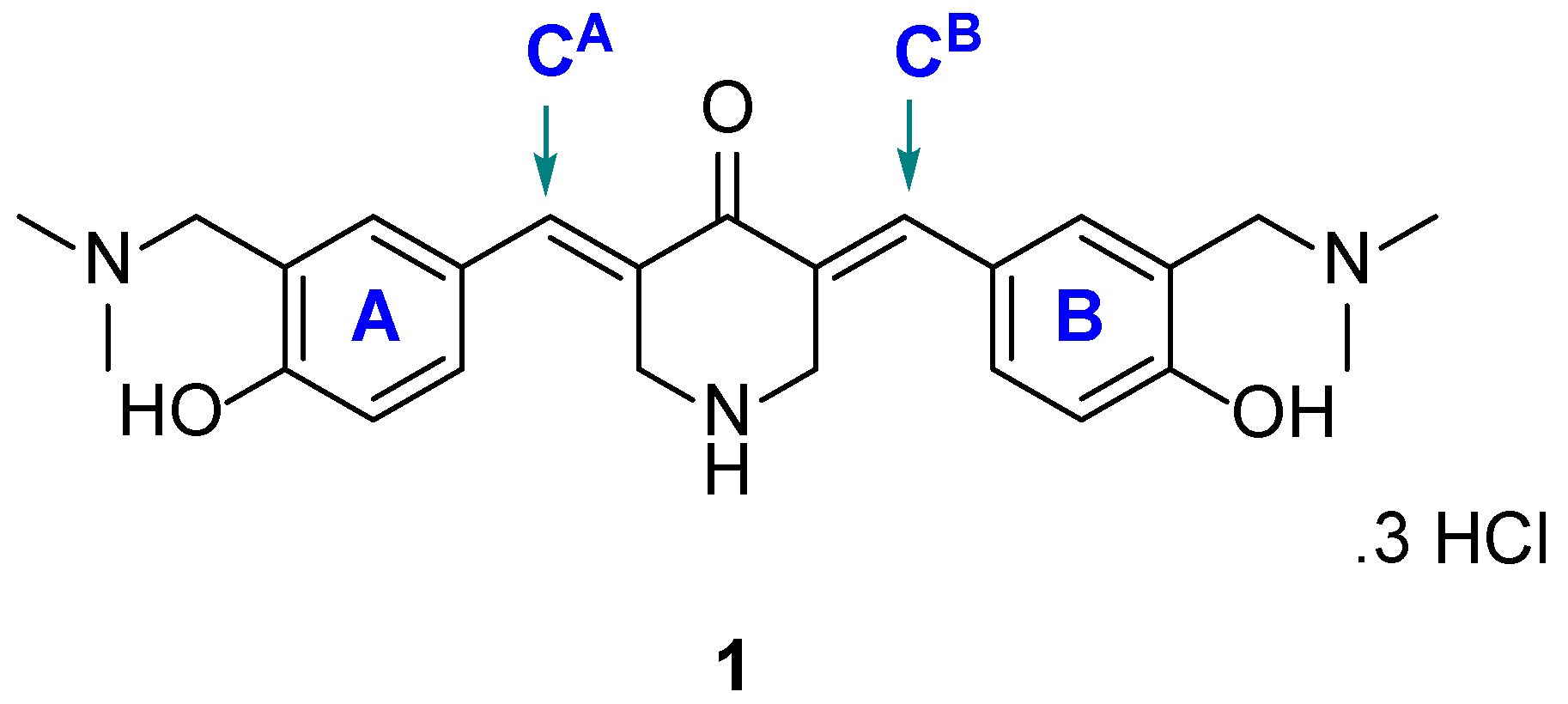
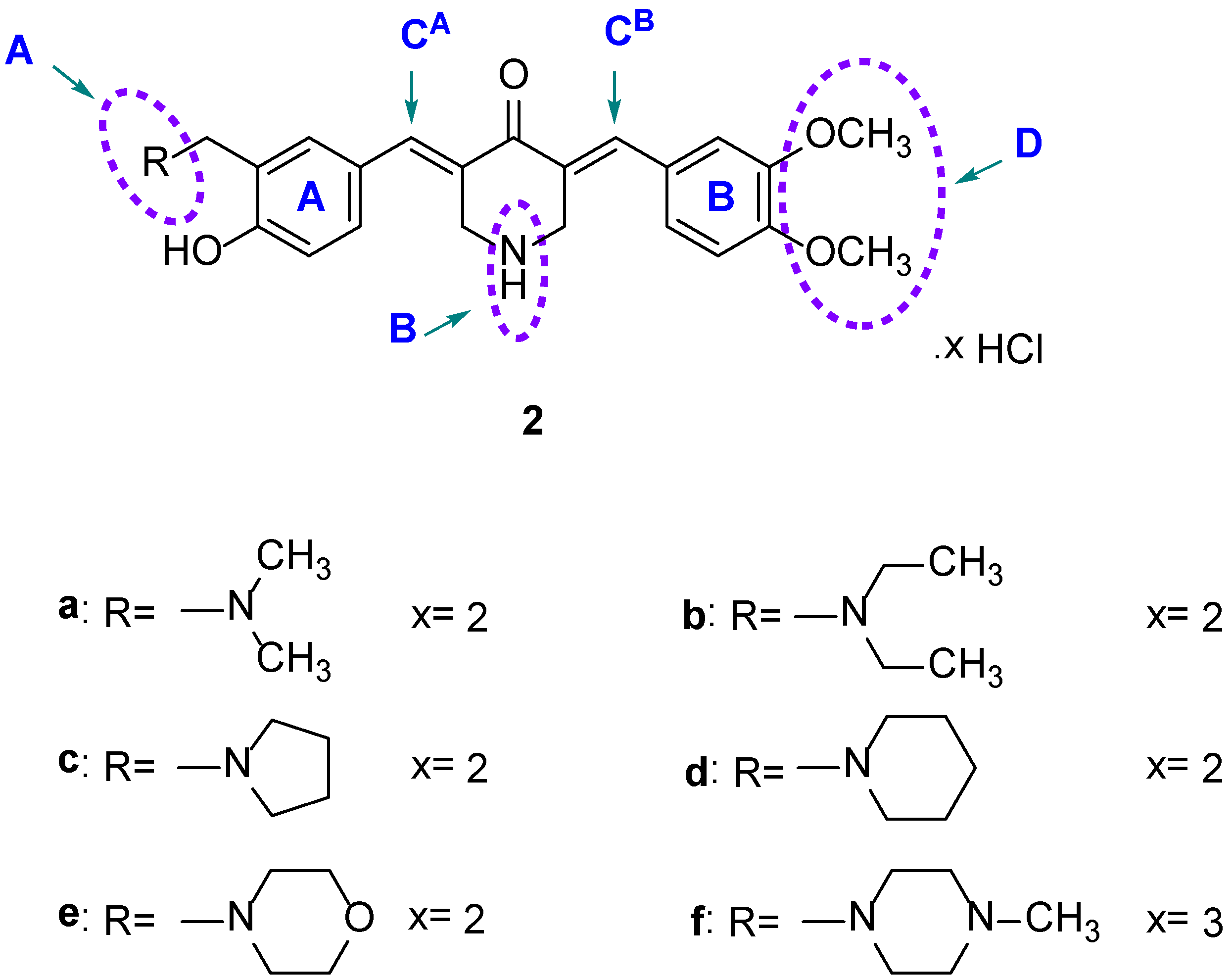
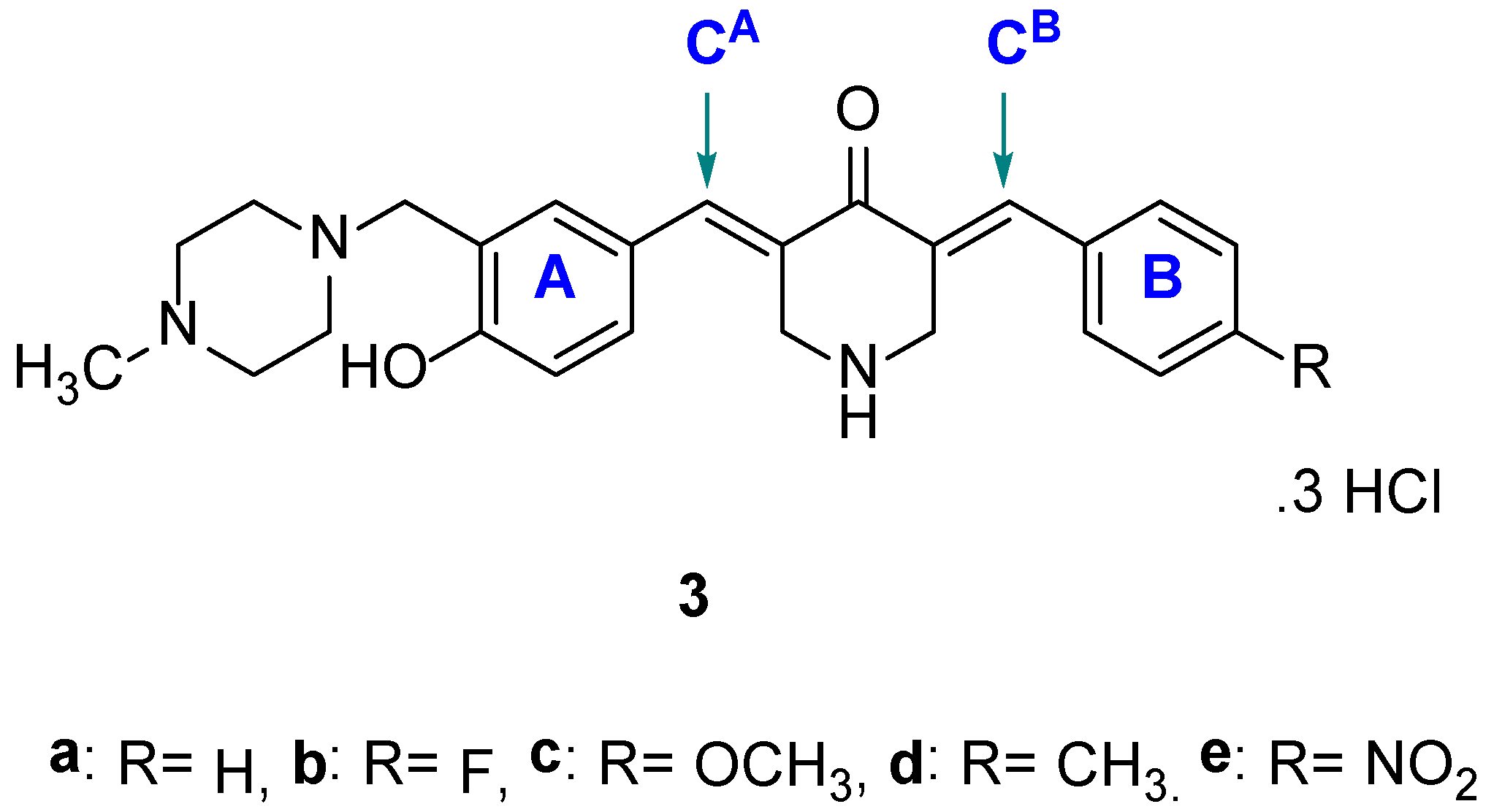
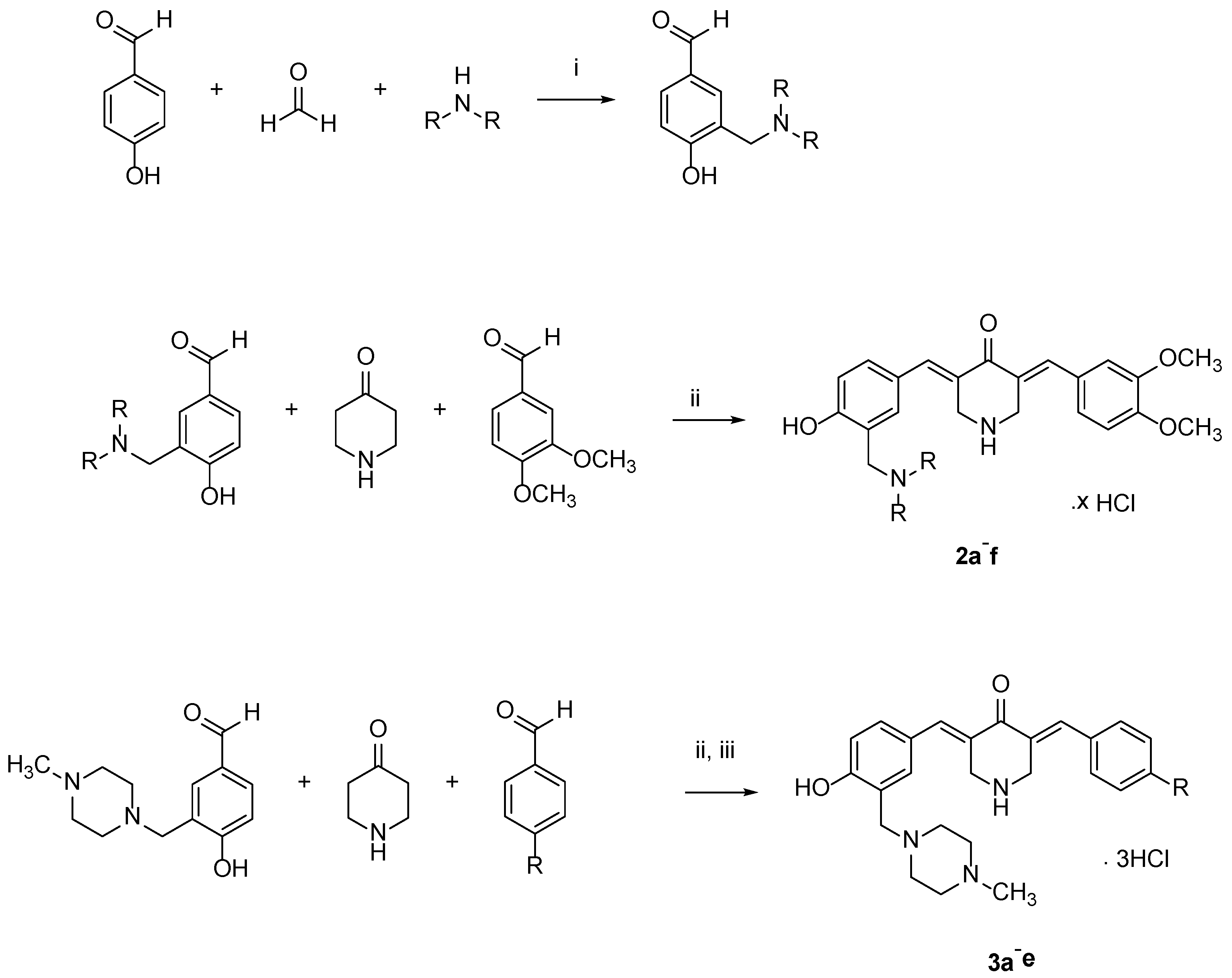
4.1. Syntheses of Compounds
A Bruker Avance AMX 500 FT spectrometer (Billerica, MA, USA) equipped with a broad band observe (BBO) probe was used to obtain 1H and 13C nuclear magnetic resonance (NMR) spectra of compounds in deuterated chloroform (CDCl3) and deuterated dimethyl sulfoxide (DMSO-d6). Mass spectra were obtained using a Jeol JMS-T100 GCv Accu tof-gcV4G instrument (Peabody, MA, USA). A DigiMelt-MPA 160 instrument (Sunnyvale, CA, USA) was used to determine melting points that are uncorrected.
The intermediate 3-aminomethyl-4-hydroxybenzaldehydes were prepared by a general method. A solution of 4-hydroxybenzaldehyde (0.05 mol) in methanol (50 mL was added to a preheated mixture of paraformaldehyde (0.06 mol) and the appropriate amine (0.05 mol) in methanol (50 mL) at 70 °C for 30 min. After the addition of 4-hydroxybenzaldehyde, the mixture was stirred at 70 °C for 2 h. On completion of the reaction, the solvent was removed, and the residue was dissolved in diethyl ether. Hydrogen chloride gas was passed through the solution for 10 min to produce the hydrochloride salt, which precipitated. The solid formed was collected by filtration and washed thoroughly with ether and dried. The
1H NMR,
13C NMR and Mass spectra are consistent with the proposed structures. The spectra of the available compounds are presented in the
Supplemental section (Spectra S1, Spectra S2 and Spectra S3).
The hydrochloride salts were treated with an aqueous potassium carbonate solution (10% w/v), and the mixture was extracted with chloroform. The removal of the organic solvent in vacuo afforded the free bases that were used in the syntheses of 2a–f and 3a–e.
The compounds in series 2 were prepared as follows. 3,4-Dimethoxybenzaldehyde (0.0025 mol) and the appropriate 3-aminomethyl-4-hydroxybenzaldehyde (0.0025 mol) were added to a suspension of 4-piperidone hydrochloride monohydrate (0.005 mol) in acetic acid (35 mL). Dry hydrogen chloride was passed through this mixture for 0.5 h. The mixture was stirred at room temperature for 24 h, and the precipitate that formed was collected, washed with water, and added to an aqueous solution of potassium carbonate (25% w/v, 25 mL). The resultant mixture was stirred for 0.5 h. The free bases were washed with water (50 mL) and dried. The compounds were purified by recrystallization from 95% ethanol and then by using a chromatography column of silica gel 60 and an eluting solvent of a mixture of methanol and chloroform (1:99). Hydrogen chloride gas was passed into a solution of the pure compound, which was dissolved in chloroform. The hydrochloride salts were collected, washed with acetone, and dried. The compounds in series 3 were prepared in the same manner as used in the syntheses of 2a–f. The melting points of 2a–f and 3a–e are over 300 °C.
(2a) 3-(3,4-Dimethoxybenzylidene)-5-(3-((dimethylamino)methyl)-4-hydroxybenzylidene)-4-piperidone dihydrochloride
Yield: 40%. 1H NMR (DMSO-d6): δ 2.39 (s, 6H, 2× NCH3), 3.74 (s, 2H, Ar-CH2-N), 3.96 (s, 3H, -OCH3), 3.98 (s, 3H, -OCH3), 4.12 (s, 2H, NCH2), 4.14 (s, 2H, NCH2), 6.75 (s, 4H, Ar-H), 6.97 (d, 2H, Ar-H), 7.75 (s, 2H, 2× =CH). 13C NMR (DMSO-d6): δ 40.55, 53.56, 56.12, 114.84, 126.92, 139.75, 149.15, 151.06, 182.44. HRMS (ESI): (m/z): 408.2057 (M+), Calc: 408.2049 for C24H28N2O4, 409.2087 (M + 1), 410.2130 (M + 2)
(2b) 3-(3-Diethylaminomethyl-4-hydroxybenzylidene)-5-(3,4-dimethoxybenzylidene)-4-piperidone dihydrochloride
Yield: 40%. 1H NMR (DMSO-d6): δ 0.95 (t, 6H, 2× N-CH2-CH3), 2.68 (q, 4H, 2× -CH2-CH3), 3.70 (s, 2H, Ar-CH2-N), 3.90 (s, 3H, -OCH3), 3.94 (s, 3H, -OCH3), 4.2 (d, 2H, -NCH2), 6.88–6.95 (m, 2H, Ar-H), 7.15 (s, 4H, Ar-H), 7.78 (s, 2H, 2× =CH). 13C NMR (DMSO-d6): 40.38, 46.65, 49.59, 56.14, 112.23, 114.84, 116.73, 117.84, 124.72, 126.14, 126.91, 139.76, 149.15, 159.17, 182.44. HRMS (ESI): (m/z): 436.2304 (M+), Calc: 436.2362 for C26H32N2O4, 437.2338 (M + 1), 438.2378 (M + 2)
(2c) 3-[3-(1-Pyrrolidinylmethyl)-4-hydroxybenzylidene]-5-(3,4-dimethoxybenzylidene)-4-piperidone dihydrochloride
Yield: 50%. 1H NMR (DMSO-d6): δ 1.85 (br. s, 4H, 2× N-CH2-CH2), 2.65 (br. s, 4H, 2× N-CH2-CH2), 3.92 (br. s, 4H, 2× NCH2), 3.95 (s, 3H, -OCH3), 3.97 (s, 3H, -OCH3), 4.17 (s, 2H, Ar-CH2-N), 6.88–7.15 (m, 4H, Ar-H), 7.28 (s, 2H, Ar-H), 7.75 (s, 2H, 2 × =CH). 13C NMR (DMSO-d6): 23.08, 44.54, 51.54, 56.14, 112.23, 114.83, 124.72, 126.14, 126.91, 139.77, 149.15, 158.82, 182.45. HRMS (ESI): (m/z): 434.2215 (M+), Calc: 434.2205 for C26H30N2O4 435.2256 (M + 1), 436.2300 (M + 2)
(2d) 3-[3-(1-Piperidinylmethyl)-4-hydroxybenzylidene]-5-(3,4-dimethoxybenzylidene)-4-piperidone dihydrochloride
Yield: 30%. 1H NMR (DMSO-d6): δ 2.45 (t, 2H, -CH2-N-CH2), 3.19 (t, 2H, -CH2-N-CH2), 3.74 (s, 2H, Ar-CH2-N), 3.92 (s, 3H, -OCH3), 3.95 (s, 3H, -OCH3), 4.2 (d, 4H, 2× NCH2), 6,83-6.96 (m, 4H, Ar-H), 7.23-7.73 (s, 2H, 2× =CH), 7.33 (s, 2H, Ar-H). 13C NMR (DMSO-d6): 22.64, 40.55, 44.52, 52.13, 56.14, 112.23, 114.84, 124.71, 126.15, 126.92, 139.75, 149.15, 159.38, 182.44. HRMS (ESI): (m/z): 448.2351 (M+), Calc: 448.2362 for C27H32N2O4, 449.2395 (M + 1), 450.2431 (M + 2)
(2e) 3-[3-(4-Morpholinomethyl)-4-hydroxybenzylidene]-5-(3,4-dimethoxybenzylidene)-4-piperidone dihydrochloride
Yield: 30%. 1H NMR (DMSO-d6): δ 2.55 (t, 4H, -CH2-N-CH2), 3.30 (t, 4H, -CH2-O-CH2), 3.64 (s, 2H, Ar-CH2-N), 3.75 (4H, 2× NCH2), 3.85–4.10 (m, 6H, 2× OCH3), 6.75–7.25 (m, 4H, Ar-H), 7.40 (s, 2H, Ar-H), 7.45 (s, 2H, 2× =CH). 13C NMR (DMSO-d6): 44.54, 56.02, 56.16, 63.49, 79.68, 112.41, 114.83, 120.04, 122.62, 127.99, 128.07, 139.87, 148.87, 149.14, 182.44. HRMS (ESI): (m/z): 450.2169 (M+), Calc: 450.2154 for C26H30N2O5, 451.2201 (M + 1), 452.2265 (M + 2)
(2f) 3-[3-(4-Methyl-1-piperazinylmethyl)-4-hydroxybenzylidene]-5-(3,4-dimethoxybenzylidene)-4-piperidone trihydrochloride
Yield: 50%. 1H NMR (DMSO-d6): δ 2.38 (s, 3H, N-CH3), 2.40–2.52, (8H, 2× -CH2-N-CH2), 3.75 (m, 4H, 2× NCH2), 3.78 (s, 2H, Ar-CH2-N), 3.85–3.90 (m, 6H, 2× OCH3), 6.84–6.90 (m, 4H, Ar-H), 7.02–7.08 (s, 2H, Ar-H), 7.75 (s, 2H, 2× =CH). 13C NMR (DMSO-d6): 39.45, 48.11, 55.02, 56.00, 58.64, 112.04, 114.43, 116.20, 123.21, 124.35, 128.23, 134.51, 148.94, 150.10, 158.59, 182.45. HRMS (ESI): (m/z): 463.2465 (M+), Calc: For C27H33N3O4, 464.2491 (M + 1), 465.2515 (M + 2)
(3a) 3-[4-Methyl-1-piperazinylmethyl-4-hydroxybenzylidene]-5-benzylidene-4-piperidone trihydrochloride
Yield: 40%. 1H NMR (DMSO-d6): δ 2.38 (s, 3H, N-CH3), 2.69 (br. s, 8H, 2× -CH2-N-CH2), 3.77 (s, 2H, Ar-CH2-N), 4.18 (s, 4H, 2× NCH2), 6.90–7.10 (m, 2H, Ar-H), 7.39 (s, 4H, Ar-H), 7.45 (m, 2H, Ar-H), 7.75–7.82 (s, 2H, 2× =CH). 13C NMR (DMSO-d6): 48.27, 61.13, 77.19, 99.08, 116.06, 128.31, 129.07, 130.60, 133.13, 135.65, 136.72, 141.92, 180.91. HRMS (ESI): (m/z): 403.2246 (M+), Calc: For C25H29N3O2, 404.2258 (M + 1), 406.2324 (M + 2)
(3b) 3-[4-Methyl-1-piperazinylmethyl-4-hydroxybenzylidene]-5-(4-fluorobenzylidene)-4-piperidone trihydrochloride
Yield: 45%. 1H NMR (DMSO-d6): δ 2.39 (s, 3H, N-CH3), 2.72 (br. s, 8H, 2× -CH2-N-CH2), 3.72 (s, 2H, Ar-CH2-N), 4.20 (br. s, 4H, 2× NCH2), 6.78–7.16 (m, 4H, Ar-H), 7.33–7.42 (s, 3H, Ar-H), 7.70–7.86 (s, 2H, 2× =CH). 13C NMR (CDCl3): 47.97, 76.58, 115.15, 116.52, 131.82, 132.44, 134.27, 135.04, 136.72, 142.00, 151.26, 154.16, 172.92, 196.10. HRMS (ESI): (m/z): 421.2155 (M+), Calc: For C25H28FN3O2, 422.2227 (M + 1), 423.2297 (M + 2)
(3c) 3-[4-Methyl-1-piperazinylmethyl-4-hydroxybenzylidene]-5-(4-methoxybenzylidene)-4-piperidone trihydrochloride
Yield: 50%. 1H NMR (DMSO-d6): δ 2.38 (s, 3H, N-CH3) 2.68, (br. s, 8H, 2× -CH2-N-CH2), 3.73 (s, 2H, Ar-CH2-N), 3.84 (s, 3H, -OCH3), 4.17 (s, 4H, 2× NCH2), 6.75–7.00 (m, 4H, Ar-H), 7.08–7.32 (m, 3H, Ar-H), 7.70 (s, 1H, Ar-H), 7.72 (s, 1H, Ar-H). HRMS (ESI): (m/z): 433.2350 (M+), Calc: For C26H31N3O3, 434.2401 (M + 1), 435.2449 (M + 2)
(3d) 3-[4-Methyl-1-piperazinylmethyl-4-hydroxybenzylidene]-5-(4-methylbenzylidene)-4-piperidone trihydrochloride
Yield: 50%. 1H NMR (DMSO-d6): δ 2.05 (s, 3H, N-CH3), 2.08 (t, 8H, 2× -CH2-N-CH2), 2.48 (s, 3H, Ar-CH3), 3.77 (s, 2H, -NCH2), 3.79 (s, H, -NCH2), 4.18 (s, H, Ar-CH2-N), 6.75-6.98 (m, 4H, Ar-H), 7.09–7.19 (m, 3H, Ar-H), 7.63–7.81 (m, 2H, 2× =CH). 13C NMR (CDCl3): 43.53, 45.82, 48.12, 60.67, 61.43, 77.35, 116.68, 122.18, 125.55, 129.22, 130.45, 131.52, 131.67, 135.65, 136.26, 190.89. HRMS (ESI): (m/z): 417.2396 (M+), Calc: For C26H31N3O2, 418.2419 (M + 1), 419.2472 (M + 2)
(3e) 3-[4-Methyl-1-piperazinylmethyl-4-hydroxybenzylidene]-5-(4-nitrobenzylidene)-4-piperidone trihydrochloride
Yield: 50%. 1H NMR (DMSO-d6): δ 2.19 (s, 3H, N-CH3), 2.32–2.61 (m, 8H, 2× -CH2-N-CH2), 3.79 (s, 2H, Ar-CH2-N), 4.18 (s, H, -NCH2), 4.19 (s, H, -NCH2), 6.78–7.16 (m, 3H, Ar-H), 7.49–7.61 (m, 2H, Ar), 7.75–7.83 (m, 2H, Ar-H), 8.22–8.33 (m, 2H, 2× =CH). 13C NMR (CDCl3): 62.8, 77.35, 116.68, 123.71, 130.91, 131.98, 132.28, 137.49, 141.77, 190.74. HRMS (ESI): (m/z): 449.2191 (M+), Calc: For C25H28N4O4, 450.2255 (M + 1), 451.2318 (M + 2)