyl-3(2H)-furanone [34,35]. The concentrations of furanones, such as 4-methoxy-2,5-dimethyl-3(2H)-furanone, increased during postharvest ripening possibly because of an increase in Fragaria × ananassa O-methyltransferase (FaOMT) activity that changes methylated 4-hydroxy-2,5-dimethyl-3(2H)-furanone into 4-methoxy-2,5-dimethyl-3(2H)-furanone [36,37]. Thus, CO2 treatment may also inhibit furanone accumulation during storage by decreasing the glucose concentration, which is responsible for furanone biosynthesis and decreasing FaOMT activity during storage.
Acetaldehyde is responsible for the pungent aroma, which is the off-odor in strawberries [38]. The acetaldehyde concentration of half-red strawberries (H0h, 899 µg/kg) was similar to that of bright-red strawberries (B0h, 881 µg/kg). The acetaldehyde concentration of half-red strawberries (H0h, 899 µg/kg) increased after 9 d of storage (H9D, 986 µg/kg). In the case of bright-red strawberries, the acetaldehyde concentration (B0h, 881 µg/kg) increased after 9 d of storage (B9D, 988 µg/kg). The acetaldehyde concentration of H9D (986 µg/kg) was 2.0-fold higher than that of HT9D (505 µg/kg). The acetaldehyde concentration of B9D (988 µg/kg) was 2.2-fold higher than that of BT9D (440 µg/kg). Thus, CO2 treatment may inhibit acetaldehyde formation and help maintain the aroma quality of strawberries during storage, regardless of maturity. A previous study reported that the acetaldehyde content was lower in strawberries (Fragaria vesca L.) stored in a controlled atmosphere with a higher CO2 concentration compared to that in the untreated control [38]. High CO2 treatment inhibited pyruvate decarboxylase activity that decarboxylates pyruvate to yield acetaldehyde during storage [39].
2,3-Butanedione is responsible for the pungent off-odor of strawberries [40]. The 2,3-butanedione concentration of bright-red strawberries (B0h, 92 µg/kg) was 2.9-fold higher than that of half-red strawberries (H0h, 32 µg/kg). The 2,3-butanedione concentration was lower in CO2-treated strawberries during storage compared to untreated strawberries at the same storage time, regardless of the ripening stage. Thus, CO2 treatment may improve the aroma quality of strawberries during storage regardless of the maturity based on lower concentrations of 2,3-butanedione compared to the control during storage.
Hexanoic acid is also related to a rancid off-odor in strawberries [40]. The concentration of hexanoic acid of half-red strawberries increased from 26 µg/kg (H0h) to 370 µg/kg (H9D) and 162 µg/kg (HT9D). In bright-red strawberries, the concentration of hexanoic acid increased from 87 µg/kg (B0h) to 664 µg/kg (B9D) and 502 µg/kg (BT9D). Thus, hexanoic acid concentrations increased during storage regardless of the ripening stage; however, CO2 treatment may inhibit hexanoic acid production during storage. Similar to our findings, the hexanoic acid concentrations of (untreated) ‘Sweet Charlie’ strawberries increased during 9 d storage at 4 °C [10]. However, the hexanoic acid concentrations of ‘Pajaro’ strawberries decreased during 12 d storage in 20% CO2 at 0 °C [41]. In addition to hexanoic acid, various fatty acids, including linoleic acid and linolenic acid, were identified in nine strawberry cultivars in a previous study [42]. Some ester compounds, such as methyl butanoate, ethyl butanoate, and methyl hexanoate, are formed by the β-oxidation of fatty acids in strawberries [18]. In this study, the ester compound production may have been inhibited during storage by CO2 treatment possibly owing to inhibited fatty acid oxidation during storage.
2.3. Principal Component Analysis (PCA) and the Heatmap of Volatiles
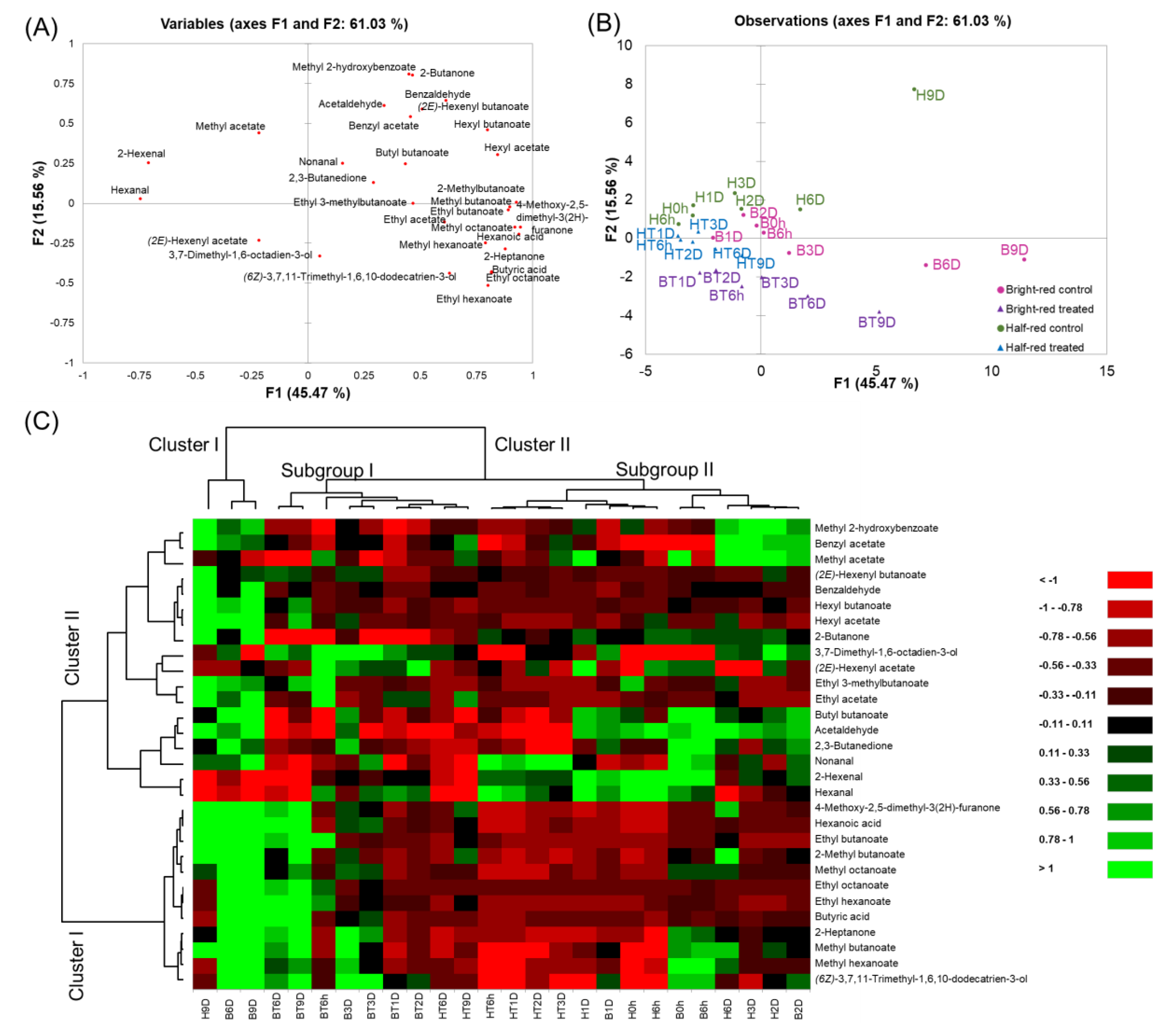
A PCA was performed using the volatile compounds data from all samples (Figure 4A and B). The first two principal components (PC1 and PC2) accounted for 61.03% of the total variation. PC1 and PC2 contributed to 45.47% and 15.56% of the total variation, respectively. The control strawberries in mostly late storage periods (H9D, B6D, and B9D) were separately grouped from other samples. As storage progressed, the strawberries were spread on the positive side of PC1. They were positively correlated with characteristic strawberry volatiles, such as 4-methoxy-2,5-dimethyl-3(2H)-furanone, methyl butanoate, ethyl butanoate, methyl hexanoate, and ethyl hexanoate. Acetaldehyde and 2,3-butanedione, and hexanoic acid, that are responsible for the off-odor of strawberries, were spread on the positive side of PC1. These volatiles correlated with H9D, B6D, and B9D. Hexanal and 2-hexenal, that are responsible for the fruity odor of strawberries, were spread on the negative side of PC1. These volatiles correlated with the half-red strawberries during the early storage period (e.g., H0h, H6h, H1D, HT6h, and HT1D). Additionally, CO2-treated strawberries were spread on the negative side of PC2. Especially, CO2-treated bright-red strawberries were spread toward the negative side of PC2 compared to CO2-treated half-red strawberries. (2E)-2-hexenyl acetate and 3,7-dimethyl-1,6-octadien-3-ol concentrations positively correlated with CO2-treated strawberry groups. Thus, these may be used as potential markers of short-term CO2 treatment in strawberries. Previously, 3,7-dimethyl-1,6-octadien-3-ol was reported as a key component of the muscat table grape [43]. The concentration of 3,7-dimethyl-1,6-octadien-3-ol was higher in CO2-treated strawberry groups compared to the control groups, regardless of the ripening stage. Ten volatiles (4-methoxy-2,5-dimethyl-3(2H)-furanone, hexanoic acid, methyl octanoate, methyl butanoate, 2-heptanone, ethyl butanoate, butyric acid, hexyl acetate, ethyl octanoate, and hexyl butanoate) were selected from the loading plot based on their high variables importance in projection (VIP) values over 1, as they contributed to the discrimination of the strawberries samples.
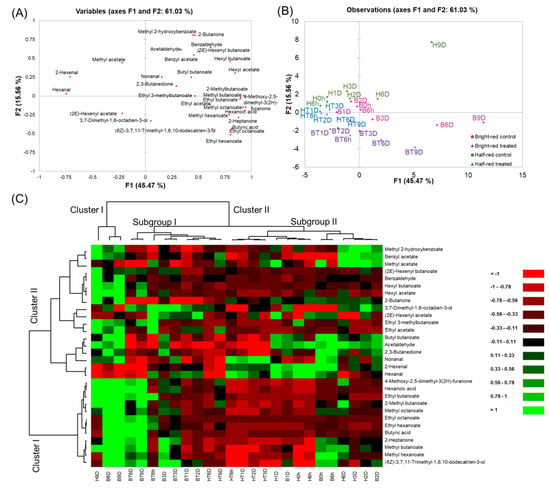
Figure 4.
(A) Score plot, (B) loading plot of the principal components analysis, and (C) hierarchical clustering and heatmap visualization of the changes in the volatile compounds in strawberries during storage. (H: half-red, B: bright-red, T: CO2 treated, h: hours, D: days). Green and red colors presented in the heatmap indicate values above and below the mean centered and scaled expression values, respectively. Black indicates values close to the mean. The total number of features was the same as the number of determined volatiles in the strawberries (n = 30), and the “non-specific filtering” option in the XLSTAT program was not used. Missing data were estimated using the nearest-neighbor algorithm. Hierarchical clustering was conducted based on Euclidian distances.
A dendrogram was obtained using cluster analysis (Figure 4C). Volatiles were grouped into two clusters. Cluster I included the characteristic strawberry volatiles (e.g., 4-methoxy-2,5-dimethyl-3(2H)-furanone, ethyl butanoate, ethyl hexanoate, methyl butanoate, and methyl hexanoate) showing higher concentrations in bright-red strawberries, regardless of CO2 treatment. Strawberries were grouped into two clusters. Cluster I included control strawberries at the late storage period (i.e., H9D, B6D, and B9D). Cluster II was divided into two subgroups. Subgroup I included CO2-treated half-red and bright-red strawberries, except B3D. Subgroup II included control half-red and bright-red strawberries, except CO2-treated half-red strawberries in the early storage period (i.e., HT6h-HT3D).
3. Materials and Methods
3.1. Chemicals and Reagents
Authentic standards of hexanal, 2-hexenal, nonanal, benzaldehyde, methyl acetate, methyl hexanoate, ethyl hexanoate, ethyl octanoate, 2-heptanone, butyric acid, hexanoic acid, and n-alkane standards were purchased from Sigma-Aldrich (St. Louis, MO, USA). Authentic standards of octanal-d16, ethyl butanoate-d3 and n-hexyl-d13 alcohol, used as internal standards, were purchased from C/D/N Isotope (Pointe-Claire, Quebec, Canada). Nanopure water used in the study was obtained from a water purification system (Milli-Q Direct 8, Merck Millipore, Billerica, MA, USA).
3.2. Strawberry Samples
‘Seolhyang’ strawberries were harvested at two different ripening stages from a strawberry farm located in Jinju, Gyeongsangnam-do, Korea (35.1800 °N, 128.1076 °E) in March 2019. The ‘Seolhyang’ strawberries were grown in a commercial greenhouse using conventional soil cultivation practices. The samples were transported to the Korea Food Research Institute (KFRI; Wanju, Korea), which was located at a distance of 175 km from the farm, using a refrigerated truck. Undamaged strawberries of uniform size and shape were selected for the study. The ripening stages were grouped into half-red (80% maturity) or bright-red (100% maturity) based on surface color development. Half of the selected fruits under each ripening group were treated with CO2 by exposing the samples to air saturated with 30% CO2 and 70% N2 for 3 h at 25 °C. Thus, the samples were divided into four subgroups (i.e., half-red control, half-red CO2-treated, bright-red control, and bright-red CO2-treated). Approximately 15–20 fruits from each subgroup were placed in a plastic bin and covered with a lid. The two bins were removed periodically at 0, 6, 24, and 48 h, and 3, 6, and 9 d to sample the strawberries stored at 10 °C. To compare the changes in the volatile profiles of strawberries following CO2 treatment, the volatile composition of strawberries after 6 h was investigated. Thus, there were 8 (time point) × 2 (half-red and bright-red) × 2 (control and CO2 treated) × 2 (duplicate) = 64 samples for strawberries stored at 10 °C. Each of the samples at the time points were immediately frozen in liquid nitrogen and stored at −80 °C until analysis.
3.3. Determination of Strawberry Volatiles using HS-SPME-GC/MS
The sample preparation method was adopted from our previous study with modifications [44]. Frozen strawberries (300 g) were ground using a blender (BL682 Auto-IQ, Nutri Ninja, Boston, MA, USA) for 1 min. Then, 5 g of the ground frozen strawberries was transferred to a glass headspace vial (Gerstel, Baltimore, MD, USA), and 5 mL of saturated sodium chloride solution was added. An internal standard mixture (octanal-d16, ethyl butanoate-d3, and n-hexyl-d13 alcohol) was added to the glass headspace vial with a final concentration of 10 µg/kg and was sealed immediately using a magnetic crimp cap (Gerstel).
After 10 min of equilibration, a 2 cm solid phase microextraction (SPME) fiber coated with divinylbenzene/carboxen/ polydimethylsiloxane (DVB/CAR/PDMS) was used to extract the volatiles. The volatiles were absorbed for 30 min at 40 °C and desorbed for 30 s at 250 °C. Volatiles in strawberry were determined using gas chromatography (7890B, Agilent technologies, Santa Clara, CA, USA) coupled with a mass selective detector (5977B, Agilent technologies) and separated on a DB-WAX capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness; Agilent technologies). The oven temperature was set to 40 °C for 2 min; the temperature was increased by 4 °C/min to 120 °C and held at 120 °C for 5 min. Then, the temperature was increased to 250 °C at a rate of 20 °C/min and held at 250 °C for 5 min. The solvent delay was set to 3 min. Helium (99.999%) was used as a carrier gas at a flow rate of 1 mL/min. The inlet temperature was 250 °C, and an inlet liner (0.75 mm i.d., Agilent technologies) was used for the splitless mode. The temperatures of the transfer line, ion source, and quadrupole were 240 °C, 230 °C, and 150 °C, respectively. The electron energy was 70 eV. Mass spectra were obtained in the range of m/z 30–350 at a rate of 3.06 scans/s.
3.4. Identification and Relative Quantification of Volatiles in Strawberries
Volatiles were identified by comparing their mass spectra and the retention time of authentic standards if available or tentatively by searching libraries (Willey and NIST 08 mass spectral libraries) for compounds with no authentic standard available. Eighty percent of the match score was used as a cut-off, and Kovats’ retention index was used to confirm the identification of volatiles. Kovats’ retention index was calculated using the retention time of each volatile compound and an authentic alkane standard mixture.
Relative volatile concentrations were calculated according to formula (1) presented below [45]. Briefly, the extracted ion peak area of each volatile was divided by the extracted ion peak area of the respective internal standard (octanal-d16 for aldehydes, ketones, and furanones; n-hexyl-d13 alcohol for alcohols and acids; ethyl butanoate-d3 for esters). The relative concentration of each volatile was converted by multiplying the area ratio with 10 µg internal standard/kg strawberry. The results were expressed on a fresh weight basis.
3.5. Statistical Analyses
Statistical analyses were conducted using SPSS statistics 23 software (SPSS, Inc., Chicago, IL, USA). Significant differences in the volatile composition among the strawberry fruits were confirmed by an analysis of variance at p < 0.05. Duncan’s test was used as a post hoc analysis. A multivariate analysis of variance (MANOVA) was conducted to evaluate the effects of maturity, storage time, CO2 treatment, and their interactions on the volatile composition of strawberries. PCA, heatmap, and hierarchical cluster analyses were performed using XLSTAT (XLSTAT, ver. 2017.03, Microsoft Excel Add-in software, New York, NY, USA). The data format for PCA was an observation/variable table, and Pearson’s correlation type was used as the PCA type. Based on the volatile profile of strawberries, heatmap and hierarchical cluster analyses were conducted to demonstrate similarities and differences among the strawberries depending on maturity, storage period, and short-term CO2 treatment. The total number of features was the same as the number of determined volatiles in the strawberries (n = 30), and the “non-specific filtering” option in the XLSTAT program was not used.
4. Conclusions
To the best of our knowledge, this is the first study investigating the effect of short-term CO2 treatment on the volatile composition of ‘Seolhyang’ strawberries. A total of thirty volatiles were identified using HS-SPME with GC-MS in ‘Seolhyang’ strawberries stored at 10 °C for 9 d. The volatile composition of the strawberries changed depending on the ripening stages, storage time, and short-term CO2 treatment. Ripening and storage increased the total volatile concentrations and CO2 treatment inhibited the production of volatiles. The production of some characteristic strawberry volatiles, such as methyl hexanoate and 4-methoxy-2,5-dimethyl-3(2H)-furanone, was inhibited in CO2-treated strawberries, especially in half-red strawberries, during cold storage. However, CO2 treatment helped maintain the hexanal and 2-hexenal concentrations, which are responsible for the fresh odor in strawberries. Interestingly, CO2 treatment decreased the formation of rancid off-odor volatiles, such as acetaldehyde, 2,3-butanedione, and hexanoic acid during storage, regardless of the ripening state. Thus, short-term CO2 treatment may help maintain a fresh odor and reduce the off-odor, improving strawberry aroma characteristics during storage. Short-term CO2 treatment may be applied to ‘Seolhyang’ strawberries for long-distance shipping to improve the strawberry aroma characteristics during shipping. In future research, the role of short-term CO2 treatment in improving the aroma quality of ‘Seolhyang’ strawberries can be confirmed using descriptive sensory analysis.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules27196599/s1, Table S1: Multivariate analysis of variance F-values and p-values for the volatile contents of all strawberries.
Author Contributions
Conceptualization, J.H.C., J.-H.L., K.-J.P. and J.L.; investigation, I.K. and D.A.; writing—original draft preparation, I.K.; writing—review and editing, J.H.C., J.-H.L., G.O. and J.L.; visualization, I.K. and D.A.; project administration, J.L.; funding acquisition, K.-J.P. and J.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Korea Food Research Institute, grant number E0211000-01. This research was supported by a grant (21153MFDS605) from the Ministry of Food and Drug Safety in 2022. This research was supported by the Chung-Ang University Graduate Research Scholarship awarded in 2019.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chungcheongnam-do Agricultural Research and Extension Services. Available online: https://cnnongup.chungnam.go.kr/board/B0015.cs?act=download&articleId=228111&fileSn=0 (accessed on 13 January 2022).
- aTKorea. Available online: https://www.kati.net/file/down.do?path=/board/2018/07/&fileName=%EB%94%B8%EA%B8%B0+%EA%B5%AD%EB%82%B4%EC%83%9D%EC%82%B0+%EB%B0%8F+%ED%95%B4%EC%99%B8%EC%8B%9C%EC%9E%A5+%EB%8F%99%ED%96%A5.pdf.pdf (accessed on 13 January 2022).
- KATI. Available online: https://www.kati.net/product/basisInfo.do?lcdCode=MD149 (accessed on 13 January 2022).
- Kim, J.-G.; Choi, J.-W.; Park, M.-H. Effect of different days of postharvest treatment and CO2 concentrations on the quality of ‘Seolhyang’ strawberry during storage. Korean J. Food Preserv. 2016, 23, 12–19. [Google Scholar] [CrossRef]
- Yan, J.w.; Ban, Z.j.; Lu, H.y.; Li, D.; Poverenov, E.; Luo, Z.s.; Li, L. The aroma volatile repertoire in strawberry fruit: A review. J. Sci. Food Agric. 2018, 98, 4395–4402. [Google Scholar] [CrossRef] [PubMed]
- Maarse, H. Volatile Compounds in Foods and Beverages, 1st ed.; Routledge: London, UK, 2017; pp. 329–387. [Google Scholar]
- Azodanlou, R.; Darbellay, C.; Luisier, J.-L.; Villettaz, J.-C.; Amadò, R. Changes in flavour and texture during the ripening of strawberries. Eur. Food Res. Technol. 2004, 218, 167–172. [Google Scholar]
- Urün, I.; Attar, S.H.; Sönmez, D.A.; Gündeşli, M.A.; Ercişli, S.; Kafkas, N.E.; Bandić, L.M.; Duralija, B. Comparison of polyphenol, sugar, organic acid, volatile compounds, and antioxidant capacity of commercially grown strawberry cultivars in Turkey. Plants 2021, 10, 1654. [Google Scholar] [CrossRef] [PubMed]
- Vandendriessche, T.; Nicolai, B.; Hertog, M. Optimization of HS SPME fast GC-MS for high-throughput analysis of strawberry aroma. Food Anal. Methods 2013, 6, 512–520. [Google Scholar] [CrossRef]
- Ozcan, G.; Barringer, S. Effect of enzymes on strawberry volatiles during storage, at different ripeness level, in different cultivars, and during eating. J. Food Sci. 2011, 76, C324–C333. [Google Scholar] [CrossRef] [PubMed]
- Pérez, A.G.; Sanz, C.; Olías, R.; Ríos, J.J.; Olías, J.M. Evolution of strawberry alcohol acyltransferase activity during fruit development and storage. J. Agric. Food Chem. 1996, 44, 3286–3290. [Google Scholar] [CrossRef]
- Myung, K.; Hamilton-Kemp, T.R.; Archbold, D.D. Biosynthesis of trans-2-hexenal in response to wounding in strawberry fruit. J. Agric. Food Chem. 2006, 54, 1442–1448. [Google Scholar] [CrossRef]
- Almenar, E.; Hernandez-Munoz, P.; Gavara, R. Evolution of selected volatiles in chitosan-coated strawberries (Fragaria × ananassa) during refrigerated storage. J. Agric. Food Chem. 2009, 57, 974–980. [Google Scholar] [CrossRef]
- Lu, H.; Wang, K.; Wang, L.; Li, D.; Yan, J.; Ban, Z.; Luo, Z.; Li, L.; Yang, D. Effect of superatmospheric oxygen exposure on strawberry (Fragaria × ananassa Fuch.) volatiles, sensory and chemical attributes. Postharvest Biol. Technol. 2018, 142, 60–71. [Google Scholar] [CrossRef]
- Öz, A.T.; Kafkas, E. Volatile compositions of strawberry fruit during shelf life using pre and postharvest hexanal treatment. J. Food Process. Preserv. 2022, 46, e16464. [Google Scholar] [CrossRef]
- Pelayo, C.; Ebeler, S.; Kader, A. Postharvest life and flavor quality of three strawberry cultivars kept at 5 °C in air or air+ 20 kPa CO2. Postharvest Biol. Technol. 2003, 27, 171–183. [Google Scholar] [CrossRef]
- Yang, M.; Ban, Z.; Luo, Z.; Li, J.; Lu, H.; Li, D.; Chen, C.; Li, L. Impact of elevated O2 and CO2 atmospheres on chemical attributes and quality of strawberry (Fragaria × ananassa Duch.) during storage. Food Chem. 2020, 307, 125550. [Google Scholar] [CrossRef]
- Pelayo-Zaldívar, C.; Abda, J.B.; Ebeler, S.E.; Kader, A.A. Quality and chemical changes associated with flavor of ‘Camarosa’strawberries in response to a CO2-enriched atmosphere. HortScience 2007, 42, 299–303. [Google Scholar] [CrossRef]
- Ahn, D.; Kim, I.; Lim, J.-H.; Choi, J.H.; Park, K.-J.; Lee, J. The effect of high CO2 treatment on targeted metabolites of ‘Seolhyang’ strawberry (Fragaria × ananassa) fruits during cold storage. LWT 2021, 143, 111156. [Google Scholar] [CrossRef]
- Dong, J.; Zhang, Y.; Tang, X.; Jin, W.; Han, Z. Differences in volatile ester composition between Fragaria × ananassa and F. vesca and implications for strawberry aroma patterns. Sci. Hortic. 2013, 150, 47–53. [Google Scholar] [CrossRef]
- Ulrich, D.; Hoberg, E.; Rapp, A.; Kecke, S. Analysis of strawberry flavour–discrimination of aroma types by quantification of volatile compounds. Eur. Food Res. Technol. 1997, 205, 218–223. [Google Scholar] [CrossRef]
- Mishra, R.; Kar, A. Effect of storage on the physicochemical and flavour attributes of two cultivars of strawberry cultivated in Northern India. Sci. World J. 2014, 2014, 794926. [Google Scholar] [CrossRef]
- Forney, C.F.; Kalt, W.; Jordan, M.A. The composition of strawberry aroma is influenced by cultivar, maturity, and storage. HortScience 2000, 35, 1022–1026. [Google Scholar] [CrossRef]
- Padilla-Jiménez, S.M.; Angoa-Pérez, M.V.; Mena-Violante, H.G.; Oyoque-Salcedo, G.; Montañez-Soto, J.L.; Oregel-Zamudio, E. Identification of organic volatile markers associated with aroma during maturation of strawberry fruits. Molecules 2021, 26, 504. [Google Scholar] [CrossRef]
- Ménager, I.; Jost, M.; Aubert, C. Changes in physicochemical characteristics and volatile constituents of strawberry (Cv. Cigaline) during maturation. J. Agric. Food Chem. 2004, 52, 1248–1254. [Google Scholar] [CrossRef]
- Perez, A.G.; Rios, J.J.; Sanz, C.; Olias, J.M. Aroma components and free amino acids in strawberry variety Chandler during ripening. J. Agric. Food Chem. 1992, 40, 2232–2235. [Google Scholar] [CrossRef]
- Wang, L.; Dou, G.; Guo, H.; Zhang, Q.; Qin, X.; Yu, W.; Jiang, C.; Xiao, H. Volatile organic compounds of Hanseniaspora uvarum increase strawberry fruit flavor and defense during cold storage. Food Sci. Nutr. 2019, 7, 2625–2635. [Google Scholar] [CrossRef]
- Perdones, Á.; Escriche, I.; Chiralt, A.; Vargas, M. Effect of chitosan–lemon essential oil coatings on volatile profile of strawberries during storage. Food Chem. 2016, 197, 979–986. [Google Scholar] [CrossRef]
- Jincy, M.; Djanaguiraman, M.; Jeyakumar, P.; Subramanian, K.; Jayasankar, S.; Paliyath, G. Inhibition of phospholipase D enzyme activity through hexanal leads to delayed mango (Mangifera indica L.) fruit ripening through changes in oxidants and antioxidant enzymes activity. Sci. Hortic. 2017, 218, 316–325. [Google Scholar] [CrossRef]
- Lanciotti, R.; Corbo, M.R.; Gardini, F.; Sinigaglia, M.; Guerzoni, M.E. Effect of hexanal on the shelf life of fresh apple slices. J. Agric. Food Chem. 1999, 47, 4769–4776. [Google Scholar] [CrossRef]
- Vandendriessche, T.; Vermeir, S.; Martinez, C.M.; Hendrickx, Y.; Lammertyn, J.; Nicolaï, B.; Hertog, M. Effect of ripening and inter-cultivar differences on strawberry quality. LWT 2013, 52, 62–70. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.F.; Wang, S.Y.; Wang, C.Y.; González-Aguilar, G.A. Effect of storage temperatures on antioxidant capacity and aroma compounds in strawberry fruit. LWT 2004, 37, 687–695. [Google Scholar] [CrossRef]
- Ke, D.; Zhou, L.; Kader, A.A. Mode of oxygen and carbon dioxide action on strawberry ester biosynthesis. J. Am. Soc. Hortic. Sci. 1994, 119, 971–975. [Google Scholar] [CrossRef]
- Bodelón, O.G.; Blanch, M.; Sanchez-Ballesta, M.T.; Escribano, M.I.; Merodio, C. The effects of high CO2 levels on anthocyanin composition, antioxidant activity and soluble sugar content of strawberries stored at low non-freezing temperature. Food Chem. 2010, 122, 673–678. [Google Scholar] [CrossRef]
- Zabetakis, I.; Gramshaw, J.; Robinson, D. 2, 5-Dimethyl-4-hydroxy-2H-furan-3-one and its derivatives: Analysis, synthesis and biosynthesis—A review. Food Chem. 1999, 65, 139–151. [Google Scholar] [CrossRef]
- Wein, M.; Lavid, N.; Lunkenbein, S.; Lewinsohn, E.; Schwab, W.; Kaldenhoff, R. Isolation, cloning and expression of a multifunctional O-methyltransferase capable of forming 2,5-dimethyl-4-methoxy-3(2H)-furanone, one of the key aroma compounds in strawberry fruits. Plant J. 2002, 31, 755–765. [Google Scholar] [CrossRef]
- Lunkenbein, S.; Salentijn, E.M.J.; Coiner, H.A.; Boone, M.J.; Krens, F.A.; Schwab, W. Up- and down-regulation of Fragaria × ananassa O-methyltransferase: Impacts on furanone and phenylpropanoid metabolism. J. Exp. Bot. 2006, 57, 2445–2453. [Google Scholar] [CrossRef] [PubMed]
- Almenar, E.; Hernández-Muñoz, P.; Lagarón, J.M.; Catalá, R.; Gavara, R. Controlled atmosphere storage of wild strawberry fruit (Fragaria vesca L.). J. Sci. Food Agric. 2006, 54, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Lara, I.; Miró, R.; Fuentes, T.; Sayez, G.; Graell, J.; López, M. Biosynthesis of volatile aroma compounds in pear fruit stored under long-term controlled-atmosphere conditions. Postharvest Biol. Technol. 2003, 29, 29–39. [Google Scholar] [CrossRef]
- Flavornet and Human Odor Space. Available online: http://www.flavornet.org (accessed on 13 January 2022).
- Larsen, M.; Watkins, C.B. Firmness and aroma composition of strawberries following short-term high carbon dioxide treatments. HortScience 1995, 30, 303–305. [Google Scholar] [CrossRef]
- Kafkas, E.; Gunaydin, S.; Ercisli, S.; Ozogu, Y.; Unlu, M.A. Fat and fatty acid composition of strawberry cultivars. Chem. Nat. Compd. 2009, 45, 861–863. [Google Scholar] [CrossRef]
- Matsumoto, H.; Ikoma, Y. Effect of postharvest temperature on the muscat flavor and aroma volatile content in the berries of ‘Shine Muscat’ (Vitis labruscana Baily × V. vinifera L.). Postharvest Biol. Technol. 2016, 112, 256–265. [Google Scholar] [CrossRef]
- Choi, S.; Seo, H.-S.; Lee, K.R.; Lee, S.; Lee, J. Effect of milling degrees on volatile profiles of raw and cooked black rice (Oryza sativa L. cv. Sintoheugmi). Appl. Biol. Chem. 2018, 61, 91–105. [Google Scholar] [CrossRef]
- Lee, J.; Xiao, L.; Zhang, G.; Ebeler, S.E.; Mitchell, A.E. Influence of storage on volatile profiles in roasted almonds (Prunus dulcis). J. Agric. Food Chem. 2014, 62, 11236–11245. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).