Influence of Oxygen Management during the Post-Fermentation Stage on Acetaldehyde, Color, and Phenolics of Vitis vinifera L. Cv. Cabernet Sauvignon Wine
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Oxygen Exposure on Dissolved Oxygen (DO), SO2 and Acetaldehyde during the PFS
2.2. The Influence of Oxygen Exposure on Phenols and Color Parameters after Five Phases of Oxidation Treatments
2.3. Evolution of Critical Wine Chemical Parameters during Five Phases of Oxidation Treatments
3. Materials and Methods
3.1. Wines and Oxygenation Trials
3.2. Sulfur Dioxide and Acetaldehyde Determination
3.3. Color Analyses
3.4. Spectrophotometric Measurements of Total Phenol, Pigments and Co-pigmented Anthocyanins
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Ribereau-Gayon, P.; Dubourdieu, D.; Doneche, B.; Lonvaud, A. Red Winemaking. In Handbook of Enology: The Microbiology of Wine and Vinifications; Ribereau-Gayon, P., Dubourdieu, D., Doneche, B., Lonvaud, A., Eds.; John Wiley & Sons, Ltd: Chichester, UK, 2006; Volume 1, pp. 327–395. ISBN 9780470010365. [Google Scholar]
- Sartini, E.; Arfelli, G.; Fabiani, A.; Piva, A. Influence of chips, lees and micro-oxygenation during aging on the phenolic composition of a red Sangiovese wine. Food Chem. 2007, 104, 1599–1604. [Google Scholar] [CrossRef]
- Han, G.; Webb, M.R.; Richter, C.; Parsons, J.; Waterhouse, A.L. Yeast alter micro-oxygenation of wine: Oxygen consumption and aldehyde production. J. Sci. Food Agric. 2017, 97, 3847–3854. [Google Scholar] [CrossRef] [PubMed]
- Sáenz-Navajas, M.P.; Henschen, C.; Cantu, A.; Watrelot, A.A.; Waterhouse, A.L. Understanding microoxygenation: Effect of viable yeasts and sulfur dioxide levels on the sensory properties of a Merlot red wine. Food Res. Int. 2018, 108, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Schmidtke, L.M.; Clark, A.C.; Scollary, G.R. Micro-Oxygenation of Red Wine: Techniques, Applications, and Outcomes. Crit. Rev. Food Sci. Nutr. 2011, 51, 115–131. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Plaza, E.; Cano-López, M. A review on micro-oxygenation of red wines: Claims, benefits and the underlying chemistry. Food Chem. 2011, 125, 1131–1140. [Google Scholar] [CrossRef]
- Riebel, M.; Sabel, A.; Claus, H.; Xia, N.; Li, H.; König, H.; Decker, H.; Fronk, P. Antioxidant capacity of phenolic compounds on human cell lines as affected by grape-tyrosinase and Botrytis-laccase oxidation. Food Chem. 2017, 229, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Vignault, A.; Gombau, J.; Jourdes, M.; Moine, V.; Canals, J.M.; Fermaud, M.; Roudet, J.; Zamora, F.; Teissedre, P.-L. Oenological tannins to prevent Botrytis cinerea damage in grapes and musts: Kinetics and electrophoresis characterization of laccase. Food Chem. 2020, 316, 126334. [Google Scholar] [CrossRef] [PubMed]
- Danilewicz, J.C. Reactions Involving Iron in Mediating Catechol Oxidation in Model Wine. Am. J. Enol. Vitic. 2013, 64, 316–324. [Google Scholar] [CrossRef]
- Danilewicz, J.C. Chemistry of Manganese and Interaction with Iron and Copper in Wine. Am. J. Enol. Vitic. 2016, 67, 377–384. [Google Scholar] [CrossRef]
- Carrascon, V.; Fernandez-Zurbano, P.; Bueno, M.; Ferreira, V. Oxygen Consumption by Red Wines. Part II: Differential Effects on Color and Chemical Composition Caused by Oxygen Taken in Different Sulfur Dioxide-Related Oxidation Contexts. J. Agric. Food Chem. 2015, 63, 10938–10947. [Google Scholar] [CrossRef]
- Qian, B.; Zhao, J.; He, Y.; Peng, L.; Ge, H.; Han, B. Miniaturized dielectric barrier discharge-molecular emission spectrometer for determination of total sulfur dioxide in food. Food Chem. 2020, 317, 126437. [Google Scholar] [CrossRef]
- Guerrero, R.F.; Cantos-Villar, E. Demonstrating the efficiency of sulphur dioxide replacements in wine: A parameter review. Trends Food Sci. Technol. 2015, 42, 27–43. [Google Scholar] [CrossRef]
- Quaglieri, C.; Jourdes, M.; Waffo-Teguo, P.; Teissedre, P.-L. Updated knowledge about pyranoanthocyanins: Impact of oxygen on their contents, and contribution in the winemaking process to overall wine color. Trends Food Sci. Technol. 2017, 67, 139–149. [Google Scholar] [CrossRef]
- Picariello, L.; Gambuti, A.; Picariello, B.; Moio, L. Evolution of pigments, tannins and acetaldehyde during forced oxidation of red wine: Effect of tannins addition. LWT 2017, 77, 370–375. [Google Scholar] [CrossRef]
- Li, S.-Y.; Duan, C.-Q. Astringency, bitterness and color changes in dry red wines before and during oak barrel aging: An updated phenolic perspective review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1840–1867. [Google Scholar] [CrossRef] [PubMed]
- Carrascón, V.; Vallverdú-Queralt, A.; Meudec, E.; Sommerer, N.; Fernandez-Zurbano, P.; Ferreira, V. The kinetics of oxygen and SO2 consumption by red wines. What do they tell about oxidation mechanisms and about changes in wine composition? Food Chem. 2018, 241, 206–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrozziello, M.; Torchio, F.; Piano, F.; Giacosa, S.; Ugliano, M.; Bosso, A.; Rolle, L. Impact of Increasing Levels of Oxygen Consumption on the Evolution of Color, Phenolic, and Volatile Compounds of Nebbiolo Wines. Front. Chem. 2018, 6, 137. [Google Scholar] [CrossRef]
- Ferreira, V.; Carrascon, V.; Bueno, M.; Ugliano, M.; Fernandez-Zurbano, P. Oxygen Consumption by Red Wines. Part I: Consumption Rates, Relationship with Chemical Composition, and Role of SO2. J. Agric. Food Chem. 2015, 63, 10928–10937. [Google Scholar] [CrossRef] [PubMed]
- Danilewicz, J.C. Mechanism of Autoxidation of Polyphenols and Participation of Sulfite in Wine: Key Role of Iron. Am. J. Enol. Vitic. 2011, 62, 319–328. [Google Scholar] [CrossRef]
- Bueno, M.; Carrascón, V.; Ferreira, V. Release and Formation of Oxidation-Related Aldehydes during Wine Oxidation. J. Agric. Food Chem. 2016, 64, 608–617. [Google Scholar] [CrossRef]
- Gambuti, A.; Han, G.; Peterson, A.L.; Waterhouse, A.L. Sulfur dioxide and glutathione alter the outcome of microoxygenation. Am. J. Enol. Vitic. 2015, 66, 411–423. [Google Scholar] [CrossRef] [Green Version]
- Elias, R.J.; Waterhouse, A.L. Controlling the fenton reaction in wine. J. Agric. Food Chem. 2010, 58, 1699–1707. [Google Scholar] [CrossRef]
- Bimpilas, A.; Panagopoulou, M.; Tsimogiannis, D.; Oreopoulou, V. Anthocyanin copigmentation and color of wine: The effect of naturally obtained hydroxycinnamic acids as cofactors. Food Chem. 2016, 197, 39–46. [Google Scholar] [CrossRef]
- Geldenhuys, L.; Oberholster, A.; du Toit, W. Monitoring the effect of Micro-oxegenation before malolactic fermentation on South African pinotage red wine with different colour and phenolic analysis. S. Afr. J. Enol. Vitic. 2012, 33, 150–160. [Google Scholar]
- Pérez-Magariño, S.; Sánchez-Iglesias, M.; Ortega-Heras, M.; González-Huerta, C.; González-Sanjosé, M.L. Colour stabilization of red wines by microoxygenation treatment before malolactic fermentation. Food Chem. 2007, 101, 881–893. [Google Scholar] [CrossRef]
- Berrueta, L.A.; Rasines-Perea, Z.; Prieto-Perea, N.; Asensio-Regalado, C.; Alonso-Salces, R.M.; Sánchez-Ilárduya, M.B.; Gallo, B. Formation and evolution profiles of anthocyanin derivatives and tannins during fermentations and aging of red wines. Eur. Food Res. Technol. 2020, 246, 149–165. [Google Scholar] [CrossRef]
- Gambuti, A.; Picariello, L.; Rinaldi, A.; Moio, L. Evolution of Sangiovese Wines With Varied Tannin and Anthocyanin Ratios During Oxidative Aging. Front. Chem. 2018, 6, 63. [Google Scholar] [CrossRef] [Green Version]
- Harbertson, J.F.; Picciotto, E.A.; Adams, D.O. Measurement of Polymeric Pigments in Grape Berry Extract sand Wines Using a Protein Precipitation Assay Combined with Bisulfite Bleaching. Am. J. Enol. Vitic. 2003, 54, 301–306. Available online: https://www.ajevonline.org/content/54/4/301 (accessed on 5 October 2022).
- Gambuti, A.; Siani, T.; Picariello, L.; Rinaldi, A.; Lisanti, M.T.; Ugliano, M.; Dieval, J.B.; Moio, L. Oxygen exposure of tannins-rich red wines during bottle aging. Influence on phenolics and color, astringency markers and sensory attributes. Eur. Food Res. Technol. 2017, 243, 669–680. [Google Scholar] [CrossRef]
- Chris Somers, T.; Evans, M.E. Spectral evaluation of young red wines: Anthocyanin equilibria, total phenolics, free and molecular SO2, “chemical age”. J. Sci. Food Agric. 1977, 28, 279–287. [Google Scholar] [CrossRef]
- Han, G.; Webb, M.R.; Waterhouse, A.L. Acetaldehyde reactions during wine bottle storage. Food Chem. 2019, 290, 208–215. [Google Scholar] [CrossRef]
- Picariello, L.; Slaghenaufi, D.; Ugliano, M. Fermentative and post-fermentative oxygenation of Corvina red wine: Influence on phenolic and volatile composition, colour and wine oxidative response. J. Sci. Food Agric. 2020. [Google Scholar] [CrossRef]
- Gutiérrez, I.H.; Lorenzo, E.S.-P.; Espinosa, A.V. Phenolic composition and magnitude of copigmentation in young and shortly aged red wines made from the cultivars, Cabernet Sauvignon, Cencibel, and Syrah. Food Chem. 2005, 92, 269–283. [Google Scholar] [CrossRef]
- Baiano, A.; De Gianni, A. A study on the effects of oxygen transmission rate of synthetic stopper on wine quality: The case of Nero di Troia. Eur. Food Res. Technol. 2016, 242, 1857–1867. [Google Scholar] [CrossRef]
- Rayne, S.; Sheppard, S.; Di Bello, T.; Eggers, N.J. Chromatic Characteristics and Optically Derived Compositional Descriptors of Micro-oxygenated Wines from Vitis vinifera cv. Merlot and Cabernet Sauvignon. Food Bioprocess Technol. 2011, 254–265. [Google Scholar] [CrossRef]
- Kontoudakis, N.; González, E.; Gil, M.; Esteruelas, M.; Fort, F.; Canals, J.M.; Zamora, F. Influence of Wine pH on Changes in Color and Polyphenol Composition Induced by Micro-oxygenation. J. Agric. Food Chem. 2011, 59, 1974–1984. [Google Scholar] [CrossRef]
- Waterhouse, A.L.; Zhu, J. A quarter century of wine pigment discovery. J. Sci. Food Agric. 2020, 100, 5093–5101. [Google Scholar] [CrossRef]
- Iland, P.; Bruer, N.; Edwards, G.; Weeks, S.; Wilkes, E. Analysis methods. In Chemical Analysis of Grape and Wine: Techniques and Concepts.; Patrick Iland Wine Promotions Pty Ltd: Campbelltown, Australia, 2004. [Google Scholar]
- Han, G.; Wang, H.; Webb, M.R.; Waterhouse, A.L. A rapid, one step preparation for measuring selected free plus SO2-bound wine carbonyls by HPLC-DAD/MS. Talanta 2015, 134, 596–602. [Google Scholar] [CrossRef]
- Glories, Y. The colour of red wines. Part II. Measurement, derivation and interpretation. Connaiss. la Vigne du Vin 1984, 18, 253–271. [Google Scholar]
- Ayala, F.; Echávarri, J.F.; Negueruela, A.I. A new simplified method for measuring the color of wines. I. Red and rose wines. Am. J. Enol. Vitic. 1997, 48, 357. Available online: https://www.ajevonline.org/content/48/3/357 (accessed on 5 October 2022).
- Waterhouse, A.L. Determination of Total Phenolics. Curr. Protoc. Food Anal. Chem. 2002, 6, I1.1.1–I1.1.8. [Google Scholar] [CrossRef]






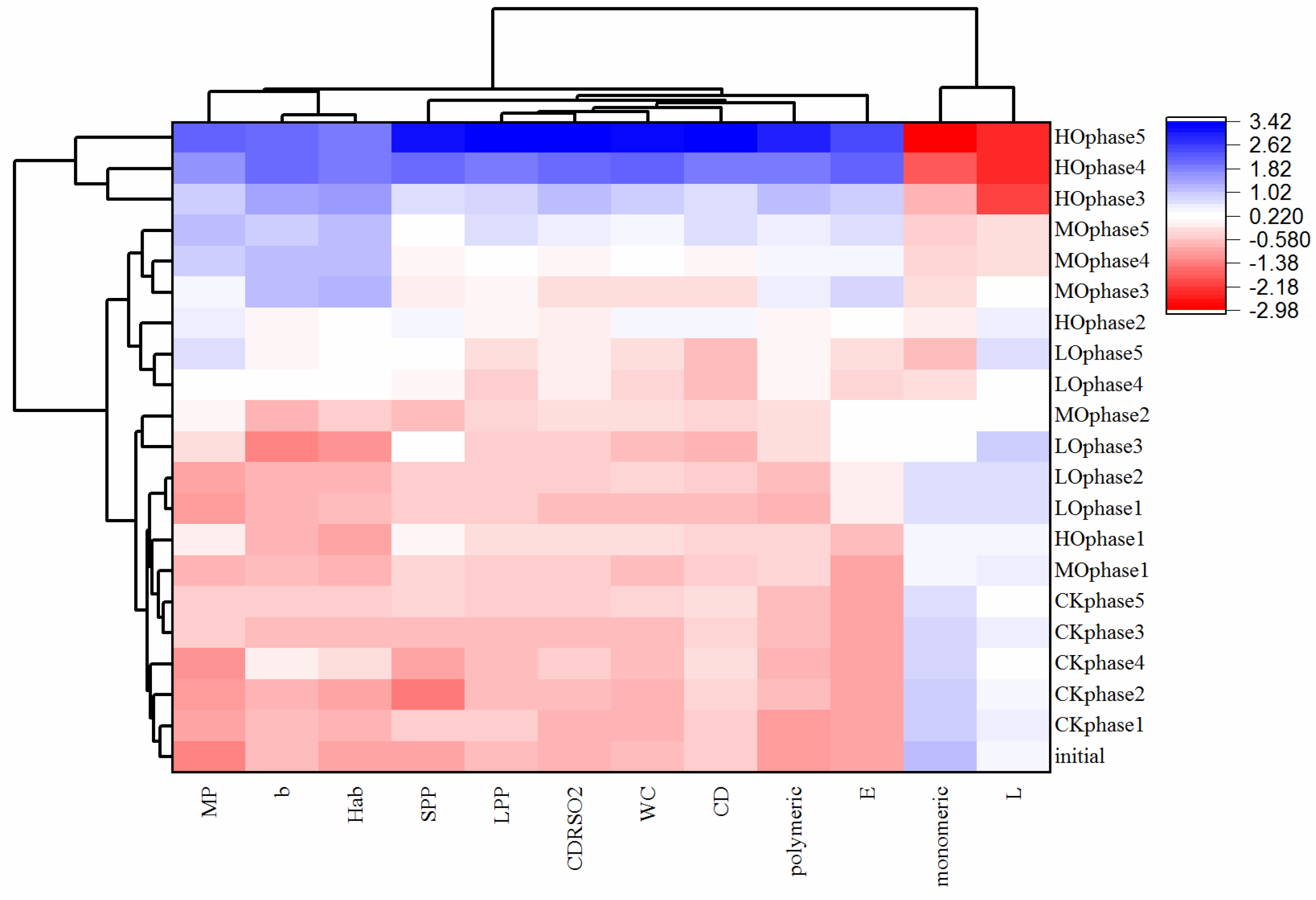
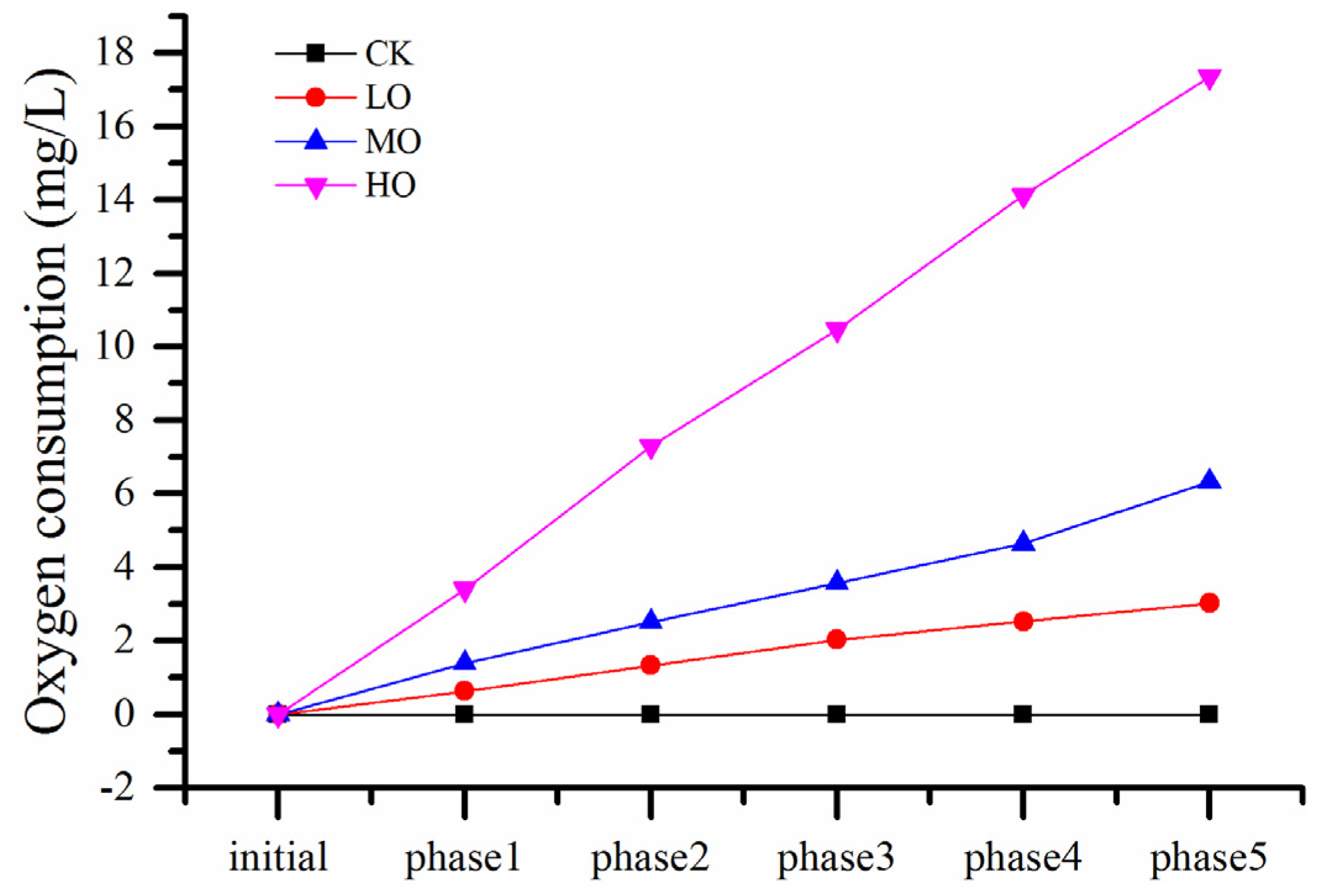
| Parameter | Oxygen Level | Initial | After Phase 1 | After Phase 2 | After Phase 3 | After Phase 5 |
|---|---|---|---|---|---|---|
| Free SO2 (mg/L) | Control | 5.6 ± 0.9 | 5.5 ± 0.6 | NT | NT | 5.2 ± 1.1 |
| LO | 5.6 ± 0.9 | 4.3 ± 0.2 | 2.6 ± 0.5 | 0 | NT | |
| MO | 5.6 ± 0.9 | 3.3 ± 0.5 | 0 | NT | NT | |
| HO | 5.6 ± 0.9 | 0 | NT | NT | NT | |
| Bound SO2 (mg/L) | Control | 28.9 ± 2.2 | NT | NT | NT | 27.9 ± 3.1 |
| LO | 28.9 ± 2.2 | NT | NT | NT | 25.4 ± 2.7 | |
| MO | 28.9 ± 2.2 | NT | NT | NT | 24.7 ± 3.3 | |
| HO | 28.9 ± 2.2 | NT | NT | NT | 21.5 ± 2.9 |
| CIELab | Control | LO | MO | HO |
|---|---|---|---|---|
| a* | 45.5 ± 0.3 a | 45.3 ± 0.3 a | 45.6 ± 0.3 a | 46.4 ± 0.3 b |
| b* | 12.5 ± 0.3 a | 13.5 ± 0.3 b | 15.4 ± 0.3 c | 17.2 ± 0.3 d |
| L* | 49.3 ± 0.4 c | 50.1 ± 0.5 c | 48.3 ± 0.5 b | 44.9 ± 0.5 a |
| C*a,b | 47.1 ± 0.4 a | 46.8 ± 0.4 a | 48.1 ± 0.4 b | 49.5 ± 0.3 c |
| H*a,b | 15.4 ± 0.2 a | 16.7 ± 0.2 b | 18.7 ± 0.2 c | 20.3 ± 0.2 d |
| ΔE | 0 a | 1.4 ± 0.001 b | 3.0 ± 0.02 c | 6.5 ± 0.01 d |
| Variable | Min | Max | Mean | SD | r (OC) | r (AD) | r (PS) |
|---|---|---|---|---|---|---|---|
| TP (mg/L) | 1342.44 | 1501.12 | 1420.51 | 41.21 | −0.460 * | −0.471 * | −0.548 * |
| Tannin (mg/L) | 533.87 | 645.66 | 601.81 | 28.01 | ns | ns | −0.582 ** |
| MP | 1.59 | 1.84 | 1.69 | 0.071 | 0.891 ** | 0.768 ** | 0.631 ** |
| SPP | 0.73 | 0.98 | 0.81 | 0.056 | 0.945 ** | 0.930 ** | 0.491 * |
| LPP | 0.60 | 1.11 | 0.69 | 0.12 | 0.962 ** | 0.975 ** | 0.483 * |
| WC | 1.12 | 1.35 | 1.16 | 0.058 | 0.979 ** | 0.975 ** | 0.464 * |
| CDRSO2 | 0.51 | 0.78 | 0.56 | 0.066 | 0.960 ** | 0.973 ** | 0.510 * |
| CD | 2.23 | 2.68 | 2.32 | 0.11 | 0.929 ** | 0.971 ** | 0.441 * |
| Hue | 0.69 | 0.73 | 0.71 | 0.0098 | ns | ns | ns |
| monomeric (%) | 20.03 | 33.52 | 29.89 | 3.33 | −0.946 ** | −0.903 ** | −0.593 ** |
| co-pigmented (%) | 21.01 | 22.9 | 21.99 | 0.49 | ns | ns | ns |
| polymeric (%) | 44.40 | 57.80 | 48.07 | 3.30 | 0.962 ** | 0.914 ** | 0.562 ** |
| a* | 44.50 | 46.70 | 45.66 | 0.69 | ns | ns | ns |
| b* | 11.20 | 17.30 | 13.54 | 1.84 | 0.853 ** | 0.754 ** | 0.583 ** |
| L* | 44.80 | 50.40 | 48.83 | 1.69 | −0.895 ** | −0.858 ** | ns |
| C* ab | 46.00 | 49.70 | 47.73 | 1.00 | 0.607 ** | 0.604 ** | ns |
| H* ab | 14.10 | 20.40 | 16.48 | 2.09 | 0.851 ** | 0.735 ** | 0.612 ** |
| ΔE | 0 | 6.50 | 1.80 | 1.91 | 0.930 ** | 0.861 ** | 0.446 * |
| Parameter | Slope | ±Standard Deviation | Change per mg/L of Consumed O2 |
|---|---|---|---|
| MP | 0.82 | 0.096 | 0.91% |
| SPP | 1.44 | 0.11 | 1.69% |
| LPP | 4.13 | 0.27 | 4.86% |
| WC | 1.04 | 0.050 | 1.12% |
| CDRSO2 | 2.58 | 0.17 | 3.06% |
| CD | 0.89 | 0.081 | 1.06% |
| monomeric (%) | −1.94 | 0.15 | −2.32% |
| polymeric (%) | 1.48 | 0.096 | 1.74% |
| b* | 2.64 | 0.37 | 2.30% |
| L* | −0.63 | 0.072 | −0.56% |
| H*ab | 2.51 | 0.36 | 2.29% |
| ΔE † | 0.37 | 0.033 | 0.37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, L.; Sun, Y.; Liu, M.; Cui, X.; Wang, J.; Li, J.; Han, G. Influence of Oxygen Management during the Post-Fermentation Stage on Acetaldehyde, Color, and Phenolics of Vitis vinifera L. Cv. Cabernet Sauvignon Wine. Molecules 2022, 27, 6692. https://doi.org/10.3390/molecules27196692
Dai L, Sun Y, Liu M, Cui X, Wang J, Li J, Han G. Influence of Oxygen Management during the Post-Fermentation Stage on Acetaldehyde, Color, and Phenolics of Vitis vinifera L. Cv. Cabernet Sauvignon Wine. Molecules. 2022; 27(19):6692. https://doi.org/10.3390/molecules27196692
Chicago/Turabian StyleDai, Lingmin, Yuhang Sun, Muqing Liu, Xiaoqian Cui, Jiaqi Wang, Jiming Li, and Guomin Han. 2022. "Influence of Oxygen Management during the Post-Fermentation Stage on Acetaldehyde, Color, and Phenolics of Vitis vinifera L. Cv. Cabernet Sauvignon Wine" Molecules 27, no. 19: 6692. https://doi.org/10.3390/molecules27196692





